CHEMICAL BIOLOGY
Glycosphingolipids, Chemistry of
Clifford A. Lingwood, Richard Abram and Murugesapillai Mylvaganum, Hospital for Sick Children, Toronto, Ontario, Canada
doi: 10.1002/9780470048672.wecb633
Eukaryotic cell glycosphingolipids (GSLs) are central components of membrane lipid microdomains that function as trans plasmamembrane signaling foci. GSLs serve as important receptors and coreceptors, primarily to mediate host/microbial pathogen interactions, and undergo unique intracellular trafficking pathways. As such, their chemical modification can generate interventive therapeutic strategies and biologic probes. The process of achieving such goals via chemistry of the carbohydrate is in its infancy, but substitution within the lipid moiety has been used extensively. A consideration of the importance of the lipid moiety in GSL function has led to novel chemical approaches that attempt to retain this property.
Glycosphingolipids (GSLs) are sugar-lipid conjugates expressed on the outer bilayer leaflet of the plasma membrane of all eukaryotic cells. The linear and branched chain carbohydrate structure is determined by a series of Golgi-lumen-located glycosyl transferases that add single sugars from nucleotide sugar donors transported into the Golgi lumen. The acceptor specificity and intraGolgi location of these glycosyl transferases determine the carbohydrate sequence of the GSLs eventually expressed on the plasma membrane. However, there still remains considerable uncertainty as to the mechanism of their sorting via the vesicular secretory pathway and their recycling from the cell surface. Differential sorting of GSLs to the apical and basolateral plasma membrane of polarized cells has been attributed to a combination of carbohydrate and lipid moieties in relation to the domain organization within the membrane (1, 2).
Glycosphingolipid Structure and Metabolism
A typical GSL, globotriaosyl ceramide galactose 1-4 galactose 1-4 glucosyl ceramide (Gb3), is shown in Fig. 1. The sugar sequence of membrane-embedded GSLs can adopt several conformational energy minima because of a restriction around the glucosyl anomeric link as a function of the relative plane of the membrane in which the GSL is contained (3). Molecular modeling of glucosyl ceramide defined nine thermodynamic minima of which the three most favorable are shown for Gb3. Modeling of blood group A GSLs showed that the internal sugar sequence greatly can affect the presentation of the A-epitope from perpendicular to parallel to the cell surface (4). Membrane parallel orientation of GSLs may be limited for longer sugar chains because a calculation of the phi/psi angles of tetra- and pentasaccharide globoseries GSLs showed that these termini can clash with the membrane for some conformers (5). These restrictions are dependent on the relative plane of the plasma membrane in relation to the glucose anomeric link. This relative plane, in turn, is defined by the composition of the ceramide moiety and the phospholipid bilayer. These are distinct in the liquid ordered vs the liquid crystalline phase so it is very likely that the partitioning of GSLs in and out of lipid microdomains significantly affects the conformation of the oligosaccharide.

Figure 1. Structure of the glycosphingolipid, globotriaosyl ceramide, Gal 1-4 Galβ. 1-4 glucosyl ceramide. The three lowest energy conformations as calculated by molecular modeling (3) are shown.
Ceramide
The synthesis of the ceramide lipid moiety of GSLs is initiated in the endoplasmic reticulum (ER) (6) via the condensation of serine and palmitoylCoA. This enzyme has been cloned (7). The product is 3-keto-sphinganine that then is reduced to sphinganine and acylated by ceramide synthase with a long-chain fatty acid to form dihydroceramide, which is converted to ceramide by ceramide desaturase, which inserts the 4,5-trans double bond.
GSLs are highly heterogeneous in terms of fatty acid composition. Fatty acid chain lengths vary from C16 to C24 and can be monounsaturated or 2'hydroxylated in mammalian systems. The functional importance of the heterogeneity has not been defined but may relate to the receptor function of GSLs and their intracellular trafficking (8). Such trafficking, in turn, is related to the membrane organization of GSLs in terms of their ordered or disordered domain structure (9). This membrane organization is itself a property of the GSL lipid moiety. The fatty acid heterogeneity was thought to be a function of the availability and the lack of specificity of the anabolic enzyme, but recently a family of ceramide synthases has been identified (Lass1-6) (10, 11) with a restricted and, in some cases, unique fatty acid specificity. This degree of synthetic control implies that the GSL fatty acid composition is more important than previously considered.
Dihydro ceramide is reduced at the 4-5 position to form ceramide. Ceramide itself is a highly bioactive molecule involved in many pathways of signal transduction, particularly those involved in apoptosis (12). Ceramide-dependent protein kinase (13) and phosphatase activity have been described. The hydroxyl of both ceramide and sphingosine can be phosphorylated. Sphingosine-1-phosphate is also important as a lipid second messenger, particularly in mitogenic responses in which it tends to oppose the action of ceramide (14, 15). A family of sphingosine-1-phosphate receptors has been defined that plays important roles particularly in vascular development (16). Ceramide can also be acylated, but functions for acyl ceramide have yet to be defined. Sphingosine is heterogeneous in terms of its chain length, C18, C20 being the primary species detected in eukaryotic cells. Sphingosine can also be hydroxylated (phytosphingose, particularly prevalent in plant cells). Fatty acid acylation additionally increases the heterogeneity of ceramide because the fatty acid chain length, saturation, and hydroxylation are also variable. Ceramide can flip between the outer and inner leaflet of the ER membrane and is transferred to the Golgi via vesicular and nonvesicular traffic (17).
Carbohydrate structure
GSLs are based on essentially five core sequences (Fig. 2). Glucosyl ceramide is first galactosylated in β1-4 link to give lactosyl ceramide. This example is the only example of a lactose-containing glycoconjugate. Lactosyl ceramide is the branch point for the major GSL series. In the Globo series galactose is added in α1-4 linkage; in the Ganglio series, N-acetylgalactosamine is added in β1-4 linkage; in the Lacto series β1-3 N-acetylglucosamine followed by β1-3gal are added and in the Neolactoseries β1-3 N-acetylglucosamine followed by β1-4 galactose. The Gala series are relatively minor GSLs based on galactosyl ceramide. Galabiosyl ceramide (galactose added in 1-4 linkage) is the most important species (18).
The carbohydrate structures of GSLs have been well characterized because of their ease of extraction and purification by organic chromatography procedures. Some 350 structures have been described (19). Only two novel mammalian GSL structures have been reported this century, which suggests that the mammalian GSL complement—the “glycome”—is approaching completion. The GSL carbohydrate sequence proximal to the lipid moiety is unique to GSLs as compared with other glycoconjugates. The more distal sugar sequences often are shared between glycoproteins and glycolipids. These proximal core carbohydrate sequences define the class to which any given GSL belongs (Fig. 2).
The majority of more recent structures relate to the sialyl Lewis x blood group carbohydrate receptor function for the selectin family of eukaryote lectins that mediate leucocyte-endothelial cell interaction during the initial stages of extravasation (“neutrophil rolling”) (20). These structures are the sialyl Lea, Lex, and Ley sequences and several 6'sulfo GlcNAc and 3'sulfo Gal derivatives found in both protein and sphingolipid glycoconjugates (21-24).

Figure 2. Structure of the major core carbohydrate sequences of neutral GSLs.
Glycosphingolipid biosynthesis
Neutral GSLs
Greater than 90% of the GSLs are based on the transfer of glucose to ceramide to give glucosyl ceramide. Dihydroceramide also can serve as a substrate (25). Glucosyl ceramide then is extended by galactosyl transferase to give lactosyl ceramide. Lactosyl ceramide is the precursor of all the major GSL series. Unlike all other glycosyl transferases involved in GSL biosynthesis, glucosyl ceramide synthesis is a cytosolic enzyme (26-28). Except for this initial enzyme reaction, glycolipid biosynthesis occurs within lumen of the Golgi (29). Glucosyl ceramide synthase transfers glucose from UDP-glucose to ceramides to form glucosyl ceramide on the outer cytosolic surface of the Golgi vesicles. Glucosyl ceramide needs to flip into the Golgi lumen to provide the precursor for the majority of GSL biosynthesis. This translocation is achieved, at least in the majority of cells, by the ABC transporter, MDR1. MDR1 (or P-glycoprotein) was discovered first because of its ability to mediate cytotoxic drug efflux in drug-resistant cells. The upregulation of Pgp has been shown subsequently to be a major mechanism for drug resistance in cancer cells (30), and MDR1 inhibitors commonly are used therapeutically (31).
MDR1 is a member of a family of ATP-dependent drug efflux pumps (32). Several of these pumps, including MDR1, have been shown to mediate lipid translocation and phospholipid bilayers (33). This translocation activity has been implicated in part as the mechanism for drug efflux by which the hydrophobic cytotoxic drug is translocated from the cytosolic to the external leaflet of the plasma membrane bilayer. MDR1 first was shown to translocate glucosyl ceramide analogs from the cytosolic to the external leaflet of the plasma membrane (33). However, the major glucosyl ceramide translocation activity of MDR1 later was shown to occur in Golgi vesicles (34). The transfection of cells with MDR1 resulted in a major increase in glycolipid biosynthesis that was prevented by MDR1 inhibition (35). In a cell-free microsomal system, exogenous glucosyl ceramide was incorporated into more complex GSLs in an MDR1-dependent manner (34). In most cells, MDR1 inhibition prevents neutral, but not acidic, GSL synthesis (34), which suggests that the lactosyl ceramide first made from the pool of glucosyl ceramide translocated into the Golgi lumen by MDR1 is unavailable for immediate ganglioside synthesis. The inhibition of MDR1 therefore represents a means to regulate selectively the neutral GSL biosynthesis (36). This regulation, however, raises a problem for the synthesis of gangliosides. Some GlcCer has been shown to be translocated directly to the cell surface without the involvement of transGolgi traffic (37, 38). Recently, LacCer and GlcCer generated by cell-surface sialidase-mediated plasma membrane GM3 turnover was found to be incorporated exclusively into gangliosides (39). Thus, potentially, gangliosides could be made from cell-surface GlcCer, derived either by direct transit from the cytosolic Golgi surface or by degradation of other plasma membrane GSLs at the cell surface (or lysosomes?).
Lactosyl ceramide synthase is a β1-4 galactosyl transferase (40). Lactosyl ceramide is the only glycoconjugate that contains lactose (as opposed to lactosamine). Lactosyl ceramide is the substrate for several glycosyl transferases that then define a core sequence of glycosphingolipids. The Globo series is defined by an α1-4 galactosyl transferase activity (41, 42) to make initially globotriaosyl ceramide [also defined as the pK antigen, see below, and CD77 (43)] and is the substrate for Gb4 synthase (a β1-3GalNac transferase) (44). Gb4 is the major neutral glycolipid of most cells including human red blood cells. Gb4 [also termed the P antigen (45)] is the precursor of the Forssman antigen formed by the action of a α1-3GalNac transferase (46, 47). Ganglio series glycolipids are made by the action of a β1-3GalNac transferase (48) on lactosyl ceramide to give gangliotriaosyl ceramide, which then is the substrate of a β1-4Gal transferase to give gangliotetraosyl ceramide. Neolacto series GSLs are defined by the action of β1-3GlcNAc transferase (49) and β1-4Gal transferase (type 2 chain); Lacto series are defined by the action of β1-3GlcNAc transferase (50) and β1-3Gal transferase (type 1 chain) (51).
Gangliosides
Gangliosides are the major acidic GSLs and are derived primarily from the α2-3 sialylation of lactosyl ceramide to give the simplest monosialylated GSL, GM3 ganglioside (52). The sialic acid of GM3 can be α2-8 sialylated further to give GD3 (53), which can be sialylated further to form GT3. GM3, GD3, and GT3 all can serve as substrates (in addition to lactosyl ceramide) for the ganglioseries of glycosyl transferases to form the “a”, “b”, and “c” series of gangliosides, respectively (Fig. 3).
The sialic acid (N-acetylneuraminic acid) of gangliosides and sialoglycoproteins can be N-glycolyl or N-acetyl, and particularly in the context of the lectins that bind such glycoconjugates, this reality is of evolutionary significance (54). Many modifications of sialic acid (primarily O-acetylation at the 7,8,9 positions) have been described (55) that are primarily α2-3 or α2-6 linked to galactose within sialoconjugates. The total synthesis of the most common gangliosides has been described (56-59). Gangliosides are abundant in neuronal tissue, and mouse knockout studies indicate a stabilizing function in axons in white matter (60). Gangliosides provide the receptors for myelin-associated glycoprotein (MAG) inhibition of neurite outgrowth (61-63), and their mimics may be of therapeutic value in neuronal injury (64).
Gangliosides within lipid rafts also can modulate growth factor receptor- [and src kinase- (65)] mediated tyrosine phosphorylation signaling cascades (66). This modulation can be a carbohydrate—carbohydrate-based interaction (67). Although the molecular basis of such flexible sugar:sugar binding is not known, a series of selective interactions have been defined (68) within the context of a “glycosignaling domain” (69, 70) or a “glycosynapse” (71). GM3 inhibits phosphorylation in such domains, but deacetylation of the sialic acid results in the promotion of signaling (72), which indicates the central importance of the sialic acid. Indeed, sialyl-sphingosine was found to be the minimum structure to block signaling (73). Ganglioside signal modification plays an important role in distinguishing motile metastic tumor cells from cessile “normal” cells (74, 75).

