THE LIVING WORLD
Unit two. The Living Cell
7. How Cells Harvest Energy from Food
7.6. Glucose Is Not the Only Food Molecule
We have considered in detail the fate of a molecule of glucose, a simple sugar, in cellular respiration. But how much of what you eat is sugar? As a more realistic example of the food you eat, consider the fate of a fast-food hamburger. The hamburger you eat is composed of carbohydrates, fats, protein, and many other molecules. This diverse collection of complex molecules is broken down by the process of digestion in your stomach and intestines into simpler molecules. Carbohydrates are broken down into simple sugars, fats into fatty acids, and proteins into amino acids. These breakdown reactions produce little or no energy themselves, but prepare the way for cellular respiration—that is, glycolysis and oxidative metabolism. Nucleic acids are also present in the food you eat and are broken down during digestion, but these macromolecules store little energy that the body actually uses.
We have seen what happens to the glucose. What happens to the amino acids and fatty acids? These subunits undergo chemical modifications that convert them into products that feed into cellular respiration.
Cellular Respiration of Protein
Proteins (the second category in figure 7.8) are first broken down into their individual amino acids. A series of deamination reactions removes the nitrogen side groups (called amino groups) and converts the rest of the amino acid into a molecule that takes part in the Krebs cycle. For example, alanine is converted into pyruvate, glutamate into α-ketoglutarate, and aspartate into oxaloacetate. The reactions of the Krebs cycle then extract the high-energy electrons from these molecules and put them to work making ATP.
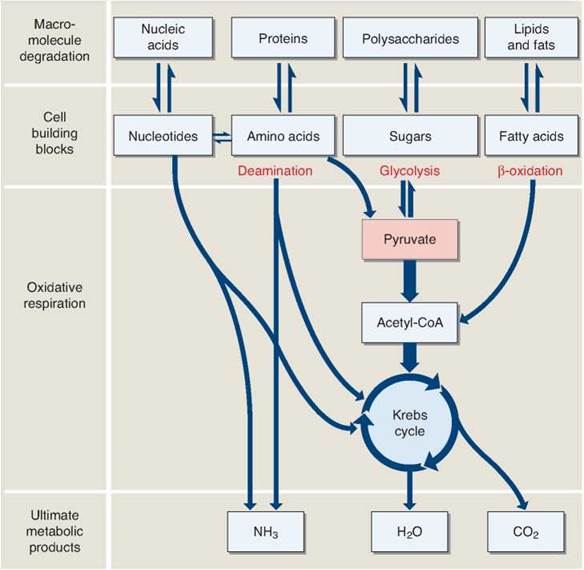
Figure 7.8. How cells obtain energy from foods.
Most organisms extract energy from organic molecules by oxidizing them. The first stage of this process, breaking down macromolecules into their subunits, yields little energy. The second stage, cellular respiration, extracts energy, primarily in the form of high-energy electrons. The subunit of many carbohydrates, glucose, readily enters glycolysis and passes through the biochemical pathways of oxidative respiration. However, the subunits of other macromolecules must be converted into products that can enter the biochemical pathways found in oxidative respiration.
Cellular Respiration of Fat
Lipids and fats (the fourth category in figure 7.8) are first broken down into fatty acids. A fatty acid typically has a long tail of sixteen or more —CH2 links, and the many hydrogen atoms in these long tails provide a rich harvest of energy. Enzymes in the matrix of the mitochondrion first remove one two-carbon acetyl group from the end of a fatty acid tail, and then another, and then another, in effect chewing down the length of the tail in two-carbon bites. Eventually the entire fatty acid tail is converted into acetyl groups. Each acetyl group then combines with coenzyme A to form acetyl-CoA, which feeds into the Krebs cycle. This process is known as β-oxidation.
Thus, in addition to the carbohydrates, the proteins and fats in the hamburger also become important sources of energy.
Key Learning Outcome 7.6. Cells garner energy from proteins and fats, which are broken down into products that feed into cellular respiration.
Biology and Stavina Healthy
Fad Diets and Impossible Dreams
Most Americans put on weight in middle age, slowly adding 30 or more pounds. They did not ask for that weight, do not want it, and are constantly looking for a way to get rid of it. It is not a lonely search—it seems like everyone past the flush of youth is trying to lose weight. Many have been seduced by fad diets, investing hope only to harvest frustration. The much discussed Atkins diet is the fad diet most have tried—Dr. Atkins' Diet Revolution is one of the 10 best-selling books in history, and was (and is) prominently displayed in bookstores. The reason this diet doesn't deliver on its promise of pain-free weight loss is well understood by science, but not by the general public. Only hope and hype make it a perpetual best seller.
