The Handy Chemistry Answer Book (2014)
THE WORLD AROUND US
CHEMICALS IN OUR WORLD
What makes a rubber band stretchy?
Rubber bands are made up of long, tangled-up, chainlike molecules called polymers. These long chains prefer to be tangled up because it maximizes their entropy or just the number of ways they can arrange themselves (see “Physical and Theoretical Chemistry” for more on entropy or “Polymer Chemistry” for more on polymers). When we stretch a rubber band, it straightens out these long chain molecules into a state with less entropy, so basically there are fewer stretched-out conformations possible than there are tangled-up ones. When we release the rubber band, it contracts to a more disordered state, primarily just because there are more possible conformations associated with the contracted length.
How does an air freshener work?
Although there are many different types of air fresheners, most of them are simply perfume dispensers. The perfume is a strong-smelling substance that has a pleasant odor, which masks the stink.
What’s hair made of?
Keratin is the major component of hair (and nails). It’s a protein that makes up the outer layer of your skin, your hair, fingernails, and toenails. Actually, keratin is everywhere in the animal world: the hooves, claws, and horns of most mammals, the scales of reptiles, the shells of turtles, and the feathers, beaks, and claws of birds. Many copies of this protein molecule assemble into a large helical structure that provides the structure and rigidity to the material. Keratin proteins contain lots of sulfur atoms in the form of cysteine amino acid residues. These sulfur atoms can form linkages between keratin strands and between the larger helices that are formed, leading ultimately to a curling of the hair strand. Yep—you guessed it: The more disulfide bonds there are, the curlier your hair will be. Hair straighteners work by breaking these disulfide bonds, relaxing the hair to a straight conformation.

We spread salt on icy roads to melt the ice, which works because the salt lowers the freezing point of frozen water.
Why does salt melt ice?
Adding salt to ice lowers the melting point of water (see freezing-point depression in “Macroscopic Properties”). So when you pour salt on the ice on your driveway, the melting point of the saltwater mixture you made by adding the salt may be lowered sufficiently such that it is below the outside temperature, which means the solid ice will start to melt.
Why do farts stink?
Farts are mostly nitrogen (N2), but that’s an odorless gas. The smelly molecules in farts are mostly sulfur-containing compounds that reek like rotten eggs. Other odoriferous compounds include skatole and indole. How did chemists figure this out? They analyzed fart gas by gas chromatography. No, really.
What is inside of a zit?
Gross. You really want to know? Okay…the major component of most pimples is a mixture of keratin and sebum. We just talked about what keratin earlier in this chapter. Sebum is an oily mixture that your skin naturally secretes. Earwax is also made up mostly of sebum.
What about blackheads and whiteheads? Are they the same?
Almost. If sebum builds up underneath the surface of the skin, it stays white. If sebum collects around a hair follicle or is in some other way exposed to the air, the sebum undergoes oxidation reactions. As sebum oxidizes it darkens, eventually appearing black.
Is it better to use “chemical-free” products?
There are no such things! Everything is made up of chemicals! Be careful when choosing which ones you eat or use, but the truth is, they’re all made up of chemicals.
How do mood rings work?
Mood rings change color based on your body temperature. The piece that changes color is a liquid crystal thermometer, which is the same technology that is used in some disposable medical thermometers as well as the adhesive thermometers commonly used in aquariums.
How does waterproof mascara work?
Like we talked about in the chapters on “Macroscopic Properties” and “Biochemistry,” hydrophobic materials do not dissolve well in water. So it makes sense to make waterproof mascaras from something hydrophobic. Waterproof mascaras contain a combination of waxes to make them resistant to washing off in water.

Mood rings were all the rage in the 1970s. They work the same way crystal thermometers work do: thermotropic liquid crystals within a hollow quartz shell respond to temperature changes by twisting, which causes them to reflect different wavelengths of light depending on temperature.
How do mirrors reflect light so well?
