CHEMISTRY THE CENTRAL SCIENCE
22 CHEMISTRY OF THE NONMETALS
22.9 CARBON
Carbon constitutes only 0.027% of Earth's crust. Although some carbon occurs in elemental form as graphite and diamond, most is found in combined form. Over half occurs in carbonate compounds, and carbon is also found in coal, petroleum, and natural gas. The importance of the element stems in large part from its occurrence in all living organisms: Life as we know it is based on carbon compounds.
Elemental Forms of Carbon
We have seen that carbon exists in several allotropic crystalline forms: graphite, diamond, fullerenes, carbon nanotubes, and graphene. Fullerenes, nanotubes, and graphene are discussed in Chapter 12; here we focus on graphite and diamond.
Graphite is a soft, black, slippery solid that has a metallic luster and conducts electricity. It consists of parallel sheets of sp2-hybridized carbon atoms held together by dispersion forces. ![]() (Section 12.7) Diamond is a clear, hard solid in which the carbon atoms form an sp3-hybridized covalent network.
(Section 12.7) Diamond is a clear, hard solid in which the carbon atoms form an sp3-hybridized covalent network. ![]() (Section 12.7) Diamond is denser than graphite (d = 2.25 g/cm3 for graphite; d = 3.51 g/cm3 for diamond). At approximately 100,000 atm at 3000 °C, graphite converts to diamond. In fact, almost any carbon-containing substance, if put under sufficiently high pressure, forms diamonds; scientists at General Electric in the 1950s used peanut butter to make diamonds. About 3 × 104 kg of industrial-grade diamonds are synthesized each year, mainly for use in cutting, grinding, and polishing tools.
(Section 12.7) Diamond is denser than graphite (d = 2.25 g/cm3 for graphite; d = 3.51 g/cm3 for diamond). At approximately 100,000 atm at 3000 °C, graphite converts to diamond. In fact, almost any carbon-containing substance, if put under sufficiently high pressure, forms diamonds; scientists at General Electric in the 1950s used peanut butter to make diamonds. About 3 × 104 kg of industrial-grade diamonds are synthesized each year, mainly for use in cutting, grinding, and polishing tools.
Graphite has a well-defined crystalline structure, but it also exists in two common amorphous forms: carbon black and charcoal. Carbon black is formed when hydrocarbons are heated in a very limited supply of oxygen, such as in this methane reaction:
![]()
Carbon black is used as a pigment in black inks; large amounts are also used in making automobile tires.
Charcoal is formed when wood is heated strongly in the absence of air. Charcoal has an open structure, giving it an enormous surface area per unit mass. “Activated charcoal,” a pulverized form of charcoal whose surface is cleaned by heating with steam, is widely used to adsorb molecules. It is used in filters to remove offensive odors from air and colored or bad-tasting impurities from water.
Oxides of Carbon
Carbon forms two principal oxides: carbon monoxide (CO) and carbon dioxide (CO2). Carbon monoxide is formed when carbon or hydrocarbons are burned in a limited supply of oxygen:
![]()
CO is a colorless, odorless, tasteless gas that is toxic because it binds to hemoglobin in the blood and thus interfere with oxygen transport. Low-level poisoning results in headaches and drowsiness; high-level poisoning can cause death. Automobile engines produce carbon monoxide, which is a major air pollutant.
Carbon monoxide is unusual in that it has a nonbonding pair of electrons on carbon: :C≡C :. It is isoelectronic with N2, so you might expect CO to be equally un-reactive. Moreover, both substances have high bond energies (1072 kJ/mol for C≡O and 941 kJ/mol for N≡N). Because of the lower nuclear charge on carbon (compared with either N or O), however, the carbon nonbonding pair is not held as strongly as that on N or O. Consequently, CO is better able to function as a Lewis base than is N2; for example, CO can coordinate its nonbonding pair to the iron of hemoglobin, displacing O2, but N2 cannot. In addition, CO forms a variety of covalent compounds, known as metal carbonyls, with transition metals. Ni(CO)4, for example, is a volatile, toxic compound formed by warming metallic nickel in the presence of CO. The formation of metal carbonyls is the first step in the transition-metal catalysis of a variety of reactions of CO.
