CHEMICAL BIOLOGY
Chromosome Formation
Patrick Ryan Potts and Hongtao Yu, University of Texas Southwestern Medical Center, Dallas, Texas
doi: 10.1002/9780470048672.wecb650
Chromosomes are large molecules of DNA that organize genetic materials of an organism. Chromosomes need to be properly organized, packaged, and compacted to ensure their integrity and segregation to daughter cells during mitosis. In eukaryotic cells, chromosomes associate with histones and other proteins to form chromatin within the nucleus. The basic packaging unit of chromatin is the nucleosome. The nucleosome core particle consists of 147 base pairs of DNA that wrap around a core histone octamer. A linear array of nucleosomes is further folded into high-order chromatin structures. In addition, chromosomes are regulated during mitosis by the structural maintenance of chromosomes (SMC) family of proteins. Sister chromatids are physically tethered during DNA replication by the SMC1/3 cohesin complex to ensure equal segregation of chromosomes to daughter cells. These sister chromatids are compacted by the SMC2/4 condensin complex to form metaphase chromosomes.
DNA is the genetic material that encodes all information necessary for life. The DNA molecules that carry this information must be highly regulated to control and protect precisely the information they encode. In addition, cellular DNA needs to be replicated and divided equally to daughter cells during mitosis. For these reasons, DNA is elegantly compacted more than 100,000 times into a nucleoprotein structure known as chromatin. This compaction allows for the protection, regulation, and segregation of the genetic information encoded in DNA.
Chromatin Structure
Genomic DNA is organized into subunits called chromosomes (1). The human genome is split into 46 chromosomes. In eukaryotes, DNA is packaged and compacted with histones and other proteins to form chromatin. DNA wraps around core histones to form nucleosomes, the most fundamental level of DNA compaction (2). In addition to compacting DNA, histones also regulate the accessibility of DNA to various biological processes that use DNA as the template, including transcription, DNA replication, and DNA repair.
Nucleosome and chromatin fiber
The nucleosome core particle is the basic building block of chromatin (Fig. 1). It consists of 147 base pairs (bp) of DNA wrapped around the histone core in 1.7 left-handed, nonuniform, superhelical turns (3). X-ray crystallography studies have revealed the structure of the nucleosome core particle to a resolution of 1.9 A (4, 5). Each nucleosome core particle is made up of eight core histone molecules, one histone H3-H4 heterotetramer and two histone H2A-H2B heterodimers (4). Each core histone contains an amino-terminal unstructured tail and a three-helix histone fold (5). The histone folds in each dimer contain three distinct DNA-binding motifs that make extensive contacts with the minor groove of the DNA (4, 5). The helical periodicity or average number of base pairs per helical turn of DNA around the nucleosome core is 10.2 bp (5, 6). The minor grooves of DNA wrapped around neighboring nucleosomes line up to form channels through which the flexible histone tails emerge from the nucleosome core particle (5). The major groove of nucleosomal DNA is exposed to allow recognition by nuclear proteins, such as transcription factors (5). The canonical core histone proteins can be replaced with histone variants, such as H2A.Z. These variant nucleosomes have similar structures, but they are functionally distinct (7).
Nucleosomes are connected to one another by linker DNA of variable length and the linker-binding histone H1 protein (Fig. 1) (7). These long arrays of nucleosomes spontaneously condense to form helical arrays of nucleosomes, termed the 30-nm fiber after its apparent diameter (Fig. 1) (8). Additional condensation and compaction of chromatin occur through internucleosomal interactions. One important internucleosomal interaction required for chromatin fiber formation is the interaction of a highly acidic patch of histone H2A with the histone H4 tail (8). Ultimately, these internucleosomal interactions form interphase chromatin with an unknown architecture (Fig. 1) (9).
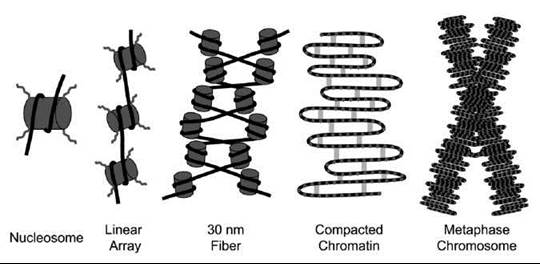
Figure 1. Schematic drawing of how chromosomes are formed from nucleosomes to the mitotic chromosome. Double-stranded DNA (black lines) wraps around core histones (gray cylinders) to form the nucleosome. Histone tails (curved lines) protrude from the histone core and can undergo various posttranslational modifications that regulate chromatin state. Helical arrays of nucleosomes form the 30-nm fiber. Scaffolding proteins (gray bars), such as condensin, further compact chromatin during mitosis to promote proper segregation to daughter cells.