Most modern mirrors are made of smooth layers of silver or aluminum along with other chemicals and coatings to aid in their construction. While the chemical properties of silver play some role in reflecting light, the fact that these layers are very, very smooth is more important here. If the surface of the silver layer were rough, the light would be reflected in a variety of different angles, which you would see as a distorted image. When the surface is perfectly smooth, the light bounces straight back into your eye and you see an accurate reflection of the object. This is why you can see clear reflections in very still water or a shiny piece of leather—both are smooth surfaces.
What are pigments?
Pigments are molecules that selectively absorb particular (ranges of) wavelengths of light. The remaining reflected wavelengths are the color that you perceive the pigment to be. So pigments can only subtract wavelengths of light from the spectrum, but they cannot generate their own light. For example, a blue pigment absorbs red and green light, but reflects every other color. Alternatively, the blue pigment might only absorb orange light, which is the color complement of blue.
Why was gold chosen as a form of currency rather than any other element in the periodic table?
What a neat question! There are very basic attributes to money that made gold the obvious element. The element should be a solid at all temperatures it might encounter over the course of its lifetime—no one wants their money to boil away on a hot day. The element needs to be very stable—we wouldn’t want our coins rusting, bursting into flame, or slowly giving off radiation. And finally, the element needs to be rare, but not too rare. After you remove elements that don’t fit these parameters, we are left with rhodium, palladium, silver, platinum, and gold. Rhodium and platinum weren’t discovered until the nineteenth century, so they weren’t an option for ancient civilizations. The furnaces of the ancient world couldn’t reach the temperature required to melt platinum (3200 °F, 1800 °C), so it couldn’t be made into coins. That leaves silver and gold. Gold has a lower melting point than silver and it doesn’t tarnish in air like silver does, making it the clear winner for a currency element on our planet.
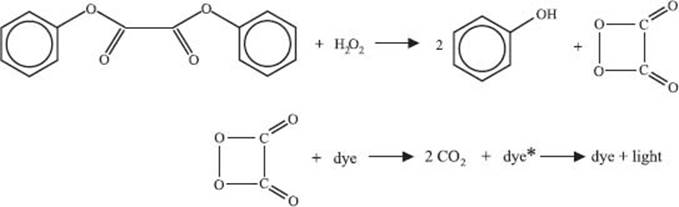
What causes the “glow” in glowsticks?
Glowsticks contain three main components—a dye, diphenyl oxalate, and hydrogen peroxide. The hydrogen peroxide is the chemical you release when you “crack” or activate the glowstick. Diphenyl oxalate reacts with the hydrogen peroxide to generate a molecule called dioxetanedione. This particular molecule decomposes to release two molecules of carbon dioxide and energy that excites a dye molecule to a higher energy state. To get back to its stable state the dye releases a photon of light, making your glow stick glow. The particular structure of the dye molecule controls the wavelength (color) of this light, which is how you can get glowsticks in different colors.
What causes nonstick pans to be nonadhesive?
Nonstick pans and other surfaces are coated with a polymer that does not interact strongly with other surfaces (hence, no sticking). Teflon®, developed by DuPont® in the 1940s, is most frequently used for this application. Teflon® is a polymer of tetrafluoroethylene, which is very hydrophobic, so water or any other substance (like food) doesn’t stick to it. More recently, other polymers have also been developed for this application. Thermolon® is a silicon oxide polymer with some similar properties to Teflon®, and EcoLon® is a nylon-based product that is reinforced with ceramics for toughness.
How does a microwave work?
Microwaves work by using a process called dielectic heating. The microwave surrounds your food in a field of electromagnetic radiation that is constantly changing directions. The polar molecules in your food, particularly water, align their dipoles with the direction of this applied field. The constant shifting of the direction of this field causes the polar molecules to tumble around, and this molecular motion warms your food.
What makes leaves change color in the fall?
Leaves are normally green because of chlorophyll. Chlorophyll absorbs blue and red wavelengths of light, so it appears green. This molecule is crucial for photosynthesis but when the days begin to get shorter as winter approaches, plants begin to produce less chlorophyll. As the level of this green chemical in leaves falls, we can start to see other highly colored molecules. In particular, carotenoids, which appear yellow, orange, or brown, start to become visible. These molecules are always present in the leaves, it’s just that the green color of chlorophyll dominates most of the year.