Carbon monoxide has several commercial uses. Because it burns readily, forming CO2, it is employed as a fuel:
![]()
Carbon monoxide is an important reducing agent, widely used in metallurgical operations to reduce metal oxides, such as the iron oxides:
![]()
Carbon dioxide is produced when carbon-containing substances are burned in excess oxygen, such as in this reaction involving ethanol:
![]()
![]() CHEMISTRY PUT TO WORK
CHEMISTRY PUT TO WORK
Carbon Fibers and Composites
The properties of graphite are anisotropic; that is, they differ in different directions through the solid. Along the carbon planes, graphite possesses great strength because of the number and strength of the carbon-carbon bonds in this direction. The bonds between planes are relatively weak, however, making graphite weak in that direction.
Fibers of graphite can be prepared in which the carbon planes are aligned to varying extents parallel to the fiber axis. These fibers are lightweight (density of about 2 g/cm3) and chemically quite unreactive. The oriented fibers are made by first slowly pyrolyzing (decomposing by action of heat) organic fibers at about 150 °C to 300 °C. These fibers are then heated to about 2500 °C to graphitize them (convert amorphous carbon to graphite). Stretching the fiber during pyrolysis helps orient the graphite planes parallel to the fiber axis. More amorphous carbon fibers are formed by pyrolysis of organic fibers at lower temperatures (1200°C to 400 °C). These amorphous materials, commonly called carbon fibers, are the type most often used in commercial materials.
Composite materials that take advantage of the strength, stability, and low density of carbon fibers are widely used. Composites are combinations of two or more materials. These materials are present as separate phases and are combined to form structures that take advantage of certain desirable properties of each component. In carbon composites the graphite fibers are often woven into a fabric that is embedded in a matrix that binds them into a solid structure. The fibers transmit loads evenly throughout the matrix. The finished composite is thus stronger than any one of its components.
Carbon composite materials are used widely in a number of applications, including high-performance graphite sports equipment such as tennis racquets, golf clubs, and bicycle wheels (![]() FIGURE 22.31). Heat-resistant composites are required for many aerospace applications, where carbon composites now find wide use.
FIGURE 22.31). Heat-resistant composites are required for many aerospace applications, where carbon composites now find wide use.

![]() FIGURE 22.31 Carbon composites in commercial products.
FIGURE 22.31 Carbon composites in commercial products.
It is also produced when many carbonates are heated:
![]()
Large quantities of CO2 are obtained as a by-product of the fermentation of sugar during the production of ethanol:
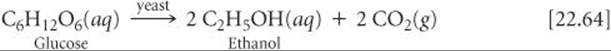
In the laboratory, CO2 can be produced by the action of acids on carbonates (![]() FIGURE 22.32):
FIGURE 22.32):
![]()

![]() FIGURE 22.32 CO2 formation from the reaction between an acid and calcium carbonate in rock.
FIGURE 22.32 CO2 formation from the reaction between an acid and calcium carbonate in rock.
Carbon dioxide is a colorless, odorless gas. It is a minor component of Earth's atmosphere but a major contributor to the greenhouse effect. ![]() (Section 18.2) Although it is not toxic, high concentrations of CO2 increase respiration rate and can cause suffocation. It is readily liquefied by compression. When cooled at atmospheric pressure, however, CO2 forms a solid rather than liquefying. The solid sublimes at atmospheric pressure at –78 °C. This property makes solid CO2, known as dry ice, valuable as a refrigerant. About half of the CO2 consumed annually is used for refrigeration. The other major use of CO2 is in the production of carbonated beverages. Large quantities are also used to manufacture washing soda (Na2CO3 · 10 H2O), used to precipitate metal ions that interfere with the cleansing action of soap, and baking soda (NaHCO3). Baking soda is so named because this reaction occurs during baking:
(Section 18.2) Although it is not toxic, high concentrations of CO2 increase respiration rate and can cause suffocation. It is readily liquefied by compression. When cooled at atmospheric pressure, however, CO2 forms a solid rather than liquefying. The solid sublimes at atmospheric pressure at –78 °C. This property makes solid CO2, known as dry ice, valuable as a refrigerant. About half of the CO2 consumed annually is used for refrigeration. The other major use of CO2 is in the production of carbonated beverages. Large quantities are also used to manufacture washing soda (Na2CO3 · 10 H2O), used to precipitate metal ions that interfere with the cleansing action of soap, and baking soda (NaHCO3). Baking soda is so named because this reaction occurs during baking:
![]()
The H+(aq) is provided by vinegar, sour milk, or the hydrolysis of certain salts. The bubbles of CO2 that form are trapped in the baking dough, causing it to rise.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Yeast are living organisms that make bread rise in the absence of baking soda and acid. What must the yeast be producing to make bread rise?
