CHEMISTRY THE CENTRAL SCIENCE
7 PERIODIC PROPERTIES OF THE ELEMENTS

Light-emitting diodes, LEDs.
WHAT'S AHEAD
7.1 DEVELOPMENT OF THE PERIODIC TABLE
We begin our discussion with a brief history of the periodic table.
7.2 EFFECTIVE NUCLEAR CHARGE
We next explore the many properties of atoms that depend on the net attraction of the outer electrons to the nucleus and on the average distance of those electrons from the nucleus. The net positive charge of the nucleus experienced by the outer electrons is called the effective nuclear charge.
7.3 SIZES OF ATOMS AND IONS
We explore the relative sizes of atoms and ions, both of which follow trends that are related to their placement in the periodic table.
7.4 IONIZATION ENERGY
We next encounter the ionization energy required to remove one or more electrons from an atom. The periodic trends in ionization energy depend on variations in effective nuclear charge and atomic radii.
7.5 ELECTRON AFFINITIES
Next we examine periodic trends in the energy released when an electron is added to an atom.
7.6 METALS, NONMETALS, AND METALLOIDS
We learn that the physical and chemical properties of metals are different from those of nonmetals. These properties arise from the fundamental characteristics of atoms, particularly ionization energy. Metalloids display properties that are intermediate between those of metals and those of nonmetals.
7.7 TRENDS FOR GROUP 1A AND GROUP 2A METALS
We examine some periodic trends in the chemistry of group 1A and group 2A metals.
7.8 TRENDS FOR SELECTED NONMETALS
We examine some periodic trends in the chemistry of hydrogen and of the elements in groups 6A, 7A, and 8A.
THE BRILLIANT COLORS OF light-emitting diodes (LEDs) arise from the composition of the materials from which they are made. The LEDs shown here are compounds of gallium and aluminum mixed with nitrogen, phosphorus, and arsenic. GaN, GaP, and GaAs can make solid solutions with each other and with AlN, AlP, and AlAs; the composition of each solid solution dictates the wavelength of light emitted by a given LED. Because these elements are in groups 3A and 5A, we say that LEDs are composed of “3-5” materials.
As we saw in Chapter 6, the periodic nature of the periodic table arises from repeating patterns in the electron configurations of the elements. Elements in the same column contain the same number of electrons in their valence orbitals, which are the occupied orbitals that hold the electrons involved in bonding. For example, O ([He]2s2 2p4) and S ([Ne]3s2 3p4) are both in group 6A. The similarity of the electron distribution in their valence s and p orbitals leads to similarities in the properties of these two elements. When we compare elemental O and elemental S, however, it is apparent that they exhibit differences as well, not the least of which is that at room temperature oxygen is a colorless gas but sulfur is a yellow solid. One major difference between O and S atoms is that the outermost electrons of O are in the second shell, whereas those of S are in the third shell. Thus, electron configurations can be used to explain differences as well as similarities in the properties of elements.
In this chapter we explore how some of the important properties of elements change as we move across a row or down a column of the periodic table. In many cases the trends in a row or column allow us to predict the physical and chemical properties of the elements.
7.1 DEVELOPMENT OF THE PERIODIC TABLE
The discovery of chemical elements has been ongoing since ancient times (![]() FIGURE 7.1). Certain elements, such as gold (Au), appear in nature in elemental form and were thus discovered thousands of years ago. In contrast, some elements, such as technetium (Tc), are radioactive and intrinsically unstable. We know about them only because of technology developed during the twentieth century.
FIGURE 7.1). Certain elements, such as gold (Au), appear in nature in elemental form and were thus discovered thousands of years ago. In contrast, some elements, such as technetium (Tc), are radioactive and intrinsically unstable. We know about them only because of technology developed during the twentieth century.
The majority of elements readily form compounds and, consequently, are not found in nature in their elemental form. For centuries, therefore, scientists were unaware of their existence. During the early nineteenth century, advances in chemistry made it easier to isolate elements from their compounds. As a result, the number of known elements more than doubled from 31 in 1800 to 63 by 1865.
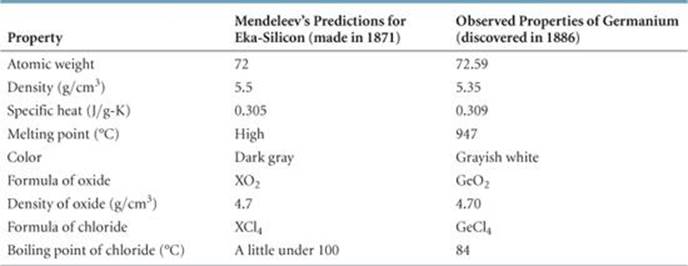
As the number of known elements increased, scientists began classifying them. In 1869, Dmitri Mendeleev in Russia and Lothar Meyer in Germany published nearly identical classification schemes. Both scientists noted that similar chemical and physical properties recur periodically when the elements are arranged in order of increasing atomic weight. Scientists at that time had no knowledge of atomic numbers. Atomic weights, however, generally increase with increasing atomic number, so both Mendeleev and Meyer fortuitously arranged the elements in proper sequence. Although Mendeleev and Meyer came to essentially the same conclusion about the periodicity of elemental properties, Mendeleev is given credit for advancing his ideas more vigorously and stimulating new work. His insistence that elements with similar characteristics be listed in the same column forced him to leave blank spaces in his table. For example, both gallium (Ga) and germanium (Ge) were unknown to Mendeleev. He boldly predicted their existence and properties, referring to them as eka-aluminum (“under” aluminum) and eka-silicon (“under” silicon), respectively, after the elements under which they appeared in his table. When these elements were discovered, their properties closely matched those predicted by Mendeleev, as shown in ![]() TABLE 7.1.
TABLE 7.1.
![]() GO FIGURE
GO FIGURE
Which row of the periodic table contains the most recently discovered elements? Can you suggest a reason?

![]() FIGURE 7.1 Discovering the elements.
FIGURE 7.1 Discovering the elements.
TABLE 7.1 • Comparison of the Properties of Eka-Silicon Predicted by Mendeleev with the Observed Properties of Germanium
In 1913, two years after Rutherford proposed the nuclear model of the atom ![]() (Section 2.2), English physicist Henry Moseley (1887-1915) developed the concept of atomic numbers. Bombarding different elements with high-energy electrons, Moseley found that each element produced X-rays of a unique frequency and that the frequency generally increased as the atomic mass increased. He arranged the X-ray frequencies in order by assigning a unique whole number, called an atomic number, to each element. Moseley correctly identified the atomic number as the number of protons in the nucleus of the atom.
(Section 2.2), English physicist Henry Moseley (1887-1915) developed the concept of atomic numbers. Bombarding different elements with high-energy electrons, Moseley found that each element produced X-rays of a unique frequency and that the frequency generally increased as the atomic mass increased. He arranged the X-ray frequencies in order by assigning a unique whole number, called an atomic number, to each element. Moseley correctly identified the atomic number as the number of protons in the nucleus of the atom. ![]() (Section 2.3)
(Section 2.3)
The concept of atomic number clarified some problems in the periodic table of Moseley's day, which was based on atomic weights. For example, the atomic weight of Ar (atomic number 18) is greater than that of K (atomic number 19), yet the chemical and physical properties of Ar are much more like those of Ne and Kr than like those of Na and Rb. However, when the elements are arranged in order of increasing atomic number, rather than increasing atomic weight, Ar and K appear in their correct places in the table. Moseley's studies also made it possible to identify “holes” in the periodic table, which led to the discovery of previously unknown elements.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Arranging the elements by atomic weight leads to an order slightly different from that in a modern periodic table, where the arrangement is by atomic number. Why does this happen? Looking at the periodic table on the inside front cover, can you find an example other than Ar and K where the order of the elements would be different if the elements were arranged in order of increasing atomic weight?