MCAT Biology Review
Chapter 8: The Immune System
8.3 The Adaptive Immune System
The adaptive immune system can identify specific invaders and mount an attack against that pathogen. The response is variable and depends on the identity of the pathogen. The adaptive immune system can be divided into two divisions: humoral immunity and cell-mediated (cytotoxic) immunity. Each involves the identification of the specific pathogen and organization of an appropriate immune response.
CELLS OF THE ADAPTIVE IMMUNE SYSTEM
The adaptive immune system consists mainly of two types of lymphocytes, B-cells and T-cells. B-cells govern the humoral response, while T-cells mount the cell-mediated response. All cells of the immune system are created in the bone marrow, but B- and T-cells mature in different locations. B-cells mature in the bone marrow (although the B in their name originally stood for the bursa of Fabricius, an organ found in birds), and T-cells mature in the thymus. When we are exposed to a pathogen, it may take a few days for the physical symptoms to be relieved. This occurs because the adaptive immune response takes time to form specific defenses against the pathogen.
KEY CONCEPT
B-cells mature in the bone marrow. T-cells mature in the thymus.
Humoral Immunity
Humoral immunity, which involves the production of antibodies, may take as long as a week to become fully effective after initial infection. These antibodies are specific to the antigens of the invading microbe. Antibodies are produced by B-cells, which are lymphocytes that originate and mature in the bone marrow and are activated in the spleen and lymph nodes.
Antibodies (also called immunoglobulins [Ig]) can carry out many different jobs in the body. Just as antigens can be displayed on the surface of cells or can float freely in blood, chyle (lymphatic fluid), or air, so too can antibodies be present on the surface of a cell or secreted into body fluids. When an antibody binds to an antigen, the response will depend on the location. For antibodies secreted into body fluids, there are three main possibilities: first, once bound to a specific antigen, antibodies may attract other leukocytes to phagocytize those antigens immediately. This is called opsonization, as described earlier. Second, antibodies may cause pathogens to clump together or agglutinate, forming large insoluble complexes that can be phagocytized. Third, antibodies can block the ability of a pathogen to invade tissues, essentially neutralizing it. For cell-surface antibodies, the binding of antigen to a B-cell causes activation of that cell, resulting in its proliferation and formation of plasma and memory cells, as described later in this chapter. In contrast, when antigen binds to antibodies on the surface of a mast cell, it causes degranulation (exocytosis of granule contents), allowing the release of histamine and causing an inflammatory allergic reaction.
Antibodies are Y-shaped molecules that are made up of two identical heavy chains and two identical light chains, as shown in Figure 8.7. Disulfide linkages and noncovalent interactions hold the heavy and light chains together. Each antibody has an antigen-binding region at the end of what is called the variable region (domain), at the tips of the Y. Within this region, there are specific polypeptide sequences that will bind one, and only one, specific antigenic sequence. Part of the reason it takes so long to initiate the antibody response is that each B-cell undergoeshypermutation of its antigen-binding region, trying to find the best match for the antigen. Only those B-cells that can bind the antigen with high affinity survive, providing a mechanism for generating specificity called clonal selection. The remaining part of the antibody molecule is known as the constant region (domain). It is this region that cells such as natural killer cells, macrophages, monocytes, and eosinophils have receptors for, and that can initiate the complement cascade. Each B-cell makes only one type of antibody, but we have many B-cells, so our immune system can recognize many antigens. Further, antibodies come in five different isotypes (IgM, IgD, IgG, IgE, and IgA). While the specific purposes of each antibody isotype is outside the scope of the MCAT, you should know that the different types can be used at different times during the adaptive immune response, for different types of pathogens, or in different locations in the body. Cells can change which isotype of antibody they produce when stimulated by specific cytokines in a process called isotype switching.