Carbonic Acid and Carbonates
Carbon dioxide is moderately soluble in H2O at atmospheric pressure. The resulting solution is moderately acidic because of the formation of carbonic acid (H2CO3):
![]()
Carbonic acid is a weak diprotic acid. Its acidic character causes carbonated beverages to have a sharp, slightly acidic taste.
Although carbonic acid cannot be isolated, hydrogen carbonates (bicarbonates) and carbonates can be obtained by neutralizing carbonic acid solutions. Partial neutralization produces HCO3–, and complete neutralization gives CO32–. The HCO3– ion is a stronger base than acid (Kb = 2.3 × 10–8; Ka = 5.6 × 10–11). The carbonate ion is much more strongly basic (Kb = 1.8 × 10–4).
The principal carbonate minerals are calcite (CaCO3), magnesite (MgCO3), dolomite [MgCa(CO3)2], and siderite (FeCO3). Calcite is the principal mineral in limestone and the main constituent of marble, chalk, pearls, coral reefs, and the shells of marine animals such as clams and oysters. Although CaCO3 has low solubility in pure water, it dissolves readily in acidic solutions with evolution of CO2:
![]()
Because water containing CO2 is slightly acidic (Equation 22.67), CaCO3 dissolves slowly in this medium:
![]()
This reaction occurs when surface waters move underground through limestone deposits. It is the principal way Ca2+ enters groundwater, producing “hard water.” If the limestone deposit is deep enough underground, dissolution of the limestone produces a cave.
One of the most important reactions of CaCO3 is its decomposition into CaO and CO2 at elevated temperatures (Equation 22.63). About 2 × 1010 kg (20 million tons) of calcium oxide, known as lime or quicklime, is produced in the United States annually. Because calcium oxide reacts with water to form Ca(OH)2, it is an important commercial base. It is also important in making mortar, the mixture of sand, water, and CaO used to bind bricks, blocks, or rocks together. Calcium oxide reacts with water and CO2 to form CaCO3, which binds the sand in the mortar:
![]()
![]()
Carbides
The binary compounds of carbon with metals, metalloids, and certain nonmetals are called carbides. The more active metals form ionic carbides, and the most common of these contain the acetylide ion (C22–). This ion is isoelectronic with N2, and its Lewis structure, [:C≡ C:]2–, has a carbon–carbon triple bond. The most important ionic carbide is calcium carbide (CaC2), produced by the reduction of CaO with carbon at high temperature:
![]()
The carbide ion is a very strong base that reacts with water to form acetylene (H—C≡C—H):
![]()
Calcium carbide is therefore a convenient solid source of acetylene, which is used in welding (Figure 22.13).
Interstitial carbides are formed by many transition metals. The carbon atoms occupy open spaces (interstices) between the metal atoms in a manner analogous to the interstitial hydrides (Section 22.2). This process generally makes the metal harder. Tungsten carbide, for example, is very hard and very heat-resistant and, thus, used to make cutting tools.
Covalent carbides are formed by boron and silicon. Silicon carbide (SiC), known as Carborundum®, is used as an abrasive and in cutting tools. Almost as hard as diamond, SiC has a diamondlike structure with alternating Si and C atoms.
Other Inorganic Compounds of Carbon
Hydrogen cyanide, HCN, is an extremely toxic gas that famously has the odor of bitter almonds. It is produced by the reaction of a cyanide salt, such as NaCN, with an acid. Aqueous solutions of HCN are known as hydrocyanic acid. Neutralization with a base produces cyanide salts, which are used in the manufacture of several plastics, including nylon and Orlon®. The CN– ion forms stable complexes with most transition metals. ![]() (Section 17.5)
(Section 17.5)
Carbon disulfide, CS2, is an important industrial solvent for waxes, greases, celluloses, and other nonpolar substances. It is a colorless, volatile liquid (bp 46.3 °C). The vapor is very poisonous and highly flammable.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Based on what you know of their physical properties, does CS2 have stronger intermolecular forces than CO2? Explain.