CHEMISTRY THE CENTRAL SCIENCE
20 ELECTRO-CHEMISTRY

A VARIETY OF BATTERIES of different sizes, composition, and voltages.
WHAT'S AHEAD
20.1 OXIDATION STATES AND OXIDATION-REDUCTION REACTIONS
We review oxidation states and oxidation-reduction (redox) reactions.
20.2 BALANCING REDOX EQUATIONS
We learn how to balance redox equations using the method of half-reactions.
20.3 VOLTAIC CELLS
We consider voltaic cells, which produce electricity from spontaneous redox reactions. Solid electrodes serve as the surfaces at which oxidation and reduction take place. The electrode where oxidation occurs is the anode, and the electrode where reduction occurs is the cathode.
20.4 CELL POTENTIALS UNDER STANDARD CONDITIONS
We see that an important characteristic of a voltaic cell is its cell potential, which is the difference in the electrical potentials at the two electrodes and is measured in units of volts. Half-cell potentials are tabulated for reduction half-reactions under standard conditions (standard reduction potentials).
20.5 FREE ENERGY AND REDOX REACTIONS
We relate the Gibbs free energy, ΔG°, to cell potential.
20.6 CELL POTENTIALS UNDER NONSTANDARD CONDITIONS
We calculate cell potentials under nonstandard conditions by using standard cell potentials and the Nernst equation.
20.7 BATTERIES AND FUEL CELLS
We describe batteries and fuel cells, which are commercially important energy sources that use electrochemical reactions.
20.8 CORROSION
We discuss corrosion, a spontaneous electrochemical process involving metals.
20.9 ELECTROLYSIS
Finally, we focus on nonspontaneous redox reactions, examining electrolytic cells, which use electricity to perform chemical reactions.
WE ARE SURROUNDED BY AN amazing array of portable electronic gadgets, including cell phones, portable music players, laptop computers, and gaming devices. In the absence of batteries, however, our electronic gadgetry would be nothing more than extra weight. Thus, a variety of batteries of different sizes, compositions, and voltages have been developed, some of which are shown in the chapter-opening photograph. Considerable research is in progress to develop new batteries with more power, faster recharging ability, lighter weight, or cheaper price. At the heart of such development are the oxidation-reduction reactions that power batteries.
As we discussed in Chapter 4, oxidation is the loss of electrons in a chemical reaction, and reduction is the gain of electrons. ![]() (Section 4.4) Thus, oxidation-reduction (redox) reactions occur when electrons are transferred from an atom that is oxidized to an atom that is reduced. Redox reactions are involved not only in the operation of batteries but also in a wide variety of important natural processes, including the rusting of iron, the browning of foods, and the respiration of animals. Electrochemistry is the study of the relationships between electricity and chemical reactions. It includes the study of both spontaneous and nonspontaneous processes.
(Section 4.4) Thus, oxidation-reduction (redox) reactions occur when electrons are transferred from an atom that is oxidized to an atom that is reduced. Redox reactions are involved not only in the operation of batteries but also in a wide variety of important natural processes, including the rusting of iron, the browning of foods, and the respiration of animals. Electrochemistry is the study of the relationships between electricity and chemical reactions. It includes the study of both spontaneous and nonspontaneous processes.
20.1 OXIDATION STATES AND OXIDATION-REDUCTION REACTIONS
We determine whether a given chemical reaction is an oxidation-reduction reaction by keeping track of the oxidation numbers (oxidation states) of the elements involved in the reaction. ![]() (Section 4.4) This procedure identifies whether the oxidation state changes for any elements involved in the reaction. For example, consider the reaction that occurs spontaneously when zinc metal is added to a strong acid (
(Section 4.4) This procedure identifies whether the oxidation state changes for any elements involved in the reaction. For example, consider the reaction that occurs spontaneously when zinc metal is added to a strong acid (![]() FIGURE 20.1):
FIGURE 20.1):
![]()
The chemical equation for this reaction can be written
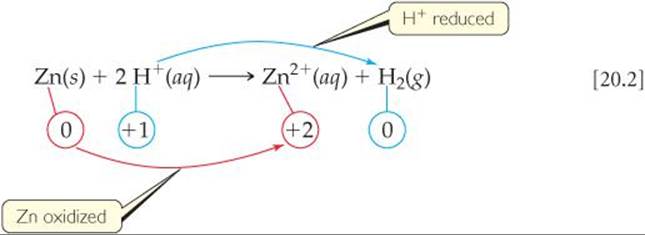
From the oxidation numbers below the equation, we see that the oxidation number of Zn changes from 0 to +2 and that of H changes from +1 to 0. Thus, this is an oxidation-reduction reaction. Electrons are transferred from zinc atoms to hydrogen ions and, therefore, Zn is oxidized and H+ is reduced.
