CHEMISTRY THE CENTRAL SCIENCE
2 ATOMS, MOLECULES, AND IONS
EXERCISES
VISUALIZING CONCEPTS
These exercises are intended to probe your understanding of key concepts rather than your ability to utilize formulas and perform calculations. Exercises with red exercise numbers have answers in the back of the book.
2.1 A charged particle is caused to move between two electrically charged plates, as shown here.
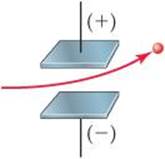
(a) Why does the path of the charged particle bend? (b) What is the sign of the electrical charge on the particle? (c) As the charge on the plates is increased, would you expect the bending to increase, decrease, or stay the same? (d) As the mass of the particle is increased while the speed of the particles remains the same, would you expect the bending to increase, decrease, or stay the same? [Section 2.2]
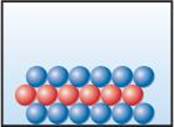
2.2 The following diagram is a representation of 20 atoms of a fictitious element, which we will call nevadium (Nv). The red spheres are 293Nv, and the blue spheres are 295Nv. (a) Assuming that this sample is a statistically representative sample of the element, calculate the percent abundance of each element. (b) If the mass of 293Nv is 293.15 amu and that of 295Nv is 295.15 amu, what is the atomic weight of Nv? [Section 2.4]

2.3 Four of the boxes in the following periodic table are colored. Which of these are metals and which are nonmetals? Which one is an alkaline earth metal? Which one is a noble gas? [Section 2.5]
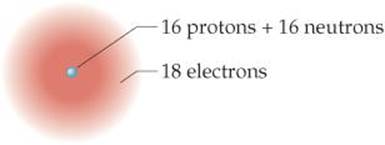
2.4 Does the following drawing represent a neutral atom or an ion? Write its complete chemical symbol including mass number, atomic number, and net charge (if any). [Sections 2.3 and 2.7]
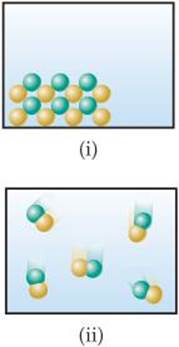
2.5 Which of the following diagrams most likely represents an ionic compound, and which represents a molecular one? Explain your choice. [Sections 2.6 and 2.7]
2.6 Write the chemical formula for the following compound. Is the compound ionic or molecular? Name the compound. [Sections 2.6 and 2.8]
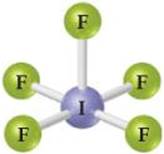
2.7 Five of the boxes in the following periodic table are colored. Predict the charge on the ion associated with each of these elements. [Section 2.7]

2.8 The following diagram represents an ionic compound in which the red spheres represent cations and blue spheres represent anions. Which of the following formulas is consistent with the drawing: KBr, K2SO4, Ca(NO3)2, Fe2(SO4)3? Name the compound. [Sections 2.7 and 2.8]
These exercises are divided into sections that deal with specific topics in the chapter. The exercises are grouped in pairs, with the answers given in the back of the book to the odd-numbered exercises, as indicated by the red exercise numbers. Those exercises whose numbers appear in brackets are more challenging than the nonbracketed exercises.
ATOMIC THEORY AND THE DISCOVERY OF ATOMIC STRUCTURE (sections 2.1–2.2)
2.9 How does Dalton's atomic theory account for the fact that when 1.000 g of water is decomposed into its elements, 0.111 g of hydrogen and 0.889 g of oxygen are obtained regardless of the source of the water?
2.10 Hydrogen sulfide is composed of two elements: hydrogen and sulfur. In an experiment, 6.500 g of hydrogen sulfide is fully decomposed into its elements. (a) If 0.384 g of hydrogen is obtained in this experiment, how many grams of sulfur must be obtained? (b) What fundamental law does this experiment demonstrate? (c) How is this law explained by Dalton's atomic theory?
______
2.11 A chemist finds that 30.82 g of nitrogen will react with 17.60 g, 35.20 g, 70.40 g, or 88.00 g of oxygen to form four different compounds. (a) Calculate the mass of oxygen per gram of nitrogen in each compound. (b) How do the numbers in part (a) support Dalton's atomic theory?
2.12 In a series of experiments, a chemist prepared three different compounds that contain only iodine and fluorine and determined the mass of each element in each compound:
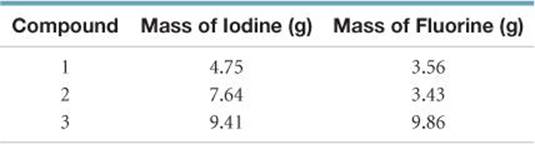
(a) Calculate the mass of fluorine per gram of iodine in each compound. (b) How do the numbers in part (a) support the atomic theory?
______
2.13 Summarize the evidence used by J. J. Thomson to argue that cathode rays consist of negatively charged particles.
2.14 An unknown particle is caused to move between two electrically charged plates, as illustrated in Figure 2.8. Its path is deflected by a smaller magnitude in the opposite direction from that of a beta particle. What can you conclude about the charge and mass of this unknown particle?
______
2.15 How did Rutherford interpret the following observations made during his α-particle scattering experiments? (a) Most a particles were not appreciably deflected as they passed through the gold foil. (b) A few a particles were deflected at very large angles. (c) What differences would you expect if beryllium foil were used instead of gold foil in the a-particle scattering experiment?
2.16 Millikan determined the charge on the electron by studying the static charges on oil drops falling in an electric field (Figure 2.5). A student carried out this experiment using several oil drops for her measurements and calculated the charges on the drops. She obtained the following data:
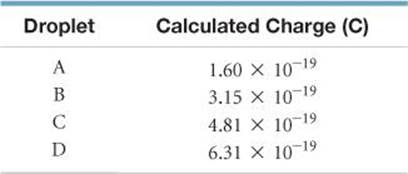
(a) What is the significance of the fact that the droplets carried different charges? (b) What conclusion can the student draw from these data regarding the charge of the electron? (c) What value (and to how many significant figures) should she report for the electronic charge?
MODERN VIEW OF ATOMIC STRUCTURE; ATOMIC WEIGHTS (sections 2.3–2.4)
2.17 The radius of an atom of gold (Au) is about 1.35 Å. (a) Express this distance in nanometers (nm) and in picometers (pm). (b) How many gold atoms would have to be lined up to span 1.0 mm? (c) If the atom is assumed to be a sphere, what is the volume in cm3 of a single Au atom?
2.18 An atom of rhodium (Rh) has a diameter of about 2.7 × 10−8 cm. (a) What is the radius of a rhodium atom in angstroms (Å) and in meters (m)? (b) How many Rh atoms would have to be placed side by side to span a distance of 6.0 μm? (c) If you assume that the Rh atom is a sphere, what is the volume in m3 of a single atom?
______
2.19 Answer the following questions without referring to Table 2.1: (a) What are the main subatomic particles that make up the atom? (b) What is the relative charge (in multiples of the electronic charge) of each of the particles? (c) Which of the particles is the most massive? (d) Which is the least massive?
2.20 Determine whether each of the following statements is true or false. If false, correct the statement to make it true: (a) The nucleus has most of the mass and comprises most of the volume of an atom. (b) Every atom of a given element has the same number of protons. (c) The number of electrons in an atom equals the number of neutrons in the atom. (d) The protons in the nucleus of the helium atom are held together by a force called the strong nuclear force.
______
2.21 (a) Define atomic number and mass number. (b) Which of these can vary without changing the identity of the element?
2.22 (a) Which two of the following are isotopes of the same element: ![]() ,
, ![]() ,
, ![]() ? (b) What is the identity of the element whose isotopes you have selected?
? (b) What is the identity of the element whose isotopes you have selected?
______
2.23 How many protons, neutrons, and electrons are in the following atoms: (a) 40Ar, (b) 65Zn, (c) 70Ga, (d) 80Br, (e) 184W, (f) 243Am?
2.24 Each of the following isotopes is used in medicine. Indicate the number of protons and neutrons in each isotope: (a) phos-phorus-32, (b) chromium-51, (c) cobalt-60, (d) technetium-99, (e) iodine-131, (f) thallium-201.
______
2.25 Fill in the gaps in the following table, assuming each column represents a neutral atom.
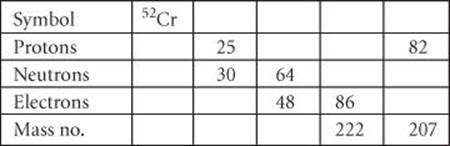
2.26 Fill in the gaps in the following table, assuming each column represents a neutral atom.
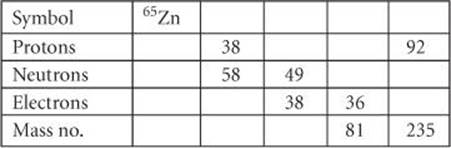
______
2.27 Write the correct symbol, with both superscript and subscript, for each of the following. Use the list of elements inside the front cover as needed: (a) the isotope of platinum that contains 118 neutrons, (b) the isotope of krypton with mass number 84, (c) the isotope of arsenic with mass number 75, (d) the isotope of magnesium that has an equal number of protons and neutrons.
2.28 One way in which Earth's evolution as a planet can be understood is by measuring the amounts of certain isotopes in rocks. One quantity recently measured is the ratio of 129Xe to 130Xe in some minerals. In what way do these two isotopes differ from one another? In what respects are they the same?
______
2.29 (a) What isotope is used as the standard in establishing the atomic mass scale? (b) The atomic weight of boron is reported as 10.81, yet no atom of boron has the mass of 10.81 amu. Explain.
2.30 (a) What is the mass in amu of a carbon-12 atom? (b) Why is the atomic weight of carbon reported as 12.011 in the table of elements and the periodic table in the front inside cover of this text?
______
2.31 Only two isotopes of copper occur naturally, 63Cu (atomic mass = 62.9296 amu; abundance 69.17%) and 65Cu (atomic mass = 64.9278 amu; abundance 30.83%). Calculate the atomic weight (average atomic mass) of copper.
2.32 Rubidium has two naturally occurring isotopes, rubidium-85 (atomic mass = 84.9118 amu; abundance = 72.15%) and rubidium-87 (atomic mass = 86.9092 amu; abundance = 27.85%). Calculate the atomic weight of rubidium.
______
2.33 (a) In what fundamental way is mass spectrometry related to Thomson's cathode-ray experiments (Figure 2.4)? (b) What are the labels on the axes of a mass spectrum? (c) To measure the mass spectrum of an atom, the atom must first lose one or more electrons. Why is this so?
2.34 (a) The mass spectrometer in Figure 2.12 has a magnet as one of its components. What is the purpose of the magnet? (b) The atomic weight of Cl is 35.5 amu. However, the mass spectrum of Cl (Figure 2.13) does not show a peak at this mass. Explain. (c) A mass spectrum of phosphorus (P) atoms shows only a single peak at a mass of 31. What can you conclude from this observation?
______
2.35 Naturally occurring magnesium has the following isotopic abundances:
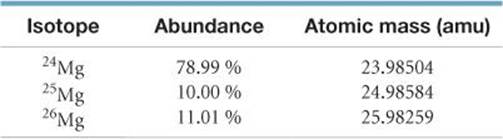
(a) What is the average atomic mass of Mg? (b) Sketch the mass spectrum of Mg.
2.36 Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The mass spectrum of H2 is taken under conditions that prevent decomposition into H atoms. The two naturally occurring isotopes of hydrogen are 1H (atomic mass = 1.00783 amu; abundance 99.9885%) and 2H (atomic mass = 2.01410 amu; abundance 0.0115%). (a) How many peaks will the mass spectrum have? (b) Give the relative atomic masses of each of these peaks. (c) Which peak will be the largest and which the smallest?
THE PERIODIC TABLE; MOLECULES AND IONS (sections 2.5–2.7)
2.37 For each of the following elements, write its chemical symbol, locate it in the periodic table, give its atomic number, and indicate whether it is a metal, metalloid, or nonmetal: (a) chromium, (b) helium, (c) phosphorus, (d) zinc, (e) magnesium, (f) bromine, (g) arsenic.
2.38 Locate each of the following elements in the periodic table; give its name and atomic number, and indicate whether it is a metal, metalloid, or nonmetal: (a) Li, (b) Sc, (c) Ge, (d) Yb, (e) Mn, (f) Sb, (g) Xe.
______
2.39 For each of the following elements, write its chemical symbol, determine the name of the group to which it belongs (Table 2.3), and indicate whether it is a metal, metalloid, or nonmetal: (a) potassium, (b) iodine, (c) magnesium, (d) argon, (e) sulfur.
2.40 The elements of group 4A show an interesting change in properties moving down the group. Give the name and chemical symbol of each element in the group and label it as a non-metal, metalloid, or metal.
______
2.41 What can we tell about a compound when we know the empirical formula? What additional information is conveyed by the molecular formula? By the structural formula? Explain in each case.
2.42 Two compounds have the same empirical formula. One substance is a gas, whereas the other is a viscous liquid. How is it possible for two substances with the same empirical formula to have markedly different properties?
______
2.43 Write the empirical formula corresponding to each of the following molecular formulas: (a) Al2Br6, (b) C8H10, (c) C4H8O2, (d) P4O10, (e) C6H4Cl2, (f) B3N3H6.
2.44 Determine the molecular and empirical formulas of the following: (a) the organic solvent benzene, which has six carbon atoms and six hydrogen atoms; (b) the compound silicon tetra-chloride, which has a silicon atom and four chlorine atoms and is used in the manufacture of computer chips; (c) the reactive substance diborane, which has two boron atoms and six hydrogen atoms; (d) the sugar called glucose, which has six carbon atoms, twelve hydrogen atoms, and six oxygen atoms.
______
2.45 How many hydrogen atoms are in each of the following: (a) C2H5OH, (b) Ca(CH3COO)2, (c) (NH4)3PO4?
2.46 How many of the indicated atoms are represented by each chemical formula: (a) carbon atoms in C2H5COOCH3, (b) oxygen atoms in Ca(ClO4)2, (c) hydrogen atoms in (NH4)2HPO4?
______
2.47 Write the molecular and structural formulas for the com pounds represented by the following molecular models:
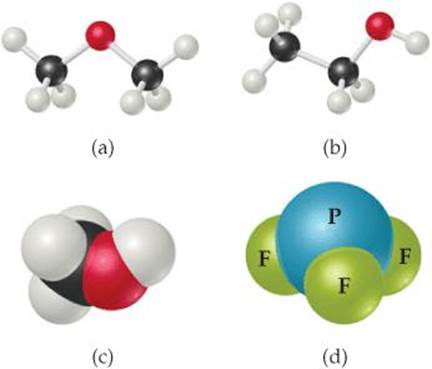
2.48 Write the molecular and structural formulas for the compounds represented by the following models:
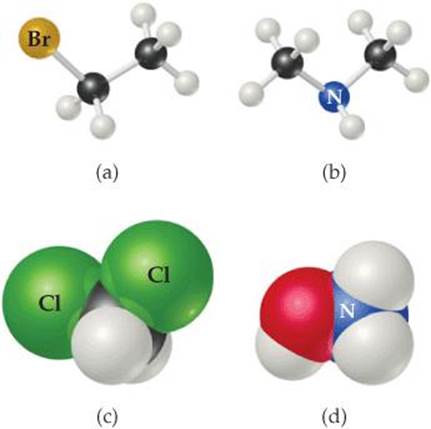
______
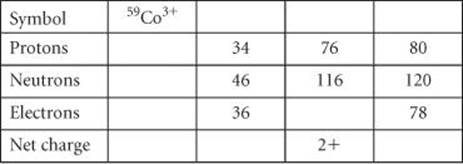
2.49 Fill in the gaps in the following table:
2.50 Fill in the gaps in the following table:
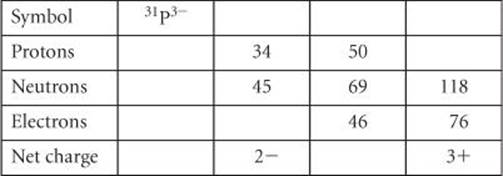
______
2.51 Each of the following elements is capable of forming an ion in chemical reactions. By referring to the periodic table, predict the charge of the most stable ion of each: (a) Mg, (b) Al, (c) K, (d) S, (e) F.
2.52 Using the periodic table, predict the charges of the ions of the following elements: (a) Ga, (b) Sr, (c) As, (d) Br, (e) Se.
______
2.53 Using the periodic table to guide you, predict the chemical formula and name of the compound formed by the following elements: (a) Ga and F, (b) Li and H, (c) Al and I, (d) K and S.
2.54 The most common charge associated with scandium in its compounds is 3+. Indicate the chemical formulas you would expect for compounds formed between scandium and (a) iodine, (b) sulfur, (c) nitrogen.
______
2.55 Predict the chemical formula for the ionic compound formed by (a) Ca2+ and Br–, (b) K+ and CO32–, (c) Al3+ and CH3COO–, (d) NH4+ and SO42–, (e) Mg2+ and PO43–.
2.56 Predict the chemical formulas of the compounds formed by the following pairs of ions: (a) Cr3+ and Br-, (b) Fe3+ and O2–, (c) Hg22+ and CO32–, (d) Ca2+ and ClO3–, (e) NH4+ and PO43–.
______
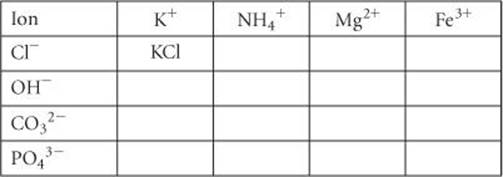
2.57 Complete the table by filling in the formula for the ionic compound formed by each pair of cations and anions, as shown for the first pair.
2.58 Complete the table by filling in the formula for the ionic compound formed by each pair of cations and anions, as shown for the first pair.
______
2.59 Predict whether each of the following compounds is molecular or ionic: (a) B2H6, (b) CH3OH, (c) LiNO3, (d) Sc2O3, (e) CsBr, (f) NOCl, (g) NF3, (h) Ag2SO4.
2.60 Which of the following are ionic, and which are molecular? (a) PF5, (b) NaI, (c) SCl2, (d) Ca(NO3)2, (e) FeCl3, (f) LaP, (g) CoCO3, (h) N2O4.
NAMING INORGANIC COMPOUNDS; ORGANIC MOLECULES (sections 2.8–2.9)
2.61 Give the chemical formula for (a) chlorite ion, (b) chloride ion, (c) chlorate ion, (d) perchlorate ion, (e) hypochlorite ion.
2.62 Selenium, an element required nutritionally in trace quantities, forms compounds analogous to sulfur. Name the following ions: (a) SeO42–, (b) Se2–, (c) HSe–, (d) HSeO3–.
______
2.63 Give the names and charges of the cation and anion in each of the following compounds: (a) CaO, (b) Na2SO4, (c) KClO4, (d) Fe(NO3)2, (e) Cr(OH)3.
2.64 Give the names and charges of the cation and anion in each of the following compounds: (a) CuS, (b) Ag2SO4, (c) Al(ClO3)3, (d) Co(OH)2, (e) PbCO3.
______
2.65 Name the following ionic compounds: (a) Li2O, (b) FeCl3, (c) NaClO, (d) CaSO3, (e) Cu(OH)2, (f) Fe(NO3)2, (g) Ca(CH3COO)2, (h) Cr2(CO3)3, (i) K2CrO4, (j) (NH4)2SO4.
2.66 Name the following ionic compounds: (a) KCN, (b) NaBrO2, (c) Sr(OH)2, (d) CoS, (e) Fe2(CO3)3, (f) Cr(NO3)3, (g) (NH4)2SO3, (h) NaH2PO4, (i) KMnO4, (j) Ag2Cr2O7.
______
2.67 Write the chemical formulas for the following compounds: (a) aluminum hydroxide, (b) potassium sulfate, (c) copper(I) oxide, (d) zinc nitrate, (e) mercury(II) bromide, (f) iron(III) carbonate, (g) sodium hypobromite.
2.68 Give the chemical formula for each of the following ionic compounds: (a) sodium phosphate, (b) zinc nitrate, (c) barium bromate, (d) iron(II) perchlorate, (e) cobalt(II) hydrogen carbonate, (f) chromium(III) acetate, (g) potassium dichromate.
______
2.69 Give the name or chemical formula, as appropriate, for each of the following acids: (a) HBrO3, (b) HBr, (c) H3PO4, (d) hypochlorous acid, (e) iodic acid, (f) sulfurous acid.
2.70 Provide the name or chemical formula, as appropriate, for each of the following acids: (a) hydroiodic acid, (b) chloric acid, (c) nitrous acid, (d) H2CO3, (e) HClO4, (f) CH3COOH
______
2.71 Give the name or chemical formula, as appropriate, for each of the following binary molecular substances: (a) SF6, (b) IF5, (c) XeO3, (d) dinitrogen tetroxide, (e) hydrogen cyanide, (f) tetraphosphorus hexasulfide.
2.72 The oxides of nitrogen are very important components in urban air pollution. Name each of the following compounds: (a) N2O, (b) NO, (c) NO2, (d) N2O5, (e) N2O4.
______
2.73 Write the chemical formula for each substance mentioned in the following word descriptions (use the front inside cover to find the symbols for the elements you don't know). (a) Zinc carbonate can be heated to form zinc oxide and carbon diox ide. (b) On treatment with hydrofluoric acid, silicon dioxide forms silicon tetrafluoride and water. (c) Sulfur dioxide reacts with water to form sulfurous acid. (d) The substance phos phorus trihydride, commonly called phosphine, is a toxic gas. (e) Perchloric acid reacts with cadmium to form cadmium(II) perchlorate. (f) Vanadium(III) bromide is a colored solid.
2.74 Assume that you encounter the following sentences in your reading. What is the chemical formula for each substance mentioned? (a) Sodium hydrogen carbonate is used as a deodorant. (b) Calcium hypochlorite is used in some bleaching solutions. (c) Hydrogen cyanide is a very poisonous gas. (d) Magnesium hydroxide is used as a cathartic. (e) Tin(II) fluoride has been used as a fluoride additive in toothpastes. (f) When cadmium sulfide is treated with sulfuric acid, fumes of hydrogen sulfide are given off.
______
2.75 (a) What is a hydrocarbon? (b) Butane is the alkane with a chain of four carbon atoms. Write a structural formula for this compound and determine its molecular and empirical formulas.
2.76 (a) What ending is used for the names of alkanes? (b) Hexane is an alkane whose structural formula has all its carbon atoms in a straight chain. Draw the structural formula for this compound and determine its molecular and empirical formulas. (Hint: You might need to refer toTable 2.6.)
______
2.77 (a) What is a functional group? (b) What functional group characterizes an alcohol? (c) With reference to Exercise 2.75, write a structural formula for 1-butanol, the alcohol derived from butane, by making a substitution on one of the carbon atoms.
2.78 (a) What do ethane and ethanol have in common? (b) How does 1-propanol differ from propane?
______
2.79 Chloropropane is a compound derived from propane by substituting Cl for H on one of the carbon atoms. (a) Draw the structural formulas for the two isomers of chloropropane. (b) Suggest names for these two compounds.
2.80 Draw the structural formulas for three isomers of pentane, C5H12.
ADDITIONAL EXERCISES
These exercises are not divided by category, although they are roughly in the order of the topics in the chapter. They are not paired.
2.81 Suppose a scientist repeats the Millikan oil-drop experiment but reports the charges on the drops using an unusual (and imaginary) unit called the warmomb (wa). The scientist obtains the following data for four of the drops:
(a) If all the droplets were the same size, which would fall most slowly through the apparatus? (b) From these data, what is the best choice for the charge of the electron in warmombs?
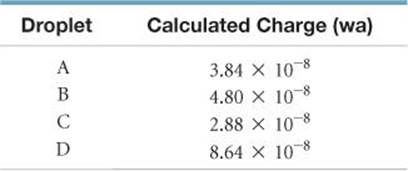
(c) Based on your answer to part (b), how many electrons are there on each of the droplets? (d) What is the conversion factor between warmombs and coulombs?
2.82 The natural abundance of 3He is 0.000137%. (a) How many protons, neutrons, and electrons are in an atom of 3He? (b) Based on the sum of the masses of their subatomic particles, which is expected to be more massive, an atom of 3He or an atom of 3H (which is also calledtritium)? (c) Based on your answer to part (b), what would need to be the precision of a mass spectrometer that is able to differentiate between peaks that are due to 3He+ and 3H+?
2.83 A cube of gold that is 1.00 cm on a side has a mass of 19.3 g. A single gold atom has a mass of 197.0 amu. (a) How many gold atoms are in the cube? (b) From the information given, estimate the diameter in Å of a single gold atom. (c) What assumptions did you make in arriving at your answer for part (b)?
2.84 The diameter of a rubidium atom is 4.95 Å. We will consider two different ways of placing the atoms on a surface. In arrangement A, all the atoms are lined up with one another to form a square grid. Arrangement B is called a close-packed arrangement because the atoms sit in the “depressions” formed by the previous row of atoms:
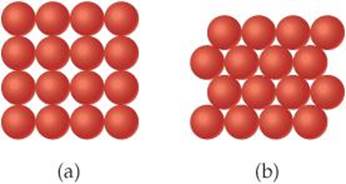
(a) Using arrangement A, how many Rb atoms could be placed on a square surface that is 1.0 cm on a side? (b) How many Rb atoms could be placed on a square surface that is 1.0 cm on a side, using arrangement B? (c) By what factor has the number of atoms on the surface increased in going to arrangement B from arrangement A? If extended to three dimensions, which arrangement would lead to a greater density for Rb metal?
2.85 (a) Assuming the dimensions of the nucleus and atom shown in Figure 2.11, what fraction of the volume of the atom is taken up by the nucleus? (b) Using the mass of the proton from Table 2.1 and assuming its diameter is 1.0 × 10−15 m, calculate the density of a proton in g/cm3.
2.86 Identify the element represented by each of the following symbols and give the number of protons and neutrons in each: (a) ![]() , (b)
, (b) ![]() , (c)
, (c) ![]() , (d)
, (d) ![]() .
.
2.87 The element oxygen has three naturally occurring isotopes, with 8, 9, and 10 neutrons in the nucleus, respectively. (a) Write the full chemical symbols for these three isotopes. (b) Describe the similarities and differences between the three kinds of atoms of oxygen.
2.88 Use Coulomb's law, F = kQ1Q2/d2, to calculate the electric force on an electron (Q = –1.6 × 10−19 C) exerted by a single proton if the particles are 0.53 × 10−10 m apart. The constant k in Coulomb's law is 9.0 × 109 N · m2/C2. (The unit abbreviated N is the newton, the SI unit of force.)
2.89 The element lead (Pb) consists of four naturally occurring isotopes with atomic masses 203.97302, 205.97444, 206.97587, and 207.97663 amu. The relative abundances of these four isotopes are 1.4, 24.1, 22.1, and 52.4%, respectively. From these data, calculate the atomic weight of lead.
2.90 Gallium (Ga) consists of two naturally occurring isotopes with masses of 68.926 and 70.925 amu. (a) How many protons and neutrons are in the nucleus of each isotope? Write the complete atomic symbol for each, showing the atomic number and mass number. (b) The average atomic mass of Ga is 69.72 amu. Calculate the abundance of each isotope.
2.91 Using a suitable reference such as the CRC Handbook of Chemistry and Physics or http://www.webelements.com, look up the following information for nickel: (a) the number of known isotopes, (b) the atomic masses (in amu) and (c) the natural abundances of the five most abundant isotopes.
2.92 There are two different isotopes of bromine atoms. Under normal conditions, elemental bromine consists of Br2 molecules, and the mass of a Br2 molecule is the sum of the masses of the two atoms in the molecule. The mass spectrum of Br2 consists of three peaks:

(a) What is the origin of each peak (of what isotopes does each consist)? (b) What is the mass of each isotope? (c) Determine the average molecular mass of a Br2 molecule. (d) Determine the average atomic mass of a bromine atom. (e) Calculate the abundances of the two isotopes.
2.93 It is common in mass spectrometry to assume that the mass of a cation is the same as that of its parent atom. (a) Using data in Table 2.1, determine the number of significant figures that must be reported before the difference in mass of 1H and 1H+ is significant. (b) What percentage of the mass of an 1H atom does the electron represent?
2.94 From the following list of elements—Ar, H, Ga, Al, Ca, Br, Ge, K, O—pick the one that best fits each description. Use each element only once: (a) an alkali metal, (b) an alkaline earth metal, (c) a noble gas, (d) a halogen, (e) a metalloid, (f) a non-metal listed in group 1A, (g) a metal that forms a 3+ ion, (h) a nonmetal that forms a 2– ion, (i) an element that resembles aluminum.
2.95 The first atoms of seaborgium (Sg) were identified in 1974. The longest-lived isotope of Sg has a mass number of 266. (a) How many protons, electrons, and neutrons are in an 266Sg atom? (b) Atoms of Sg are very unstable, and it is therefore difficult to study this element's properties. Based on the position of Sg in the periodic table, what element should it most closely resemble in its chemical properties?
2.96 The explosion of an atomic bomb releases many radioactive isotopes, including strontium-90. Considering the location of strontium in the periodic table, suggest a reason for the fact that this isotope is particularly harmful to humans.