How does hand sanitizer work?
The active ingredient in hand sanitizer is usually an alcohol like isopropanol. This chemical is also used in antiseptic wipes and pads because of its ability to kill bacteria, fungi, and viruses.
How does soap work?
Soap molecules have polar end groups and long hydrophobic tails. In the presence of water, they arrange themselves into spheres called micelles. These structures can transport greasy particles in their interiors, helping remove the bits that water can’t remove on its own from your hands or clothes.
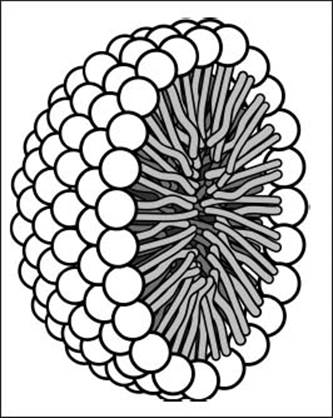
Soap molecules arrange themselves into micelles like this one (shown as a cross-section), which can move grease into the center of the structure.
Why does bleach kill everything?
The active ingredient in bleach, sodium hypochlorite (NaOCl), has a few ways of killing off microbes. One method involves causing particular proteins in the microbes to unfold, preventing their normal function and eventually killing the bacteria. Alternatively, bleach can disrupt the membrane that forms the outer shell of a bacteria. Since most bacterial membranes are very similar, bleach is very effective against a whole host of different types of bacteria. The human body actually produces hypochlorous acid (HOCl) itself to combat bacterial infections.
Iodine is useful as a disinfectant that can kill a wide variety of pathogens.
Why do you put iodine on cuts?
The iodine you buy at the drugstore is usually an ethanol solution of elemental iodine (I2). Iodine is a general disinfectant, meaning that it kills all sorts of pathogens, including spores, which are notoriously difficult to kill.
How do fireworks work?
Fireworks, those that explode in the sky, technically called pyrotechnic stars or aerial shells, have only a few essential components. Once the shell gets up in the air, the main event is fueled by aluminum metal or sometimes a mix of aluminum and magnesium metal. By themselves, these elements don’t burn quickly enough in the atmosphere and the flames they produce are just boring white. So this fuel component is mixed with another chemical that helps the aluminum burn (technically oxidize) faster. Different oxidizing compounds make different-colored flames when they react: purple (KNO3), blue (CsNO3), green (BaCl2), yellow (NaNO3), or red (SrCO3). There are lots of other components of modern fireworks (like gunpowder to help disperse these chemicals to make big shapes), but all of the chemistry is based on these simple reactions.
Why is ultraviolet light from the Sun potentially dangerous?
Most of us like being out in the Sun, but we’ve also all heard to be careful not to get sunburned since too much sun can cause serious problems like skin cancer, along with less serious problems like wrinkles and dry skin. The Sun can damage your skin because the ultraviolet rays from the Sun are relatively high-energy photons that can damage the elastin fibers in your skin. These can then lose their ability to go back to their original position after they are stretched and also lose their ability to heal as quickly when wounded or bruised.
As we discussed briefly in “Biochemistry,” the cause of cancers generally involve damage to the genetic material (DNA) of your cells, which interferes with their ability to replicate (or stop replicating) normally. With regard to cancers arising from too much exposure to ultraviolet radiation, DNA can be damaged in two ways. The first, probably more obvious, route is that the ultraviolet radiation could be absorbed by the DNA directly, causing changes in its chemical structure. The second possibility is that ultraviolet radiation can be absorbed by other molecules first, forming reactive, damaging radical species (like hydroxyl radicals or singlet oxygen), which then diffuse through cells and can damage DNA.
How does sunscreen work?
Sunscreens either reflect or absorb ultraviolet light from the Sun. To reflect the light sunscreens contain either titanium or zinc oxides, which are both very white solids (so all wavelengths of visible light are being reflected). To absorb light, sunscreens can contain organic chemicals that interact with harmful UV wavelengths. While almost all sunscreens use titanium and/or zinc oxides, the particular organic compounds that are used vary widely across brands and countries.