Figure 3. Structure of the major acidic GSLs (gangliosides). Lactosylceramide, GM3, GD3, and GT3 are substrates for the same series of glycosyl transferases reactions, namely, GalNac transferase, galactosyl transferase II, and sialyl transferase V.
Galactosyl ceramide-based GSLs
A few GSLs are based on galactosyl ceramide rather than glucosyl ceramide. Galactosyl ceramide synthase, unlike glucosyl ceramide synthase is found within the ER lumen (76). This enzyme is regulated by an unusual enhancing activity (77).
Sulfatide (3'sulfogalactosyl ceramide or cerebroside sulfate) is the most important galactosyl ceramide-based GSL. Galactosyl ceramide and sulfatide are found primarily in neuronal tissue, and sulfatide synthesis is a key component in the myelination of the oligodendrocyte (78, 79). The galactosyl ceramide synthase knockout mouse resulted in no gross abnormalities in mouse development. However, tremor hind limb paralysis and long-term neurological effects were found in that myelin compaction was compromised (80, 81). Deletion of the galactosyl cerebroside sulfotransferase similarly resulted in enhanced terminal oligodendrocyte differentiation (82). Sulfogalactoglycerolipid (seminolipid), a product of the same sulfotransferase, also is a key component in mammalian spermatogenesis (83), and these knockout mice were found to be male infertile (84). Sulfatide also is expressed highly in the kidney, but the sulfotransferase knockout mice were without overt renal phenotype.
Inhibitors of glycosphingolipid biosynthesis
L-cycloserine is an inhibitor of serine palmitoyl transferase (85), the first committed step in GSL biosynthesis. However, because this inhibitor prevents the synthesis of the highly bioactive ceramide, an interpretation of GSL depletion is equivocal.
Fumonisin B1 (Fig. 4) is a fungal inhibitor of ceramide synthase because of structural similarity to sphinganine (86). This inhibition also depletes ceramide levels, however. Moreover, the identified fatty acid selective ceramide synthases have proven to be fumonisin resistant (10, 11). From the structure, it is evident that fatty acid chain length-specific ceramide synthases might have a binding site that is too restricted to accommodate the branched terminus of this inhibitor. Sensitivity to fumonisin also is dependent on the nature of the sphingoid base because acylation of sphingosine rather than sphinganine in vitro is resistant to fumonisin (87), which is consistent with the saturated alkyl chain of this amino alcohol.
The natural form of ceramide is the D-erythro isomer, and this form is the substrate for glucosyl ceramide synthase, which forms the precursor to the majority of GSLs. The imino glucopyranose analog, butyl deoxynijirimycin (DNJ), is a potent inhibitor of glucosyl ceramide synthase and thereby prevents the synthesis of the majority of GSLs. This inhibitor has been used as a substrate reduction approach to several GSL storage diseases (88, 89). However, DNJ also inhibits glucosidases and thereby affects intracellular glycoprotein carbohydrate processing (90) and glucose storage (91); therefore, more selective inhibitors have been developed. An inhibitor of glucosyl ceramide synthase was designed to mimic the product of this reaction structurally. 1-Phenyl-2-decanoylamino-3-morpholino-1-propanol (PDMP) (Fig. 5) is membrane permeable and has been used extensively to examine the role of glucose ceramide-based GSLs in a variety of physiologic processes (92). However, PDMP was found to have additional inhibitory activities (93), for example, on other enzymes for which ceramide is a substrate such as ceramide acyl transferase. Several generations of derivatives of PDMP have been designed subsequently with improved specific activity for glucosyl ceramide synthase. The first was to lengthen the hydrocarbon chain from 10 to 16 carbons (94) (PPMP) and then to replace the morpholino ring with a pyrrolidino ring (P4) (95). Later substitutions within the phenyl ring were examined (96) and the 4'hydroxy derivative was found to be the most potent inhibitor (IC50 for the GC synthase is 90nM).
Although the use of these inhibitors has been highly informative, the functional aspects of GSLs in plasma membranes have been difficult to define. Changes in growth status, differentiation, and malignancy are associated with altered GSL profiles (97). Ganglioside modification of growth factor receptor kinase activity (a coreceptor function) may relate to these changes (98). This difficulty in defining the functional aspects of GSLs in plasma membranes is largely because of the lack of methodologies for varying the expression of any specific GSL.
The overall importance of GSLs to cell physiology has been demonstrated clearly by using molecular biology techniques. Many glycosyl transferases involved in their synthesis have been cloned. As indicated above, the glucosyl ceramide synthase is the first committed step in the synthesis of most GSLs. This enzyme has been knocked out in mice and shown to be embryonic lethal (99). Interestingly, cultured cells survive well without this enzyme (100), which indicates that GSLs are required for the more differentiated functions in embryogenesis. Other enzymes involved in ganglioside biosynthesis, for example, GM3 synthase and GM2 synthase (60), have provided a more selective approach to reduce ganglioside biosynthesis, and knockout mice that lack these enzymes have been shown to have a less severe phenotype, primarily confined to CNS defects in long-term neuronal cell function.
Knocking out the galactosyl ceramide synthase gene in mice results in the formation of dysfunctional and unstable myelin (78). A similar phenotype was observed for the 3'sulfotransferase knockout mouse, which indicates that SGC is the major species responsible for these defects (84). Several previous studies had shown the importance of SGC and its glycerol-based analog, sulfogalactoglycerolipid in spermatogenesis (83) and fertilization (101). Similarly, the sulfation of galactosyl ceramide in the brain is a function of myelination (102). The calcium-mediated interaction between galactosyl ceramide and SGC demonstrated to occur in vitro (103) is implicated strongly in myelin compaction. Ganglioside binding to myelin-associated glycoprotein (MAG) has been shown to be responsible for some negative signals that prevent neuronal cell regeneration (61).
GSL carbohydrate binding complementarity has been proposed as a mechanism for signal transduction, primarily by Hakomori (71), who has shown the binding interaction between GM3 ganglioside and gangliotriaosyl ceramide/lactosyl ceramide, SSEA3 and Gb4. These in vitro binding selectivities correlate with cellular adhesion between cells that express high levels of these GSLs (68). Nevertheless, the mechanism by which the flexible GSL carbohydrate chain can interact specifically with another flexible carbohydrate chain in these GSLs is unknown. The Hakomori group also has shown that gangliosides can modulate the growth factor receptor function and has proposed a cis-interaction between the receptor and the ganglioside within the plane of the plasma membrane (75). Similar lateral interactions between receptors and GSLs have been proposed for the α2-interferon receptor (104, 105) and CD19 (106) with globotriaosyl ceramide. Both these transmembrane protein receptors contain an extracellular N-terminal sequence with similarity to the sequence within the receptor binding domain of the verotoxin (Shiga toxin) B subunit (107). Interestingly, α2-interferon receptor signaling has been compared with that of γ-interferon. Both these receptors are internalized by clathrin-coated pits, but only α2-interferon signaling is prevented by the inhibition of internalization (108).
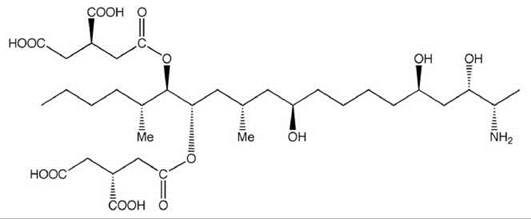
Figure 4. Structure of Fumonisin B1.

Figure 5. Structure of chemical inhibitors of glucosyl inhibitors of glucosyl ceramide synthase: PDMP and hydroxy P4.
The concept of cellular lipid rafts was introduced first by Simons (1, 109) to explain differential GSL trafficking in polarized epithelial cells. These domains are cholesterol and GSL enriched. The H -bond donor and acceptor capability of sphingolipids result in a close, rigid cholesterol complex—a lipid-ordered domain—as compared with the liquid crystalline, predominantly glycerolipid bilayer. In addition, membrane proteins anchored through glycophosphoinositol linkages preferentially accumulate in these domains. Palmitoylated or myristylated cytosolic enzymes involved in downstream signal transduction pathways [e.g., src family kinases (110, 111)] accumulate in the corresponding cytosolic domains of such cell surface lipid rafts.
Lipid rafts serve as foci for a variety of signal transduction pathways (112) and also as access points for many pathogenic microorganisms (113). Direct GSL-mediated signaling within such rafts has been documented by using bacterial toxins that specifically recognize GSLs within these domains (114, 115). Indeed, cholera toxin binding to its receptor GSL, GM1 ganglio- side, is the gold standard marker for lipid rafts (116) in virtually all studies. However, lipid rafts as monitored by CT binding can be excluded from clathrin-coated pits (117). Moreover, recent studies have shown that the binding of cholera toxin does not correlate always with GM1 content (118) but rather fucosylated GM1 to which cholera toxin also binds (119). This finding certainly will require a reevaluation of the basis of the association of cholera toxin with lipid rafts.
Phase separation in model lipid membranes is established clearly (120), but the significance of lipid rafts in cell membranes has been controversial (121) largely because of their isolation by harsh conditions of detergent resistance. More recently, methods have been developed to visualize lipid rafts in living cells (122, 123) and to identify proteins within them by less invasive methods (124).
The functional role of GSLs within rafts is speculative but may involve linkage to cytosolic, raft-associated signal transduction cascades such as src family kinases (125). In this regard, we have demonstrated recently the structural similarity of 3'sulfogalactose, found in GSLs, and tyrosine phosphate (126). This structural similarity is represented in Fig. 6. Ligands that bound tyrosine phosphate and/or tyrosine sulfate (including SH2 domains from src) were found also to bind 3'sulfogalactose GSLs. Furthermore, the binding of such ligands to tyrosine sulfate lipid conjugates was found to depend on the lipid structure, a property typical of GSL recognition. We interpret this finding to indicate that the recognition of GSL carbohydrate within the context of the cell membrane and the recognition of the selective ligand binding of specific tyrosine phosphate groups in the context of a specific polypeptide background may share common principles. In modeling studies, we showed that a galactose hydroxyl group could replace a bound water molecule in tyrosine phosphate-ligand complexes, as determined by crystallography. As such, substitute binding of the carbohydrate could provide an entropic advantage such that carbohydrate-based inhibitors of specific tyrosine phosphatases/kinases might be feasible.

Figure 6. Superimposition of the minimum energy conformation of 3'sulfogalactose (dark gray) on that of tyrosine phosphate (light gray). Partial charges for the sulfate and phosphate oxygens are shown (taken from Reference 126). (a) From above and (b) side view.
Chemical Modification of Glycosphingolipids
Studies on the chemical modification of GSLs are not extensive. The sialic acid of gangliosides is liable to selective oxidation, for example, using periodate, and this has been used to couple gangliosides to various moieties (127). In addition, procedures were developed for the selective oxidation of cell surface sialoconjugates by periodate at a low temperature (128). Cell surface carbohydrate modification can be achieved also via the metabolic route by culturing cells with unnatural per-acetylated sugar analogs that become incorporated into growing glycoconjugate carbohydrate chains, for example, azido sialic acid (129), to allow the probing of lectin binding specificity. However, chemical modification of the carbohydrate moiety of GSLs to generate higher affinity ligand receptors or antagonists is largely an untapped area. Amino substitution within the globotriaose moiety of Gb3 by coupling azido monosaccharides during trisaccharide assembly (130) was found to reduce, rather than enhance, VT1 binding (131).
Terminal N-acetyl hexosamine
We described a procedure for the selective removal of terminal N-acetyl hexosamines from GSLs (132). In this procedure, the N-acetyl group first is deacetylated selectively with an aqueous base. Then, in the presence of H2O2, the amino function at the 2 position of the terminal amino sugar undergoes a free radical rearrangement reaction that results in the loss of the terminal sugar and some additional peeling reaction, with the likely release of NO. Internal N-acetyl hexosamine residues are unaffected. We propose a mechanism in Fig. 7. Peroxide oxidation of the amine to a nitroso group (133) results in the electronic reconfiguration to cleave the glycosidic bond and liberate NO. Although free amino sugars are rare, they might occur in lysosomes in the context of peroxide radicals, which might make this reaction a biologic source of NO.

Figure 7. H2O2-mediated, nonreducing terminal hexosamine cleavage. Terminal amino sugar loss is the major product (but some peeling of the n-1, n-2 sugar is seen), possibly via the rearrangement of a nitroso oxidation product to liberate NO and generate a free radical that rearranges to induce the partial loss of the next sugar, and so forth.
Sphingosine double bond
Oxidative cleavage of the sphingosine double bond by ozonolysis was reported some 30 years ago (134). However, this reaction could be carried out only in large scale, and yields of the truncated GSL were very poor and required an ozone generator. This procedure was carried out with globoside and hematoside, and the equivalent glycoceramidic acid was characterized as the truncated product.
