Chemistry Essentials for Dummies
Chapter 2. What's In an Atom?
Centering on the Nucleus
In 1911, Ernest Rutherford discovered that atoms have a nucleus — a center — containing protons. Scientists later discovered that the nucleus also houses the neutron.
The nucleus is very, very small and very, very dense when compared to the rest of the atom. Typically, atoms have diameters that measure around 10-10 meters (that’s small!). Nuclei are around 10-15 meters in diameter (that’s really small!). If the Superdome in New Orleans represented a hydrogen atom, the nucleus would be about the size of a pea.
The protons of an atom are all crammed together inside the nucleus. Now you may be thinking, “Okay, each proton carries a positive charge, and like charges repel each other. So if all the protons are repelling each other, why doesn’t the nucleus simply fly apart?” It’s the Force, Luke. Forces in the nucleus counteract this repulsion and hold the nucleus together. Physicists call these forces nuclear glue. (Note: Sometimes this “glue” isn’t strong enough, and the nucleus does break apart. This process is called radioactivity, and I cover it in Chapter 4.)
Not only is the nucleus very small, but it also contains most of the mass of the atom. In fact, for all practical purposes, the mass of the atom is the sum of the masses of the protons and neutrons. (I ignore the minute mass of the electrons unless I’m doing very, very precise calculations.)
REMEMBER. The sum of the number of protons plus the number of neutrons in an atom is called the mass number. And the number of protons in a particular atom is given a special name, the atomic number. Chemists commonly use the symbolization in Figure 2-1 to represent these amounts for a particular element.
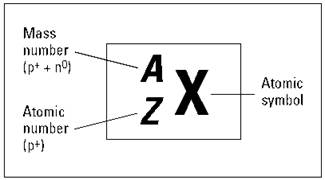
Figure 2-1: Representing a specific element.
As Figure 2-1 shows, chemists use the placeholder X to represent the chemical symbol. You can find an element’s chemical symbol on the periodic table or in a list of elements. The placeholder Z represents the atomic number — the number of protons in the nucleus. And A represents the mass number, the sum of the number of protons plus neutrons. The mass number is listed in amu.
For example, you can represent a uranium atom that has 92 protons and a mass number of 238 as in Figure 2-2.

Figure 2-2: Representing uranium.
TIP. You can find the number of neutrons in an atom by subtracting the atomic number (number of protons) from the mass number (protons plus neutrons). For instance, you know that uranium has an atomic number of 92 and mass number of 238.
So if you want to know the number of neutrons in uranium, all you have to do is subtract the atomic number (92 protons) from the mass number (238 protons plus neutrons). The answer shows that uranium has 146 neutrons.
But how many electrons does uranium have? Because the atom is neutral (it has no electrical charge), there must be equal numbers of positive and negative charges inside it, or equal numbers of protons and electrons. So there are 92 electrons in each uranium atom.
You can find both the element symbol and its atomic number on the periodic table, but the mass number for a particular element is not shown there. What is shown is the average atomic mass or atomic weight for all forms of that particular element, taking into account the percentages of each found in nature. See the later section “Isotopes: Varying neutrons” for details on other forms of an element.