What is a CD made of?
All types of optical discs (CDs, DVDs, Blu-Ray® Discs, etc.) have basically the same components. The outer layer of clear polycarbonate plastic protects the inner layers that contain the data from damage. A layer of a highly reflective metal, usually aluminum, is used to reflect the laser that is used to read the data. The data itself is stored on another polycarbonate layer that contains teeny-tiny little pits. The pits are arranged in spiral tracks, just like a vinyl record (if you’ve ever seen one of those), and are about 100 nm deep and about 500 nm wide. The laser can detect the change in height by measuring changes in how the light is reflected.
Why is arsenic so poisonous?
Arsenic interrupts some of your body’s most basic and common biochemical pathways. Arsenic, particularly As3+ and As5+ oxides, interferes with the citric acid cycle and respiration (specifically reduction of NAD+ and ATP synthesis—see “Biochemistry”). If that weren’t enough to kill you, and it probably is, arsenic also boosts the level of hydrogen peroxide in your body, which causes another whole set of problems. Unfortunately, these toxic forms of arsenic are not only water soluble but can be found in well water from natural sources and man-made contamination from mining.
How do our brains tell time?
Until very recently, scientists presumed (and it’s not clear that there were many experiments to support this) that our brain had a stopwatch of sorts built into its machinery. By stopwatch, scientists meant some biological system that created some signal at regular intervals. If this were true, we would be equally good at estimating short and long periods of time, but that’s totally not the case. Humans are pretty awful at guessing how long extended periods of time are.
Instead, Dean Buonomano at UCLA proposed in 2007 that our brains tell time in a different way. To steal his analogy, imagine a rock being tossed into a lake, which creates a series of ripples in the surface of the water. If you were to throw a second rock in, the ripples from the two rocks would interact with each other. The pattern of this interaction depends on the time between the two rocks hitting the lake. The firing of neurons (the rocks hitting the lake) create these unique patterns in their signals—and neurons can use the different patterns of signals interacting to tell time between events. The wonderful piece of this theory is that it explains why we’re good at telling time over short durations, but not long ones—over a long time, the ripples in the water just fade away.
What chemical process leads to the formation of fossils?
There are of course many different types of fossils, but most form through some sort of mineralization process. What’s that? In water that has lots of dissolved minerals, after an organism dies, those minerals can slowly deposit in the tiniest of spaces within that organism, even within cell walls. Well-preserved fossils require the organism to be covered with sediment quickly after death (like on the bottom of a lake) so that the body doesn’t decay before the slower mineralization process can take place.
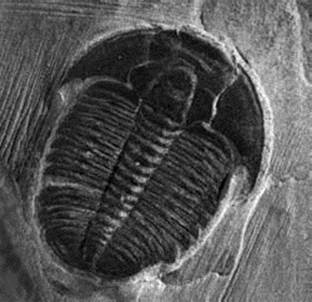
This fossil of an extinct trilobyte was formed millions of years ago when the animal was covered by sediments at the bottom of an ocean or lake. Over time, minerals replaced the decaying flesh and solidified into the shape of the original animal.
Is sodium laureth sulfate toxic?
You’re probably asking this because you’ve seen this chemical listed on your shampoo or toothpaste—and at the levels in those products, no, it is not toxic. This molecule is a surfactant, very similar to many soaps. And like most soaps, if you get them in your eyes, it hurts. The chemical is an irritant, but it does not cause cancer, like you might have heard.
What’s in toothpaste that makes our teeth cleaner?
If you get your teeth whitened at the dentist or buy some over-the-counter whitening products, the active ingredient is usually hydrogen peroxide. The hydrogen peroxide reacts with the colored molecules that are staining your teeth to remove them or at least make them colorless. This can be a slow reaction, so at the dentist’s office, sometimes they use a bright light to speed up the breakdown of the H2O2.
If you’re wondering about whitening toothpastes, those contain an abrasive compound that simply rubs the stain molecules off of your teeth—no fancy chemistry at all.
How does fluoride work in toothpaste?