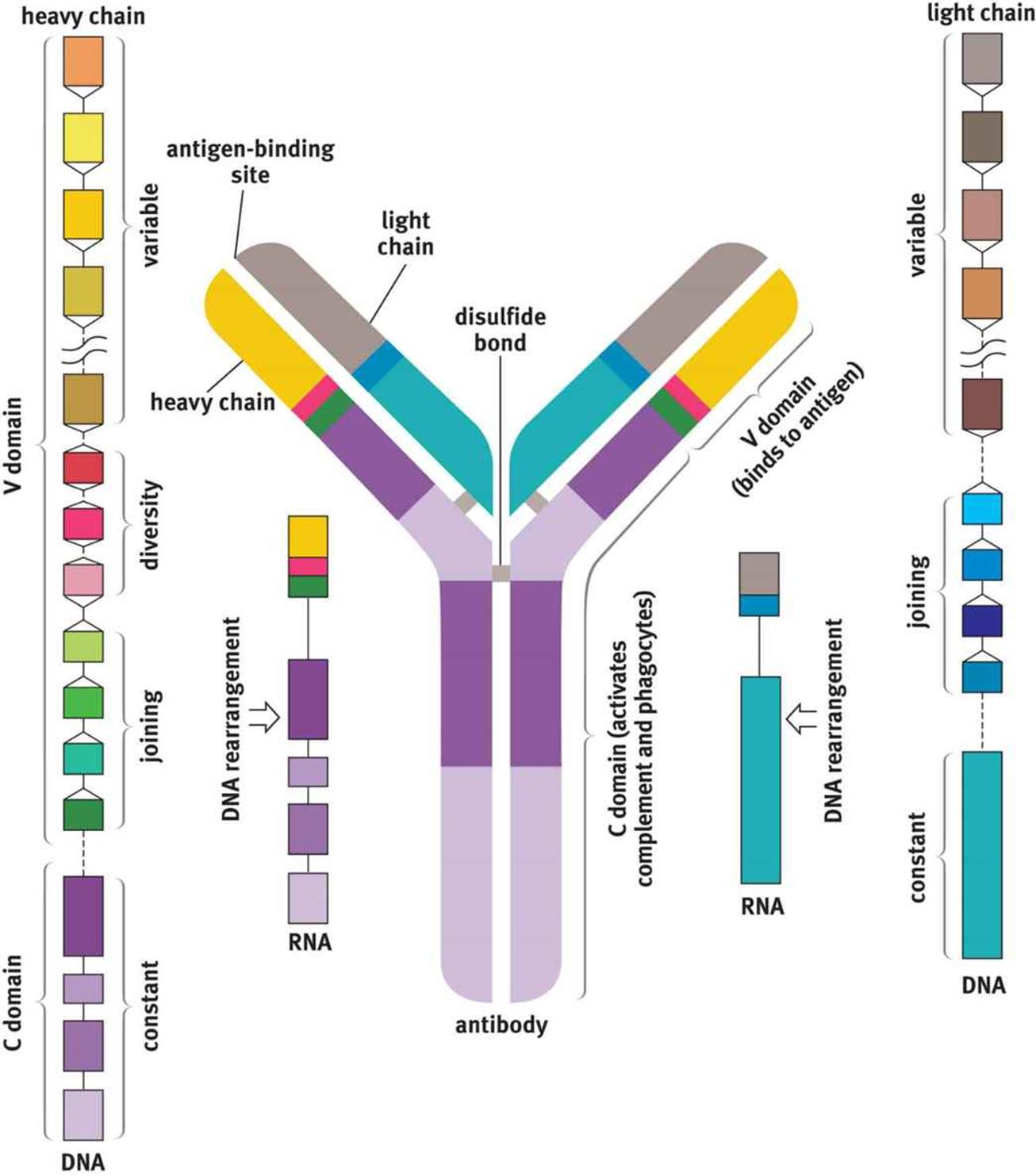 Figure 8.7. Structure of an Antibody Molecule
Figure 8.7. Structure of an Antibody Molecule
Not all B-cells that are generated actively or constantly produce antibodies. Antibody production is an energetically expensive process, and there is no reason to expend energy producing antibodies that are not needed. Instead, naïve B-cells (those that have not yet been exposed to an antigen) wait in the lymph nodes for their particular antigen to come along. Upon exposure to the correct antigen, a B-cell will proliferate and produce two types of daughter cells. Plasma cells produce large amounts of antibodies, whereas memory B-cells stay in the lymph node, awaiting reexposure to the same antigen. This initial activation takes approximately seven to ten days and is known as the primary response. The plasma cells will eventually die, but the memory cells may last the lifetime of the organism. If the same microbe is ever encountered again, the memory cells jump into action and produce the antibodies specific to that pathogen. This immune response, called the secondary response, will be more rapid and robust. The development of these lasting memory cells is the basis of the efficacy of vaccinations.
