CHEMISTRY THE CENTRAL SCIENCE
12 SOLIDS AND MODERN MATERIALS
12.6 MOLECULAR SOLIDS
Molecular solids consist of atoms or molecules held together by dipole-dipole forces, dispersion forces, and/or hydrogen bonds. Because these intermolecular forces are weak, molecular solids are soft and have relatively low melting points (usually below 200 °C). Most substances that are gases or liquids at room temperature form molecular solids at low temperature. Examples include Ar, H2O, and CO2.
The properties of molecular solids depend in large part on the strengths of the forces between molecules. Consider, for example, the properties of sucrose (table sugar, C12H22O11). Each sucrose molecule has eight –OH groups, which allows for the formation of multiple hydrogen bonds. Consequently, sucrose exists as a crystalline solid at room temperature, and its melting point, 184 °C, is relatively high for a molecular solid. Molecular shape is also important because it dictates how efficiently molecules pack together in three dimensions. Benzene (C6H6), for example, is a highly symmetrical planar molecule. ![]() (Section 8.6) It has a higher melting point than toluene, a compound in which one of the hydrogen atoms of benzene has been replaced by a CH3 group (
(Section 8.6) It has a higher melting point than toluene, a compound in which one of the hydrogen atoms of benzene has been replaced by a CH3 group (![]() FIGURE 12.29). The lower symmetry of toluene molecules prevents them from packing as efficiently as benzene molecules. As a result, the intermolecular forces that depend on close contact are not as effective and the melting point is lower. In contrast, the boiling point of toluene is higher than that of benzene, indicating that the intermolecular attractive forces are larger in liquid toluene than in liquid benzene. The melting and boiling points of phenol, another substituted benzene shown in Figure 12.29, are higher than those of benzene because the OH group of phenol can form hydrogen bonds.
FIGURE 12.29). The lower symmetry of toluene molecules prevents them from packing as efficiently as benzene molecules. As a result, the intermolecular forces that depend on close contact are not as effective and the melting point is lower. In contrast, the boiling point of toluene is higher than that of benzene, indicating that the intermolecular attractive forces are larger in liquid toluene than in liquid benzene. The melting and boiling points of phenol, another substituted benzene shown in Figure 12.29, are higher than those of benzene because the OH group of phenol can form hydrogen bonds.
![]() GO FIGURE
GO FIGURE
In which substance, benzene or toluene, are the intermolecular forces stronger? In which substance do the molecules pack more efficiently?
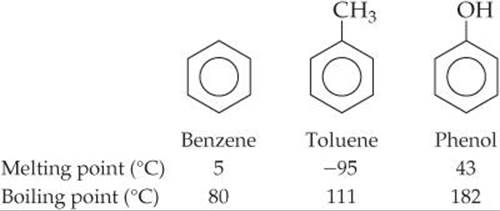
![]() FIGURE 12.29 Melting and boiling points for benzene, toluene, and phenol.
FIGURE 12.29 Melting and boiling points for benzene, toluene, and phenol.