MCAT Biology Review
Chapter 12: Genetics and Evolution
Introduction
European royal families, for generations, practiced what is known as royal intermarriage. For purposes of establishing or continuing political alliances, maintaining bloodline purity, or smoothing out diplomatic relations, marriages between royal families were arranged, resulting in such an interweaving of bloodlines that eventually most European royalty was—and still is—genetically related.
Such marriage unions led to rather severe restrictions on the gene pool—all the alleles represented in the royal family lines. Offspring of parents who were also related to each other through blood lineage (consanguinity) came to have greater similarities in their genotypes, and certain alleles became so frequent that their phenotypic expression became almost a hallmark of royal descent. The House of Habs-burg, which ruled a number of European kingdoms from the 11th century until the late 18th century was perhaps the most infamous for its inbreeding practices, and members of this royal family bore the unmistakable mark of their restricted genes through a jaw malformation that even came to be known as the Habsburg lip. Medically termed prognathism (Greek: “forward jaw”), the condition is a misalignment of the mandible and maxilla. The Habsburg family portraits present individuals with prominent, forward-thrusting lower jaws and chins, characteristic of mandibular prognathism. The genetic condition has more than just aesthetic implications; it can lead to serious disfigurement and disability. Charles II of Spain suffered from the worst case of the Habsburg lip on record—his lower teeth protruded so much further than his upper teeth that he was not able to chew his food.
In this chapter, we will explore the concepts of classical genetics, which were originally described in the mid-1800s. Consider this chapter in tandem with the discussions of molecular genetics in Chapters 6 and 7 of MCAT Biochemistry Review. Then, we will explore the changes in the gene pool that occur over time with a discussion of evolution. We’ll also quantify populations that are not undergoing evolution with the Hardy–Weinberg principle.
12.1 Fundamental Concepts of Genetics
The physical and biochemical characteristics of every living organism are determined by genes, which are DNA sequences that code for heritable traits that can be passed from one generation to the next. Taken together, all genes (as well as a large supply of noncoding DNA) are organized into chromosomes to ensure that genetic material is passed easily to daughter cells during mitosis and meiosis. Each gene may have alternative forms called alleles. We’ve already explored the ABO blood antigens as an example of three alleles for the same gene (IA, IB, and i). The genetic combination possessed by an individual is known as a genotype, and the manifestation of a given genotype as an observable trait is known as a phenotype.
BRIDGE
Blood type A individuals carry the A antigen on their erythrocytes and have circulating anti-B antibodies. Blood type B individuals carry the B antigen on their erythrocytes and have circulating anti-A antibodies. Those with type AB have both antigens and neither antibody; those with type O have neither antigen and both antibodies. That makes type O individuals universal donors and type AB individuals universal recipients. Blood typing is discussed in Chapter 7 of MCAT Biology Review.
Each human being possesses two copies of each chromosome, called homologues, except for male sex chromosomes (in which there is one X chromosome and one Y chromosome). Each gene has a particular locus, or location on a specific chromosome. The normal locus of a particular gene is consistent among human beings, such that a gene can be described by its location. Because each chromosome is part of a homologous pair, a person will inherit two alleles for all genes (again, except for male sex chromosomes). Alleles can be categorized based on their expression. If only one copy of an allele is needed to express a given phenotype, the allele is said to be dominant and is usually represented with a capital letter. If two copies are needed, the allele is said to be recessive and is usually represented with a lowercase letter. If both alleles are the same for a given gene, the individual is said to have a homozygous genotype. If the alleles are different, the individual has a heterozygous genotype. A hemizygous genotype describes a situation in which only one allele is present for a given gene, as is the case for parts of the X chromosome in males.
PATTERNS OF DOMINANCE
When only one dominant and one recessive allele exist for a given gene, there is said to be complete dominance. In this case, the presence of one dominant allele will mask the recessive allele, if present. When more than one dominant allele exists for a given gene, there is codominance. For example, a person with one allele for the A blood antigen and one allele for the B blood antigen will express both antigens simultaneously. Finally, incomplete dominance occurs when a heterozygote expresses a phenotype that is intermediate between the two homozygous genotypes. A classic example of incomplete dominance is the mating of certain flowers, in which a red flower crossed with a white flower results in pink flowers, as shown in Figure 12.1.