Cytotoxic Immunity
Whereas humoral immunity is based on the activity of B-cells, cell-mediated immunity involves the T-cells. T-cells mature in the thymus, where they undergo both positive and negative selection. Positive selection refers to maturing only cells that can respond to the presentation of antigen on MHC (cells that cannot respond to MHC undergo apoptosis because they will not be able to respond in the periphery). Negative selection refers to causing apoptosis in cells that are self-reactive (activated by proteins produced by the organism itself). The maturation of T-cells is facilitated by thymosin, a peptide hormone secreted by thymic cells. Once the T-cell has left the thymus, it is mature but naïve. Upon exposure to antigen, T-cells will also undergo clonal selection so that only those with the highest affinity for a given antigen proliferate.
There are three major types of T-cells: helper T-cells, suppressor T-cells, and killer (cytotoxic) T-cells. Helper T-cells (Th), also called CD4+ T-cells, coordinate the immune response by secreting chemicals known as lymphokines. These molecules are capable of recruiting other immune cells (such as plasma cells, cytotoxic T-cells, and macrophages) and increasing their activity. The loss of these cells, as occurs in human immunodeficiency virus (HIV) infection, prevents the immune system from mounting an adequate response to infection; in advanced HIV infection, also called acquired immunodeficiency syndrome (AIDS), even weak pathogens can cause devastating consequences as opportunistic infections. CD4+ T-cells respond to antigens presented on MHC-II molecules. Because MHC-II presents exogenous antigens, CD4+ T-cells are most effective against bacterial, fungal, and parasitic infections.
REAL WORLD
“CD” in immunology stands for cluster of differentiation and includes cell-surface markers that can be detected by the lab technique called flow cytometry; these markers give an indication of the types of leukocytes under investigation, how many are present, and in what state of maturity they are.
Cytotoxic T-cells (Tc or CTL, for cytotoxic T-lymphocytes), also called CD8+ T-cells, are capable of directly killing virally infected cells by injecting toxic chemicals that promote apoptosis into the infected cell. CD8+ T-cells respond to antigens presented on MHC-I molecules. Because MHC-I presents endogenous antigens, CD8+ T-cells are most effective against viral (and intracellular bacterial or fungal) infections.
KEY CONCEPT
CD4+ T-cells are better at fighting extracellular infections, while CD8+ T-cells are better at targeting intracellular infections.
Suppressor or regulatory T-cells (Treg) also express CD4, but can be differentiated from helper T-cells because they also express a protein called Foxp3. These cells help to tone down the immune response once infection has been adequately contained. These cells also turn off self-reactive lymphocytes to prevent autoimmune diseases: this is termed self-tolerance.
MNEMONIC
· CD × MHC = 8
· CD4+ cells respond to MHC-II (4 × 2 = 8)
· CD8+ cells respond to MHC-I (8 × 1 = 8)
Finally, memory T-cells can be generated. Similar to memory B-cells, these cells lie in wait until the next exposure to the same antigen. When activated, they result in a more robust and rapid response.
REAL WORLD
Many suppressor T-cells were formerly self-reactive T-cells that have been turned off. When a suppressor T-cell inactivates another lymphocyte, it can either target it for destruction or promote its conversion into another suppressor T-cell.
