Chemistry Essentials for Dummies
Chapter 6. Covalent Bonding
In This Chapter
· Seeing how one hydrogen atom bonds to another hydrogen atom
· Defining covalent bonding
· Finding out about the different types of chemical formulas
· Taking a look at polar covalent bonding and electronegativity
What holds together sugar, vinegar, and even DNA? Not ionic bonds! In this chapter, I discuss the other major type of bonding: covalent bonding. I explain the basics with an extremely simple covalent compound, hydrogen.
Covalent Bond Basics
Atoms form compounds to achieve a filled valence energy level (see Chapter 2 for more on energy levels). But instead of achieving it by gaining or losing electrons, as in ionic bonding (Chapter 5), the atoms in some compounds share electrons. That’s the basis of a covalent bond.
Sharing electrons: A hydrogen example
Hydrogen is number 1 on the periodic table — upper-left corner. Hydrogen has one valence electron. It’d love to gain another electron to fill its 1s energy level, which would make it isoelectronic with helium (because the two would have the same electronic configuration), the nearest noble gas. Energy level 1 can hold only two electrons in the 1s orbital, so gaining another electron would fill it. That’s the driving force of hydrogen: filling the valence energy level and achieving the same electron arrangement as the nearest noble gas.
Why atoms have to share
Why can’t the simple gain or loss of electrons explain the stability of H2? Imagine one hydrogen atom transferring its single electron to another hydrogen atom. The hydrogen atom receiving the electron fills its valence shell and reaches stability while becoming an anion (H-). However, the other hydrogen atom now has no electrons (H+) and moves further away from stability. This process of electron loss and gain simply won’t happen, because the goal of both atoms is to fill their valence energy levels. So the H2 compound can’t result from the loss or gain of electrons.
What can happen is that the two atoms can share their electrons. At the atomic level, this sharing is represented by the electron orbitals (sometimes called electron clouds) overlapping. The two electrons (one from each hydrogen atom) “belong” to both atoms. Each hydrogen atom feels the effect of the two electrons; each has, in a way, filled its valence energy level. A covalent bond is formed — a chemical bond that comes from the sharing of one or more electron pairs between two atoms.
That’s why the hydrogen found in nature is often not comprised of an individual atom. It’s primarily found as H2, a diatomic (two-atom) compound. Taken one step further, because a molecule is a combination of two or more atoms, H2 is called a diatomic molecule.
REMEMBER. In addition to hydrogen, six other elements are found in nature in the diatomic form: oxygen (O2), nitrogen (N2), fluorine (F2), chlorine (Cl2), bromine (Br2), and iodine (I2). So when I talk about oxygen gas or liquid bromine, I’m talking about the diatomic compound (diatomic molecule).
Representing covalent bonds
The overlapping of the electron orbitals and the sharing of an electron pair is represented in Figure 6-1a.
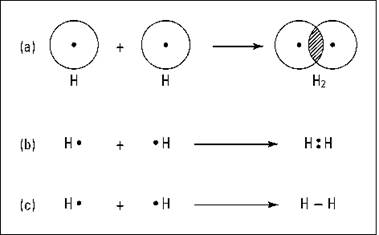
Figure 6-1: The formation of a covalent bond in hydrogen.
Another way to represent this process is through the use of an electron-dot formula. In this type of formula, valence electrons are represented as dots surrounding the atomic symbol, and the shared electrons are shown between the two atoms involved in the covalent bond. Figure 6-1b shows the electron- dot formula representations of H2.
Most of the time, I use a slight modification of the electron- dot formula called the Lewis structural formula; it’s basically the same as the electron-dot formula, but the shared pair of electrons (the covalent bond) is represented by a dash. Figure 6-1c shows the Lewis structural formula.
Comparing covalent bonds with other bonds
The properties of ionic and covalent compounds are different. Table 6-1 shows how the compounds compare. (Note: For the classification between metals and nonmetals, see Chapter 3.)
Table 6-1. Properties of Ionic and Covalent Compounds
|
Property |
Ionic Compounds (Salts) |
Covalent Compounds |
|
Bonds occur between |
A metal and a nonmetal |
Two nonmetals |
|
State of the compound at room temperature |
Usually solid |
Can be solid, liquid, or gas |
|
Melting point |
Higher than for covalent compounds |
Lower than for ionic compounds |
|
Electrolytes (they form ions and conduct electricity when dissolved) or nonelectrolytes |
Tend to be electrolytes |
Tend to be nonelectrolytes |
I know just what you’re thinking: If metals react with nonmetals to form ionic bonds, and nonmetals react with other nonmetals to form covalent bonds, do metals react with other metals? The answer is yes and no.
Metals don’t really react with other metals to form compounds. Instead, the metals combine to form alloys, solutions of one metal in another. But there is such a situation as metallic bonding, and it’s present in both alloys and pure metals. In metallic bonding, the valence electrons of each metal atom are donated to an electron pool, commonly called a sea of electrons, and are shared by all the atoms in the metal. These valence electrons are free to move throughout the sample instead of being tightly bound to an individual metal nucleus. The ability of the valence electrons to flow throughout the entire metal sample is why metals tend to be conductors of electricity and heat.
Dealing with multiple bonds
I define covalent bonding as the sharing of one or more electron pairs. In hydrogen and most other diatomic molecules, only one electron pair is shared. But in many covalent bonding situations, the atoms share more than one electron pair. For instance, nitrogen (N2) is a diatomic molecule in which the atoms share more than one electron pair.
The nitrogen atom is in the VA family on the periodic table, meaning that it has five valence electrons (see Chapter 3 for details on the periodic table). So nitrogen needs three more valence electrons to complete its octet. A nitrogen atom can fill its octet by sharing three electrons with another nitrogen atom, forming three covalent bonds, a so-called triple bond.
Figure 6-2 shows the triple bond formation of nitrogen.
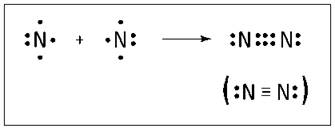
Figure 6-2: Triple bond formation in N2.
A triple bond isn’t quite three times as strong as a single bond, but it’s a very strong bond. In fact, the triple bond in nitrogen is one of the strongest bonds known. This strong bond is what makes nitrogen very stable and resistant to reaction with other chemicals. It’s also why many explosive compounds (such as TNT and ammonium nitrate) contain nitrogen: When these compounds break apart in a chemical reaction, nitrogen gas (N2) is formed, and a large amount of energy is released.
Carbon dioxide (CO2) is another example of a compound containing a multiple bond. Carbon can react with oxygen to form carbon dioxide. Carbon has four valence electrons, and oxygen has six. Carbon can share two of its valence electrons with each of the two oxygen atoms, forming two double bonds. Figure 6-3 shows these double bonds.
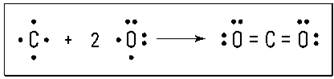
Figure 6-3: Formation of carbon dioxide.