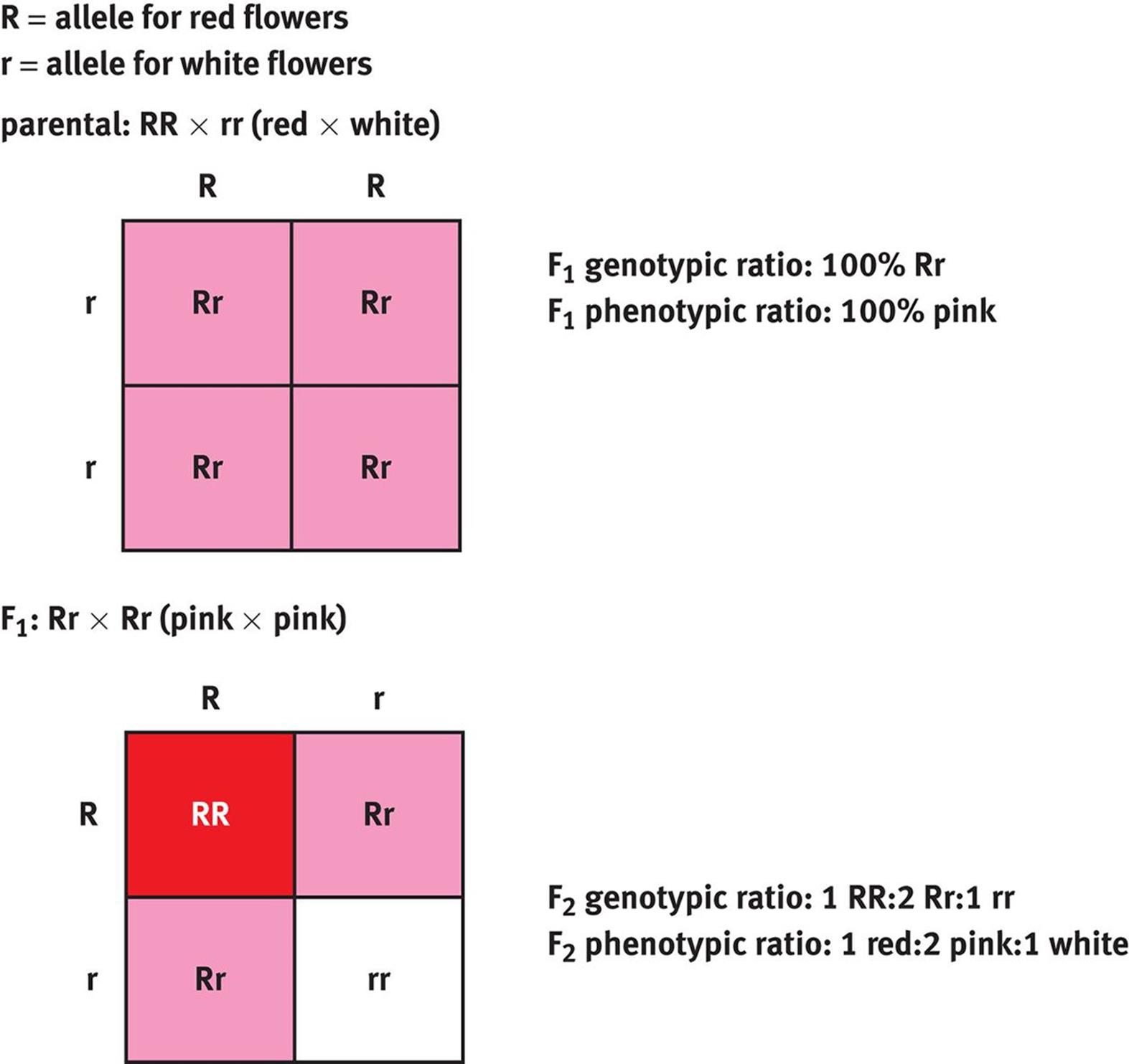 Figure 12.1. Incomplete Dominance Snapdragons display incomplete dominance, in which neither allele is dominant and the heterozygous phenotype is a mixture of the two homozygous phenotypes.