A summary of the different types of lymphocytes in adaptive (specific) immunity is shown in Figure 8.8.
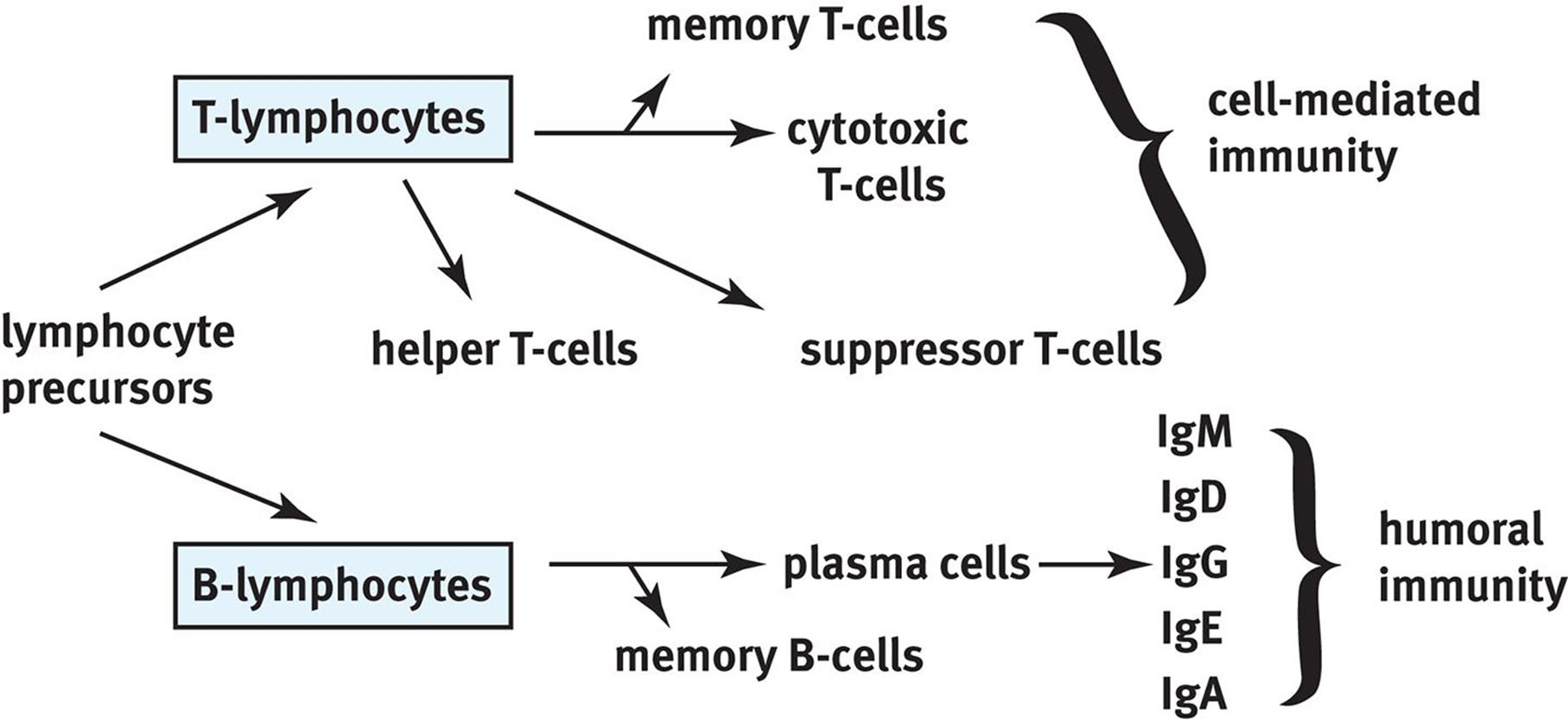 Figure 8.8. Lymphocytes of Specific Immunity This diagram shows the differentiation of lymphocyte precursors and the cell types involved in specific immunity.