The secret of the Atkins diet, stated simply, is to avoid carbohydrates. Atkins's basic proposition is that your body, if it does not detect blood glucose (from metabolizing carbohydrates), will think it is starving and start to burn body fat, even if there is lots of fat already circulating in your bloodstream. You may eat all the fat and protein you want, all the steak and eggs and butter and cheese, and you will still burn fat and lose weight—just don't eat any carbohydrates, any bread or pasta or potatoes or fruit or candy. Despite the title of Atkins's book, this diet is hardly revolutionary. A basic low-carbohydrate diet was first promoted over a century ago in the 1860s by William Banting, an English casket maker, in his best-selling book Letter on Corpulence. Books promoting low-carbohydrate diets have continued to be best sellers ever since.
Those who try the Atkins diet often lose 10 pounds in two to three weeks. In three months it is all back, and then some. So what happened? Where did the pounds go, and why did they come back? The temporary weight loss turns out to have a simple explanation. Carbohydrates act as water sponges in your body, and so forcing your body to become depleted of carbohydrates causes your body to lose water. The 10 pounds lost on this diet was not fat weight but water weight, quickly regained with the first starchy foods eaten.
The Atkins' diet is the sort of diet the American Heart Association tells us to avoid (all those saturated fats and cholesterol), and it is difficult to stay on. If you do hang in there, you will lose weight, simply because you eat less. Other popular diets these days, The Zone diet of Dr. Barry Sears and The South Beach Diet of Dr. Arthur Agatston, are also low-carbohydrate diets, although not as extreme as the Atkins diet. Like the Atkins diet, they work not for the bizarre reasons claimed by their promoters, but simply because they are low-calorie diets.
There are two basic laws that no diet can successfully violate:
1. All calories are equal.
2. (calories in) - (calories out) = fat.
The fundamental fallacy of the Atkins diet, the Zone diet, the South Beach diet, and indeed of all fad diets, is the idea that somehow carbohydrate calories are different from fat and protein calories. This is scientific foolishness. Every calorie you eat contributes equally to your eventual weight, whether it comes from carbohydrate, fat, or protein.
To the extent these diets work at all, they do so because they obey the second law. By reducing calories in, they reduce fat. If that were all there was to it, we should all go out and buy a diet book. Unfortunately, losing weight isn't that simple, as anyone who has seriously tried already knows. The problem is that your body will not cooperate.
If you try to lose weight by exercising and eating less, your body will attempt to compensate by metabolizing more efficiently. It has a fixed weight, what obesity researchers call a "set point,” a weight to which it will keep trying to return. A few years ago, a group of researchers at Rockefeller University in New York, in a landmark study, found that if you lose weight, your metabolism slows down and becomes more efficient, burning fewer calories to do the same work—your body will do everything it can to gain the weight back! Similarly, if you gain weight, your metabolism speeds up. In this way your body uses its own natural weight control system to keep your weight at its set point. No wonder it's so hard to lose weight!
Clearly our bodies don't keep us at one weight all our adult lives. It turns out your body adjusts its fat thermostat—its set point—depending on your age, food intake and amount of physical activity. Adjustments are slow, however, and it seems to be a great deal easier to move the body's set point up than to move it down. Apparently higher levels of fat reduce the body's sensitivity to the leptin hormone that governs how efficiently we burn fat. That is why you can gain weight, despite your set point resisting the gain—your body still issues leptin alarm calls to speed metabolism, but your brain doesn't respond with as much sensitivity as it used to. Thus the fatter you get, the less effective your weight control system becomes.
This doesn't mean that we should give up and learn to love our fat. Rather, now that we are beginning to understand the biology of weight gain, we must accept the hard fact that we cannot beat the requirements of the two diet laws. The real trick is not to give up. Eat less and exercise more, and keep at it. In one year, or two, or three, your body will readjust its set point to reflect the new reality you have imposed by constant struggle. There simply isn't any easy way to lose weight.
Inquiry & Analysis
How Do Swimming Fish Avoid Low Blood pH?
Animals that live in oxygen-poor environments, like worms living in the oxygen-free mud at the bottom of lakes, are not able to obtain the energy required for muscle movement from the Krebs cycle. Their cells lack the oxygen needed to accept the electrons stripped from food molecules. Instead, these animals rely on glycolysis to obtain ATP, donating the electron to pyruvate, forming lactic acid. While much less efficient than the Krebs cycle, glycolysis does not require oxygen. Even when oxygen is plentiful, the muscles of an active animal may use up oxygen more quickly than it can be supplied by the bloodstream and so be forced to temporarily rely on glycolysis to generate the ATP for continued contraction.
