Cracking the SAT Chemistry Subject Test
Part IV
Practice SAT Chemistry Subject
Tests and Answers and Explanations
Chapter 19
Practice SAT Chemistry Subject Test 3
Click here to download a PDF of the Practice SAT Chemistry Subject Test 3.
PRACTICE SAT CHEMISTRY SUBJECT TEST 3
You are about to take the third practice SAT Chemistry Subject Test. After answering questions 1–32, which constitute Part A, you'll be directed to answer questions 101–116, which constitute Part B. Then, begin again at question 33. Questions 33–69 constitute Part C.
When you're ready to score yourself, refer to the scoring instructions and answer key on this page. Full explanations regarding the correct answers to all questions start on this page.
CHEMISTRY SUBJECT TEST
MATERIAL IN THE FOLLOWING TABLE MAY BE USEFUL IN ANSWERING THE QUESTIONS IN THIS EXAMINATION.
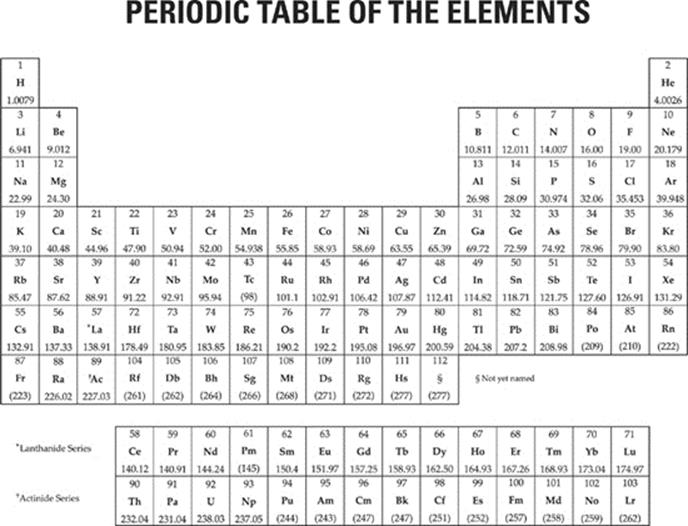
Click here to see full page view
CHEMISTRY SUBJECT TEST 3
Note: For all questions involving solutions and/or chemical equations, assume that the system is in pure water unless otherwise stated.
Part A
Directions: Each set of lettered choices below refers to the numbered statements or questions immediately following it. Select the one lettered choice that best fits each statement or answers each question, and then fill in the corresponding oval on the answer sheet. A choice may be used once, more than once, or not at all in each set.
Questions 1–5 refer to the following.
(A) Carbon
(B) Nitrogen
(C) Oxygen
(D) Neon
(E) Argon
1. Is the third most abundant gas in Earth's atmosphere
2. At standard conditions, has an allotrophic form that is a good electrical conductor
3. Regardless of its electron configuration, it must always be paramagnetic when it's a single, neutrally charged atom
4. The key element delivered in soil fertilizer
5. Allotrope of this element is the primary absorber of UV solar radiation in Earth's atmosphere
Questions 6–9 refer to the following.
(A) Chemical pH indicator
(B) Acid/base buffer
(C) Anhydrous solution
(D) Hypotonic solution
(E) Supersaturated solution
6. A conjugate acid/base pair with differing spectral absorbencies
7. An example of a solution not in equilibrium
8. Term used in reference to an aqueous solution's osmotic pressure
9. Addition of water to this solution will not change [H3O+]
Questions 10–14 refer to the following.
(A) Standard voltaic potential
(B) Entropy
(C) Enthalpy
(D) Reaction rate
(E) Gibbs free energy
10. Increased with the addition of a catalyst
11. Abbreviated as “H”
12. A property that must decrease when a gas condenses into a liquid
13. Is always positive for a spontaneous chemical reaction
14. Is zero for a crystalline solid that is elementally pure at 0 K
Questions 15–19 refer to the following.
(A) Alkali metals
(B) Alkaline earth metals
(C) Noble gases
(D) Halogens
(E) Transition metals
15. The most unreactive family of elements
16. Form negative ions in an ionic bond
17. Consist of atoms that have valence electrons in a d subshell
18. Exist as diatomic molecules at room temperature
19. Members possess the lowest first ionization energy in their respective period
Questions 20–24 refer to the following.
(A) N2
(B) KI
(C) CCl4
(D) AgNO3
(E) CaCO3
20. A product of a neutralization of a strong acid with a strong base
21. A volatile covalent liquid at 25°C and 1 atm
22. Releases a gas with the addition of dilute acid
23. Forms a white precipitate when added to a solution of NaCl
24. Treatment of the dry solid with a mild oxidizing agent produces a purple solid
Questions 25–28 refer to the following.
(A) Gamma decay
(B) Nuclear fusion
(C) Alpha decay
(D) Positron emission
(E) Nuclear fission
25. Is the principle reaction responsible for the energy output of the sun
26. Is a nuclear process that results in no change in the mass number and atomic number of a nuclide
27. Responsible for most helium found on Earth
28. The nuclear process that transmutes uranium-238 into thorium-234
Questions 29–32 refer to the following.
(A) 0.1 M MgCl2
(B) 0.1 M HClO4
(C) 0.1 M NH4OH
(D) 0.1 M KOH
(E) 0.1 M LiNO3
29. Has a pH of 13
30. The solution with the lowest freezing point temperature
31. The solution with the highest boiling point temperature
32. Indicates a red flame when ionized with a Bunsen burner
PLEASE GO TO THE SPECIAL SECTION LABELED CHEMISTRY AT THE LOWER RIGHT-HAND CORNER OF THE ANSWER SHEET YOU ARE WORKING ON AND ANSWER QUESTIONS 101–116 ACCORDING TO THE FOLLOWING DIRECTIONS.