Fluoride, usually in the form of sodium fluoride (NaF) in toothpaste, strengthens the enamel in your teeth. But how does it do that? Let’s back up a bit first.
Enamel is the outer layer of your teeth, and it’s made of a mineral called hydroxyapatite. It’s a calcium phosphate structure with one hydroxyl group (Ca5(PO4)3(OH)). This mineral dissolves in the presence of acids, which is exactly what bacteria generate when they metabolize sugars in your mouth. This is how cavities form when you drink soda.
Fluoride ions help rebuild your tooth’s enamel by replacing the hydroxyl group in the apatite mineral. The new mineral ((Ca5(PO4)3F), or fluorapatite, is more stable in the presence of acids, so your teeth are more resistant to decay.
What is the hardest material in your body?
Tooth enamel is the hardest material in your body. It’s even harder than bone!
Why do old library books begin to smell after sitting on shelves for years?
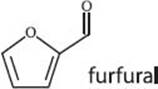
The smell of old books is due to hundreds of volatile organic compounds that form from the slow degradation of the book’s paper and other materials used in its construction. Acetic acid (vinegar) and furfural (smells like almonds) are two common chemicals ascribed to the smell of old books. Scientists can analyze the volatile compounds to identify the materials used without having to destroy a part of the historical document.
Why does the inhalation of helium make voices higher?
After you inhale helium your voice might sound higher, but the pitch (or frequency of the sound waves) is exactly the same. Your vocal cords vibrate at the same frequency because your body doesn’t adjust for the presence of a less-dense gas in your throat. What does change is the speed of sound in helium versus air—because helium has a lower molecular weight than air, the speed of sound is higher. You’ve probably heard this is because helium is less dense—that’s not technically correct, but let’s not go there.
So the speed of sound is faster, but why does that make your voice sound weird? The tone is actually identical; what’s different is the timbre. Specifically, the lower frequencies of your voice have less power, so your voice sounds squeaky—like a duck.
What is ink made of?
Inks can be very complicated mixtures, but two key ingredients are the pigment used to color the ink and the solvent used to dissolve (or at least suspend) the pigment particles. While modern inks come in every color imaginable for a host of different pen types, historically inks fell into one of two major categories.
The first is carbon-based inks. Residues from burning wood or oil, like soot, were used as coloring in these inks, which were suspended in the sap of the acacia tree (known today as gum arabic).
The other type of ink used historically is called iron gall. The iron was usually added in the form of iron sulfate (Fe2+SO42-); “gall” refers to gallotannic acid that was extracted from growths, or galls, on oak trees. Iron gall ink slowly darkens as the iron ions undergo oxidation from Fe2+ to Fe3+. The acidity of the ink solution can cause damage to the paper it is used on, so preservation of historical documents that were written with iron gall ink is challenging.

Pencil “lead” is actually made of graphite, a form of carbon that is easy to write and draw with, as well as easy to erase.
Why is graphite so good to write with?
Graphite is an attractive chemical for writing for a few reasons. Graphite, unlike most inks, is not dissolved by water or affected by moisture, but it’s easy to erase.
Fun fact: We commonly refer to the graphite in pencils as “pencil lead,” but there’s no lead (Pb) in there. The Romans did use lead for writing, but the practice didn’t make it much farther in history than that. The paint on the outside of pencils did, however, contain lead up through the 1900s.
How do thermometers work?
There are actually many kinds of thermometers available, but let’s talk about the two types that you probably have in your home.
The first type is a glass tube filled with either alcohol or mercury. As the temperature rises the volume of the liquid also increases, so it rises up the tube. The height of the liquid is calibrated with a scale so you can read the temperature value easily.
The second type is known as a bimetallic strip thermometer. While you’ve probably never heard this name before, you’ve likely used this type of thermometer. They are the most common models of thermostats (before they went digital), used as meat thermometers, oven thermometers, and the little thermometers that you see baristas using at coffee shops when they’re steaming milk for your latte. You can tell from the name bimetallic that there are two metals involved here (usually steel and copper). In order to measure temperatures, these two metals need to expand at different rates when they are heated. If you make a strip of these two metals and wind that strip up into a coil, the difference in their thermal expansion will cause the coil to wind tighter or unwind as the temperature changes (depending on which side of the coil you place the material that expands more). This coil then turns a needle to indicate that the temperature is rising or falling.