![]() GO FIGURE
GO FIGURE
Explain (a) the vigorous bubbling in the beaker on the right and (b) the formation of steam above that beaker.

![]() FIGURE 20.1 Oxidation of zinc by hydrochloric acid.
FIGURE 20.1 Oxidation of zinc by hydrochloric acid.
In a reaction such as Equation 20.2, a clear transfer of electrons occurs. In some reactions, however, the oxidation numbers change, but we cannot say that any substance literally gains or loses electrons. For example, in the combustion of hydrogen gas,
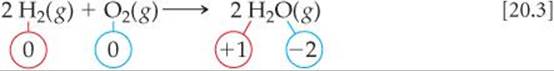
hydrogen is oxidized from the 0 to the +1 oxidation state and oxygen is reduced from the 0 to the –2 oxidation state. Therefore, Equation 20.3 is an oxidation-reduction reaction. Water is not an ionic substance, however, and so there is not a complete transfer of electrons from hydrogen to oxygen as water is formed. Thus, keeping track of oxidation states is a convenient form of “bookkeeping,” but you should not generally equate the oxidation state of an atom with its actual charge in a chemical compound. ![]() (Section 8.5 “A Closer Look: Oxidation Numbers, Formal Charges, and Actual Partial Charges”)
(Section 8.5 “A Closer Look: Oxidation Numbers, Formal Charges, and Actual Partial Charges”)
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What are the oxidation numbers of the elements in the nitrite ion, NO2–?
In any redox reaction, both oxidation and reduction must occur. If one substance is oxidized, another must be reduced. The substance that makes it possible for another substance to be oxidized is called either the oxidizing agent or the oxidant. The oxidizing agent acquires electrons from the other substance and so is itself reduced. A reducing agent, or reductant, is a substance that gives up electrons, thereby causing another substance to be reduced. The reducing agent is therefore oxidized in the process. In Equation 20.2, H+(aq), the species that is reduced, is the oxidizing agent and Zn(s), the species that is oxidized, is the reducing agent.
SAMPLE EXERCISE 20.1 Identifying Oxidizing and Reducing Agents
The nickel-cadmium (nicad) battery uses the following redox reaction to generate electricity:
![]()
Identify the substances that are oxidized and reduced, and indicate which is the oxidizing agent and which is the reducing agent.
SOLUTION
Analyze We are given a redox equation and asked to identify the substance oxidized and the substance reduced and to label the oxidizing agent and the reducing agent.
Plan First, we assign oxidation states, or numbers, to all the atoms and determine which elements change oxidation state. Second, we apply the definitions of oxidation and reduction.
Solve
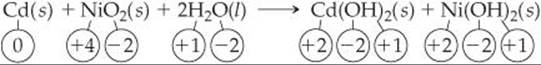
The oxidation state of Cd increases from 0 to +2, and that of Ni decreases from +4 to +2. Thus, the Cd atom is oxidized (loses electrons) and is the reducing agent. The oxidation state of Ni decreases as NiO2 is converted into Ni(OH)2. Thus, NiO2 is reduced (gains electrons) and is the oxidizing agent.
Comment A common mnemonic for remembering oxidation and reduction is “LEO the lion says GER”: losing electrons is oxidation; gaining electrons is reduction.
PRACTICE EXERCISE
Identify the oxidizing and reducing agents in the reaction
![]()
Answer: Al(s) is the reducing agent; MnO4–(aq) is the oxidizing agent.