CHEMISTRY THE CENTRAL SCIENCE
2 ATOMS, MOLECULES, AND IONS
2.4 ATOMIC WEIGHTS
Atoms are small pieces of matter, so they have mass. In this section we discuss the mass scale used for atoms and introduce the concept of atomic weights.
The Atomic Mass Scale
Scientists of the nineteenth century were aware that atoms of different elements have different masses. They found, for example, that each 100.0 g of water contains 11.1 g of hydrogen and 88.9 g of oxygen. Thus, water contains 88.9/11.1 = 8 times as much oxygen, by mass, as hydrogen. Once scientists understood that water contains two hydrogen atoms for each oxygen atom, they concluded that an oxygen atom must have 2 × 8 = 16 times as much mass as a hydrogen atom. Hydrogen, the lightest atom, was arbitrarily assigned a relative mass of 1 (no units). Atomic masses of other elements were at first determined relative to this value. Thus, oxygen was assigned an atomic mass of 16.
Today we can determine the masses of individual atoms with a high degree of accuracy. For example, we know that the 1H atom has a mass of 1.6735 × 10–24 g and the 16O atom has a mass of 2.6560 × 10–23 g. As we noted in Section 2.3, it is convenient to use the atomic mass unit(amu) when dealing with these extremely small masses:
1 amu = 1.66054 × 10–24 g and 1 g = 6.02214 × 1023 amu
The atomic mass unit is presently defined by assigning a mass of exactly 12 amu to an atom of the 12C isotope of carbon. In these units, an 1H atom has a mass of 1.0078 amu and an 16O atom has a mass of 15.9949 amu.
Atomic Weight
Most elements occur in nature as mixtures of isotopes. We can determine the average atomic mass of an element, usually called the element's atomic weight, by using the masses of its isotopes and their relative abundances:
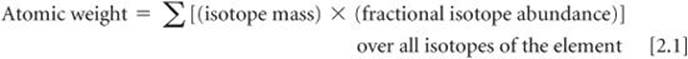
Naturally occurring carbon, for example, is composed of 98.93% 12C and 1.07% 13C. The masses of these isotopes are 12 amu (exactly) and 13.00335 amu, respectively, making the atomic weight of carbon
(0.9893)(12 amu) + (0.0107)(13.00335 amu) = 12.01 amu
The atomic weights of the elements are listed in both the periodic table and the table of elements inside the front cover of this text.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
A particular atom of chromium has a mass of 52.94 amu, whereas the atomic weight of chromium is 51.99 amu. Explain the difference in the two masses.
SAMPLE EXERCISE 2.4 Calculating the Atomic Weight of an Element from Isotopic Abundances
Naturally occurring chlorine is 75.78% 35Cl (atomic mass 34.969 amu) and 24.22% 37Cl (atomic mass 36.966 amu). Calculate the atomic weight of chlorine.
SOLUTION
We can calculate the atomic weight by multiplying the abundance of each isotope by its atomic mass and summing these products. Because 75.78% = 0.7578 and 24.22% = 0.2422, we have
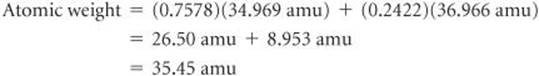
This answer makes sense: The atomic weight, which is actually the average atomic mass, is between the masses of the two isotopes and is closer to the value of 35Cl, the more abundant isotope.
PRACTICE EXERCISE
Three isotopes of silicon occur in nature: 28Si (92.23%), atomic mass 27.97693 amu; 29Si (4.68%), atomic mass 28.97649 amu; and 30Si (3.09%), atomic mass 29.97377 amu. Calculate the atomic weight of silicon.
Answer: 28.09 amu
 A CLOSER LOOK
A CLOSER LOOK
THE MASS SPECTROMETER
The most accurate means for determining atomic weights is provided by the mass spectrometer (![]() FIGURE 2.12). A gaseous sample is introduced at A and bombarded by a stream of high-energy electrons at B. Collisions between the electrons and the atoms or molecules of the gas produce positively charged particles that are then accelerated toward a negatively charged grid (C). After the particles pass through the grid, they encounter two slits that allow only a narrow beam of particles to pass. This beam then passes between the poles of a magnet, which deflects the particles into a curved path. For particles with the same charge, the extent of deflection depends on mass—the more massive the particle, the less the deflection. The particles are thereby separated according to their masses. By changing the strength of the magnetic field or the accelerating voltage on the grid, charged particles of various masses can be selected to enter the detector.