Why do we need to sleep?
Even though this sounds like a pretty straightforward question, the truth is that scientists still don’t know the whole answer! There are several theories, including the idea that sleep promotes restorative functions, that sleep promotes development and structural changes in the brain, that we sleep because it helps us to conserve energy, or simply that it may have been safer for our evolutionary predecessors to remain inactive at night. While there is evidence to support each of these (and other) theories, it remains a very difficult question to answer conclusively.
How do bees make honey?
The first step is searching out a flower to pick up some nectar. Nectar is a mixture of sugar and water. Specifically, the sugar in nectar used to make honey is sucrose, a dis-accharide (see “Biochemistry”). A honeybee produces enzymes in its body that can break down the sucrose into monosaccharides, fructose, and glucose as well as gluconic acid. These sugars are the primary constituents of honey. Most of the water evaporates, which is what makes honey so viscous and sticky.
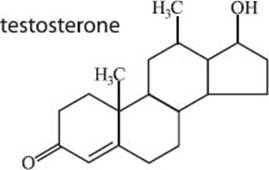
What is testosterone?
Testosterone is a steroid hormone molecule that is found in human males and females as well as many other species. In humans testosterone serves as the primary male sex hormone, and it plays a crucial role in the development of the male reproductive system. In males it is secreted from the tesisticles, and in females it is secreted from within the ovaries. Males use significantly more testosterone than females, and for this reason males produce testosterone at about twenty times the rate of females. Oddly enough, males are also less sensitive to testosterone than females.
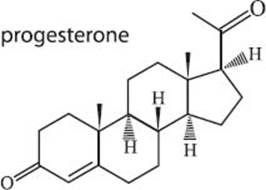
What is progesterone?
Progesterone is a steroid hormone that is crucial to regulating the female menstrual cycle and pregnancy cycle in human females and in some other species as well. It is produced in the ovaries and the adrenal glands and is stored in fat tissue.
What causes the tides in the ocean?
The ocean tides are caused by the gravitational forces between the Earth, the Sun, and the Moon, along with the centrifugal force imposed by the Earth’s rotation. As the Earth rotates, and as the three bodies move relative to one another, the gradual and recurring shifts in the balance of gravitational forces cause the water in the oceans to tend to move toward one coast or the other.
How does fertilizer work?
Fertilizers are used to get elements that plants need into the soil when the natural environment doesn’t provide sufficient quantities. The typical elements are nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg), and sulfur (S). They’re actually labelled by the elements they contain. Next time you’re at a garden store, look for “NPK” or “NPKS” on a bag of fertilizer. The numbers after these codes tell you the weight percent of these elements in the bag.
What nutrients do plants obtain from the soil?
There are thirteen mineral nutrients that plants obtain from the soil, and these are divided into the categories of macronutrients and micronutrients. The primary macronutrients include nitrogen, phosphorus, and potassium. Plants require these primary macronutrients in relatively large quantities and deplete them from the soil more rapidly than others. The secondary macronutrients include calcium, magnesium, and sulfur. Micronutrients are required in smaller quantities than the macronutrients, and these include boron, copper, iron, chloride, molybdenum, manganese, and zinc.
What happens when something biodegrades in a landfill?
Biodegradation describes the process by which microorganisms consume a material and convert it to compounds that are found in nature. This process can happen either aerobically (with oxygen involved in the process), or anaerobically (without oxygen involved in the process). A related term is “compostable”—this specifically indicates that a material will biodegrade/break down when it is placed in a compost pile.
What is smoke?
Smoke is a cloud of particles given off by a material that undergoes combustion. Its chemical composition will vary depending on what material is being burned. Smoke may consist of hydrocarbons, haloalkanes, hydrogen fluoride, hydrogen chloride, and a variety of sulfur-containing compounds, among others. These compounds can vary widely in their toxicity, so the severity of the health hazards associated with the smoke from a fire will depend on what is being burned.