Figure 12.1. Incomplete Dominance Snapdragons display incomplete dominance, in which neither allele is dominant and the heterozygous phenotype is a mixture of the two homozygous phenotypes.
PENETRANCE AND EXPRESSIVITY
Penetrance and expressivity both reveal the complex interplay between genes and the environment. Penetrance is a population measure defined as the proportion of individuals in the population carrying the allele who actually express the phenotype. In other words, it is the probability that, given a particular genotype, a person will express the phenotype. Alleles can be classified by their degree of penetrance; Huntington’s disease, caused by an expansion of a repetitive sequence in the huntingtin gene, is a classic example. Individuals with over 40 sequence repeats have full penetrance—100 percent of individuals with this allele show symptoms of Huntington’s disease. Individuals with fewer sequence repeats show high penetrance, wherein most—but not all—of those with the allele show symptoms of the disease. With fewer sequence repeats, the gene comes to have reduced penetrance, low penetrance, or even nonpenetrance.
A related, but distinct, concept is expressivity, which is defined as varying phenotypes despite identical genotypes. If expressivity is constant, then all individuals with a given genotype express the same phenotype. However, if expressivity is variable, then individuals with the same genotype may have different phenotypes. Whereas penetrance is a population parameter (what percent of individuals with a given genotype express the phenotype?), expressivity explores the grey area in expression and is more commonly considered at the individual level. For example, the disease neurofibromatosis type II is an autosomal dominant disease that results from a mutation of the gene NF2 (merlin). Interestingly, a range of phenotypes is associated with carrying the affected allele. Many patients have debilitating tumors of the vestibulocochlear nerve, which is needed for hearing and balance. Some have cataracts. Others have tumors in the skin called neuromas. Still others have spinal lesions. A small proportion of the population is nonpenetrant. The disease shows variable expressivity because there is a range of presentations between no clinical effect and severe disability.
KEY CONCEPT
· Penetrance—the proportion of the population with a given genotype who actually express the phenotype
· Expressivity—the different manifestations of the same genotype across the population
MENDELIAN CONCEPTS
Gregor Mendel, an Augustinian friar, developed several of the tenets of genetics in the 1860s based on his work with pea plants. While the study of genetics has come a long way, from pedigree analysis to DNA probes to whole-genome sequencing, many of Mendel’s original ideas still hold.
Mendel’s First Law: Law of Segregation
There are four basic tenets of the modern interpretation of Mendel’s first law (of segregation), some of which have already been discussed:
· Genes exist in alternative forms (alleles).
· An organism has two alleles for each gene, one inherited from each parent.
· The two alleles segregate during meiosis, resulting in gametes that carry only one allele for any inherited trait.
· If two alleles of an organism are different, only one will be fully expressed and the other will be silent. The expressed allele is said to be dominant, the silent allele recessive. (Keep in mind that codominance and incomplete dominance are exceptions to this rule.)
The key cellular correlate to draw here is the separation of homologous chromosomes during anaphase I of meiosis. By separating—segregating—these chromosomes into different cells, each gamete carries only one allele for any given trait.
Mendel’s Second Law: Law of Independent Assortment
Mendel’s second law (of independent assortment) states that the inheritance of one gene does not affect the inheritance of another gene. Remember from Chapter 2 of MCAT Biology Review that, before meiosis I, spermatogonia and oogonia undergo genome replication. The daughter DNA strand is held to the parent strand at the centromere. Together, these DNA strands are known as sister chromatids. During prophase I of meiosis, homologous chromosomes pair up to form tetrads, which derive their name from the four chromatids involved (two chromatids in each of two homologous chromosomes). Small segments of genetic material are swapped between chromatids in homologous chromosomes, resulting in novel combinations of alleles that were not present in the original chromosomes (recombination). This allows the inheritance of one gene to be independent of the inheritance of all others.
Mendel’s second law has been problematized by the discovery of linked genes. We will discuss nonindependent assortment and linkage later in this chapter in the section on analytical approaches in genetics.
Both segregation of homologous chromosomes and independent assortment of alleles increase the genetic diversity of gametes and, subsequently, the genetic diversity of offspring. This has been demonstrated to improve the ability of a species to evolve to adapt to environmental stresses.