Histone tails
One striking characteristic of the nucleosome core particle is the protrusion of flexible amino-terminal tails from the core histone proteins (5). These histone tails make ideal targets for the regulation of the nucleosome core particle. During events such as transcription, the chromatin must be opened up to allow the extensive protein machinery to access DNA. Acetylation of lysine residues on the protruding core histone tails is one major posttranslational modification that promotes chromatin opening (10). Lysine acetylation of histone tails is associated with active transcriptional regions of the genome, whereas hypoacetylation correlates with transcriptional repression (11). Histone acetyl transferases (HATs) promote transcription by catalyzing acetylation of histone tails (11). Histone deacetylases (HDACs) mediate histone tail deacetylation and, therefore, repress transcription. In addition to acetylation, core histone tails are posttranslationally modified by a number of other moieties, such as serine/threonine phosphorylation, arginine/lysine methylation, lysine ubiquitination, lysine sumoylation, glutamic acid ADP ribosylation, arginine deimination, and proline isomerization (10). These modifications either promote or suppress access to chromatin during transcription, DNA repair, or chromatin condensation in unique ways (12). Recent work has identified several proteins that catalyze these histone modifications, such as the methyl transferases that methylate lysines and arginines, as well as the histone demethylases such as, Jumonji C (JmjC) domain-containing proteins and lysine specific demethylase 1 (LSD1) (13).
Chromatin assembly
Chromatin assembly is coupled with DNA replication. Immediately after DNA replication, the parental histones from the original chromosome are randomly divided between the two daughter strands of DNA (14). The remaining complements of histones are assembled from newly synthesized histones. Newly synthesized histones H3 and H4 are acetylated, and these acetyl marks are removed after their incorporation into DNA (15). Assembly of chromatin requires the functions of histone chaperones and ATP-dependent chromatin remodeling factors (16). Histone chaperones bind histones and facilitate their proper deposition onto DNA by preventing nonspecific histone-DNA interactions (17). Two major histone chaperones are CAF-1 and NAP-1. CAF-1 localizes to the replication fork by binding PCNA and facilitates the deposition of histones H3 and H4 onto the newly synthesized DNA strands (18,19). Subsequently, NAP-1 facilitates the deposition of histones H2A and H2B to complete the nucleosome (20). Using in vitro nucleosome assembly and nuclease digestion mapping assays, it was shown that the periodic spacing of nucleosomes requires the function of ATP-dependent chromatin remodeling factors, such as the ACF/ISWI complex (16, 21).
Chromosome Cohesion
The propagation of genetic information during cell division is a complex and highly regulated process that involves extensive changes to the interphase chromatin. The somatic cell cycle is divided into four phases: G1, S, G2, and M. During S phase, DNA is replicated to form sister chromatids (22). Each sister chromatid contains the exact same genetic information. Therefore, it is imperative to ensure equal segregation of sister chromatids to daughter cells during mitosis (M phase). Unequal segregation of chromosomes results in daughter cells with wrong chromosome numbers, a condition known as aneuploidy. One important mechanism that ensures proper segregation of chromosomes to daughter cells is sister chromatid cohesion.
The cohesin complex
Sister chromatid cohesion is primarily established by the cohesin complex (23). The cohesin complex is a multiprotein complex that contains the structural maintenance of chromosomes (SMC) family of proteins (24). SMC proteins contain the Walker A motif at their N-termini and the Walker B motif at their C-termini (Fig. 2a). These motifs are brought together by an intramolecular coiled-coil domain to form a functional ATPase domain, which is similar to other ATP-binding cassette (ABC) ATPases, such as RAD50 (25). SMC proteins heterodimerize with one another through their hinge domains to form three unique complexes: cohesin (SMC1-SMC3), condensin (SMC2-SMC4), and SMC5-SMC6 (16, 19, 20). The head domains of the heterodimeric pairs of SMC proteins are linked by proteins known as kleisins (23). In addition to kleisins, SMC complexes contain additional non-SMC proteins. The cohesin complex is a multi-subunit complex that consists of the heterodimeric SMC1-SMC3 core, SCC1/MCD1/RAD21, and SA2 (SCC3 in yeast) (Fig. 2b) (23, 24). The function of SA2 is unknown. SCC1 is the kleisin subunit of the cohesin complex. SCC1 contains a winged helix domain at its C-terminus that mediates binding to the head domain of SMC1 (21). The binding of SCC1 to SMC1 facilitates the binding of SCC1’s N-terminal domain to the SMC3 head domain (26). In so doing, SCC1 links the two head domains of the SMC1-SMC3 heterodimer to form a ring-shaped structure. Cryoelectron microscopy has confirmed the ability of the cohesin complex to form a ring-shaped structure with an internal diameter of 30-40 nm (27).

Figure 2. (a) Architecture of the SMC protein complexes. A functional ATPase head domain is formed by N-terminal Walker A and C-terminal Walker B motifs that are brought together by an intramolecular coiled coil. Two SMC proteins heterodimerize through their hinge domains. Kleisin proteins link the two head domains of the SMC heterodimer. (b) Architecture of the cohesin complex. The SCC1 kleisin protein links together the head domains of the SMC1-SMC3 heterodimer to form a ring. (c) Two models for how cohesin establishes functional sister-chromatid cohesion. Sister chromatids are shown as cylinders.
The cohesin complex is loaded onto chromatin during late G1 phase of the cell cycle (28). The ATPase activity of SMC1-SMC3 is required for cohesin loading onto chromatin (29). The mechanism by which the ATPase activity promotes cohesin loading is unclear. In addition to the ATPase activity of SMC1-SMC3, cohesin loading is dependent on the SCC2-SCC4 complex (30). Both SCC2 and SCC4 contain Huntington, elongation A subunit, TOR (HEAT) repeats. The exact mechanism by which SCC2-SCC4 promotes cohesion loading is unclear. It has been proposed that SCC2-SCC4 stimulates the ATPase activity of SMC1-SMC3 (23, 29). The cohesin complex is concentrated around centromeres and at intergenic regions (31-33). Interestingly, in budding yeast, the chromosomal localization of SCC2-SCC4 does not overlap with that of cohesin, suggesting that cohesin redistributes to intergenic regions after SCC2-SCC4 facilitates its loading onto chromatin (27). The enrichment of cohesin at intergenic regions is thought to function in regulation of gene expression by insulating promoters from distant enhancers.
Loading of cohesin onto chromatin is itself insufficient to establish functional cohesion between sister chromatids. Establishment of sister chromatid cohesion occurs subsequent to cohesin loading and is tightly coupled to DNA replication in S phase. Several factors are required for the establishment of functional sister chromatid cohesion after the loading of cohesin onto chromatin. These factors include the acetyl transferase ECO1/CTF7, the replication factor C (RFC) complex (CTF18/DCC1/CTF8), and the chromatin structure remodeling (RSC) complex (34-36). The precise mechanism by which chromatin-bound cohesin establishes sister chromatid cohesion is unresolved. Elegant studies have shown that cohesin binds chromatin by entrapping DNA within its ring-shaped structure. These studies inserted an artificial tobacco etch virus (TEV) protease cleavage site into one cohesin subunit to allow inducible cleavage. Upon expression of TEV, the cohesin complex was rapidly removed from chromatin and sister chromatid cohesion was destroyed (27). In addition, using circular minichromosomes in which cohesin is loaded, cleavage of the circular DNA results in the removal of cohesin from these chromosomes (37). These studies show that DNA is entrapped within the cohesin ring. How DNA is encircled by the cohesin ring is unclear. Several possible mechanisms have been proposed, most of which involve the transient dissociation of proteins within the cohesin complex to open the ring structure. These include dissociation of the SMC1 and SMC3 hinge domains, SMC1 head domain and SCC1 C-terminus, or SMC3 head domain and SCC1 N-terminus. Another outstanding question is how cohesin actually mediates sister chromatid cohesion. Two equally viable models have been proposed (Fig. 2c). The embrace model proposes that a single cohesin complex encircles both sister chromatids within its ring-shaped structure as the DNA replication fork progresses (Fig. 2c, left). With a diameter of 30-40 nm, the cohesin ring is large enough to accommodate both sister chromatids (10 nm each) (32). However, it is unclear how the cohesin ring copes with the massive DNA replication machinery that would presumably need to move through the cohesin ring. Alternatively, a cohesin dimerization model proposes that each sister chromatid is trapped by separate cohesin complexes. In this model, the individually trapped sister chromatids are held together by the dimerization of the two cohesin complexes (Fig. 2c, right). However, intermolecular interaction between cohesin complexes has not been detected.