Figure 8.8. Lymphocytes of Specific Immunity This diagram shows the differentiation of lymphocyte precursors and the cell types involved in specific immunity.
ACTIVATION OF THE ADAPTIVE IMMUNE SYSTEM
When the human body encounters an antigen, the immune system must be able to respond. It is important to note that the innate and adaptive immune systems are not really disparate entities that function separately. The proper functioning of the entire immune system depends on the interactions between these two systems. There are five types of infectious pathogens: bacteria, viruses, fungi, parasites (including protozoa, worms, and insects), and prions (for which there are no immune defenses). While the immune system’s response depends on the specific identity of the pathogen, we present two classic examples: a bacterial (extracellular pathogen) infection and a viral (intracellular pathogen) infection. Keep in mind that this categorization is imperfect; for example, some bacteria, like Mycobacterium tuberculosis and Listeria monocytogenes, actually live intracellularly.
Bacterial (Extracellular Pathogen) Infections
Macrophages are like the sentinels of the human body, always on the lookout for potential invaders. Let’s say a person suffers a laceration and bacteria are introduced into the body via this laceration. First, macrophages (and other antigen-presenting cells) engulf the bacteria and subsequently release inflammatory mediators. These cells also digest the bacteria and present antigens from the pathogen on their surfaces in conjunction with MHC-II. The cytokines attract inflammatory cells, including neutrophils and additional macrophages. Mast cells are activated by the inflammation and degranulate, resulting in histamine release and increased leakiness of the capillaries. This allows for immune cells to leave the bloodstream to travel to the affected tissue. The dendritic cell then leaves the affected tissue and travels to the nearest lymph node, where it presents the antigen to B-cells. B-cells that produce the correct antibody proliferate through clonal selection to create plasma cells and memory cells. Antibodies then travel through the bloodstream to the affected tissue, where they tag the bacteria for destruction.
At the same time, dendritic cells are also presenting the antigen to T-cells, activating a T-cell response. In particular, CD4+ T-cells are activated. These cells come in two types, called Th1 and Th2. Th1 cells release interferon gamma (IFN-γ), which activates macrophages and increases their ability to kill bacteria. Th2 cells help activate B-cells.
After the pathogen has been eliminated, plasma cells die, but memory B- and T-cells remain. These memory cells allow for a much faster secondary response upon exposure to the pathogen at a later time.
Viral (Intracellular Pathogen) Infections
In a viral infection, the virally infected cell will begin to produce interferons, which reduce the permeability of nearby cells (decreasing the ability of the virus to infect these cells), reduce the rate of transcription and translation in these cells (decreasing the ability of the virus to multiply), and cause systemic symptoms (malaise, muscle aching, fever, and so on). These infected cells also present intracellular proteins on their surface in conjunction with MHC-I; in a virally infected cell, at least some of these intracellular proteins will be viral proteins.
CD8+ T-cells will recognize the MHC-I and antigen complex as foreign and will inject toxins into the cell to promote apoptosis. In this way, the infection can be shut down before it is able to spread to nearby cells. In the event that the virus downregulates the production and presentation of MHC-I molecules, natural killer cells will recognize the absence of MHC-I and will accordingly cause apoptosis of this cell.
Again, once the pathogen has been cleared, memory T-cells will be generated that can allow a much faster response to be mounted upon a second exposure.
RECOGNITION OF SELF AND NONSELF
Self-antigens are the proteins and carbohydrates present on the surface of every cell of the body. Under normal circumstances, these self-antigens signal to immune cells that the cell is not threatening and should not be attacked. However, when the immune system fails to make the distinction between self and foreign, it may attack cells expressing particular self-antigens, a condition known as autoimmunity. Note that autoimmunity is only one potential problem with immune functioning: another problem arises when the immune system misidentifies a foreign antigen as dangerous when, in fact, it is not. Pet dander, pollen, and peanuts are not inherently threatening to human life, yet some people’s immune systems are hypersensitive to these antigens and become overactivated when these antigens are encountered in what is called an allergic reaction. Allergies and autoimmunity are part of a family of immune reactions classified as hypersensitivity reactions.