We developed a new, more versatile procedure for the oxidative cleavage of the double bond of the sphingosine of glycolipids by using a more controllable potassium perman-ganate/potassium iodate oxidation system. This procedure allowed the oxidation of GSLs at the microgram level and under neutral conditions and generated the ceramidic acid in quantitative yield. This occurrence allowed a subsequent coupling to various inert supports (135). This coupling route has been used to conjugate glycolipids to protein carriers for generating GSL immunogens as potential anticancer therapies. However, under basic conditions this oxidation resulted in greater truncation of the sphingoid base to give the glycosyl serine acid rather than the ceramidic acid produced under neutral conditions (136). A hydroxy-acyl intermediate was identified, the oxidation of which was pH dependent. Unsaturated fatty acids also were cleaved such that the oxidation procedure could be used to define the aglycone composition of natural GSLs. The mass spectrometry analysis of the oxidative cleavage of glucosyl ceramide is shown in Table 1.
Table 1. Mass spectroscopy of oxidized glucosyl ceramide that shows that the oxidation procedure allows the quantitation of a degree of unsaturation within the fatty acid moiety (taken from Reference 136)
|
MS Comparison of GkC(OAc)5 and the (Neutral) Oxidized Material GlcC(OAc)5∙CCOOHa |
|||||||||||
|
|
before |
after |
hvdroxv acyl |
ceramide cionoacids |
ceramide diacids |
carboxy acvl |
|||||
|
glycan |
oxidation |
oxidation |
intermediate |
M + Na, M - H + 2Na, etc. |
M + Na, M – H + 2Na |
M + Na, M - H + 2Na |
|||||
|
Glc16C |
933.0 (100) |
|
965.0 (45) |
768.6 (96), 790.8 (100) |
|
||||||
|
Gk18C |
961.0 (37) |
|
993.0 (21) |
796.6 (50) |
|
||||||
|
Glc20C |
939 (28) |
|
1021.0 (23) |
824.3 (36), 846.6 (36) 868.8 (23) |
|
||||||
|
Gk22C |
1017.2 (66)1 |
|
1049.0 (53)5 |
852.8 (92), 874.8 (72) |
|
||||||
|
Glc24C |
1045 (<50)2 |
|
1077.0 (53)6 |
880.8 (81), 902.8 (75) |
|
||||||
|
Glc22:1 |
1015.2 (20) |
|
1047.0 (46)3 1079.0 (44) |
}C-15: 784.6 (98) 806.6 (89) |
|
||||||
|
Glc24:1C |
1043.2 (78) |
|
1075.2 (34)4 1107.2 (33) |
|
|||||||
|
Glc16+C |
|
935.0 (53) |
|
|
|
||||||
|
Glc18+C |
|
963.0 (39) |
|
|
|
||||||
|
Glc20+C |
|
991.0 (21) |
|
|
|
||||||
|
Glc22+C |
|
1019.0 (29) |
|
|
|
||||||
|
Glc24+C |
|
1047.2 (37)3 |
|
|
|
||||||
|
Glc26+C Glc22:1+C |
1017.2 (66)1 |
1075.2 (37)4 |
1049.0 (5S)5 |
|
}C-12: 903.8 (24) 930.8(24) |
||||||
|
Glc24:1+C |
1045.2 (50)2 |
|
1077.2 (53f |
|
|||||||
aRelative intensities are given in parentheses, and isobaric masses are indicated by superscript numbers. Ceramide monoacid, ceramide diacid, and carboxyacyl refer to GlcC(OAc)5∙CCOOH, GlcC(OAc)5∙CCOOHnCOOH, and Clc*C(OAc)5∙nCOOH. respectively.
Ganglioside coupling reactions
Procedures for the selective deacylation of the ceramide moiety of gangliosides (137) have been used as routes to form ganglioside conjugates, primarily for immunogen development. Thio-linked sialic acid has been used to reduce immunogen degradation (138). Gangliosides are shed by tumor cells and antibodies to such GSLs are considered of potential therapeutic benefit, and these have been raised via ganglioside-protein conjugate vaccines (139, 140).
The increased sensitivity of the sialic acid glycerol moiety to periodate oxidation as compared with other sugars has been used as a coupling method to label gangliosides selectively (141).
The tolerance of the metabolic pathway for the synthesis of CMP-sialic acid and the subsequent sialyl transferases to N-substitution of the amino sugar precursor permits the innovative use of N-acetyl mannosamine analogs as metabolic precursors of cellular sialoproteins (129), which has enabled the novel selective coupling of these glycoconjugates. This approach recently has been extended to gangliosides (142) such that cell surface azidosialo GSLs, which can be labeled via Staudinger ligation to generate a free amine (deacetyl neuraminic acid) under mild conditions, are generated more efficiently than azi- dosialo proteins. Presumably, the metabolism of amino sugar precursors for GalNAc/GlcNAc glycoconjugate synthesis are not so tolerant.
GSL fatty acid substitutions
Many studies have substituted the fatty acid moieties of GSLs with various chromaphores to study GSL cellular trafficking and ligand binding properties. Nitrobenzoxadiazol (NBD) has been a popular choice (143). Early studies use the direct coupling of NBD via N-hydroxy succinimidyl ester condensation with the sphingosine amine of lyso-sphingolipids. NBD with various fatty acid chain lengths were coupled, but C6 was most useful in terms of cell uptake. C6 NDB ceramide was used as a fluorescent probe of retrograde transport from the cell surface to the Golgi apparatus and metabolized to NBD-GSLs (144). Procedures were developed later to couple the fluor within the GSL lipid moiety selectively. With neutral GSL, this coupling is achieved easily by base hydrolysis (145). For gangliosides, selective cleavage of the base labile ceramide was required first (137).
These studies were superceded largely by the use of boron dipyrromethene difluoride (BODIPY), which has the useful property of showing a differential fluorescence emission spectrum according to concentration (146). The BODIPY moiety could be coupled with a variety of hydrocarbon spacer groups, and as with NBD the 5-carbon fatty acid analog has been used frequently. BODIPY analogs of gal-cer, lac-cer sulfatide, GM1 ceramide, and sphingomyelin have been synthesized. The structure of examples of NBD- and BODIPY-labeled GSLs is shown in Fig. 8. However, these fluorophore substitutions considerably alter the lipid characteristics of these analogs such that their trafficking and partition into membranes does not mimic necessarily that of the natural parental GSL because GSL intracellular trafficking can be affected markedly by the lipid moiety within the membrane (9, 148). This effect is illustrated by the fact that these BODIPY- or NBD-labeled GSLs, when incorporated into cell membranes, can be back-extracted readily by the treatment of the cells with BSA. In addition, whereas BODIPY-labeled Gb3 was internalized by a clathrin-independent mechanism in cultured cells, Shiga toxin internalization via endogenous Gb3 was clathrin dependent in the same cells (149).
Nevertheless, the use of fluorescent GSL analog trafficking within cells to define aberrant intracellular trafficking pathways in cells from lysosomal storage disease patients has proven valuable in establishing the link between cholesterol and GSL trafficking. Indeed, the aberrant trafficking of BODIPY-GSLs can be used as a diagnostic tool in these genetic diseases (150).
A recent alternative procedure for the fluorescent labeling of GSLs has been described; it attempts to reduce the impact of the fluor on the structure of the GSL lipid moiety (147). A strong alkaline hydrolysis of gangliosdes was reported to cleave the ceramide fatty acid and deacetylate the sialic acid group without affecting the N-acetyl galactosamine residue. (However, in our hands we find that the strong base simultaneously deacetylates N-acetyl amino sugars while deacylating the ceramide moiety of neutral GSLs; see Fig. 9.) The protection of the sphingosine amine with fmoc then can allow the selective reacylation of the sialic acid with acetic anhydride. The removal of the fmoc group with piperidine in DMF allows the selective coupling of the lyso-ganglioside to azido octodecanoate. The selective reduction of the azido group without reducing the sphingosine double bond was achieved by using hydrogen sulfide. NBD fluoride then was coupled to this 2-amino function within the lipid moiety of the ganglioside (Fig. 8). In previous coupling procedures for the generation of fluorescent glycosphingolipids, the NBD has been coupled directly to the sphingosine amine. In this procedure, the advantage is that the lipid moiety of the fluorescent ganglioside is more similar to that of the native species. Because many receptor functions of GSLs are modulated by the lipid moieties, this approach provides a mechanism by which such effects will be retained, at least in part, within the deriva- tized GSL. This conversion is supported by the effect that the GM2 activator was shown to bind the fluorescent GM2 analog as effectively as the natural GM2 ganglioside. The disturbing effect of such chromaphore conjugations within the lipid moiety has been reduced more by the use of polyene substitution within fatty acids and coupling to sphingosine (151).

Figure 8. Comparison of NDB- and BODIPY-labeled lyso glucosyl ceramide and NBD-labeled amino acyl glucosyl ceramide (147).
Radiolabeling of GSL
GSL radiolabeling is achieved commonly by the catalytic reduction of the sphingosine double bond using palladium and sodium borotritide (152), although this, of course, generates the dihydro species that may behave differently from the native ceramide-containing glycosphingolipid. Radiolabeling with galactose oxidase is a feasible alternative that produces an aldehyde at the C6 position of terminal galactose residues, which can be reduced again with sodium borotritide (153, 154). This procedure can be adapted to label only cell surface GSLs (155). Interestingly, this reaction was found to fail when the glycolipid had a glycerol, as opposed to a sphingosine, backbone (156), which indicates the that availability of the C6 position of galactose to the enzyme is restricted in the case of the glycoglycerol species.
Surprisingly, a good chemical method for radiolabeling all glycosphingolipids has yet to be devised. The deacylation of the sphingosine under basic conditions and the reacylation of generated amino function with radiolabeled fatty acid is a feasible method, although this method does eliminate the fatty acid heterogeneity of the native species. However, this approach is problematic when it comes to gangliosides because the sialic acid also is base labile. Similarly, in amino sugar-containing GSLs, basic hydrolysis will result in the deacetylation of the N-acetyl amino sugar, which thereby precludes the subsequent selective acylation of the sphingosine amine. We have overcome this problem in terms of amino sugar-containing GSLs by developing a procedure for the selective reacylation of either the sphingosine amine or the galactose (glucose) amino function (Fig. 9).

Figure 9. Selective ceramide deacylation of N-acetylhexosamine that contains GSLs. Gangliotriaosyl ceramide is shown as example. In organic solvent, 1 N base hydrolysis results in the cleavage of both amides, but acetylation in aqueous buffer only affects the amino sugar.
Chemical Glycosphingolipid Synthesis
The carbohydrate moieties of GSLs have been synthesized, but these procedures are essentially no different from the chemical synthesis of oligosaccharides found on glycoproteins (157). The major difference is the coupling of the oligosaccharide to the aglycone. Complex GSLs have been synthesized by the assembly of appropriately protected oligosaccharide donors and the coupling to a ceramide acceptor and deprotection under mild conditions, for examples, see References 158-160. These approaches are restricted by the complexity of the protection chemistry, intermediate purification, and low product yields. Combined enzymatic and chemical syntheses can address these problems. A recent innovative approach has been to reverse the hydrolytic activity of the endoglycoceramidase that normally removes the carbohydrate from GSLs, to generate a novel glycosynthase (161). In this approach, the catalytic nucleophile residue of the hydrolase is mutated to an inactive amino acid, and the reverse reaction with the appropriate fluoride sugar donor then becomes readily apparent. This process removes problems with protecting group chemistry and ensures the correct stereochemistry of the adduct.
Thioglucosyl ceramide and thiolactosyl ceramide also have been synthesized to study cellular glycolipid traffic and metabolism (162). Both βthiolactosyl ceramide and lactosyl βthioceramide were made. The thioglycocidic link is resistant to glycosidase degradation to ceramide, and these analogs therefore can be used as precursors to monitor cellular GSL anabolism alone.
The pthiolactosyl ceramide was made by coupling a protected thio-D-galactose to the C-4 of a protected galactosyl sphingosine. The invertion of configuration during the SN2 reaction generated the correct pthiolactosyl ceramide. Lactosyl Pthioceramide and glucosyl pthioceramide were made by condensation of the protected thiourea glucoside or the thioacetyl lactoside donor with a protected iodosphingosine.
The synthesis of glycolipid analogs suitable for microarrays is an important component for defining carbohydrate-binding phenotypes in “glycomics.” These conjugation procedures that allow the solid-phase presentation of the glycolipid carbohydrate moiety are most physiologic when as much of the intact ceramide moiety of the parent GSL as possible is maintained. Serine-based GSL analogs, at least in part, accomplish this. This has been achieved by the use of fluoro hydroxy methophenoxyl acidic acid as the linker to conjugate to microtitre plates (163).
A new procedure for GSL synthesis via olefin cross metathesis (164) is highly versatile in terms of the hydrophobic aglycone. A protected 5 carbon amino alkene diol is the central building block to which the protected carbohydrate donor, long chain fatty acid, or, by olefin cross metathesis, the long alkenyl chain of the base can be coupled, in a variety of sequences. This atypical synthetic flexibility should allow a structural approach to dissecting the role of the lipid moiety in GSL receptor function and intracellular trafficking.
Glycosphingolipid mimics
GSLs often serve a receptor function in microbial pathogenesis. Therefore, one therapeutic approach to such infections is the development of soluble inhibitors of this interaction, and such inhibitors are for the most part based on the generation of high-affinity receptor mimics. Ligands that bind GSL sugars that are distal to the aglycone are less likely to be affected by the modification or removal of the lipid moiety. Cholera toxin binding to GM1 ganglioside is affected marginally by the nature of the lipid moiety (165). This occurrence has allowed the development of GM1 mimics in which the ceramide is absent. CT binds to a conformer that is similar to the global energy minimum of the free GM1 oligosaccharide (166), and the sugar has been paired away such that the sialic acid is reduced to lactic acid (167) linked to an appropriate conformationally restricted 1,2 dicarboxy-4,5-cyclohexanediol (168) that serves as a simplified mimic of the 3,4 substituted core galactose. This has been multimerized by using a tetravalent scaffold to improve the affinity for the multivalent toxin B subunit pentamer (169).
Although the dendritic presentation of carbohydrates for glycoprotein binding lectins has been well established (170, 171), the potential multivalency of GSLs within the plasma membrane bilayer is far greater than for glycoproteins, and thus the chemical solution to the generation of synthetic oligosaccharide multimers that retain a binding affinity of the order of the natural membrane GSL has been particularly difficult. In terms of the Escherichia coli-derived verotoxin (also termed Shiga toxin), which is responsible for the development of hemolytic uremic syndrome primarily in children (172), the importance of the lipid moiety of its GSL receptor, globotriaosyl ceramide (Gb3), is clearly apparent (173). Moreover, the pentameric receptor binding B subunit indicates at least five binding sites, which are suggested in the original crystal structure of the VT1 B subunit to be within the intersubunit cleft (174). Modeling studies supported this conclusion (175). However, subsequent cocrystallographic studies with the Gb3 oligosaccharide indicated as many as three binding thermodynamic minima/monomer (15 in total) (176), which makes the design of inhibitors difficult. The interplay of these potential receptor sites still remains uncertain (177).
Although the binding affinity of the lipid-free Gb3 oligosaccharide was of the order of 5 log weaker than that of the intact GSL (178), this affinity was considered an avidity problem, which could be solved by the design of an appropriate dendrimer presentation. The first, and probably the most elegant, approach was the synthesis of a pentamer of globotriaose dimers. This decavalent pentamer was based on coupling a globotriaose dimer to a central glucose moiety (179). The intersubunit cleft site was not the major binding site but rather a shallow trough on the surface opposite the bound membrane of each subunit monomer. This surface binding site, site 2, was targeted for occupancy by the pentameric globotriaose array. This “starfish” array proved an effective neutralizing ligand for VT1 but less so for VT2 in terms of both binding inhibition and prevention of cell cytotoxicity in vitro. Subsequent modifications of this pentameric array such that the separation between the globotriaose diamers was increased to accommodate potentially the distance between site 2 and the inter subunit cleft site (site 1) improved efficacy, and this was attributed to dual binding site occupancy (180); however, this was not seen in cocrystals. Rather, each globotriose within each dimer occupied equivalent sites in adjacent B subunit pentamers—a pentameric sugar sandwich—and crosslinked adjacent B subunit pentamers to inhibit cytotoxicity. Subsequent studies found that a trimer, rather than the pentamer, of diamers was a more effective ligand substitute for VT2 (181). The crystal structure of VT2 (182) has shown that the C-terminal extension of the A subunit that penetrates the B subunit pentamer would obstruct receptor binding in the site 3 equivalent of this toxin. The loop that contains the equivalent of site 2 is in a different conformation that also would not allow Gb3 carbohydrate binding. Thus, VT2 may show receptor binding in site 1 only.
The other procedure described for the generation of multivalent globotriaose dendrimers for protection against VT1 or VT2 cytotoxicity in vivo is based on carbosilane multimers (183). These multimers are more systematic expansions of trimeric structures that are based on multiples of the tetrameric carbosilane (184). In these studies, coupling the two terminal silicon residues of a linear carbosilane trimer (“super twig”) to either 3 or 9 globotriaose oligosaccharide units (9 requires the termini to be substituted with three additional carbosilane units) proved the most effective in neutralizing verotoxin in mice. Site-specific mutational studies on the B subunits of VT1 and VT2 showed that sites 1 and 2 were required for “super twig” VT1 binding but that site 3 was central for VT2 binding (184). However, this finding does not reconcile with the VT2 crystal structure that shows that site 3 is inaccessible (182). It is significant that whereas these more densely displayed globotriaose dendrimers proved more effective than the “rational designed” pentameric displays discussed above, both of these synthetic dendrimeric presentations proved less effective than the random coupling of globotriaose to a polyacrylamide support (185), which indicates that a considerable gap still exists in relating the structure of the Gb3 oligosaccharide-verotoxin B subunit complex to the Gb3 GSL binding on target cells (177).
The other major GSL receptor function is that of GM1 ganglioside for cholera toxin. In contrast to the verotoxin-Gb3 system, no attempts to generate GM1 receptor mimics to prevent cholera toxin binding have been reported. Although the lipid moiety of GM1 ganglioside plays a less significant role in cholera toxin binding than that which Gb3 plays in VT1 recognition, a chemical substitution of the lipid moiety of GM1 ganglioside has been shown to modify cholera toxin cytotoxicity (165). Substitution of the ceramide moiety for cholesterol, aliphatic amines, or aminophospholipids has been shown to modify the cholera toxin response. The increased aliphatic chain length increased efficacy. The lower functional limit (C12) is similar to the fatty acid chain length dependency for the verotoxin receptor GSL function (186). The GM1-cholesterol analog proved more effective than the native GM1, which may relate to the GM1 raft requirement for cholera toxin cytopathology (116), and implies differential intracellular trafficking of the toxin receptor mimic complexes. GM1 ganglioside is the gold standard marker of lipid raft assembly in plasma membranes (187). A mutation of the cholera toxin B subunit such that a nonraft ganglioside was bound preferentially resulted in the oblation of cholera toxin cytopathology (116, 188). Thus, although the lipid moiety may not be crucial for cholera toxin binding, it is crucial for plasma membrane receptor organization, which in turn defines toxin efficacy.
HIV envelope adhesin gp120 binding to several GSLs has been reported (189, 190), and GSL depletion prevents HIV infection (191). HIV binding to galactosyl ceramide is implicated in the infection of CD4 negative cells (192), which has spurred the synthesis of several analogs as a means to control this infection (193-195). The glucose ring of lactose was opened and coupled to aminoundecanoic sodium carboxylate. The amino group then was C16 acylated more. This soluble analog inhibited HIV infection and syncytium formation in vitro (193). Gal Cer analogs were made in which the sphingosine base was truncated via the construction of galactosyl “serinol” (196). This was not bound by gp120, showing the importance of the hydrocarbon chains. Although conjugates that contain a C9 sphinganine did not bind gp120 (196), these could inhibit gp120/GalCer binding according to aglycone structure. Hydroxylation at the C'2 position of the fatty acid (even for short chain fatty acids) promoted this inhibition (196). OH and increased hydrocarbon chain length correlated with the calculated head group conformation—at a right angle to the hydrocarbon axis, which indicates that this is the major aglycone effect. Water-soluble C-glycoside analogs of lyso-galactosyl ceramide were made (197) via C-glycosyl aldehyde condensation with Wittig reagent to form the trans double bond that was then reduced; the ring was opened and oxidized to form a protected C-glycosyl amino acid. This C-glycosyl amino acid then was coupled to aliphatic amines of increasing chain length. Gp120 binding of the C16 species was equivalent to lyso-galactosyl ceramide, and binding was hydrocarbon chain length-dependent.
The GSL carbohydrate binding specificity of gp120 in vitro is lax. The binding to N-stearyl-deoxygalactonojirimycin (198) prompted the synthesis of stearyl C-glycoside and aza-C-glycoside analogs of galactosyl ceramide (195). A common precursor, a C1 stearyl galactal, was constructed; it was hy- droborated to give the C-glycoside, or it was transformed to a diketone from which the aza-C-glycoside was derived by reductive amination. By Langmuir trough measurements, these stearyl glycosides showed equal or greater binding to gp120 than galactosyl ceramide. An assay of their efficacy against HIV infection will be of interest. In contrast to the O-glycosides, fatty acid-OH and head group conformation were not found important. This finding could relate to the -OH/anomeric oxygen H-bond in the O-glycosides that restricts the headgroup conformation (196). In the C-glycosides, rotation will be reduced, and this may not be necessary. In addition, a substitution of the anomeric oxygen will make the interface between the “sugar” and the hydrocarbon chain more hydrophobic, which will alter packing and thereby might reduce the effect of the hydrocarbon chain length seen for the O -glycoside. Galactosyl ceramide analogs with hydrophilic (ethylene glycol) spacer groups of increasing length between the sugar and the lipid chains have been constructed (199), but gp120 binding has yet to be reported.
Adamantyl GSLs
Our approach to the generation of Gb3-based therapeutics to prevent verotoxin cytopathology has been to optimize the monomeric unit for inhibition with a view to generating polymers, dendrimers, and so forth at a later date. We have considered that the lipid moieties of GSLs play more than a multimerization function in terms of their ligand binding within a plasma membrane, although it is clear that multimeric binding plays a significant role in enhancing binding avidity. The lipid-free GSL-derived carbohydrate may be unable to inhibit ligand GSL binding even if that ligand only has a single GSL binding site (200). We therefore consider that the lipid moiety plays a crucial role in the presentation of the carbohydrate in an appropriate manner for ligand binding (201). This may be in the context of lipid rafts because these can be considered less hydrated regions of the plasma membrane (122). It is certainly true that for ligand/carbohydrate binding, water molecules that solvate the sugar must be displaced, and it is possible that the solvation of the carbohydrate of raft-associated GSLs, particularly those sugar sequences adjacent to the ceramide moiety, might be restricted to facilitate ligand binding. We have replaced the fatty acid of several GSLs with rigid carbon frames such as adamantane (202). These structures are surprisingly water-soluble and yet, unlike the lipid-free carbohydrate, retain the receptor function of the native membrane-bound GSL. Our premise is that selection of appropriate chemical substitutions within these frames can promote ligand binding and indeed modulate intracellular trafficking pathways. Adamantyl Gb3 (Fig. 10) is an effective inhibitor of verotoxin/Gb3 binding (203) and can protect cells against VT cytopathology in culture. However, adamantyl Gb3 is not effective in protecting against VT2 cytopathology in the mouse model (204). Adamantyl Gb3 is, nevertheless, a highly effective ligand for the HIV envelope-protein gp120 (205) and prevents HIV infection in vitro, irrespective of strain or drug-resistant status (206), via inhibition of viral-host gp120-dependent cell fusion. Using a Langmuir trough, we established that the molecular area of adamantyl Gb3 is greater than that of native Gb3. Moreover, an adamantyl Gb3 monolayer was more rigid than that of Gb3. HIV gp120 interaction with Gb3 monolayers was slow and with sigmoidal kinetics, whereas that with adamantyl Gb3 monolayers was rapid and exponential (205). Gp120 interaction with Gb3 became exponential in the presence of cholesterol, which suggests that adamantyl Gb3 may mimic a Gb3:cholesterol complex.
Members of the hsp70 family of stress proteins bind the GSL 3'sulfogalactosyl ceramide (SGC) (207). The binding site is in the N-terminal ATPase domain (208). Adamantyl SGC, similarly generated via fatty acid replacement, similarly has proven water soluble and is an effective inhibitor of hsp70-SGC binding (200). Adamantyl SGC has been shown to inhibit hsp70 ATPase activity (209) in vitro and therefore may modulate its chaperone function in cells. Such an effect also has therapeutic potential (210).
The high water solubility of these adamantyl GSL derivatives was surprising considering that the adamantane frame is at least as hydrophobic as the fatty acid it replaced. It is possible, however, that the adamantane frame together with the sphingosine acyl chain may pack sufficiently to be below the hydrophobic size that is necessary to disturb the molecular organization of water (211). This situation may provide the basis of their aqueous solubility characteristics.
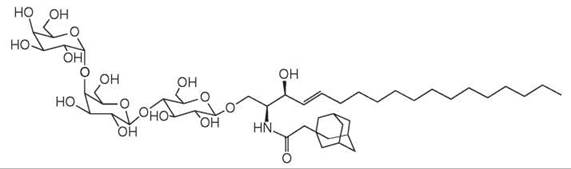
Figure 10. Adamantyl Gb3 is a soluble mimic of Gb3 GSL.
Studies from our laboratory have been supported by grants from CIHR and the Ontario HIV Treatment Network.
References
1. van Meer G, Simons K. Lipid polarity and sorting in epithelial cells. J. Cell Biochem. 1988; 36:51-58.