How much salt (NaCl) is in the average human body?
The average adult human body contains about 250 g, or roughly half of a pound, of salt.
What fraction of the oxygen on Earth is produced by the Amazon rainforest?
It is estimated that about 20% of the diatomic oxygen (O2) on Earth at a given time was produced by the Amazon rainforest!
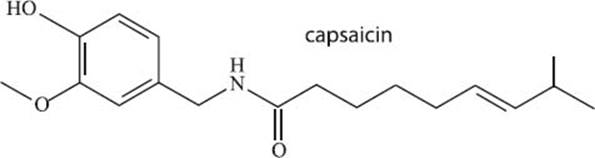
What makes hot peppers so spicy?
The molecule that makes hot peppers so spicy is called capsaicin (see its chemical structure below). This molecule behaves as an irritant to humans and other mammals, but some of us still really like its flavor!
What is blood doping in sports?
Blood doping is the act of artificially increasing the number of red blood cells in a person’s blood for the purpose of improving athletic performance. This works based on the fact that red blood cells are responsible for carrying oxygen to muscles, and thus more red blood cells can provide more endurance against muscle fatigue. This was originally done by transfusions of red blood cells, either from another person or by collecting and storing a person’s own red blood cells to be used later. In the past couple of decades, a new type of blood doping has come about. This is based on the hormone erythropoietin, which stimulates the body to produce red blood cells. Erythropoietin is produced artificially in mass quantities and is commonly used to treat anemia, but is also sometimes used by athletes for the purposes of blood doping.
What makes carbon monoxide dangerous?
When carbon monoxide is inhaled, it is readily absorbed through the lungs and into the bloodstream where it binds to the Fe center in hemoglobin (see also “Biochemistry”). Unfortunately, hemoglobin binds to carbon monoxide much more strongly than it does to oxygen, so carbon monoxide rapidly interferes with the ability of hemoglobin to deliver oxygen throughout your body. If this happens, then your muscles and your brain will begin to run out of oxygen, similar to what happens when a person is drowning! Carbon monoxide is a colorless, odorless gas, which makes it difficult to detect unless you have a carbon monoxide detector around. Carbon monoxide levels of 100 parts per million or higher can be hazardous or fatal to humans.
Why is the water in the ocean salty?
One source of salts in the ocean comes from minerals on land dissolving in rainwater and streams, which eventually make their way into the ocean. Since salts do not tend to evaporate along with the water in the ocean, their concentration can build up over time. Another source of salts in the ocean are hydrothermal vents; seawater can flow into these vents, where it becomes warm and dissolves minerals before flowing back out. Underwater volcano eruptions also contribute to the presence of minerals in seawater.
The majority of the salt ions in the ocean are sodium and chloride—these make up about 90% of dissolved ions in seawater. The remainder of the ions present are mainly magnesium, sulfate, and calcium. The concentration of salts in seawater is fairly high, and, on average, seawater is about 3.5% salt by weight.
Poisoning from carbon monoxide can result in brain damage, damage to the endocrine system, to the nervous system, and to the heart and other organs. It represents the leading cause of accidental poisoning-related death and poisoning-related injury globally. We should mention that even if a person survives carbon monoxide poisoning, there may be long-lasting effects. Early symptoms of carbon monoxide poisoning can include headaches or nausea, and the treatment for carbon monoxide poisoning typically involves having a person breathe 100% O2 (recall that air only contains ca. 20% O2), so that O2 can more competitively bind hemoglobin to replace carbon monoxide.
What is permafrost?
Permafrost is soil that is below the freezing point of water for two consecutive years or more. Most of it is located near the North or South Poles or at very high altitudes.
What chemicals are commonly found in mosquito repellent?
A chemical known as N,N-diethyl-meta-toluamide (more commonly known as DEET) is the most commonly used insect/mosquito repellent. This chemical can be applied directly to the skin to prevent mosquito and other bug bites. It is believed to work because mosquitoes dislike the smell of DEET, so they try to stay away from it. In some cases DEET can be an irritant and, in very rare cases, it has been associated with more serious health problems such as seizures.