Cracking the AP Chemistry Exam
Part IV
Content Review for the AP Chemistry Exam
Chapter 6
Big Idea #4: Chemical Reactions and their Rates
Rates of chemical reactions are determined by details of the molecular collisions.
RATE LAW USING INITIAL CONCENTRATIONS
The rate law for a reaction describes the dependence of the initial rate of a reaction on the concentrations of its reactants. It includes the Arrhenius constant, k, which takes into account the activation energy for the reaction and the temperature at which the reaction occurs. The rate of a reaction is described in terms of the rate of appearance of a product or the rate of disappearance of a reactant. The rate law for a reaction cannot be determined from a balanced equation; it must be determined from experimental data, which is presented on the test in table form.
Here’s How It’s Done
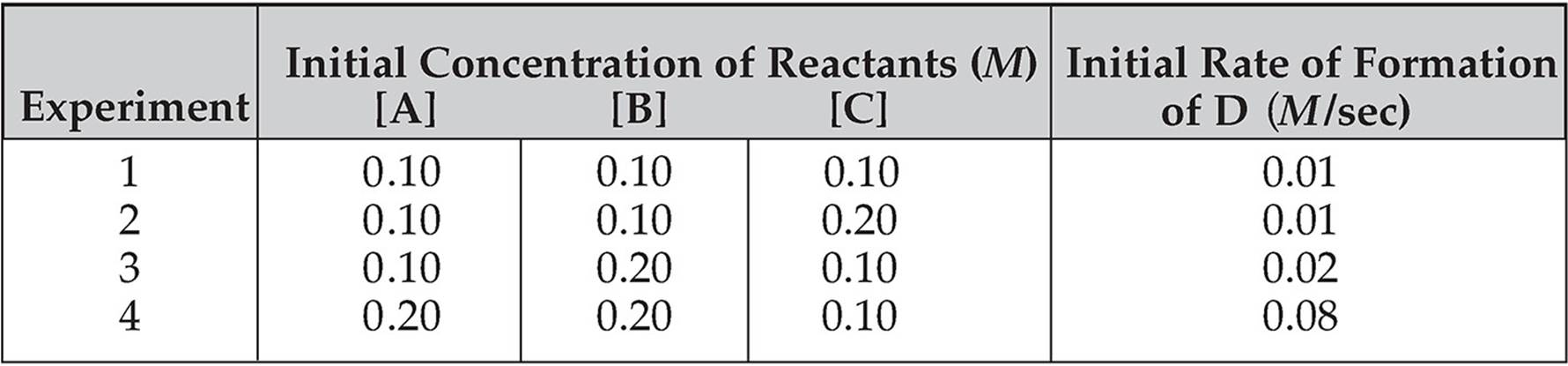
The data below were collected for the following hypothetical reaction:
A + 2B + C → D
The rate law always takes the following form, using the concentrations of the reactants:
Rate = k[A]x[B]y[C]z
The greater the value of a reactant’s exponent, the more a change in the concentration of that reactant will affect the rate of the reaction. To find the values for the exponents x, y, and z, we need to examine how changes in the individual reactants affect the rate. The easiest way to find the exponents is to see what happens to the rate when the concentration of an individual reactant is doubled.
Let’s Look at [A]
From experiment 3 to experiment 4, [A] doubles while the other reactant concentrations remain constant. For this reason, it is useful to use the rate values from these two experiments to calculate x (the order of the reaction with respect to reactant A).
As you can see from the table, the rate quadruples from experiment 3 to experiment 4, going from 0.02 M/sec to 0.08 M/sec.
We need to find a value for the exponent x that relates the doubling of the concentration to the quadrupling of the rate. The value of x can be calculated as follows:
(2)x = 4, so x = 2
Because the value of x is 2, the reaction is said to be second order with respect to A.
Rate = k[A]2[B]y[C]z
Let’s Look at [B]
From experiment 1 to experiment 3, [B] doubles while the other reactant concentrations remain constant. For this reason it is useful to use the rate values from these two experiments to calculate y (the order of the reaction with respect to reactant B).
As you can see from the table, the rate doubles from experiment 1 to experiment 3, going from 0.01 M/sec to 0.02 M/sec.
We need to find a value for the exponent y that relates the doubling of the concentration to the doubling of the rate. The value of y can be calculated as follows:
(2)y = 2, so y = 1
Because the value of y is 1, the reaction is said to be first order with respect to B.
Rate = k[A]2[B][C]z
Let’s Look at [C]
From experiment 1 to experiment 2, [C] doubles while the other reactant concentrations remain constant.
The rate remains the same at 0.01 M.
The rate change is (2)z = 1, so z = 0.
Because the value of z is 0, the reaction is said to be zero order with respect to C.
Rate = k[A]2[B]
Because the sum of the exponents is 3, the reaction is said to be third order overall.
Once the rate law has been determined, the value of the rate constant can be calculated using any of the lines of data on the table. The units of the rate constant are dependent on the order of the reaction, so it’s important to carry along units throughout all rate constant calculations.
Let’s use experiment 3.
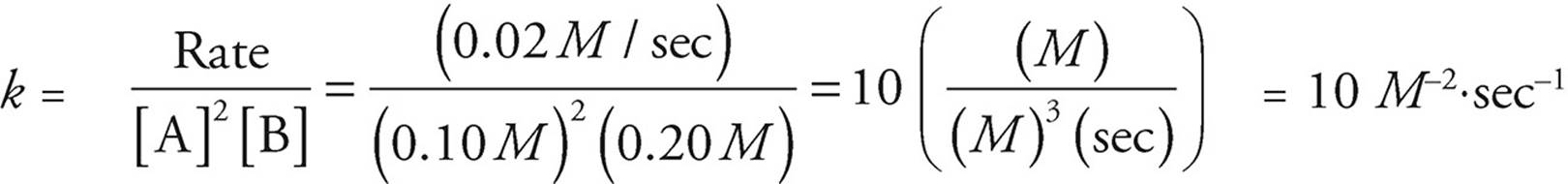
You should note that we can tell from the coefficients in the original balanced equation that the rate of appearance of D is equal to the rate of disappearance of A and C because the coefficients of all three are the same. The coefficient of D is half as large as the coefficient of B, however, so the rate at which D appears is half the rate at which B disappears.