CONCEPTS IN BIOLOGY
PART II. CORNERSTONES: CHEMISTRY, CELLS, AND METABOLISM
5. Enzymes, Coenzymes, and Energy
5.4. How the Environment Affects Enzyme Action
An enzyme forms a complex with one substrate molecule, encourages a reaction to occur, detaches itself, and then forms a complex with another molecule of the same substrate. The number of molecules of substrate with which a single enzyme molecule can react in a given time (e.g., reactions per minute) is called the turnover number.
Sometimes, the number of jobs an enzyme can perform during a particular time period is incredibly large—ranging between a thousand (103) and 10 thousand trillion (1016) times faster per minute than uncatalyzed reactions. Without the enzyme, perhaps only 50 or 100 substrate molecules might be altered in the same time. With this in mind, let’s identify the ideal conditions for an enzyme and consider how these conditions influence the turnover number.
Temperature
An important condition affecting enzyme-controlled reactions is the environmental temperature (figure 5.5), which has two effects on enzymes: (1) It can change the rate of molecular motion, and (2) it can cause changes in the shape of an enzyme. An increase in environmental temperature increases molecular motion. Therefore, as the temperature of an enzyme-substrate system increases, the amount of product molecules formed increases, up to a point. The temperature at which the rate of formation of enzyme-substrate complex is fastest is termed the optimum temperature. Optimum means the best or most productive quantity or condition. In this case, the optimum temperature is the temperature at which the product is formed most rapidly. As the temperature decreases below the optimum, molecular motion slows, and the rate at which the enzyme- substrate complexes form decreases. Even though the enzyme is still able to operate, it does so very slowly. Foods can be preserved by storing them in freezers or refrigerators because the enzyme-controlled reactions of the food and spoilage organisms are slowed at lower temperatures.
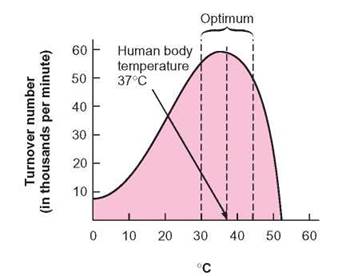
FIGURE 5.5. The Effect of Temperature on Turnover Number
As the temperature increases, the turnover number increases. The increasing temperature increases molecular motion and may increase the number of times an enzyme contacts and combines with a substrate molecule. Temperature may also influence the shape of the enzyme molecule, making it fit better with the substrate. At high temperatures, the enzyme molecule is irreversibly changed, so that it can no longer function as an enzyme. At that point, it has been denatured. Notice that the enzyme represented in this graph has an optimum (best) temperature range of between 30°C and 45°C.
When the temperature is raised above the optimum, some of the enzyme molecules are changed in such a way that they can no longer form the enzyme-substrate complex; thus, the reaction slows. If the temperature continues to increase, more and more of the enzyme molecules become inactive. If the temperature is high enough, it causes permanent changes in the three-dimensional shape of the molecules. The surface geometry of the enzyme molecule is not recovered, even when the temperature is reduced. Recall the wrench analogy. When a wrench is heated above a certain temperature, the metal begins to change shape. The shape of the wrench is changed permanently, so that, even if the temperature is reduced, the surface geometry of the end of the wrench is permanently lost. When this happens to an enzyme, it has been denatured. A denatured enzyme is one whose protein structure has been permanently changed, so that it has lost its original biochemical properties. Because enzymes are molecules and are not alive, they are not killed but, rather, denatured. For example, although egg white is not an enzyme, it is a protein and provides a common example of what happens when denaturation occurs as a result of heating. As heat is applied to the egg white, it is permanently changed from a runny substance to a rubbery solid (denatured).
Many people have heard that fevers cause brain damage. Brain damage from a fever can result from the denaturation of proteins if the fever is over 42°C (107.6°F). However, denaturation and brain damage from fevers is rare, because untreated fevers seldom go over 40.5°C (105°F) unless the child is overdressed or trapped in a hot place. Generally, the brain’s thermostat will stop the fever from going above (41.1°C) 106°F. Children with a rectal temperature of 106°F or higher also have a greater risk for serious bacterial infection and for viral illness, or both.
pH
Another environmental condition that influences enzyme action is pH. The three-dimensional structure of a protein leaves certain side chains exposed. These side chains may attract ions from the environment. Under the right conditions, a group of positively charged hydrogen ions may accumulate on certain parts of an enzyme. In an environment that lacks these hydrogen ions, this would not happen. Thus, variation in the enzyme’s shape could be caused by a change in the number of hydrogen ions present in the solution. Because the environmental pH is so important in determining the shapes of protein molecules, there is an optimum pH for each specific enzyme. The enzyme will fit with the substrate only when it has the proper shape, and it has the proper shape only when it is at the right pH. Many enzymes function best at a pH close to neutral (7). However, a number of enzymes perform best at pHs quite different from 7. Pepsin, an enzyme found in the stomach, works well at an acid pH of 1.5 to 2.2, whereas arginase, an enzyme in the liver, works well at a basic pH of 9.5 to 9.9 (figure 5.6).
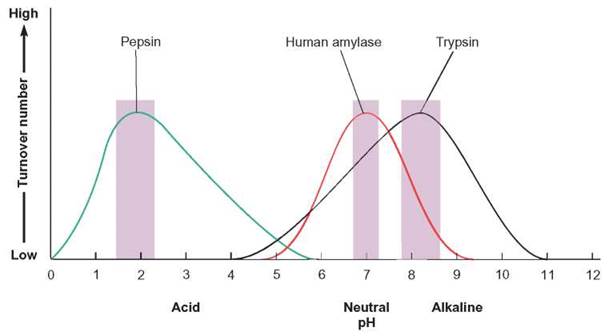
FIGURE 5.6. The Effect of pH on the Turnover Number
As the pH changes, the turnover number changes. The ions in solution alter the environment of the enzyme’s active site and the overall shape of the enzyme. The enzymes illustrated here are human amylase, pepsin, and trypsin. Amylase is found in saliva and is responsible for hydrolyzing starch to glucose. Pepsin is found in the stomach and hydrolyzes protein. Trypsin is produced in the pancreas and enters the small intestine, where it also hydrolyzes protein. Notice that each enzyme has its own pH range of activity, the optimum (shown in the color bars) being different for each.
Enzyme-Substrate Concentration
In addition to temperature and pH, the concentration of enzymes, substrates, and products influences the rates of enzymatic reactions. Although the enzyme and the substrate are in contact with one another for only a short time, when there are huge numbers of substrate molecules it may happen that all the enzymes present are always occupied by substrate molecules. When this occurs, the rate of product formation cannot be increased unless the number of enzymes is increased. Cells can do this by synthesizing more enzymes. However, just because there are more enzyme molecules does not mean that any one enzyme molecule will work any faster. The turnover number for each enzyme stays the same. As the enzyme concentration increases, the amount of product formed increases in a specified time. A greater number of enzymes are turning over substrates; they are not turning over substrates faster. Similarly, if enzyme numbers are decreased, the amount of product formed declines.
We can also look at this from the point of view of the substrate. If substrate is in short supply, enzymes may have to wait for a substrate molecule to become available. Under these conditions, as the amount of substrate increases, the amount of product formed increases. The increase in product is the result of more substrate being available to be changed. When there is a very large amount of substrate, all the enzymes will be occupied all the time. However, if given enough time, even a small amount of enzyme can eventually change all the substrate to product; it just takes longer.
5.4. CONCEPT REVIEW
9. What is the turnover number? Why is it important?
10. How does changing temperature affect the rate of an enzyme-controlled reaction?
11. What factors in a cell can speed up or slow down enzyme reactions?
12. What effect might a change in pH have on enzyme activity?