KEY CONCEPT
Segregation and independent assortment allow for greater genetic diversity in the offspring.
DNA AS GENETIC MATERIAL
While Mendel noticed that there were certain patterns of inheritance, what he did not know was that DNA was the genetic material transferred to offspring and that genes were made of DNA. In fact, the scientific community rejected Mendel’s initial papers on inheritance. It wasn’t until the early 1900s that his work was rediscovered. In the early to mid-1900s, it was largely believed that protein was the heritable material. In the mid-1900s, there were three experiments conducted that largely pointed to DNA’s role in genetic inheritance.
In the 1920s, Frederick Griffith was a scientist working with the British government to study Streptococcus pneumoniae, a bacteria that causes pneumonia. Two strains of S. pneumoniae were identified: a virulent (disease-causing) strain and a non-virulent strain. In successive trials, Griffith exposed mice to these strains of bacteria under different conditions and observed whether the mouse lived or died, as shown in Figure 12.2. The virulent S. pneumonia has a smooth capsule that helps the bacterium eschew the immune system and cause disease. He injected this strain into mice, which resulted in death of the mice. However, if the virulent bacteria were killed prior to injection, no disease resulted. Likewise, exposure of the mice to the nonvirulent strain, which has a rough capsule, did not cause disease. However, when both dead virulent bacteria and live nonvirulent bacteria were injected into the mouse, the mouse died, and live bacteria with smooth capsules could be found in the mice. He theorized that the live, nonvirulent bacteria must have acquired the ability to form smooth capsules from the dead virulent bacteria. This was known as the transforming principle.
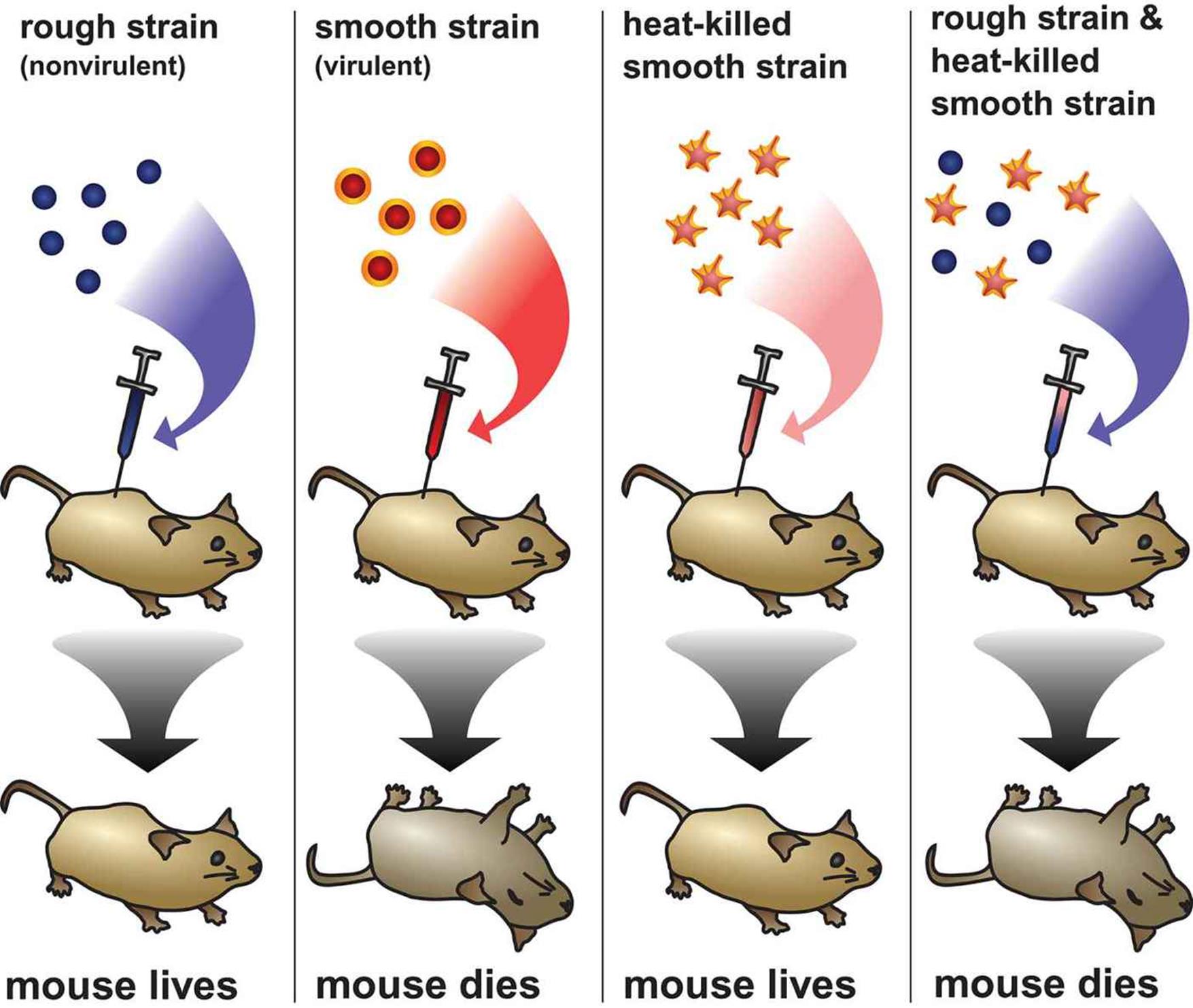 Figure 12.2. The Griffith Experiment
Figure 12.2. The Griffith Experiment
BRIDGE
The transforming principle described in Griffith’s experiment is the same as bacterial transformation discussed in Chapter 1 of MCAT Biology Review. Remember that transformation is one of three main ways bacteria increase genetic variability; the other two are conjugation and transduction.