2. Rajendran L, Simons K. Lipid rafts and membrane dynamics. J. Cell Sci. 2005; 118:1099-1102.
3. Nyholm P-G, Pascher I. Orientation of the saccharide chains of glycolipids at the membrane surface: Conformational analysis of the glucose-ceramide and the glucose-glyceride linkages using molecular mechanics (MM3). Biochemistry 1993; 32:1225-1234.
4. Nyholm P-G, Samuelsson BE, Breimer M, Pascher I. Conformational analysis of blood group A-active glycosphingolipids using HSEA-calculations. The possible significance of the core oligosaccharide chain for the presentation and recognition of the A-determinant. J. Mol. Recog. 1989; 2:103-113.
5. Stromberg N, Nyholm P-G, Pascher I, Normark S. Saccharide orientation at the cell surface affects glycolipid receptor function. Proc. Natl. Acad. Sci. U.S.A. 1991; 88:9340-9344.
6. Perry RJ, Ridgway ND. Molecular mechanisms and regulation of ceramide transport. Biochim. Biophys. Acta 2005; 1734:220-234.
7. Weiss B, Stoffel W. Human and murine serine-palmitoyl-CoA transferase-cloning, expression and characterization of the key enzyme in sphingolipid synthesis. Eur. J. Biochem. 1997; 249:239-247.
8. Lingwood CA. Aglycone modulation of glycolipid receptor function. Glycoconj. J. 1996; 13:495-503.
9. Falguieres T, Mallard F, Baron C, Hanau D, Lingwood C, Goud B, Salamero J, Johannes L. Targeting of Shiga toxin b-subunit to retrograde transport route in association with detergent- resistant membranes. Mol. Biol. Cell 2001; 12:2453-2468.
10. Venkataraman K, Riebeling C, Bodennec J, Riezman H, Allegood JC, Sullards MC, Merrill AH, Jr., Futerman AH. Upstream of growth and differentiation factor 1 (uog1), a mammalian homolog of the yeast longevity assurance gene 1 (LAG1), regulates N-stearoyl-sphinganine (C18-(dihydro)ceramide) synthesis in a fumonisin B1-independent manner in mammalian cells. J. Biol. Chem. 2002; 277:35642-35649.
11. Mizutani Y, Kihara A, Igarashi Y. Mammalian Lass6 and its related family members regulate synthesis of specific ceramides. Biochem. J. 2005; 390:263-271.
12. Hannun YA, Obeid LM. The ceramide-centric universie of lipid-mediated cell regulation: stress encounters of the lipid kind. J. Biol. Chem. 2002; 277:25847-25850.
13. Joseph CK, Wright SD, Bornmann WG, Randolph JT, Kumar ER, Bittman R, Liu J, Kolesnick RN. Bacterial lipopolysac-charide has structural similarity to ceramide and stimulates ceramide-activated protein kinase in myeloid cells. J. Biol. Chem. 1994; 269:17606-17610.
14. Spiegel SM, S. Sphingosine 1-phosphate, a key cell signaling molecule. J. Biol. Chem. 2002; 277:25851-25854.
15. Yang J, Yu Y, Sun S, Duerksen-Hughes PJ. Ceramide and other sphingolipids in cellular responses. Cell Biochem. Biophys. 2004; 40:323-350.
16. Spiegel S, Milstien S. Functions of a new family of sphingosine-1-phosphate receptors. Biochim. Biophys. Acta 2000; 1484:107-116.
17. Funato K, Riezman H. Vesicular and nonvesicular transport of ceramide from ER to the Golgi apparatus in yeast. J. Cell Biol. 2001; 155:949-959.
18. Hayashi A, Nishimura Y, Matsubara T. Occurrence of ceramide digalactoside as the main glycosphingolipid in the marine sponge Halichondria japonica. Biochim. Biophys. Acta 1991; 1083:179-186.
19. Stults CLM, Sweeley CH, Macher BA. Glycosphingolipids; structure biological source and properties. Methods Enzymol. 1989; 179:167-214.
20. Sperandio M. Selectins and glycosyltransferases in leukocyte rolling in vivo. FEBS J. 2006; 273:4377-4389.
21. Sanders WJ, Katsumoto TR, Bertozzi CR, Rosen SD, Kiessling LL. L-selectin-carbohydrate interactions: relevant modifications of the Lewis x trisaccharide. Biochemistry 1996; 35:14862- 14867.
22. Kawashima H. Roles of sulfated glycans in lymphocyte homing. Biol. Pharm. Bull. 2006; 29:2343-2349.
23. Galustian C, Lubineau A, le Narvor C, Kiso M, Brown G, Feizi T. L-selectin interactions with novel mono- and multisul-fated Lewisx sequences in comparison with the potent ligand 3'-sulfated Lewisa. J. Biol. Chem. 1999; 274:18213-18217.
24. Lowe JB. Glycosyltransferases and glycan structures contributing to the adhesive activities of L-, E- and P-selectin counterreceptors. Biochem. Soc. Symp. 2002:33-45.
25. Kok JW, Nikolova-Karakashian M, Klappe K, Alexander C, Merrill AH Jr. Dihydroceramide biology. Structure-specific metabolism and intracellular localization. J. Biol. Chem. 1997; 272:21128-21136.
26. Futerman AH, Pagano RE. Determination of the intracellular sites and topology of glucosylceramide synthesis in rat liver. Biochem. J. 1991; 280:295-302.
27. Jeckel D, Karrenbauer A, Burger KNJ, van Meer G, Wieland F. Glucosylceramide is synthesized at the cytosolic surface of various Golgi subfractions. J. Cell Biol. 1992; 117:59-267.
28. Lannert H, Bunning C, Jeckel D, Wieland F. Lactosyl ceramide is synthesized in the lumen of the Golgi apparatus. FEBS Lett. 1994; 342:91-96.
29. Lannert H, Gorgas K, Meitner I, Wieland FT, Jeckel D. Functional organization of the Golgi apparatus in glycosphingolipid biosynthesis. Lactosylceramide and subsequent glycosphingolipids are formed in the lumen of the late Golgi. J. Biol. Chem. 1998; 273:2939-2946.
30. Leith C, Kopecky K, Chen I, Eijdems L, Slovak M, McConnell T, Head D, Weick J, Grever M, Appelbaum F, Willman C. Frequency and clinical significance of the expression of the multidrug resistance proteins MDR1/P-glycoprotein, MRP1, and LRP in acute myeloid leukemia: a Southwest Oncology Group Study. Blood 1999; 94:1086-1099.
31. Takara K, Sakaeda T, Okumura K. An update on overcoming MDR1-mediated multidrug resistance in cancer chemotherapy. Curr. Pharm. Des. 2006; 12:273-286.
32. Riordan J, Deuchars K, Kartner N, Alon N, Trent J, Ling V. Amplification of P-glycoprotein genes in multidrug-resistant mammalian cell lines. Nature 1985; 316:817-819.
33. van Helvoort A, Smith A, Sprong H, Fritzsche I, Schinkel A, Borst P, van Meer G. MDR1 P-Glycoprotein is a lipid translocase of broad specificity, while MDR3 P-glycoprotein specifically translocates phosphatidyl choline. Cell 1996; 87:507-517.
34. De Rosa MF, Sillence D, Ackerley C, Lingwood C. Role of Multiple Drug Resistance Protein 1 in neutral but not acidic glycosphingolipid biosynthesis. J. Biol. Chem. 2004; 279:7867-7876.
35. Lala P, Ito S, Lingwood CA. Transfection of MDCK cells with the MDR1 gene results in a major increase in globotriaosyl ceramide and cell sensitivity to verocytotoxin: role of P-gp in glycolipid biosynthesis. J. Biol. Chem. 2000; 275:6246-6251.
36. Mattocks M, Bagovich M, De Rosa M, Bond S, Binnington B, Rasaiah V, Medin J, Lingwood C. Treatment of neutral glycosphingolipid storage disease via inhibition of the ABC Drug Transporter, MDR1: Cyclosporin A can lower serum and some tissue globotriaosyl ceramide levels in the Fabry’s mouse model. FASEB J. 2006; 273:2064-2075.
37. van IJzendoorn SC, Zegers MM, Kok JW, Hoekstra D. Segregation of glucosylceramide and sphingomyelin occurs in the apical to basolateral transcytotic route in HepG2 cells. J. Cell Biol. 1997; 137:347-357.
38. Wojtal KA, De Vries E, Hoekstra D, van Ijzendoorn SC. Efficient trafficking of MDRl/P-glycoprotein to apical canalicular plasma membranes in HepG2 cells requires PKA-RIIalpha anchoring and glucosylceramide. Mol. Biol. Cell 2006; 17:3638-3650.
39. Valaperta R, Chigorno V, Basso L, Prinetti A, Bresciani R, Preti A, Miyagi T, Sonnino S. Plasma membrane production of ceramide from ganglioside GM3 in human fibroblasts. FASEB J. 2006; 20:1227-1229.
40. Amado M, Almeida R, Schwientek T, Clausen H. Identification and characterization of large galactosyltransferase gene families: galactosyltransferases for all functions. Biochim. Biophys. Acta 1999; 1473:35-53.
41. Keusch J, Manzella SM, Nyame KA, Cummings RD, Baenziger JU. Cloning of Gb3 synthase, the key enzyme in globo-series glycosphingolipid synthesis, predicts a family of 1,4 glycosyn- transferases conserved in plants, insects and mammals. J. Biol. Chem. 2000; 275:25315-25321.
42. Steffensen R, Carlier K, Wiels J, Levery SB, Stroud M, Cederen B, Nilsson SB, Bennett EP, Jersild C, Clausen H. Cloning and expression of the histo-blood group Pk UDP-galactose: Galbeta 1-4Glcbeta 1-Cer alpha 1, 4-galactosyltransferase. Molecular genetic basis of the p phenotype. J. Biol. Chem. 2000; 275:16723-16729.
43. Mangeney M, Richard Y, Coulaud D, Tursz T, Wiels J. CD77: an antigen of germinal center B cells entering apoptosis. Eur. J. Immunol. 1991; 21:1131-1140.
44. Okajima T, Nakamura Y, Uchikawa M, Haslam DB, Numata SI, Furukawa K, Urano T. Expression cloning of human globoside synthase cDNAs. Identification of beta 3Gal-T3 as UDP-N-acetylgalactosamine:globotriaosylceramide beta 1,3-N-acetylgalactosaminyltransferase. J. Biol. Chem. 2000; 275:40498-40503.
45. Spitalnik P, Spitalnik S. The P blood group system: Biochemical, serological and clinical aspects. Transfus. Med. Rev. 1995; 9:110-122.
46. Haslam DB, Baenziger JU. Expression cloning of Forssman glycolipid synthetase: a novel member of the histo-blood group ABO gene family. Proc. Natl. Acad. Sci. U.S.A. 1996; 93:10697-10702.
47. Fujii Y, Numata SI, Nakamura Y, Honda T, Furukawa K, Urano T, Wiels J, Uchikawa M, Ozaki N, Matsuo SI, Sugiura Y. Murine glycosyltransferases responsible for the expression of globo-series glycolipids: cDNA structures, mRNA expression, and distribution of their products. Glycobiology 2005.
48. Nagata Y, Yamashiro S, Yodoi J, Lloyd KO, Shiku H, Furukawa K. Expression cloning of β1,4N-acetylgalactosaminyltransferase cDNAs that determine the expression of GM2 and GD2 gangliosides. J. Biol. Chem. 1992; 267:12082.
49. Togayachi A, Akashima T, Ookubo R, Kudo T, Nishihara S, Iwasaki H, Natsume A, Mio H, Inokuchi J, Irimura T, Sasaki K, Narimatsu H. Molecular cloning and characterization of UDP-GlcNAc:lactosylceramide beta 1,3-N-acetylglucosaminyltransferase (beta 3Gn-T5), an essential enzyme for the expression of HNK-1 and Lewis X epitopes on glycolipids. J. Biol. Chem. 2001; 276:22032-22040.
50. Henion TR, Zhou D, Wolfer DP, Jungalwala FB, Hennet T. Cloning of a mouse beta 1,3N-acetylglucosaminyltransferase GlcNAc(beta 1,3)Gal(beta 1,4) Glc-ceramide synthase gene encoding the key regulator of lacto-series glycolipid biosynthesis. J. Biol. Chem. 2001; 276:30261-30269.
51. Miyazaki H, Fukumoto S, Okada M, Hasegawa T, Furukawa K. Expression cloning of rat cDNA encoding UDP-galactose:GD2 beta1,3-galactosyltransferase that determines the expression of GD1b/GM1/GA1. J. Biol. Chem. 1997; 272:24794-24799.
52. Kono M, Takashima S, Liu H, Inoue M, Kojima N, Lee YC, Hamamoto T, Tsuji S. Molecular cloning and functional expression of a fifth-type alpha 2,3-sialyltransferase (mST3Gal V: GM3 synthase). Biochem. Biophys. Res. Commun. 1998; 253-175.
53. Daniotti JL, Martina JA, Giraudo CG, Zurita AR, Maccioni HJ. GM3 alpha2,8-sialyltransferase (GD3 synthase): protein characterization and sub-golgi location in CHO-K1 cells. J. Neurochem. 2000; 74:1711-1720.
54. Altheide TK, Hayakawa T, Mikkelsen TS, Diaz S, Varki N, Varki A. System-wide genomic and biochemical comparisons of sialic acid biology among primates and rodents: Evidence for two modes of rapid evolution. J. Biol. Chem. 2006; 281:25689-25702.
55. Varki A, Angata T. Siglecs-the major subfamily of I-type lectins. Glycobiology 2006; 16:1R-27R.
56. Prabhanjan H, Ishida H, Kiso M, Hasegawa A. A facile total synthesis of ganglioside GM1b and its positional analog. Carbohydr Res 1991; 211: C1-5.
57. Ishida H, Ohta Y, Tsukada Y, Isogai Y, Kiso M, Hasegawa A. A facile total synthesis of ganglioside GD2. Carbohydr. Res. 1994; 252:283-290.
58. Ando H, Ishida H, Kiso M, Hasegawa A. A synthetic approach to the c-series gangliosides containing sialyl-alpha(2-8)sialyl-alpha (2-8)sialic acid: synthesis of ganglioside GT4, alpha(2-6) GT4 and GT3. Carbohydr. Res. 1997; 300:207-217.
59. Duclos RI, Jr. The total synthesis of ganglioside GM3. Carbohydr. Res. 2000; 328:489-507.
60. Yamashita T, Wu YP, Sandhoff R, Werth N, Mizukami H, Ellis JM, Dupree JL, Geyer R, Sandhoff K, Proia RL. Interruption of ganglioside synthesis produces central nervous system degeneration and altered axon-glial interactions. Proc. Natl. Acad. Sci. U.S.A. 2005; 102:2725-2730.
61. Vyas A, Patel H, Fromholt S, Heffer-Lauc M, Vyas K, Dang J, Schachner M, Schnaar R. Gangliosides are functional nerve cell ligands for myelin-associated glycoprotein (MAG), an inhibitor of nerve regeneration. Proc. Natl. Acad. Sci. U.S.A. 2002; 99:8412-8417.
62. Fujitani M, Kawai H, Proia RL, Kashiwagi A, Yasuda H, Yamashita T. Binding of soluble myelin-associated glycoprotein to specific gangliosides induces the association of p75NTR to lipid rafts and signal transduction. J. Neurochem. 2005; 94:15-21.
63. Yang LJ, Lorenzini I, Vajn K, Mountney A, Schramm LP, Schnaar RL. Sialidase enhances spinal axon outgrowth in vivo. Proc. Natl. Acad. Sci. U.S.A. 2006; 103:11057-11062.
64. Vyas AA, Blixt O, Paulson JC, Schnaar RL. Potent glycan inhibitors of myelin-associated glycoprotein enhance axon outgrowth in vitro. J. Biol. Chem. 2005; 280:16305-16310.
65. Iwabuchi K, Zhang Y, Handa K, Withers DA, Sinay P, Hakomori S. Reconstitution of membranes simulating “glycosignaling domain” and their susceptibility to lyso-GM3. J. Biol. Chem. 2000; 275:15174-15181.
66. Toledo MS, Suzuki E, Handa K, Hakomori S. Effect of ganglioside and tetraspanins in microdomains on interaction of integrins with fibroblast growth factor receptor. J. Biol. Chem. 2005; 280:16227-16234.
67. Yoon SJ, Nakayama K, Hikita T, Handa K, Hakomori SI. Epidermal growth factor receptor tyrosine kinase is modulated by GM3 interaction with N-linked GlcNAc termini of the receptor. Proc. Natl. Acad. Sci. U.S.A. 2006; 103:18987-18991.
68. Hakomori S. Carbohydrate-to-carbohydrate interaction, through glycosynapse, as a basis of cell recognition and membrane organization. Glycoconj. J. 2004; 21:125-137.
69. Hakomori S, Handa K, Iwabuchi K, Yamamura S, Prinetti A. New insights in glycosphingolipid function: “glycosignaling domain,” a cell surface assembly of glycosphingolipids with signal transducer molecules, involved in cell adhesion coupled with signaling. Biochemistry 1999; 38:3369-3378.
70. Prinetti A, Iwabuchi K, Hakomori S. Glycosphingolipid-enriched signaling domain in mouse neuroblastoma Neuro2a cells. Mechanism of ganglioside-dependent neuritogenesis. J. Biol. Chem. 1999; 274:20916-20924.
71. Hakomori SI. The glycosynapse. Proc. Natl. Acad. Sci. U.S.A. 2002; 99:225-232.
72. Hanai N, Dohi T, Nores GA, Hakomori S. A novel ganglioside, de-N-acetyl-GM3 (II3NeuNH2LacCer), acting as a strong promoter for epidermal growth factor receptor kinase and as a stimulator for cell growth. J. Biol. Chem. 1988; 263:6296-6301.
73. Zhang Y, Iwabuchi K, Nunomura S, Hakomori S. Effect of synthetic sialyl 2→1 sphingosine and other glycosylsphingosines on the structure and function of the “glycosphingolipid signaling domain (GSD)” in mouse melanoma B16 cells. Biochem 2000; 39:2459-2468.
74. Kawakami Y, Kawakami K, Steelant WF, Ono M, Baek RC, Handa K, Withers DA, Hakomori S. Tetraspanin CD9 is a “proteolipid,” and its interaction with alpha 3 integrin in microdomain is promoted by GM3 ganglioside, leading to inhibition of laminin-5-dependent cell motility. J. Biol. Chem. 2002; 277:34349-34358.
75. Toledo MS, Suzuki E, Handa K, Hakomori S. Cell growth regulation through GM3-enriched microdomain (glycosynapse) in human lung embryonal fibroblast WI38 and its oncogenic transformant VA13. J. Biol. Chem. 2004; 279:34655-34664.
76. Burger K, van der Bijl P, van Meer G. Topology of sphingolipid galactosyl transferase in ER and Golgi: transbilayer movement of monohexyl sphingolipids is required for higher glycosphingolipid biosynthesis. J. Cell Biol. 1996; 133:15-28.
77. Ogura K, Tai T. Characterization of the functional domains of galactosylceramide expression factor 1 in MDCK cells. Glycobiology 2001; 11:751-758.
78. Coetzee T, Fujita N, Dupree J, Shi R, Blight A, Suzuki K, Popko B. Myelination in the absence of galactocerebroside and sulfatide: normal structure with abnormal function and regional instability. Cell 1996; 86:209-219.
79. Ishibashi T, Dupree JL, Ikenaka K, Hirahara Y, Honke K, Peles E, Popko B, Suzuki K, Nishino H, Baba H. A myelin galactolipid, sulfatide, is essential for maintenance of ion channels on myelinated axon but not essential for initial cluster formation. J. Neurosci. 2002; 22:6507-6514.
80. Dupree JL, Coetzee T, Blight A, Suzuki K, Popko B. Myelin galactolipids are essential for proper node of Ranvier formation in the CNS. J. Neurosci. 1998; 18:1642-1649.
81. Dupree JL, Coetzee T, Suzuki K, Popko B. Myelin abnormalities in mice deficient in galactocerebroside and sulfatide. J. Neurocytol. 1998; 27:649-659.
82. Hirahara Y, Bansal R, Honke K, Ikenaka K, Wada Y. Sulfatide is a negative regulator of oligodendrocyte differentiation: development in sulfatide-null mice. Glia 2004; 45:269-277.
83. Lingwood CA. Developmental regulation of glycolipid sulfotransferase during mammalian spermatogenesis. Evidence for a substrate-selective inhibitor of testicular sulphotransferase activity in the rat. Biochem. J. 1985; 231:393-400.
84. Honke K, Hirahara Y, Dupree J, Suzuki K, Popko B, Fukushima K, Fukushima J, Nagasawa T, Yoshida N, Wada Y, Taniguchi N. Paranodal junction formation and spermatogenesis require sulfo-glycolipids. Proc. Natl. Acad. Sci. U.S.A. 2002; 99:4227-4232.
85. Sundaram KS, Lev M. The long-term administration of L-cycloserine to mice: specific reduction of cerebroside level. Neurochem. Res. 1989; 14:245-248.
86. Soriano JM, Gonzalez L, Catala AI. Mechanism of action of sphingolipids and their metabolites in the toxicity of fumonisin B1. Prog. Lipid Res. 2005; 44:345-356.
87. Bionda C, Portoukalian J, Schmitt D, Rodriguez-Lafrasse C, Ardail D. Subcellular compartmentalization of ceramide metabolism: MAM (mitochondria-associated membrane) and/or mitochondria? Biochem J 2004; 382:527-533.
88. Platt FM, Jeyakumar M, Andersson U, Heare T, Dwek RA, Butters TD. Substrate reduction therapy in mouse models of the glycosphingolipidoses. Philos. Trans. R. Soc. Lond B Biol. Sci. 2003; 358:947-954.
89. Zimran A, Elstein D. Gaucher disease and the clinical experience with substrate reduction therapy. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2003; 358:961-966.
90. Hettkamp H, Bause E, Legler G. Inhibition by nojirimycin and 1-deoxynojirimycin of microsomal glucosidases from calf liver acting on the glycoprotein oligosaccharides Glc1-3Man9Glc NAc2. Biosci. Rep. 1982; 2:899-906.
91. Andersson U, Reinkensmeier G, Butters TD, Dwek RA, Platt FM. Inhibition of glycogen breakdown by imino sugars in vitro and in vivo. Biochem. Pharmacol. 2004; 67:697-705.
92. Radin NS, Shayman JA, Inokuchi J-I. Metabolic effects of inhibiting glucosylceramide synthesis with PDMP and other substances. In: Advances in Lipid Research, vol. 26. Bell RM, Hannun YA, Merrill Jr AH, eds. 1993. Academic Press, New York. pp. 183-213.
93. Kok JW, Babia T, Filipeanu CM, Nelemans A, Egea G, Hoekstra D. PDMP blocks Brefeldin A-induced retrograde membrane transport from Golgi to ER: Evidence for involvement of calcium homeostasis and dissociation from sphingolipid metabolism. J. Cell Biol. 1998; 142:25-38.
94. Abe A, Inokuchi J, Jimbo M, Shimeno H, Nagamatsu A, Shayman JA, Shukla GS, Radin NS. Improved inhibitors of glucosyl-ceramide synthase. J. Biochem. 1992; 111:191-196.
95. Abe A, Radin NS, Shayman JA, Wotring LL, Zipkin RE, Sivakumar R, Ruggieri JM, Carson KG, Ganem B. Structural and stereochemical studies of potent inhibitors of glucosylceramide synthase and tumor cell growth. J. Lipid Res. 1995; 36:611-621.
96. Lee L, Abe A, Shayman JA. Improved inhibitors of glucosylceramide synthase. J. Biol. Chem. 1999; 274:14662-14669.
97. Hakomori S-I. Cancer-associated glycosphingolipid antigens: their structure, organization, and function. Acta Anat. 1998; 161: 79-90.
98. Nishio M, Tajima O, Furukawa K, Urano T. Over-expression of GM1 enhances cell proliferation with epidermal growth factor without affecting the receptor localization in the microdomain in PC12 cells. Int. J. Oncol. 2005 ;26:191-199.
99. Yamashita T, Wada R, Sasaki T, Deng C, Bierfreund U, Sandhoff K, Proia RL. A vital role for glycosphingolipid synthesis during development and differentiation. Proc. Natl. Acad. Sci. U.S.A. 1999; 96:9142-9147.
100. Ichikawa S, Nakajo N, Sakiyama H, Hirabayashi Y. A mouse B16 melanoma mutant deficient in glycolipids. Proc. Natl. Acad. Sci. U.S.A. 1994; 91:2703-2707.
101. Furimsky A, Vuong N, Xu H, Kumarathasan P, Xu M, Weerachatyanukul W, Bou Khalil M, Kates M, Tanphaichitr N. Percoll gradient-centrifuged capacitated mouse sperm have increased fertilizing ability and higher contents of sulfogalactosyl-glycerolipid and docosahexaenoic acid-containing phosphatidylcholine compared to washed capacitated mouse sperm. Biol. Reprod. 2005; 72:574-583.
102. Burkart T, Caimi L, Herschkowitz NN, Wiesmann UN. Metabolism of sulfogalactosyl glycerolipids in the myelinating mouse. Devel. Biol. 1983; 98:182-186.
103. Boggs J, Menikh A, Rangaraj G. Trans interactions between galactosylceramide and cerebroside sulfate across apposed bilayers. Biophys. J. 2000; 78:874-885.
104. Ghislain J, Lingwood CA, Fish EN. Evidence for glycosphin- golipid modification of the type 1 IFN receptor. J. Immunol. 1994; 153:3655-3663.
105. Khine AA, Lingwood CA. Functional significance of globotriao- sylceramide in 2 interferon/ type I interferon receptor mediated anti viral activity'. J. Cell. Physiol. 2000; 182:97-108.
106. Maloney MD, Lingwood CA. CD19 has a potential CD77 (globotriaosylceramide)-binding site with sequence similarity to verotoxin B-subunits: implications of molecular mimicry for B cell adhesion and enterohemorrhagic Escherichia coli pathogenesis. J. Exp. Med. 1994; 180:191-201.
107. Lingwood CA, Yiu SCK. Glycolipid modification of 2-interferon binding:sequence similarity between 2-interferon receptor and the verotoxin (Shiga-like toxin) B-subunit. Biochem. J. 1992; 283: 25-26.
108. Marchetti M, Monier MN, Fradagrada A, Mitchell K, Baychelier F, Eid P, Johannes L, Lamaze C. Stat-mediated signaling induced by type I and type II interferons (IFNs) is differentially controlled through lipid microdomain association and clathrin-dependent endocytosis of IFN receptors. Mol. Biol. Cell 2006; 17:2896-2909.
109. Fiedler K, Kobayashi T, Kurzchalia TV, Simons K. Glycosphingolipid-enriched, detergent-insoluble complexes in protein sorting in epithelial cells. Biochemistry 1993; 32:6365-6373.
110. Mukherjee A, Arnaud L, Cooper JA. Lipid-dependent recruitment of neuronal Src to lipid rafts in the brain. J. Biol. Chem. 2003; 278:40806-40814.
111. Palacios EH, Weiss A. Function of the Src-family kinases, Lck and Fyn, in T-cell development and activation. Oncogene 2004; 23:7990-8000.
112. Simons K, Toomre D. Lipid rafts and signal transduction. Nature Rev. 2000; 1:31-39.
113. Rosenberger CM, Brumell JH, Finlay BB. Microbial pathogenesis: lipid rafts as pathogen portals. Curr. Biol. 2000; 10:R823-825.
114. Katagiri Y, Mori T, Nakajima H, Katagiri C, Taguchi T, Takeda T, Kiyokawa N, Fujimoto J. Activation of Src family kinase induced by Shiga toxin binding to globotriaosyl ceramide (Gb3/CD77) in low density, detergent-insoluble microdomains. J. Biol. Chem. 1999; 274:35278-35282.
115. Takenouchi H, Kiyokawa N, Taguchi T, Matsui J, Katagiri YU, Okita H, Okuda K, Fujimoto J. Shiga toxin binding to globotriaosyl ceramide induces intracellular signals that mediate cytoskeleton remodeling in human renal carcinoma-derived cells. J. Cell Sci. 2004; 117:3911-3922.
116. Fujinaga Y, Wolf AA, Rodighiero C, Wheeler H, Tsai B, Allen L, Jobling MG, Rapoport T, Holmes RK, Lencer WI. Gangliosides that associate with lipid rafts mediate transport of cholera and related toxins from the plasma membrane to ER. Mol. Biol. Cell 2003.
117. Nichols B. GM1-containing lipid rafts are depleted within clathrin-coated Pits. Curr. Biol. 2003; 13:686-690.
118. Yanagisawa M, Ariga T, Yu RK. Cholera toxin B subunit binding does not correlate with GM1 expression: a study using mouse embryonic neural precursor cells. Glycobiology 2006; 16:19G-22G.
119. Yanagisawa M, Ariga T, Yu RK. Fucosyl-GM1 expression and amyloid-beta protein accumulation in PC12 cells. J. Neurosci. Res. 2006; 84:1343-1349.
120. Connell SD, Smith DA. The atomic force microscope as a tool for studying phase separation in lipid membranes. Mol. Membr. Biol. 2006; 23:17-28.
121. Jacobson K, Mouritsen OG, Anderson RG. Lipid rafts: at a crossroad between cell biology and physics. Nat. Cell Biol. 2007; 9:7-14.
122. Gaus K, Gratton E, Kable EP, Jones AS, Gelissen I, Kritharides L, Jessup W. Visualizing lipid structure and raft domains in living cells with two-photon microscopy. Proc. Natl. Acad. Sci. U.S.A. 2003; 100:15554-15559.
123. Lenne PF, Wawrezinieck L, Conchonaud F, Wurtz O, Boned A, Guo XJ, Rigneault H, He HT, Marguet D. Dynamic molecular confinement in the plasma membrane by microdomains and the cytoskeleton meshwork. EMBO J. 2006; 25:3245-3256.
124. Baumgart T, Hammond AT, Sengupta P, Hess ST, Holowka DA, Baird BA, Webb WW. Large-scale fluid/fluid phase separation of proteins and lipids in giant plasma membrane vesicles. Proc. Natl. Acad. Sci. U.S.A. 2007; 104:3165-3170.
125. Kasahara K, Sanai Y. Functional roles of glycosphingolipids in signal transduction via lipid rafts. Glycoconj. J. 2000; 17:153-162.
126. Lingwood C, Mylvaganam M, Minhas F, Binnington B, Branch D, Pomes R. The sulfogalactose moiety of sulfoglycosphingolipids serves as a mimic of tyrosine phosphate in many recognition processes: prediction and demonstration of SH2 domain/sulfogalactose binding. J. Biol. Chem. 2005; 280:12542-12547.
127. Spiegel S. Fluorescent derivatives of ganglioside GM1 function as receptors for cholera toxin. Biochemistry 1985; 24:5947-5952.
128. Gahmberg CG, Andersson LC. Selective radioactive labeling of cell surface sialoglycoproteins by periodate-tritiated borohydride. J. Biol. Chem. 1977; 252:5888-5894.
129. Luchansky SJ, Bertozzi CR. Azido sialic acids can modulate cell-surface interactions. ChemBioChem. 2004; 5:1706-1709.
130. Hansen HC, Magnusson G. Synthesis of selected aminodeoxy analogues of galabiose and globotriose. Carb. Res. 1999; 322:166-180.
131. Mylvaganam M, Binnington B, Hansen H, Magnusson G, Lingwood C. Interaction of verotoxin 1 B subunit with globotriaosyl ceramide analogues: Aminosubstituted (aminodeoxy) adamantylGb3Cer provides insight into the nature of the Gb3Cer binding sites. Biochem. J. 2002; 368:769-776.
132. Lingwood CA, Nutikka A. A novel chemical procedure for the selective removal of nonreducing terminal N-acetyl residues from glycolipids. Anal. Biochem. 1994; 217:119-123.
133. Gowenlock BG, Richter-Addo GB. Preparations of C-nitroso compounds. Chem. Rev. 2004; 104:3315-3340.
134. Laine R, Yogeeswaren G, Hakomori S. Glycosphingolipids covalently linked to agarose gel or glass beads. J. Biol. Chem. 1974; 249:4460-4466.
135. Mylvaganam M, Lingwood CA. A convenient oxidation of natural glycosphingolipids to their “ceramide acids”for neoglycoconjugation: bovine serum albumin-glycoceramide acid conjugates as investigative probes for HIV coat protein gp120-glycosphingolipid interactions. J. Biol. Chem. 1999; 274:20725-20732.
136. Mylvaganam M, Meng L-J, Lingwood CA. Oxidation of glycosphingolipids under basic conditions: Synthesis of glycosylserine acids as opposed to “ceramide acids”. Precursors for neogly- coconjugates with increased ligand binding affinity. Biochemistry 1999; 38:10885-10897.
137. Sonnino S, Kirschner G, Ghidoni R, Aquotti D, Tettamanti G. Preparation of GM1 ganglioside molecular species having homogeneous fatty acid and long chain base moieties. J. Lipid Res. 1985; 26:248-257.
138. Rich JR, Wakarchuk WW, Bundle DR. Chemical and chemoenzymatic synthesis of S-linked ganglioside analogues and their protein conjugates for use as immunogens. Chemistry 2006;12: 845-858.
139. Helling F, Shang A, Calves M, Zhang S, Ren S, Yu RK, Oettgen HF, Livingston PO. GD3 vaccines for melanoma: Superior immunogenicity of keyhole limpet hemocyanin conjugate vaccines. CancerRes. 1994; 54:197-203.
140. Livingston PO, Ragupathi G. Cancer vaccines targeting carbohydrate antigens. Hum. Vaccin. 2006; 2:137-143.
141. Buk SJ, High OB. Periodate oxidation of glycolipids: a boro-hydride-periodate-Schiff method for ganglioside demonstration in tissue sections. Histochem. J. 1986; 18:228-232.
142. Bussink AP, van Swieten PF, Ghauharali K, Scheij S, van Eijk M, Wennekes T, van der Marel GA, Boot RG, Aerts JM, Overkleeft HS. N-Azidoacetylmannosamine mediated chemical tagging of gangliosides. J. Lipid Res. 2007.
143. Pagano R, Martin O. A series of fluorescent N-acylsphingosines: synthesis, physical properties and studies on cultured cells. Biochem. 1988; 27:4439-4445.
144. Lipsky NG, Pagano RE. Sphingolipid metabolism in clutured fibroblasts: microscopic and biochemical studies employing a florescent ceramide analogue. Proc. Natl. Acad. Sci. U.S.A. 1983; 80:2608-2612.
145. Chen CS, Bach G, Pagano RE. Abnormal transport along the lysosomal pathway in mucolipidosis, type IV disease. Proc. Natl. Acad. Sci. U.S.A. 1998; 95:6373-6378.
146. Pagano RE, Watanabe R, Wheatley C, Dominguez M. Applications of BODIPY-sphingolipid analogs to study lipid traffic and metabolism in cells. Methods Enzymol. 2000; 312:523-534.
147. Schwarzmann G, Wendeler M, Sandhoff K. Synthesis of novel NBD-GM1 and NBD-GM2 for the transfer activity of GM2-activator protein by a FRET-based assay system. Glycobiology 2005; 15:1302-1311.
148. Arab S, Lingwood C. Intracellular targeting of the endoplasmic reticulum/nuclear envelope by retrograde transport may determine cell hypersensitivity to Verotoxin: sodium butyrate or selection of drug resistance may induce nuclear toxin targeting via globotriaosyl ceramide fatty acid isoform traffic. J. Cell Physiol. 1998; 177:646-660.
149. Puri V, Watanabe R, Singh RD, Dominguez M, Brown JC, Wheatley CL, Marks DL, Pagano RE. Clathrin-dependent and -independent internalization of plasma membrane sphingolipids initiates two Golgi targeting pathways. J. Cell Biol. 2001; 154:535-547.
150. Chen C, Patterson M, Wheatley C, O’Brien J, Pagano R. Broad screening test for sphingolipid-storage diseases. Lancet 1999; 354:901-905.
151. Kuerschner L, Ejsing CS, Ekroos K, Shevchenko A, Anderson KI, Thiele C. Polyene-lipids: a new tool to image lipids. Nat. Methods 2005; 2:39-45.
152. Schwarzmann G. A simple and novel method for tritium labeling of gangliosides and other sphingolipids. Biochim Biophys Acta 1978; 529:106-114.
153. Suzuki Y, Suzuki K. Specific radioactive labeling of terminal n-acetylgalactosamine of glycosphingolipids by the galactose oxidase-sodium borohydride method. J. Lipid Res. 1972; 13:687-690.
154. Ghidoni R, Tettamanti G, Zambotti V. Labeling of natural substrates for the radiochemical assay of enzymes involved in lipid storage diseases: a general procedure for tritiation of ganglio- sides. Biochem. Exp. Biol. 1977; 13:61-69.
155. Gahmberg CG, Hakomori S. Extermal labeling of cell surface galactose and galactosamine in glycolipid and glycoprotein of human erythrocytes. J. Biol. Chem. 1973; 248:4311-4317.
156. Lingwood CA. Action of galactose oxidase on galactolipids. Can. J. Biochem. 1979; 57:1138-1143.
157. Holemann A, Seeberger PH. Carbohydrate diversity: synthesis of glycoconjugates and complex carbohydrates. Curr Opin Biotechnol 2004; 15:615-622.
158. Ito H, Ishida H, Collins BE, Fromholt SE, Schnaar RL, Kiso M. Systematic synthesis and MAG-binding activity of novel sulfated GM1b analogues as mimics of Chol-1 (alpha-series) gangliosides: highly active ligands for neural siglecs. Carbohydr. Res. 2003; 338:1621-1639.
159. Nilsson U, Ray AK, Magnusson G. Synthesis of the globotriaose tetrasaccharide and terminal tri- and di-saccharide fragments. Carb. Res. 1994; 252:117-136.
160. Hansen HC, Magnusson G. Synthesis of some aglycon analogs of globotriosylceramide. Carbohydrate Res. 1999; 322:181-189.
161. Vaughan MD, Johnson K, DeFrees S, Tang X, Warren RA, Withers SG. Glycosynthase-mediated synthesis of glycosphingolipids. J. Am. Chem. Soc. 2006; 128:6300-6301.
162. Albrecht B, Putz U, Schwarzmann G. Synthesis of fluorescent and radioactive analogues of two lactosylceramides and glucosyl-ceramide containing beta-thioglycosidic bonds that are resistant to enzymatic degradation. Carbohydr. Res. 1995; 276:289-308.
163. Wallner FK, Norberg HA, Johansson AI, Mogemark M, Elofsson M. Solid-phase synthesis of serine-based glycosphingolipid analogues for preparation of glycoconjugate arrays. Org. Biomol. Chem. 2005; 3:309-315.
164. Rai AN, Basu A. Synthesis of the glycosphingolipid beta-galactosyl ceramide and analogues via olefin cross metathesis. J. Org. Chem. 2005; 70:8228-8230.
165. Pacuszka T, Bradley RM, Fishman PH. Neoglycolipid analogues of ganglioside GM1 as functional receptors of cholera toxin. Biochemistry 1991; 30:2563-2570.
166. Bernardi A, Potenza D, Capelli AM, Garcia-Herrero A, Canada FJ, Jimene-Barbero J. Second-generation mimics of ganglioside GM1 oligosaccharide: a three-dimensional view of their interactions with bacterial enterotoxins by NMR and computational methods. Chemistry 2002; 8:4598-4612.
167. Bernardi A, Carrettoni L, Ciponte AG, Monti D, Sonnino S. Second generation mimics of ganglioside GM1 as artificial receptors for cholera toxin: replacement of the sialic acid moiety. Bioorg. Med. Chem. Lett. 2000; 10:2197-2200.
168. Bernardi A, Arosio D, Manzoni L, Micheli F, Pasquarello A, Seneci P. Stereoselective synthesis of conformationally constrained cyclohexanediols: a set of molecular scaffolds for the synthesis of glycomimetics. J. Org. Chem. 2001; 66:6209-6216.
169. Arosio D, Vrasidas I, Valentini P, Liskamp RM, Pieters RJ, Bernardi A. Synthesis and cholera toxin binding properties of multivalent GM1 mimics. Org. Biomol. Chem. 2004; 2:2113- 2124.
170. Page D, Roy R. Synthesis and biological properties of manno-sylated starburst poly (amidoamine) dendrimers. Bioconj. Chem. 1997; 8:714-723.
171. Dam TK, Roy R, Page D, Brewer CF. Negative cooperativity associated with binding of multivalent carbohydrates to lectins. Thermodynamic analysis of the “multivalency effect”. Biochemistry 2002; 41:1351-1358.
172. Karmali MA. Infection by Shiga toxin-producing Escherichia coli: an overview. Mol. Biotechnol. 2004; 26:117-122.
173. Boyd B, Zhiuyan Z, Magnusson G, Lingwood CA. Lipid modulation of glycolipid receptor function: Presentation of galactose 1-4 galactose disaccharide for Verotoxin binding in natural and synthetic glycolipids. Eur. J. Biochem. 1994; 223:873-878.
174. Stein PE, Boodhoo A, Tyrrell GJ, Brunton JL, Read RJ. Crystal structure of the cell-binding B oligomer of verotoxin-1 from E. coli. Nature 1992; 355:748-750.
175. Nyholm PG, Magnusson G, Zheng Z, Norel R, Binnington-Boyd B, Lingwood CA. Two distinct binding sites for globotriaosyl ceramide on verotoxins: molecular modelling and confirmation by analogue studies and a new glycolipid receptor for all verotoxins. Chem. Biol. 1996; 3:263-275.
176. Ling H, Boodhoo A, Hazes B, Cummings M, Armstrong G, Brunton J, Read R. Structure of the shiga toxin B-pentamer complexed with an analogue of its receptor Gb3. Biochemistry 1998; 37:1777-1788.
177. Soltyk AM, MacKenzie CR, Wolski VM, Hirama T, Kitov PI, Bundle DR, Brunton JL. A mutational analysis of the Globotriaosylceramide binding sites of Verotoxin VT1. J. Biol. Chem. 2002; 277:5351-5359.
178. St. Hilaire PM, Boyd MK, Toone EJ. Interaction of the Shiga-like toxin type 1 B-subunit with its carbohydrate receptor. Biochemistry 1994; 33:14452-14463.
179. Kitov PI, Sadowska JM, Mulvey G, Armstrong GD, Lingaw H, Pannu NS, Read RJ, Bundle DR. Shiga-like toxins are neutralized by tailored multivalent carbohydrate ligands. Nature 2000; 403:669-672.
180. Kitov PI, Shimizu H, Homans SW, Bundle DR. Optimization of tether length in nonglycosidically linked bivalent ligands that target sites 2 and 1 of a Shiga-like toxin. J. Am. Chem. Soc. 2003; 125:3284-3294.
181. Mulvey GL, Marcato P, Kitov PI, Sadowska J, Bundle DR, Armstrong GD. Assessment in mice of the therapeutic potential of tailored, multivalent Shiga toxin carbohydrate ligands. J. Infect. Dis. 2003; 187:640-649.
182. Fraser ME, Fujinaga M, Cherney MM, Melton-Celsa AR, Twiddy EM, O’Brien AD, James MN. Structure of shiga toxin type 2 (Stx2) from escherichia coli O157:H7. J. Biol. Chem. 2004; 279:27511-27517.
183. Nishikawa K, Matsuoka K, Kita E, Okabe N, Mizuguchi M, Hino K, Miyazawa S, Yamasaki C, Aoki J, Takashima S, Yamakawa Y, Nishijima M, Terunuma D, Kuzuhara H, Natori Y. A therapeutic agent with oriented carbohydrates for treatment of infections by Shiga toxin-producing Escherichia coli O157:H7. Proc. Natl. Acad. Sci. U.S.A. 2002; 99:7669-7674.
184. Nishikawa K, Matsuoka K, Watanabe M, Igai K, Hino K, Hatano K, Yamada A, Abe N, Terunuma D, Kuzuhara H, Natori Y. Identification of the optimal structure required for a shiga toxin neutralizer with oriented carbohydrates to function in the circulation. J. Infect. Dis. 2005; 191:2097-2105.
185. Watanabe M, Matsuoka K, Kita E, Igai K, Higashi N, Miyagawa A, Watanabe T, Yanoshita R, Samejima Y, Terunuma D, Natori Y, Nishikawa K. Oral therapeutic agents with highly clustered globotriose for treatment of Shiga toxigenic Escherichia coli infections. J. Infect. Dis. 2004; 189:360-368.
186. Kiarash A, Boyd B, Lingwood CA. Glycosphingolipid receptor function is modified by fatty acid content: Verotoxin 1 and Vero- toxin 2c preferentially recognize different globotriaosyl ceramide fatty acid homologues. J. Biol. Chem. 1994; 269:11138-11146.
187. Wilson BS, Steinberg SL, Liederman K, Pfeiffer JR, Surviladze Z, Zhang J, Samelson LE, Yang LH, Kotula PG, Oliver JM. Markers for detergent-resistant lipid rafts occupy distinct and dynamic domains in native membranes. Mol. Biol. Cell. 2004; 15:2580-2592.
188. Wolf AA, Jobling MG, Wimer-Mackin S, FErguson-Maltzman M, Madara JL, Holmes RK, Lencer WI. Ganglioside structure dictates signal transduction by cholera toxin and association with caveolae-like membrane domains in polarized epithelia. J. Cell. Biol. 1998; 141:917-927.
189. Bhat S, Mettus R, Reddy E, Ugen K, Srikanthan V, Williams W, Weiner D. The galactosyl ceramide/sulfatide receptor binding region of HIV-1gp120 maps to amino acids 206-275. Aids Res. Hum. Retrovirus. 1993; 9:175-181.
190. Fantini J, Hammache D, Delezay O, Pieroni G, Tamalet C, Yahi N. Sulfatide inhibits HIV-1 entry into CD4-/CXCR4+ cells. Virology 1998; 246:211-220.
191. Puri A, Rawat SS, Lin HM, Finnegan CM, Mikovits J, Ruscetti FW, Blumenthal R. An inhibitor of glycosphingolipid metabolism blocks HIV-1 infection of primary T-cells. Aids 2004; 18:849- 858.
192. Fantini J, Cook DG, Nathanson N, Spitalnik SL, Gonzalez- Scarano F. Infection of colonic epithelial cell lines by type 1 human immunodeficiency virus is associated with cell surface expression of galactosylceramide, a potential alternative gp120 receptor. Proc. Natl. Acad. Sci. U.S.A. 1993; 90:2700-2704.
193. Fantini J, Hammache D, Delezay O, Yahi N, Andre-Barres C, Rico-Lattes I, Lattes A. Synthetic soluble analogs of galacto-sylceramide (GalCer) bind to the V3 domain of HIV-1 gp 120 and Inhibit HIV-1-induced fusion and entry. J. Biol. Chem. 1997; 272:7245-7252.
194. Faroux-Corlay B, Greiner J, Terreux R, Cabrol-Bass D, Aubertin AM, Vierling P, Fantini J. Amphiphilic anionic analogues of galactosylceramide: synthesis, anti-HIV-1 activity, and gp120 binding. J. Med. Chem. 2001; 44:2188-2203.
195. Augustin LA, Fantini J, Mootoo DR. C-Glycoside analogues of beta-galactosylceramide with a simple ceramide substitute: synthesis and binding to HIV-1 gp120. Bioorg. Med. Chem. 2006; 14:1182-1188.
196. Villard R, Hammache D, Delapierre G, Fotiadu F, Buono G, Fantini J. Asymmetric synthesis of water-soluble analogues of galactosylceramide, an HIV-1 receptor: new tools to study virus-glycolipid interactions. Chembiochem 2002; 3:517-525.
197. Bertozzi CR, Cook DG, Kobertz WR, Gonzalez-Scarano F, Bednarski MD. Carbon-linked galactosphingolipid analogs bind specifically to HIV-1 gp 120. J. Am. Chem. Soc. 1992; 114:10639-10641.
198. Weber KT, Hammache D, Fantini J, Ganem B. Synthesis of glycolipid analogues that disrupt binding of HIV-1 gp120 to galactosylceramide. Bioorg. Med. Chem. Lett. 2000; 10:1011-1014.
199. LaBell RY, Jacobsen NE, Gervay-Hague J, O’Brien DF. Synthesis of novel glycolipids that bind HIV-1 Gp120. Bioconjug. Chem. 2002; 13:143-149.
200. Mamelak D, Mylvaganam M, Tanahashi E, Ito H, Ishida H, Kiso M, Lingwood C. The aglycone of sulfogalactolipids can alter the sulfate ester substitution position required for hsc70 recognition. Carbohydr. Res. 2001; 335:91-100.
201. Mylvaganam M, Lingwood CA. A preamble to aglycone reconstruction for membrane-presented glycolipids. In: Carbohydrate-based Drug Discovery, Volume 2. Wong C-H, ed. 2003. Wiley- VCH Press, Weinheim, Germany. pp. 761-780.
202. Lingwood CA, Mylvaganam M. Lipid modulation of glycosphin-golipid (GSL) receptors: Soluble glycolipid mimics provide new probes of glycolipid receptor function. In: Methods in Enzymol- ogy, Volume 363, pt B. Lee YCLRT, ed. 2003. Academic Press, New York. pp. 264-284.
203. Mylvaganam M, Lingwood C. Adamantyl globotriaosyl ceramide—a monovalent soluble glycolipid mimic which inhibits verotoxin binding to its glycolipid receptor. Biochem. Biophys. Res. Commun. 1999; 257:391-394.
204. Rutjes N, Binnington B, Smith C, Maloney M, Lingwood C. Differential tissue targeting and pathogenesis of Verotoxins 1 and 2 in the mouse animal model. Kid. Intl. 2002; 62:832-845.
205. Mahfoud R, Mylvaganam M, Lingwood CA, Fantini J. A novel soluble analog of the HIV-1 fusion cofactor, globotriaosyl-ceramide(Gb3), eliminates the cholesterol requirement for high affinity gp120/Gb3 interaction. J. Lipid Res. 2002; 43:1670-1679.
206. Lund N, Branch D, Mylvaganam M, Chark D, Ma X, Sakac D, Binnington B, Fantini J, Puri A, Blumenthal R, Lingwood C. A novel soluble mimic of the glycolipid globotriaosylceramide inhibits HIV infection. AIDS 2006; 20:1-11.
207. Mamelak D, Mylvaganam M, Whetstone H, Hartmann E, Lennarz W, Wyrick P, Raulston J, Han H, Hoffman P, Lingwood C. Hsp70s contain a specific sulfogalactolipid binding site. Differential aglycone influence on sulfogalactosyl ceramide binding by prokaryotic and eukaryotic hsp70 family members. Biochemistry 2001; 40:3572-3582.
208. Mamelak D, Lingwood C. The ATPase Domain of Hsp70 Possesses a Unique Binding Specificity for 3'Sulfogalactolipids. J. Biol. Chem. 2001; 276:449-456.
209. Whetstone D, Lingwood C. 3’Sulfogalactolipid binding specifically inhibits Hsp70 ATPase activity in vitro. Biochem 2003; 42:1611-1617.
210. Brodsky JL, Chiosis G. Hsp70 molecular chaperones: emerging roles in human disease and identification of small molecule modulators. Curr. Top. Med. Chem. 2006; 6:1215-1225.
211. Chandler D. The two faces of water. Nature 2002; 417:491.