The human body strives to prevent autoimmune reactions very early in T-cell and B-cell maturation processes. T-cells are educated in the thymus. Part of this education involves the elimination of T-cells that respond to self-antigens, called negative selection. Immature B-cells that respond to self-antigens are eliminated before they leave the bone marrow. However, this process is not perfect, and occasionally a cell that responds to self-antigens is allowed to survive. Most autoimmune diseases can be treated with a number of therapies; one common example is administration ofglucocorticoids (modified versions of cortisol), which have potent immunosuppressive qualities.
REAL WORLD
Autoimmune diseases can result in destruction of tissues, causing various deficiencies. Type I diabetes mellitus results from autoimmune destruction of the β-cells of the pancreas. This results in an inability to produce insulin, characterized by high blood sugars and excessive utilization of fats and proteins for energy. Other examples of autoimmune diseases include multiple sclerosis, myasthenia gravis, psoriasis, systemic lupus erythematosus, rheumatoid arthritis, Graves’ disease, and Guillain–Barré syndrome.
IMMUNIZATION
Often, diseases can have significant, long-term consequences. Infection with the poliovirus, for example, can leave a person disabled for the remainder of his or her life. Polio used to be a widespread illness; however, today we hardly hear about it outside of the Indian subcontinent because of a highly effective vaccination program, which led to the eradication of polio from the Western hemisphere.
Immunization can be achieved in an active or passive fashion. In active immunity, the immune system is stimulated to produce antibodies against a specific pathogen. The means by which we are exposed to this pathogen may either be natural or artificial. Through natural exposure, antibodies are generated by B-cells once an individual becomes infected. Artificial exposure (through vaccines) also results in the production of antibodies; however, the individual never experiences true infection. Instead, he or she receives an injection or intranasal spray containing an antigen that will activate B-cells to produce antibodies to fight the specific infection. The antigen may be a weakened or killed form of the microbe, or it may be a part of the microbe’s protein structure.
Immunization may also be achieved passively. Passive immunity results from the transfer of antibodies to an individual. The immunity is transient because only the antibodies, and not the plasma cells that produce them, are given to the individual. Natural examples are the transfer of antibodies across the placenta during pregnancy to protect the fetus and the transfer of antibodies from a mother to her nursing infant through breast milk. In some cases of exposure, such as to the rabies virus or tetanus, intravenous immunoglobulin may be given to prevent the pathogen from spreading.
REAL WORLD
In 1998, a paper published in The Lancet claimed to have found a link between vaccines and autism. This paper has since been withdrawn from The Lancet after it was demonstrated to be fraudulent and scientifically inaccurate. In fact, no well-designed scientific study has yet shown this link to exist. However, the sensationalist reporting of this connection in the lay population has led many parents to avoid immunizing their children. Since 1998, outbreaks of measles and mumps in the United States and other industrialized nations have raised concerns about the resurgence of illnesses that were previously almost eradicated. Vaccines do carry risks, including rare cases of encephalitis (brain inflammation) and Guillain–Barré syndrome (an autoimmune disease in which the myelin of peripheral nerves is attacked), but so too do the pathogens these vaccines protect against.
MCAT Concept Check 8.3:
Before you move on, assess your understanding of the material with these questions.
1. For each of the lymphocytes listed below, what are its main functions?
· Plasma cell:
· Memory B-cell:
· Helper T-cell:
· Cytotoxic T-cell:
· Suppressor (regulatory) T-cell:
· Memory T-cell:
2. What are the three main effects circulating antibodies can have on a pathogen?
·
·
·
3. How do antibodies become specific for a given antigen?
4. What is meant by positive and negative selection?
· Positive selection:
· Negative selection:
5. Which cells account for the fact that the secondary response to a pathogen is much more rapid and robust than the primary response?
6. What is the difference between active and passive immunity?
· Active immunity:
· Passive immunity: