CHEMISTRY THE CENTRAL SCIENCE
20 ELECTRO-CHEMISTRY
20.9 ELECTROLYSIS
Voltaic cells are based on spontaneous redox reactions. It is also possible for nonspontaneous redox reactions to occur, however, by using electrical energy to drive them. For example, electricity can be used to decompose molten sodium chloride into its component elements Na and Cl2. Such processes driven by an outside source of electrical energy are called electrolysis reactions and take place in electrolytic cells.
An electrolytic cell consists of two electrodes immersed either in a molten salt or in a solution. A battery or some other source of electrical energy acts as an electron pump, pushing electrons into one electrode and pulling them from the other. Just as in voltaic cells, the electrode at which reduction occurs is called the cathode, and the electrode at which oxidation occurs is called the anode.
In the electrolysis of molten NaCl, Na+ ions pick up electrons and are reduced to Na at the cathode ![]() FIGURE 20.25. As Na+ ions near the cathode are depleted, additional Na+ ions migrate in. Similarly, there is net movement of Cl– ions to the anode where they are oxidized. The electrode reactions for the electrolysis are
FIGURE 20.25. As Na+ ions near the cathode are depleted, additional Na+ ions migrate in. Similarly, there is net movement of Cl– ions to the anode where they are oxidized. The electrode reactions for the electrolysis are
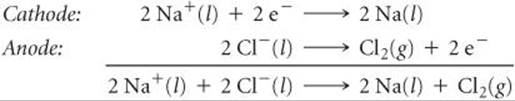
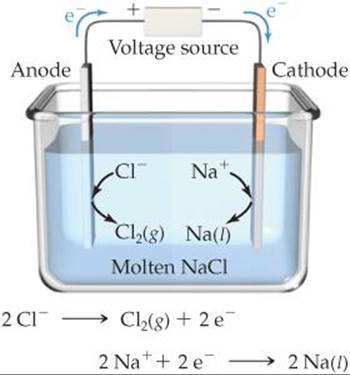
![]() FIGURE 20.25 Electrolysis of molten sodium chloride. Pure NaCl melts at 801°C.
FIGURE 20.25 Electrolysis of molten sodium chloride. Pure NaCl melts at 801°C.
Notice how the energy source is connected to the electrodes in Figure 20.25. The positive terminal is connected to the anode and the negative terminal is connected to the cathode, which forces electrons to move from the anode to the cathode.
Because of the high melting points of ionic substances, the electrolysis of molten salts requires very high temperatures. Do we obtain the same products if we electrolyze the aqueous solution of a salt instead of the molten salt? Frequently the answer is no because water itself might be oxidized to form O2 or reduced to form H2 rather than the ions of the salt.
In our examples of the electrolysis of NaCl, the electrodes are inert; they do not react but merely serve as the surface where oxidation and reduction occur. Several practical applications of electrochemistry, however, are based on active electrodes—electrodes that participate in the electrolysis process. Electroplating, for example, uses electrolysis to deposit a thin layer of one metal on another metal to improve beauty or resistance to corrosion. Examples include electroplating nickel or chromium onto steel and electroplating a precious metal like silver onto a less expensive one.
![]() FIGURE 20.26 illustrates an electrolytic cell for electroplating nickel onto a piece of steel. The anode is a strip of nickel metal, and the cathode is the steel. The electrodes are immersed in a solution of NiSO4(aq). When an external voltage is applied, reduction occurs at the cathode. The standard reduction potential of Ni2+ (
FIGURE 20.26 illustrates an electrolytic cell for electroplating nickel onto a piece of steel. The anode is a strip of nickel metal, and the cathode is the steel. The electrodes are immersed in a solution of NiSO4(aq). When an external voltage is applied, reduction occurs at the cathode. The standard reduction potential of Ni2+ (![]() = –0.28 V) is less negative than that of H2O (
= –0.28 V) is less negative than that of H2O (![]() = –0.83 V), so Ni2+ is preferentially reduced, depositing a layer of nickel metal on the steel cathode.
= –0.83 V), so Ni2+ is preferentially reduced, depositing a layer of nickel metal on the steel cathode.
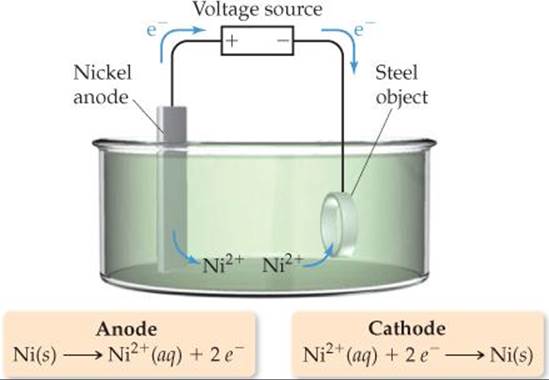
![]() FIGURE 20.26 Electrolytic cell with an active metal electrode. Nickel dissolves from the anode to form Ni2+(aq). At the cathode Ni2+(aq) is reduced and forms a nickel “plate” on the steel cathode.
FIGURE 20.26 Electrolytic cell with an active metal electrode. Nickel dissolves from the anode to form Ni2+(aq). At the cathode Ni2+(aq) is reduced and forms a nickel “plate” on the steel cathode.
At the anode, the nickel metal is oxidized. To explain this behavior, we need to compare the substances in contact with the anode, H2O and NiSO4(aq), with the anode material, Ni. For the NiSO4(aq) solution, neither Ni2+ nor SO42– can be oxidized (because both already have their elements in their highest possible oxidation state). The H2O solvent and the Ni atoms in the anode, however, can both undergo oxidation:
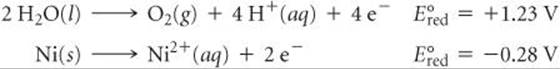
We saw in Section 20.4 that the half-reaction with the more negative ![]() undergoes oxidation more readily. (Remember Figure 20.12: The strongest reducing agents, which are the substances oxidized most readily, have the most negative
undergoes oxidation more readily. (Remember Figure 20.12: The strongest reducing agents, which are the substances oxidized most readily, have the most negative ![]() values.) Thus, it is the Ni(s), with its
values.) Thus, it is the Ni(s), with its ![]() = –0.28 V, that is oxidized at the anode rather than the H2O. If we look at the overall reaction, it appears as if nothing has been accomplished. However, this is not true because Ni atoms are transferred from the Ni anode to the steel cathode, plating the steel with a thin layer of nickel atoms.
= –0.28 V, that is oxidized at the anode rather than the H2O. If we look at the overall reaction, it appears as if nothing has been accomplished. However, this is not true because Ni atoms are transferred from the Ni anode to the steel cathode, plating the steel with a thin layer of nickel atoms.
The standard emf for the overall reaction is
![]()
Because the standard emf is zero, only a small emf is needed to cause the transfer of nickel atoms from one electrode to the other.
Quantitative Aspects of Electrolysis
The stoichiometry of a half-reaction shows how many electrons are needed to achieve an electrolytic process. For example, the reduction of Na+ to Na is a one-electron process:
![]()
Thus, 1 mol of electrons plates out 1 mol of Na metal, 2 mol of electrons plate out 2 mol of Na metal, and so forth. Similarly, 2 mol of electrons are required to produce 1 mol of Cu from Cu2+, and 3 mol of electrons are required to produce 1 mol of Al from Al3+:
![]()
For any half-reaction, the amount of substance reduced or oxidized in an electrolytic cell is directly proportional to the number of electrons passed into the cell.
The quantity of charge passing through an electrical circuit, such as that in an electrolytic cell, is generally measured in coulombs. As noted in Section 20.5, the charge on 1 mol of electrons is 96,485 C. A coulomb is the quantity of charge passing a point in a circuit in 1 s when the current is 1 ampere (A). Therefore, the number of coulombs passing through a cell can be obtained by multiplying the current in amperes by the elapsed time in seconds.
![]()
![]() FIGURE 20.27 shows how the quantities of substances produced or consumed in electrolysis are related to the quantity of electrical charge used. The same relationships can also be applied to voltaic cells. In other words, electrons can be thought of as “reagents” in electrolysis reactions.
FIGURE 20.27 shows how the quantities of substances produced or consumed in electrolysis are related to the quantity of electrical charge used. The same relationships can also be applied to voltaic cells. In other words, electrons can be thought of as “reagents” in electrolysis reactions.
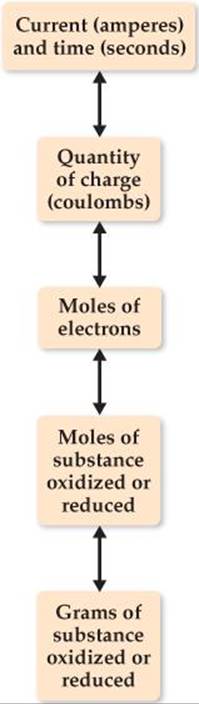
![]() FIGURE 20.27 Relationship between charge and amount of reactant and product in electrolysis reactions.
FIGURE 20.27 Relationship between charge and amount of reactant and product in electrolysis reactions.
SAMPLE EXERCISE 20.14 Relating Electrical Charge and Quantity of Electrolysis
Calculate the number of grams of aluminum produced in 1.00 h by the electrolysis of molten AlCl3 if the electrical current is 10.0 A.
SOLUTION
Analyze We are told that AlCl3 is electrolyzed to form Al and asked to calculate the number of grams of Al produced in 1.00 h with 10.0 A.
Plan Figure 20.27 provides a road map for this problem. First, the product of the amperage and the time in seconds gives the number of coulombs of electrical charge being used (Equation 20.21). Second, the coulombs can be converted with Faraday's constant (F = 96,485 C/mol electrons) to tell us the number of moles of electrons being supplied. Third, reduction of 1 mol of Al3+ to Al requires 3 mol of electrons. Hence, we can use the number of moles of electrons to calculate the number of moles of Al metal it produces. Finally, we convert moles of Al into grams.
Solve First, we calculate the coulombs of electrical charge passed into the electrolytic cell:

Second, we calculate the number of moles of electrons that pass into the cell:
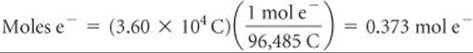
Third, we relate number of moles of electrons to number of moles of aluminum formed, using the half-reaction for the reduction of Al3+:
![]()
Thus, 3 mol of electrons are required to form 1 mol of Al:
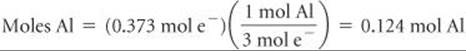
Finally, we convert moles to grams:
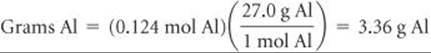
Because each step involves multiplication by a new factor, we can combine all the steps:

PRACTICE EXERCISE
(a) The half-reaction for formation of magnesium metal upon electrolysis of molten MgCl2 is Mg 2+ + 2e– → Mg. Calculate the mass of magnesium formed upon passage of a current of 60.0 A for a period of 4.00 × 103 s. (b) How many seconds would be required to produce 50.0 g of Mg from MgCl2 if the current is 100.0 A?
Answers: (a) 30.2 g of Mg, (b) 3.97 × 103s
 CHEMISTRY PUT TO WORK
CHEMISTRY PUT TO WORK
Electrometallurgy of Aluminum
Many processes used to produce or refine metals are based on electrolysis. Collectively these processes are referred to as electrometallurgy. Electrometallurgical procedures can be broadly differentiated according to whether they involve electrolysis of a molten salt or of an aqueous solution.
Electrolytic methods using molten salts are important for obtaining the more active metals, such as sodium, magnesium, and aluminum. These metals cannot be obtained from aqueous solution because water is more easily reduced than the metal ions. The standard reduction potentials of water under both acidic (![]() = 0.00 V) and basic (
= 0.00 V) and basic (![]() = –0.83 V) conditions are more positive than those of Na+ (
= –0.83 V) conditions are more positive than those of Na+ (![]() = –2.71 V), Mg2+ (
= –2.71 V), Mg2+ (![]() = –2.37 V), and Al3+ (
= –2.37 V), and Al3+ (![]() = –1.66 V).
= –1.66 V).
Historically, obtaining aluminum metal has been a challenge. It is obtained from bauxite ore, which is chemically treated to concentrate aluminum oxide (Al2O3). The melting point of aluminum oxide is above 2000°C, which is too high to permit its use as a molten medium for electrolysis.
The electrolytic process used commercially to produce aluminum is the Hall-Héroult process, named after its inventors, Charles M. Hall and Paul Héroult. Hall (![]() FIGURE 20.28) began working on the problem of reducing aluminum in about 1885 after he had learned from a professor of the difficulty of reducing ores of very active metals. Before the development of an electrolytic process, aluminum was obtained by a chemical reduction using sodium or potassium as the reducing agent, a costly procedure that made aluminum metal expensive. As late as 1852, the cost of aluminum was $545 per pound, far greater than the cost of gold. During the Paris Exposition in 1855, aluminum was exhibited as a rare metal, even though it is the third most abundant element in Earth's crust.
FIGURE 20.28) began working on the problem of reducing aluminum in about 1885 after he had learned from a professor of the difficulty of reducing ores of very active metals. Before the development of an electrolytic process, aluminum was obtained by a chemical reduction using sodium or potassium as the reducing agent, a costly procedure that made aluminum metal expensive. As late as 1852, the cost of aluminum was $545 per pound, far greater than the cost of gold. During the Paris Exposition in 1855, aluminum was exhibited as a rare metal, even though it is the third most abundant element in Earth's crust.

![]() FIGURE 20.28 Charles M. Hall (1863–1914) as a young man.
FIGURE 20.28 Charles M. Hall (1863–1914) as a young man.
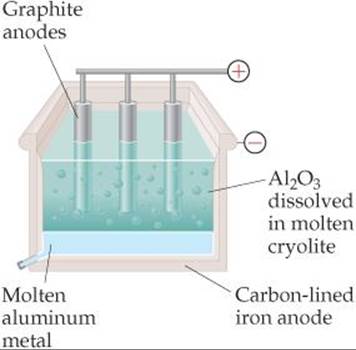
![]() FIGURE 20.29 The Hall-Héroult process. Because molten aluminum is denser than the mixture of cryolite (Na3AlF6) and Al2O3, the metal collects at the bottom of the cell.
FIGURE 20.29 The Hall-Héroult process. Because molten aluminum is denser than the mixture of cryolite (Na3AlF6) and Al2O3, the metal collects at the bottom of the cell.
Hall, who was 21 years old when he began his research, utilized handmade and borrowed equipment in his studies and used a woodshed near his Ohio home as his laboratory. In about a year's time he developed an electrolytic procedure using an ionic compound that melts to form a conducting medium that dissolves Al2O3 but does not interfere with the electrolysis reactions. The ionic compound he selected was the relatively rare mineral cryolite (Na3AlF6). Héroult, who was the same age as Hall, independently made the same discovery in France at about the same time. Because of the research of these two unknown young scientists, large-scale production of aluminum became commercially feasible, and aluminum became a common and familiar metal. Indeed, the factory that Hall subsequently built to produce aluminum evolved into Alcoa Corporation.
In the Hall-Héroult process, Al2O3 is dissolved in molten cryolite, which melts at 1012°C and is an effective electrical conductor (![]() FIGURE 20.29). Graphite rods are employed as anodes and are consumed in the electrolysis:
FIGURE 20.29). Graphite rods are employed as anodes and are consumed in the electrolysis:
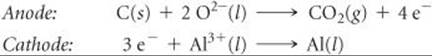
A large amount of electrical energy is needed in the Hall-Héroult process with the result that the aluminum industry consumes about 2% of the electrical energy generated in the United States. Because recycled aluminum requires only 5% of the energy needed to produce “new” aluminum, considerable energy savings can be realized by increasing the amount of aluminum recycled. Approximately 42% of postcon-sumer aluminum is recycled in the United States.
SAMPLE INTEGRATIVE EXERCISE Putting Concepts Together
The Ksp at 298 K for iron(II) fluoride is 2.4 ×10–6. (a) Write a half-reaction that gives the likely products of the two-electron reduction of FeF2(s) in water. (b) Use the Ksp value and the standard reduction potential of Fe2+(aq) to calculate the standard reduction potential for the half-reaction in part (a). (c) Rationalize the difference between the reduction potential in part (a) and the reduction potential for Fe2+(aq).
SOLUTION
Analyze We are going to have to combine what we know about equilibrium constants and electrochemistry to obtain reduction potentials.
Plan For (a) we need to determine which ion, Fe2+ or F–, is more likely to be reduced by two electrons and complete the overall reaction FeF2 + 2e– →?. For (b) we need to write the chemical equation associated with the Ksp and see how it relates to E° for the reduction half-reaction in (a). For (c) we need to compare E° from (b) with the value for the reduction of Fe2+.
Solve
(a) Iron(II) fluoride is an ionic substance that consists of Fe2+ and F– ions. We are asked to predict where two electrons could be added to FeF2. We cannot envision adding the electrons to the F– ions to form F2–, so it seems likely that we could reduce the Fe2+ ions to Fe(s). We therefore predict the half-reaction
![]()
(b) The Ksp for FeF2 refers to the following equilibrium: (Section 17.4)
![]()
We were also asked to use the standard reduction potential of Fe2+, whose half-reaction and standard reduction potentials are listed in Appendix E:
![]()
According to Hess's law, if we can add chemical equations to get a desired equation, then we can add their associated thermodynamic state functions, like ΔH or ΔG, to determine that thermodynamic quantity for the desired reaction. ![]() (Section 5.6) So we need to consider whether the three equations we are working with can be combined in a similar fashion. Notice that if we add the Ksp reaction to the standard reduction half-reaction for Fe2+, we get the half-reaction we want:
(Section 5.6) So we need to consider whether the three equations we are working with can be combined in a similar fashion. Notice that if we add the Ksp reaction to the standard reduction half-reaction for Fe2+, we get the half-reaction we want:
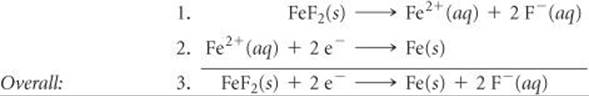
Reaction 3 is still a half-reaction, so we do see the free electrons.
If we knew ΔG° for reactions 1 and 2, we could add them to get ΔG° for reaction 3. We can relate ΔG° to E° by ΔG° = –nFE° (Equation 20.12) and to K by ΔG° = –RT ln K (Equation 19.20, see also Figure 20.14). Furthermore, we know that K for reaction 1 is the Ksp of FeF2, and we know E° for reaction 2. Therefore we can calculate ΔG° for reactions 1 and 2:

(Recall that 1 volt is 1 joule per coulomb.)
Then ΔG° for reaction 3, the one we want, is the sum of the ΔG° values for reactions 1 and 2:
![]()
We can convert this to E° from the relationship ΔG° = –nFE°:
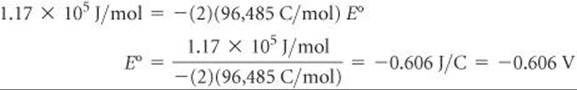
(c) The standard reduction potential for FeF2(–0.606 V) is more negative than that for Fe2+(–0.440 V), telling us that the reduction of FeF2 is the less favorable process. When FeF2 is reduced, we not only reduce the Fe2+ ions but also break up the ionic solid. Because this additional energy must be overcome, the reduction of FeF2 is less favorable than the reduction of Fe2+.