Mitotic Chromosome Condensation
In addition to cohesion between sister chromatids established during the S phase of the cell cycle, chromosomes are further condensed to allow for the movement and segregation of sister chromatids to daughter cells during mitosis. The formation of stable, rod-like chromosomes during prometaphase of mitosis is mediated by several scaffolding proteins, such as topoisomerase II and the condensin complex (38, 39).
Chromosome condensation during mitosis is mediated by the condensin complex. Condensin has two subtypes: condensin I and condensin II (Fig. 3a) (23, 24). Both condensin I and condensin II contain the core SMC2-SMC4 heterodimer (34). Similar to SMC1 and SMC3, SMC2 and SMC4 dimerize through their hinge domains. An intramolecular coiled-coil domain brings together the Walker A and Walker B motifs at the N- and C-termini of SMC2 and SMC4. These functional AT-Pase heads are linked by kleisin proteins, CAP-H for condensing I and CAP-H2 for condensin II (24). The condensin complexes also contain additional non-SMC proteins. Condensin I contains CAP-D2 and CAP-G, whereas condensin II contains CAP-D3 and CAP-G2 (34). The functions of these non-SMC proteins are unclear. Electron microscopy studies showed that the condensin complex can adopt a ring-shaped structure similar to cohesin. Condensin can also adopt a lollipop-like structure, with the coiled-coil and hinge domains of SMC2-SMC4 projecting away from a protein mass that includes the head domains of SMC2-SMC4 and the non-SMC proteins (35). How or whether condensin I and condensin II vary in function is unknown. Both condensin I and condensin II are excluded from the nucleus during interphase. Condensin I associates on chromatin after nuclear envelop breakdown during mitosis, whereas condensing II associates with chromatin before nuclear envelop breakdown (40). By cytological examination, chromosome-bound condensin I and condensin II do not overlap (36). Depletion of condensin I or condensin II causes distinct abnormalities in chromosome morphology (41), suggesting that the two complexes may not be functionally redundant.
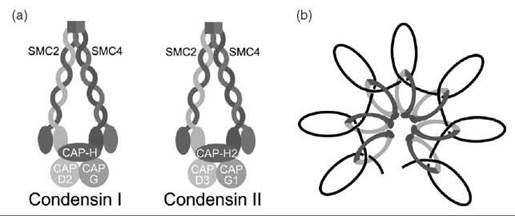
Figure 3. (a) Architecture of the condensin complexes. Both condensin I and condensin II contain the SMC2-SMC4 heterodimer. CAP-H and CAP-H2 are the kleisin subunits that link the head domains of SMC2-SMC4 in condensin I and condensin II, respectively. Condensin I contains the non-SMC proteins, CAP-D2 and CAP-G, whereas condensin II contains CAP-D3 and CAP-G1. (b) Model for condensin-mediated chromosome condensation. A linear strand of DNA is shown as a black line.
The mechanism by which the condensin complex organizes chromosomes during mitosis into their fully compacted and stable structure is currently unresolved. Several studies in multiple organisms have established that inactivation of condensin does not result in completely decondensed chromosomes but in compacted chromosomes that are not properly structured into rigid rods (42). The identity of those factors that mediate chromosome compaction in the absence of condensin is unknown. These studies suggest that condensin mediates the organization of compacted chromosomes into higher order structures that give rise to familiar metaphase chromosomes (Fig. 1). Clues into how condensin organizes chromosomes come from its activities in vitro. The addition of DNA to purified Xenopus condensin stimulates its ATPase activity (43). Condensin promotes the positive supercoiling of closed circular DNA when in the presence of topoisomerase I and ATP, but not the nonhydrolyzable ATP analog, AMP-PNP (43). The supercoiling of DNA by condensin could promote mitotic chromosome formation, but whether this activity of condensin occurs in vivo is unclear.
The activity of condensin is cell cycle regulated. Condensin purified from mitotic extracts is much more active in promoting DNA supercoiling than condensin from interphase extracts (43). In yeast, cdc2 stimulates condensin activity by phosphorylation of condensin (44). Furthermore, cdc2 is necessary for chromosome condensation (45). Likewise, inhibition of regulatory phosphatases and activation of cdc2 is sufficient to promote premature chromosome condensation (46). In addition to condensin, cdc2 phosphorylates histone H1, which is also important for chromosome condensation (47).
Other evidence suggests condensin functions with topoisomerase II, another component of the scaffold fraction of chromosomes (39). In vitro studies have shown that condensin can promote knotting of nicked circular DNA in the presence of topoisomerase II (48). Electron microscopy revealed that most of these knots had a trefoil structure (48). Based on this knot architecture, it has been proposed that topoisomerase II intertwines condensin-induced positive supercoils to form the knot (24). By looping DNA in this manner, condensin would promote DNA compaction (Fig. 3b).
References
1. Widom J. Structure, dynamics, and function of chromatin in vitro. Annu. Rev. Biophys. Biomol. Struct. 1998; 27:285-327.
2. Chakravarthy S, et al. Structure and dynamic properties of nucleosome core particles. FEBS Lett. 2005; 579:895-898.
3. Richmond TJ, et al. Structure of the nucleosome core particle at 7 A resolution. Nature 1984; 311:532-537.
4. Arents G, et al. The nucleosomal core histone octamer at 3.1 A resolution: a tripartite protein assembly and a left-handed superhelix. Proc. Natl. Acad. Sci. U.S.A. 1991; 88:10148-10152.
5. Luger K, et al. Crystal structure of the nucleosome core particle at 2.8 A resolution. Nature 1997; 389:251-260.
6. Richmond TJ, Davey CA. The structure of DNA in the nucleosome core. Nature 2003; 423:145-150.
7. Hansen JC. Conformational dynamics of the chromatin fiber in solution: determinants, mechanisms, and functions. Annu. Rev. Biophys. Biomol. Struct. 2002; 31:361-392.
8. Schalch T, et al. X-ray structure of a tetranucleosome and its implications for the chromatin fibre. Nature 2005; 436:138-141.
9. Tremethick DJ. Higher-order structures of chromatin: the elusive 30nm fiber. Cell 2007; 128:651-654.
10. Kouzarides T. Chromatin modifications and their function. Cell 2007; 128:693-705.
11. Sterner DE, Berger SL. Acetylation of histones and transcription-related factors. Microbiol. Mol. Biol. Rev. 2000; 64:435-459.
12. Li B, Carey M, Workman JL. The role of chromatin during transcription. Cell 2007; 128:707-719.
13. Martin C, Zhang Y. The diverse functions of histone lysine methylation. Nat. Rev. Mol. Cell Biol. 2005; 6:838-849.
14. Krude T, Keller C. Chromatin assembly during S phase: contributions from histone deposition, DNA replication and the cell division cycle. Cell. Mol. Life Sci. 2001; 58:665-672.
15. Sobel RE, et al. Conservation of deposition-related acetylation sites in newly synthesized histones H3 and H4. Proc. Natl. Acad. Sci. U.S.A. 1995; 92:1237-1241.
16. Haushalter KA, Kadonaga JT. Chromatin assembly by DNA- translocating motors. Nat. Rev. Mol Cell. Biol. 2003; 4:613-620.
17. Mello JA, Almouzni G. The ins and outs of nucleosome assembly. Curr. Opin. Genet. Dev. 2001; 11:136-141.
18. Shibahara K, Stillman B. Replication-dependent marking of DNA by PCNA facilitates CAF-1-coupled inheritance of chromatin. Cell 1999; 96:575-585.
19. Verreault A, et al. Nucleosome assembly by a complex of CAF-1 and acetylated histones H3/H4. Cell 1996; 87:95-104.
20. Ito T, et al. Drosophila NAP-1 is a core histone chaperone that functions in ATP-facilitated assembly of regularly spaced nucleosomal arrays. Mol. Cell. Biol. 1996; 16:3112-3124.
21. Ito T, et al. ACF, an ISWI-containing and ATP-utilizing chromatin assembly and remodeling factor. Cell 1997; 90:145-155.
22. Blow JJ, Tanaka TU. The chromosome cycle: coordinating replication and segregation. Second in the cycles review series. EMBO Rep. 2005; 6:1028-1034.
23. Nasmyth K, Haering CH. The structure and function of SMC and kleisin complexes. Annu. Rev. Biochem. 2005; 74:595-648.
24. Hirano T. At the heart of the chromosome: SMC proteins in action. Nat. Rev. Mol. Cell. Biol. 2006; 7:311-322.
25. Melby TE, et al. The symmetrical structure of structural maintenance of chromosomes (SMC) and MukB proteins: long, antiparallel coiled coils, folded at a flexible hinge. J. Cell. Biol. 1998; 142:1595-1604.
26. Haering CH, et al. Structure and stability of cohesin’s Smc1-kleisin interaction. Mol. Cell. 2004; 15:951-964.
27. Gruber S, Haering CH, Nasmyth K. Chromosomal cohesin forms a ring. Cell 2003; 112:765-777.
28. Michaelis C, Ciosk R, Nasmyth K. Cohesins: chromosomal proteins that prevent premature separation of sister chromatids. Cell 1997; 91:35-45.
29. Arumugam P, et al. ATP hydrolysis is required for cohesin’s association with chromosomes. Curr. Biol. 2003; 13:1941-1953.
30. Ciosk R, et al. Cohesin’s binding to chromosomes depends on a separate complex consisting of Scc2 and Scc4 proteins. Mol. Cell. 2000; 5:243-254.
31. Parelho V, et al. Cohesins Functionally associate with CTCF on mammalian chromosome arms. Cell 2008; 132:422-433.
32. Tanaka T, et al. Identification of cohesin association sites at centromeres and along chromosome arms. Cell 1999; 98:847-858.
33. Wendt KS, et al. Cohesin mediates transcriptional insulation by CCCTC-binding factor. Nature 2008; 451:796-801.
34. Baetz KK, et al. The ctf13-30/CTF13 genomic haploinsufficiency modifier screen identifies the yeast chromatin remodeling complex RSC, which is required for the establishment of sister chromatid cohesion. Mol. Cell. Biol. 2004; 24:1232-1244.
35. Hanna JS, et al. Saccharomyces cerevisiae CTF18 and CTF4 are required for sister chromatid cohesion. Mol. Cell. Biol. 2001; 21:3144-3158.
36. Kenna MA Skibbens RV. Mechanical link between cohesion establishment and DNA replication: Ctf7p/Eco1p, a cohesion establishment factor, associates with three different replication factor C complexes. Mol. Cell. Biol. 2003; 23:2999-3007.
37. Ivanov D, Nasmyth K. A topological interaction between cohesin rings and a circular minichromosome. Cell 2005; 122:849-860.
38. Gassmann R, et al. Mitotic chromosome formation and the condensin paradox. Exp. Cell. Res. 2004; 296:35-42.
39. Maeshima K, Laemmli UK. A two-step scaffolding model for mitotic chromosome assembly. Dev. Cell. 2003; 4:467-480.
40. Ono T, et al. Spatial and temporal regulation of Condensins I and II in mitotic chromosome assembly in human cells. Mol. Biol. Cell. 2004; 15:3296-3308.
41. Ono T, et al. Differential contributions of condensin I and condensin II to mitotic chromosome architecture in vertebrate cells. Cell 2003; 115:109-121.
42. Hudson DF, et al. Condensin is required for nonhistone protein assembly and structural integrity of vertebrate mitotic chromosomes. Dev. Cell. 2003; 5:323-336.
43. Kimura K, Hirano T. ATP-dependent positive supercoiling of DNA by 13S condensin: a biochemical implication for chromosome condensation. Cell 1997; 90:625-634.
44. Kimura K, et al. Phosphorylation and activation of 13S condensin by Cdc2 in vitro. Science 1998; 282:487-490.
45. Nurse P. Universal control mechanism regulating onset of M-phase. Nature 1990; 344:503-508.
46. Gotoh E, Durante M. Chromosome condensation outside of mitosis: mechanisms and new tools. J. Cell. Physiol. 2006; 209:297- 304.
47. Maresca TJ, Heald R. The long and the short of it: linker histone H1 is required for metaphase chromosome compaction. Cell Cycle 2006; 5:589-591.
48. Kimura K, et al. 13S condensin actively reconfigures DNA by introducing global positive writhe: implications for chromosome condensation. Cell 1999; 98:239-248.
Nucleosome: Topics in Chemical Biology
Cell Cycle
Cell Cycle: Regulation of
Topoisomerases, Chemistry of