FIGURE 2.12). A gaseous sample is introduced at A and bombarded by a stream of high-energy electrons at B. Collisions between the electrons and the atoms or molecules of the gas produce positively charged particles that are then accelerated toward a negatively charged grid (C). After the particles pass through the grid, they encounter two slits that allow only a narrow beam of particles to pass. This beam then passes between the poles of a magnet, which deflects the particles into a curved path. For particles with the same charge, the extent of deflection depends on mass—the more massive the particle, the less the deflection. The particles are thereby separated according to their masses. By changing the strength of the magnetic field or the accelerating voltage on the grid, charged particles of various masses can be selected to enter the detector.
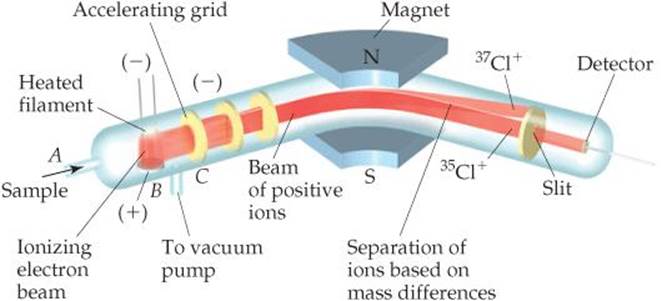
![]() Figure 2.12 A mass spectrometer. Cl atoms are introduced at A and are ionized to form Cl+ ions, which are then directed through a magnetic field. The paths of the ions of the two Cl isotopes diverge as they pass through the field.
Figure 2.12 A mass spectrometer. Cl atoms are introduced at A and are ionized to form Cl+ ions, which are then directed through a magnetic field. The paths of the ions of the two Cl isotopes diverge as they pass through the field.
A graph of the intensity of the detector signal versus particle atomic mass is called a mass spectrum (![]() FIGURE 2.13). Analysis of a mass spectrum gives both the masses of the charged particles reaching the detector and their relative abundances, which are obtained from the signal intensities. Knowing the atomic mass and the abundance of each isotope allows us to calculate the atomic weight of an element, as shown in Sample Exercise 2.4.
FIGURE 2.13). Analysis of a mass spectrum gives both the masses of the charged particles reaching the detector and their relative abundances, which are obtained from the signal intensities. Knowing the atomic mass and the abundance of each isotope allows us to calculate the atomic weight of an element, as shown in Sample Exercise 2.4.
Mass spectrometers are used extensively today to identify chemical compounds and analyze mixtures of substances. Any molecule that loses electrons can fall apart, forming an array of positively charged fragments. The mass spectrometer measures the masses of these fragments, producing a chemical “fingerprint” of the molecule and providing clues about how the atoms were connected in the original molecule. Thus, a chemist might use this technique to determine the molecular structure of a newly synthesized compound or to identify a pollutant in the environment.
RELATED EXERCISES: 2.33, 2.34, 2.35(b), 2.36, 2.92, and 2.93
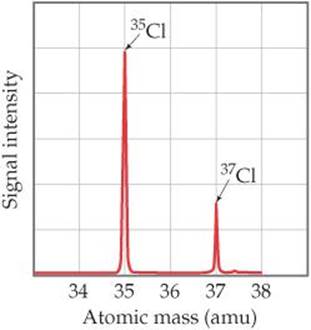
![]() Figure 2.13 Mass spectrum of atomic chlorine. The fractional abundances of the isotopes 35Cl and 37Cl are indicated by the relative signal intensities of the beams reaching the detector of the mass spectrometer.
Figure 2.13 Mass spectrum of atomic chlorine. The fractional abundances of the isotopes 35Cl and 37Cl are indicated by the relative signal intensities of the beams reaching the detector of the mass spectrometer.