This presents a particular problem for fish. Fish blood is much lower in carbon dioxide than yours is, and as a consequence, the amount of sodium bicarbonate acting as a buffer in fish blood is also quite low. Now imagine you are a trout, and need to suddenly swim very fast to catch a mayfly for dinner. The vigorous swimming will cause your muscles to release large amounts of lactic acid into your poorly-buffered blood; this could severely disturb the blood's acid-base balance and so impede contraction of your swimming muscles before the prey is captured.
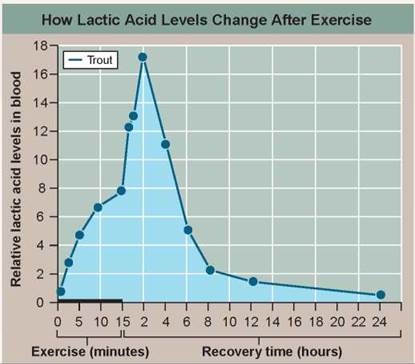
The graph to the right presents the results of an experiment designed to explore how a trout solves this dilemma. In the experiment, the trout was made to swim vigorously for 15 minutes in a laboratory tank, and then allowed a day's recovery. The lactic acid concentration in its blood was monitored periodically during swimming and recovery phases.
1. Applying Concepts
a. Variable. What is the dependent variable?
b. Recording Data. Lactic acid levels are presented for both swimming and recovery periods. In what time units are the swimming data presented? The recovery data?
2. Interpreting Data
a. What is the effect of exercise on the level of lactic acid in the trout's blood?
b. Does the level of lactic acid change after exercise stops? How?
3. Making Inferences About how much of the total lactic acid created by vigorous swimming is released after this exercise stops? [Hint: Notice the x axis scale changes from minutes to hours, so replot all points to minutes and compare areas under curve.]
4. Drawing Conclusions Is this result consistent with the hypothesis that fish maintain blood pH levels by delaying the release of lactic acid from muscles? Why might this be beneficial to the fish?

Test Your Understanding
1. In animals, the energy for life is obtained by cellular respiration. This involves
a. breaking down the organic molecules that were consumed.
b. capturing photons from plants.
c. obtaining ATP from plants.
d. breaking down CO2 that was produced by plants.
2. During glycolysis, ATP forms by
a. the breakdown of pyruvate.
b. chemiosmosis.
c. substrate-level phosphorylation.
d. NAD+.
3. Which of the following processes can occur in the absence of oxygen?
a. the Krebs cycle
b. glycolysis
c. chemiosmosis
d. All of the above.
4. Every living creature on this planet is capable of carrying out the rather inefficient biochemical process of glycolysis, which
a. makes glucose, using the energy from ATP.
b. makes ATP by splitting glucose and capturing the energy.
c. phosphorylates ATP to make ADP.
d. makes glucose, using oxygen and carbon dioxide and water.
5. The electrons generated from the Krebs cycle are transferred to _____ , which then carries them to _____.
a. NAD+, oxygen
b. NAD+, the electron transport chain
c. NADH, oxygen
d. NADH, the electron transport chain
6. After glycolysis, the pyruvate molecules go to the
a. nucleus of the cell and provide energy.
b. membranes of the cell and are broken down in the presence of CO2 to make more ATP.
c. mitochondria of the cell and are broken down in the presence of O2 to make more ATP.
d. Golgi bodies and are packaged and stored until needed.
7. The vast majority of the ATP molecules produced within a cell are produced
a. during pyruvate oxidation.
b. during glycolysis.
c. during the Krebs cycle.
d. during the electron transport chain.
8. NAD+ is recycled during
a. glycolysis.
b. fermentation.
c. the Krebs cycle.
d. the formation of acetyl-CoA.
9. The final electron acceptor in lactic acid fermentation is
a. pyruvate.
b. NAD+.
c. lactic acid.
d. O2.
10. Cells can extract energy from foodstuffs other than glucose because
a. proteins, fatty acids, and nucleic acids get converted to glucose and then enter oxidative respiration.
b. each type of macromolecule has its own oxidative respiration pathway.
c. each type of macromolecule is broken down into its subunits, which enter the oxidative respiration pathway.
d. they can all enter the glycolytic pathway.