Cracking the AP Biology Exam
4
Cellular Energetics
TYPES OF REACTIONS
Exergonic reactions are those in which the products have less energy than the reactants. Simply put, energy is given off during the reaction.
Let’s look at an example. The course of a reaction can be represented by an energy diagram. Here’s an energy diagram for an exergonic reaction:
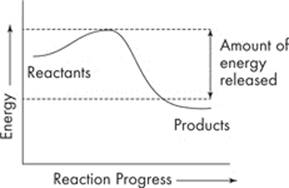
You’ll notice that energy is represented along the y-axis. Based on the diagram, our reaction released energy. An example of an exergonic reaction is when food is oxidized in mitochondria of cells and then releases the energy stored in the chemical bonds.
Reactions that require an input of energy are called endergonic reactions. You’ll notice that the products have more energy than the reactants.
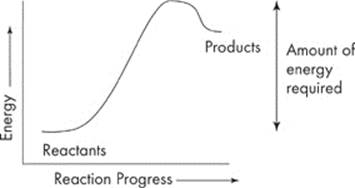
The products gained energy in the form of heat. An example is when plants use carbon dioxide and water to form sugars.
ACTIVATION ENERGY
Although we said that exergonic reactions release energy, this does not mean that they do not require any energy to get started. Take a look at this energy diagram of a typical exergonic reaction:
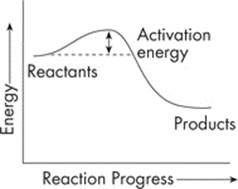
You’ll notice that we needed a little energy to get us going. That’s because chemical bonds must be broken before new bonds can form. This energy barrier—the hump in the graph—is called the activation energy. Once a set of reactants has reached its activation energy, the reaction can occur much faster than it would in the absence of the enzyme.
GETTING BACK TO ENZYMES…
Why are enzymes so important in biology? They’re important because many reactions would never occur in the cell if it weren’t for the help of enzymes. As we saw earlier, enzymes, by definition, catalyze reactions: They activate them. What this means in chemical terms is that theylower the activation energy of a reaction, enabling the reaction to occur much faster than it would in the absence of the enzyme.
ENZYME SPECIFICITY
Most of the crucial reactions that occur in the cell require enzymes. Yet enzymes themselves are highly specific—in fact, each enzyme catalyzes only one kind of reaction. This is known as enzyme specificity. Since this is true, enzymes are usually named after the molecules they target. In enzymatic reactions, the targeted molecules are known as substrates. For example, maltose, a disaccharide, can be broken down into two glucose molecules. Our substrate, maltose, gives its name to the enzyme that catalyzes this reaction: maltase.
Many enzymes are named simply by replacing the suffix of the substrate with –ase. Using this nomenclature, maltose becomes maltase.
ENZYME-SUBSTRATE COMPLEX
Enzymes have a unique way of helping reactions along. As we just saw, the reactants in an enzyme-assisted reaction are known as substrates. During a reaction, the enzyme’s job is to bring the substrates together. It accomplishes this due to a special region on the enzyme known as anactive site.
The enzyme temporarily binds the substrates to its active site, and forms an enzyme-substrate complex. Let’s take a look:
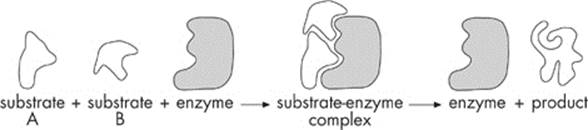
Once the reaction has occurred and the product is formed, the enzyme is released from the complex and restored to its original state. Now, the enzyme is free to react again with another bunch of substrates.
By binding and releasing over and over again, the enzyme speeds the reaction along, enabling the cell to release much-needed energy from various molecules. Here is a quick review on the function of enzymes.
Enzymes Do
- Increase the rate of a reaction by lowering the reaction’s activation energy
- Form temporary enzyme-substrate complexes
- Remain unaffected by the reaction
Enzymes Don’t
- Change the reaction
- Make reactions occur that would otherwise not occur at all
INDUCED FIT
However, scientists have discovered that enzymes and substrates don’t fit together quite so seamlessly. It appears that the enzyme has to change its shape slightly to accommodate the shape of the substrates. This is called induced fit.
ENZYMES DON’t ALWAYS WORK ALONE
Enzymes sometimes need a little help in catalyzing a reaction. Those factors are known as coenzymes. Vitamins are examples of organic coenzymes. Your daily dose of vitamins is important for just this reason: The vitamins are “active and necessary participants” in crucial chemical reactions. The function of coenzymes is to accept electrons and pass them along to another substrate. Two examples of such enzymes are NAD+ and NADP+.
In addition to organic coenzymes, inorganic elements—called cofactors—help catalyze reactions. These elements are usually metal ions, such as Fe+2.
FACTORS AFFECTING REACTION RATES
Enzymatic reactions can be influenced by a number of factors, such as temperature, pH, and the relative amounts of enzyme and substrate.
Temperature
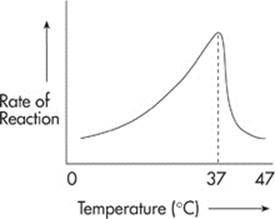
The rate of a reaction increases with increasing temperature, up to a point, because an increase in the temperature of a reaction increases the chance of collisions among the molecules. But too much heat can damage an enzyme. If a reaction is conducted at an excessively high temperature (above 42°C), the enzyme loses its three-dimensional shape and becomes inactivated. Enzymes damaged by heat and deprived of their ability to catalyze reactions are said to be denatured.
Here’s one thing to remember: All enzymes operate at an ideal temperature. For most human enzymes, this temperature is body temperature, 37°C.
pH
Enzymes also function best at a particular pH. For most enzymes, the optimal pH is at or near a pH of 7:
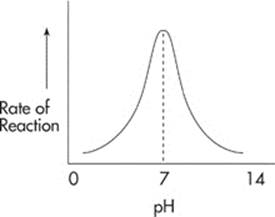
Other enzymes operate at a low pH. For instance, pepsin, the digestive enzyme found in the stomach, is most effective at an extremely acidic pH of 2.
Here’s another important piece of information:
Most enzymes are active over a narrow range of pH.
ENZYME REGULATION
We know that enzymes control the rates of chemical reactions. But what regulates the activity of enzymes? It turns out that a cell can control enzymatic activity by regulating the conditions that influence the shape of the enzyme. For example, some enzymes have allosteric sites, a region of the enzyme other than the active site to which a substance can bind. Substances called allosteric regulators can either inhibit or activate enzymes. An allosteric inhibitor will bind to an allosteric site and keep the enzyme in its inactive form while an allosteric activator will bind to an enzyme and induce its active form. Allosteric enzymes are subject to feedback inhibition in which the formation of an end product inhibits an earlier reaction in the sequence.
Most enzymes can be inhibited by certain chemical substances. If the substance has a shape that fits the active site of an enzyme, it can compete with the substrate and effectively inactivate the enzyme. This is called competitive inhibition. Usually a competitive inhibitor is structurally similar to the normal substrate. In noncompetitive inhibition, the inhibitor binds with the enzyme at a site other than the active site and inactivates the enzyme by altering its shape. This prevents the enzyme from binding with the substrate at the active site.