Researchers at the Rockefeller Institute confirmed the transformation principle. Three American scientists—Oswald Avery, Colin MacLeod, and Maclyn McCarty—were attempting to determine the exact material underlying the transformation principle. These scientists purified a very large quantity of heat-killed virulent S. pneumoniae bacteria and separated the subcellular components of the bacteria into different extracts. It was noted that the addition of one particular extract to non-virulent S. pneumoniae transformed the bacteria and enabled them to kill the mouse when injected. When this substance was treated with enzymes known to degrade DNA, the bacteria were not transformed and the mice lived. However, when the substance was treated with enzymes known to degrade proteins, the bacteria were still transformed and the mice died. Thus, the group concluded that the transforming substance must be DNA.
In 1952 (one year before the description of the Watson–Crick model), Alfred Hershey and Martha Chase worked to confirm the idea that DNA could independently carry genetic information. These scientists created bacteriophages with radiolabeled DNA and protein. One group of bacteriophages contained radiolabeled sulfur, which is found in protein but not in DNA. Another group contained radiolabeled phosphorus, which is found in DNA but not in protein. Each of these bacteriophages was permitted to infect a group of nonlabeled bacteria. Recall from Chapter 1 of MCAT Biology Review that when bacteriophages infect a bacterium, they inject their genetic material into the cell and leave their capsid outside. After the phages and bacteria were incubated, the sample was centrifuged to separate the material that remained outside the cell from the bacterial cells themselves. It was determined that no radiolabeled protein entered the cells, but that radiolabeled DNA had. It was known that viruses must enter a cell to cause disease and replicate, so this experiment once again confirmed that DNA was the heritable genetic material.
BRIDGE
Radiolabeling sulfur was an appropriate choice to tag proteins in the Hershey–Chase experiment. Recall from Chapter 1 of MCAT Biochemistry Review that two amino acids—cysteine and methionine—contain sulfur in their R group, while no nucleotides contain any sulfur.
MCAT Concept Check 12.1:
Before you move on, assess your understanding of the material with these questions.
1. What does it mean for an allele to be dominant? Recessive?
· Dominant:
· Recessive:
2. What does it mean for a genotype to be homozygous? Heterozygous? Hemizygous?
· Homozygous:
· Heterozygous:
· Hemizygous:
3. What is the difference between complete dominance, codominance, and incomplete dominance?
· Complete dominance:
· Codominance:
· Incomplete dominance:
4. What is the difference between penetrance and expressivity?
· Penetrance:
· Expressivity:
5. With which phase of meiosis does each of Mendel’s laws most closely correlate?
· Mendel’s first law:
· Mendel’s second law: