The Handy Chemistry Answer Book (2014)
ORGANIC CHEMISTRY
REACTIONS OF ORGANIC COMPOUNDS
What are “curved arrows” in organic chemistry?
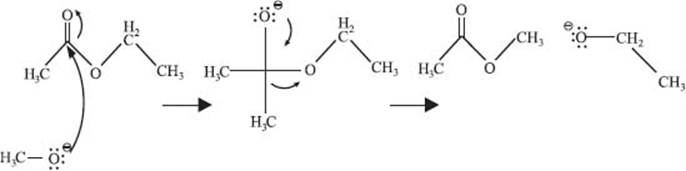
Chemists used curved arrows to depict the flow of electrons in a chemical reaction. The arrow starts at the nucleophile (a lone pair of electrons, a π bond, or a σ bond), and points toward the electrophile (an atom, or bond, with a full or partial positive charge). Here’s an example of a transesterification reaction (i.e., changing one ester for another).
What is a nucleophile?
A nucleophile is a molecule that donates electrons (to an electrophile) in a chemical reaction. These are typically functional groups with lone pairs of electrons, but can also be π bonds, or, in some rare cases, σ bonds.
What is an electrophile?
Electrophiles are species that accept electrons (from a nucleophile) in a chemical reaction. Usually, electrophiles have either a full- or partial-positive charge, or in some cases (like BH3) have an unfilled octet of electrons.
What is a substitution reaction?
The exchange of one functional group, or atom, for another is called a substitution reaction. The transesterification reaction above is an example of substituting a -OCH2CH3 group for a -OCH3 group. Another simple substitution reaction is the exchange of one halogen for another on a methyl group:
![]()
What is a unimolecular substitution reaction?
We’ve already talked about substitution reactions, so what makes one “unimolecular”? If the transition state (remember, this is the highest energy state of an individual chemical reaction) involves one (uni-) molecule (-molecular), then it is referred to as a uni-molecular reaction. This might seem like an odd thing to distinguish, but there are many differences between uni- and bimolecular substitution reactions. These differences all result from how many species are involved in the transition state.
For an example, here’s the reaction of tert–butyl chloride with hydroxide ion:
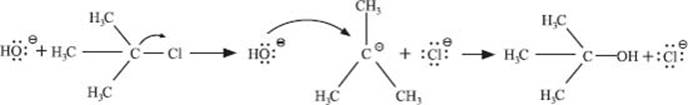
The first step is breaking the C–Cl bond, and this involves only the (CH3)3C–Cl molecule. Then the hydroxide ion reacts with the tert–butyl carbocation in the second step. Since there is only one molecule in the slower first step (just assume that’s true so we can illustrate this point), this is a unimolecular substitution reaction.
What is a bimolecular substitution reaction?
From the last answer, you can probably guess that a bimolecular substitution reaction has two (bi-) molecules (-molecular) in the transition state. This means that the slowest step involves two molecules interacting. In the previous example, there was only one.
Let’s look at a similar substitution reaction, but instead of tBuCl, we’ll react hydroxide with MeCl:
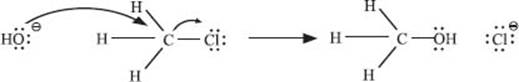
Here the nucleophile (OH−) directly displaces the leaving group (Cl−), without forming a carbocation intermediate. This is because the methyl cation (CH3+) is much less stable than the tert–butyl carbocation formed in the previous question.
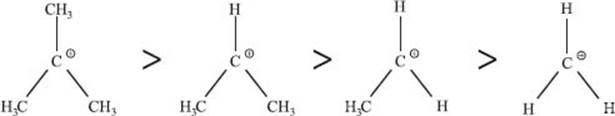
Why are more substituted carbocations more stable?
Great question! First, let’s review what led you to this question. In the unimolecular substitution reaction we looked at, an intermediate tert–butyl cation was formed, but in the bimolecular substitution reaction, the cation (this time a methyl cation, CH3+) was too high in energy. In this case the hydroxide ion directly displaced the chloride ion.
Generally, the more highly substituted a carbocation is, the more stable it is. There are a number of ways to explain why this is true. The first is that carbon substituents are more electron-donating than hydrogen atoms. Electrons on neighboring carbon atoms can help stabilize the cationic center. Simply put, the order of carbocation stability matches the number of carbons bonded to the cationic carbon, with higher numbers leading to greater stability.
What is hyperconjugation?
Hyperconjugation is another way to explain why substituted carbocations are more stable. The electrons in either the C–C or C–H bonds that are near the cationic center can interact with the empty p-orbital. It’s not the bonds that are directly connected, but rather one bond removed from the center carbon, that are the most important (see the illustration below). At the simplest level, this provides an explanation for the statement in the previous answer that neighboring carbon atoms are more electron-donating than hydrogen atoms: it’s really the neighboring C–H bonds that help stabilize the empty p-orbital.
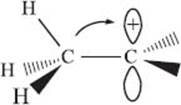
Note: The arrow above is just to illustrate the overlap of the C–H σ bond with the empty p orbital. It’s not to suggest that the hydrogen atom actually moves…though this does happen sometimes!
What is an addition reaction?
In an addition reaction, two or more molecules combine to make one molecule. This is different than the substitution reactions we have been looking at, where two molecules combine to make two different molecules.
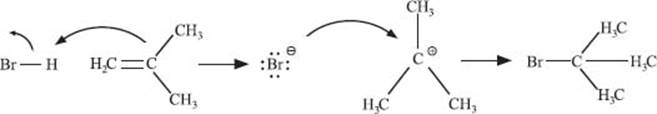
The addition of an acid to an alkene is the simplest example. Here, an acid proto-nates a carbon–carbon double bond. The carbocation that is formed will be the more substituted one of the possible products that could form. The conjugate base of the acid then reacts with this carbocation.
What is Markovnikov’s rule?
We actually just looked at an example of Markovnikov’s rule! This rule states that when you add a protic acid (like HBr) to an alkene, the proton ends up attached to the carbon with fewer alkyl substituents, while the conjugate base ends up bonded to the carbon with more alkyl substituents. This is because of the stability of the carbocation intermediate that is formed (remember, more substituted means more stable).
What is an elimination reaction?
If an addition reaction adds two groups to a molecule, then an elimination reaction takes them away. The textbook example again involves carbon-carbon double bonds, but this time we’re making that double bond.
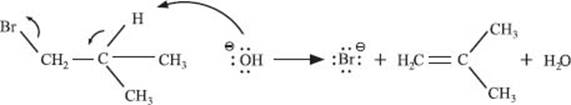
A molecule of base (hydroxide ion, −OH) causes the elimination of HBr. In this case, the process happens in one step, as shown.
Are there uni- and bimolecular elimination reaction mechanisms, like there were for substitution reactions?
Yes! And what do you think controls whether an elimination reaction happens in one step or two? Right—the stability of the carbocation. In the previous example, if bromide ion had dissociated first, a primary carbocation would have formed:
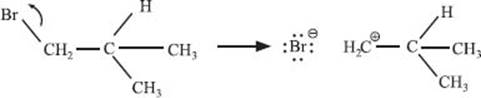
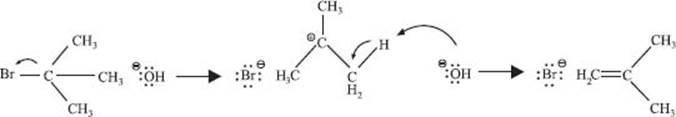
This is a much more difficult reaction than the bimolecular process where the elimination of HBr takes place in a single step.
But if an alkyl halide can form a stable carbocation, the unimolecular elimination reaction is faster. It’s referred to as “unimolecular” because the slow step has only one molecule in the transition state, just like substitution reactions.
Who were Diels and Alder?
The Diels-Alder reaction, like many reactions in organic chemistry, is named after the chemists who discovered it—in this case Otto Paul Hermann Diels (1876–1954) and Kurt Alder (1902–1958). Kurt Alder was actually Diels’ student at the University of Kiel, and Alder was awarded a PhD in 1926. Alder and his advisor Diels jointly received the Nobel Prize in Chemistry in 1950.
What’s a cycloaddition reaction?
In the most general terms, a cycloaddition reaction is one that forms a ring from multiple π-bonds. To illustrate some of the concepts and ways that these reactions are classified, let’s look at the cycloaddition reaction shown below. This reaction, known as the Diels-Alder reaction, is between a conjugated diene (double alkene) and a another alkene that likes to react with dienes (hence dienophile, or diene lover). It is classified as a [4 + 2] cycloaddition, referring to the number of atoms directly involved in bond-forming events.
(There’s a second system for classifying cycloaddition reactions that uses the number of π-electrons involved in ring formation, but let’s just leave that aside.)
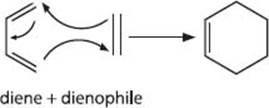
What’s a pericyclic reaction?
A cycloaddition reaction is actually a type of pericyclic reaction, but the term “pericyclic” includes other types of reactions. The textbook definition of pericyclic is a reaction whose transition state has a cyclic structure (i.e., the electrons are flowing in a closed loop). In addition to cycloaddition reactions (which exchange two π-bonds for two σ-bonds, or vice versa), pericyclic reactions include sigmatropic reactions, electrocyclic reactions, and cheletropic reactions (and a few others which we’ll ignore).
What’s a sigmatropic reaction?
While a cycloaddition reaction uses two π-bonds to make two new σ-bonds, a sigmatropic reaction takes one σ-bond and makes…well…one σ-bond. One of the most well-known sigmatropic rearrangements is known as the Cope rearrangement. Using the system we talked about earlier, this reaction is classified as a [3,3]-sigmatropic reaction (the methyl substituent is not directly involved, so we don’t count it).
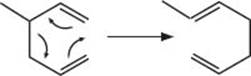
What’s an electrocyclic reaction?
Okay, so a cycloaddition reaction was 2π![]() 2σ, and we just saw that sigmatropic reactions are 1σ
2σ, and we just saw that sigmatropic reactions are 1σ![]() 1σ, so what about a pericyclic reaction that is 1π
1σ, so what about a pericyclic reaction that is 1π![]() 1σ? That’s an electrocyclic reaction. Like most pericylic reactions, electrocyclic reactions can either make or break a σ-bond. If a σ-bond is made in the process, it’s an electrocyclic ring-closing reaction. If the σ-bond is consumed in the process, the process is named an electrocyclic ring-opening reaction, but it’s the same process.
1σ? That’s an electrocyclic reaction. Like most pericylic reactions, electrocyclic reactions can either make or break a σ-bond. If a σ-bond is made in the process, it’s an electrocyclic ring-closing reaction. If the σ-bond is consumed in the process, the process is named an electrocyclic ring-opening reaction, but it’s the same process.
One noteworthy example of an electrocyclic ring-closing reaction is the Nazarov cyclization, which converts divinyl ketones into cyclopentenes, usually in the presence of an acid.
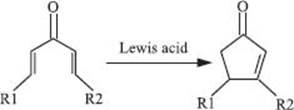
What’s a cheletropic reaction?
Finally, there’s a second type of reaction that involves two π-bonds being transformed into two σ-bonds. The difference here is that two bonds are formed (or broken) at a single atom, while in cycloaddition reactions, only one bond is made at each reactive atom. An example involving SO2 is shown below (surprisingly this reaction isn’t named after anyone!). In this reaction both σ-bonds are made (or broken) at the sulfur atom. Compare this with the Diels-Alder reaction mentioned above, where the two new σ-bonds are connected to the two different ends of the dieneophile.
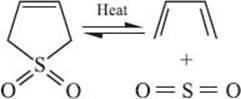
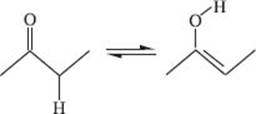
What is a tautomerization reaction?
A tautomerization reaction interconverts two constitutional isomers of an organic molecule, referred to as tautomers, which differ only by the positions of hydrogen atoms. The scheme below shows how the reaction typically takes place at the ketone functional group of an organic molecule. The ketone form of the molecule is commonly referred to as the “keto” form of the molecule, while the alcohol form is commonly referred to as the “enol” form of the molecule.