2.97 From the molecular structures shown here, identify the one that corresponds to each of the following species: (a) chlorine gas, (b) propane, (c) nitrate ion, (d) sulfur trioxide, (e) methyl chloride, CH3Cl.
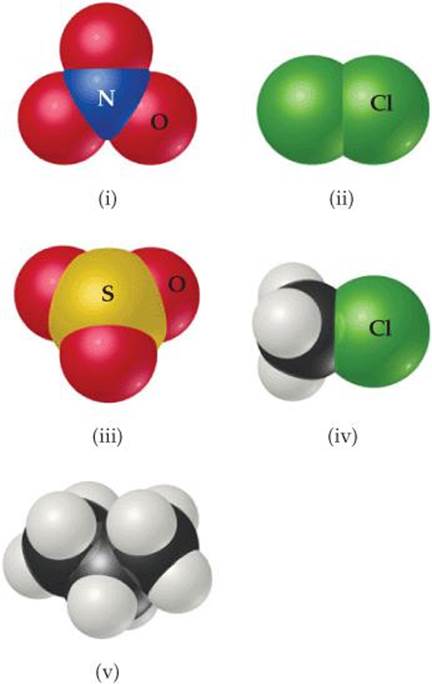
2.98 Name each of the following oxides. Assuming that the compounds are ionic, what charge is associated with the metallic element in each case? (a) NiO, (b) MnO2, (c) Cr2O3, (d) MoO3.
2.99 Fill in the blanks in the following table:
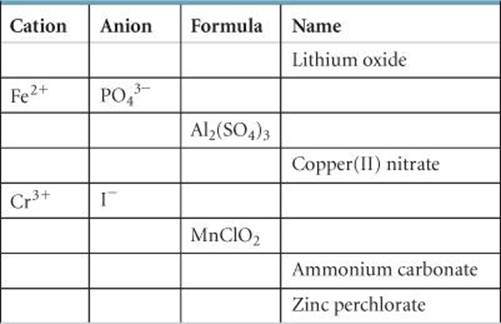
2.100 Iodic acid has the molecular formula HIO3. Write the formulas for the following: (a) the iodate anion, (b) the periodate anion, (c) the hypoiodite anion, (d) hypoiodous acid, (e) periodic acid.
2.101 Elements in the same group of the periodic table often form oxyanions with the same general formula. The anions are also named in a similar fashion. Based on these observations, suggest a chemical formula or name, as appropriate, for each of the following ions: (a) BrO4–, (b)SeO32–, (c) arsenate ion, (d) hydrogen tellurate ion.
2.102 Carbonic acid occurs in carbonated beverages. When allowed to react with lithium hydroxide it produces lithium carbonate. Lithium carbonate is used to treat depression and bipolar disorder. Write chemical formulas for carbonic acid, lithium hydroxide, and lithium carbonate.
2.103 Give the chemical names of each of the following familiar compounds: (a) NaCl (table salt), (b) NaHCO3 (baking soda), (c) NaOCl (in many bleaches), (d) NaOH (caustic soda), (e) (NH4)2CO3 (smelling salts), (f) CaSO4 (plaster of Paris).
2.104 Many familiar substances have common, unsystematic names. For each of the following, give the correct systematic name: (a) saltpeter, KNO3; (b) soda ash, Na2CO3; (c) lime, CaO; (d) muriatic acid, HCl; (e) Epsom salts, MgSO4; (f) milk of magnesia, Mg(OH)2.
2.105 Because many ions and compounds have very similar names, there is great potential for confusing them. Write the correct chemical formulas to distinguish between (a) calcium sulfide and calcium hydrogen sulfide, (b) hydrobromic acid and bromic acid, (c) aluminum nitride and aluminum nitrite, (d) iron(II) oxide and iron(III) oxide, (e) ammonia and ammonium ion, (f) potassium sulfite and potassium bisulfite, (g) mercurous chloride and mercuric chloride, (h) chloric acid and perchloric acid.
2.106 The compound cyclohexane is an alkane in which six carbon atoms form a ring. The partial structural formula of the compound is as follows:
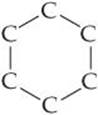
(a) Complete the structural formula for cyclohexane. (b) Is the molecular formula for cyclohexane the same as that for n-hexane, in which the carbon atoms are in a straight line? If possible, comment on the source of any differences. (c) Propose a structural formula for cyclohexanol, the alcohol derived from cyclohexane.