Chemistry Essentials for Dummies
Chapter 11. Acids and Bases
Phun with the pH Scale
The acidity of a solution is related to the concentration of the hydronium ion in the solution: The more acidic the solution is, the higher the concentration. In other words, a solution in which the [H3O+] equals 1.0 x 10-2 is more acidic than a solution in which the [H3O+] equals 1.0 x 10-7.
REMEMBER. Scientists developed the pH scale, a scale based on the [H3O+], to more easily tell, at a glance, the relative acidity of a solution. The pH is defined as the negative logarithm (abbreviated as log) of [H3O+]. Mathematically, it looks like this:
![]()
In pure water, the [H3O+] equals 1.0 x 10-7, based on the water dissociation constant, Kw (see “Acting as either an acid or a base: Amphoteric water,” earlier in this chapter. Using this mathematical relationship, you can calculate the pH of pure water:
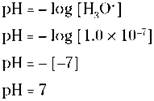
REMEMBER. The pH of pure water is 7. Chemists call this point on the pH scale neutral. A solution is acidic if it has a larger [H3O+] than water and a smaller pH value than 7. A basic solution has a smaller [H3O+] than water and a larger pH value than 7.
The pH scale really has no end. You can have a solution of pH that registers less than 0. (A 10 M HCl solution, for example, has a pH of -1.) However, the 0 to 14 range is a convenient range to use for weak acids and bases and for dilute solutions of strong acids and bases. Figure 11-2 shows the pH scale.
The [H3O+] of a 2.0 M acetic acid solution is 6.0 x 10-3. Looking at the pH scale, you see that this solution is acidic. Now calculate the pH of this solution:
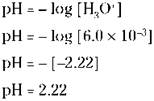
The pOH is the negative logarithm of the [OH-], and it can be useful in calculating the pH of a solution. You can calculate the pOH of a solution just like the pH by taking the negative log of the hydroxide ion concentration. If you use the Kw expression (which enables you to calculate the [H3O+] if you have the [OH-]; see the section “Acting as either an acid or base: Amphoteric water”) and take the negative log of both sides, you get 14 = pH + pOH. This equation makes it easy to go from pOH to pH.

Figure 11-2: The pH scale.
Just as you can you convert from [H3O+] to pH, you can also go from pH to [H3O+]. To do this, you use what’s called the antilog relationship, which is
![]()
Human blood, for example, has a pH of 7.3. Here’s how you calculate the [H3O+] from the pH of blood:
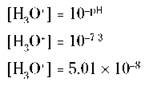
You can use the same procedure to calculate the [OH-] from the pOH.