Part B
Directions: Each question below consists of two statements, I in the left-hand column and II in the right-hand column. For each question, determine whether statement I is true or false and whether statement II is true or false, and fill in the corresponding T or F ovals on your answer sheet.Fill in oval CE only if statement II is a correct explanation of statement I.
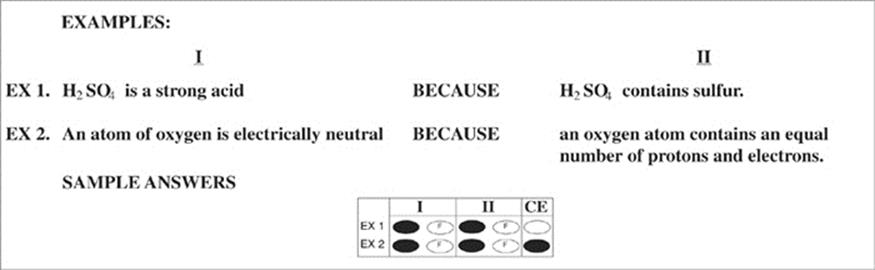
![]()
101. ![]()
102. 
103. ![]()
104. ![]()
105. ![]()
106. ![]()
107. ![]()
108. ![]()
109. ![]()
110. ![]()
111. ![]()
112. ![]()
113. ![]()
114. 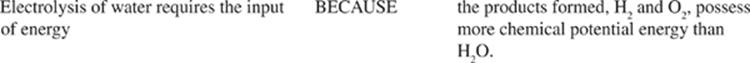
115. ![]()
116. ![]()
RETURN TO THE SECTION OF YOUR ANSWER SHEET YOU STARTED FOR CHEMISTRY AND ANSWER QUESTIONS 33–69.
Part C
Directions: Each of the questions or incomplete statements below is followed by five suggested answers or completions. Select the one that is best in each case and then fill in the corresponding oval on the answer sheet.
33. Choose the answer below that accurately describes the correct molecular shape for the molecule XeOF4.
(A) Tetrahedral
(B) Trigonal pyramidal
(C) Trigonal bipyramidal
(D) Square pyramidal
(E) Flat
34. For the radioactive atom 99Tc, what is the correct number of protons and neutrons?
(A) 43 protons and 56 neutrons
(B) 43 protons and 99 neutrons
(C) 56 protons and 43 neutrons
(D) 56 protons and 99 neutrons
(E) Cannot be determined
35. Which one of the following acids is NOT strong?
(A) HCl
(B) HBr
(C) HNO3
(D) H3PO4
(E) H2SO4
36. Identify the equation used to determine the amount of heat required to melt 10 grams of ice.
(A) Q = mCspΔT
(B) Q = nΔH
(C) KE = ![]() mv2
mv2
(D) PE = mgh
(E) PV = nRT
37. Identify the correct ground state electron configuration for Cr.
(A) [Ar] 3s23d4
(B) [Ar] 3s23d5
(C) [Ar] 4s23d5
(D) [Ar] 4s23d4
(E) [Ar] 4s13d5
38. What is the hydroxide concentration for a solution with a pH of 10 at 25°C?
(A) 10−14 M
(B) 10−10 M
(C) 10−7 M
(D) 10−4 M
(E) 10−1 M
39. Five hundred milliliters of solution of 0.1 M NaBr has how many milligrams of bromine?
(A) 200 mg
(B) 400 mg
(C) 2,000 mg
(D) 4,000 mg
(E) 20,000 mg
40. According to the ideal gas law, what is the approximate volume that will be occupied by 0.5 mole of an ideal gas at 30°C and 3 atm pressure (gas constant R = 0.0821 L•atm/mol•K)?
(A) Less than 1 L
(B) 5 L
(C) 10 L
(D) 15 L
(E) More than 20 L
41. Given that ΔG = ΔH – TΔS, how is the spontaneity of an endothermic reaction expected to change with decreasing T?
(A) Becomes less spontaneous
(B) Becomes more spontaneous
(C) Does not change
(D) Decreases at first but then increases
(E) Insufficient information to make a conclusion
42. Identify the element with the greatest first ionization energy.
(A) Ce
(B) C
(C) Cl
(D) Ca
(E) Cs
43. Identify the molecule/ion with the greatest potential to act as a Lewis acid.
(A) CH3+
(B) CN–
(C) NH3
(D) BF4–
(E) CO2
2 Ca3(PO4)2 + 6 SiO2 + 10 C → P4 +…CaSiO3 + 10 CO
44. Which coefficient balances the reaction given above?
(A) 2
(B) 4
(C) 5
(D) 6
(E) 8
45. A 100-milliliter solution containing AgNO3 was treated with excess NaCl to completely precipitate the silver as AgCl. If 5.7 g AgCl was obtained, what was the concentration of Ag+ in the original solution?
(A) 0.03 M
(B) 0.05 M
(C) 0.12 M
(D) 0.30 M
(E) 0.40 M
46. Identify which of the following statements is FALSE.
(A) The vapor pressure of a liquid decreases with increasing atmospheric pressure.
(B) The value of an equilibrium constant is dependent on temperature.
(C) The rate of a spontaneous reaction cannot be determined solely by its Gibbs free energy.
(D) During a phase transition, the temperature of a substance must be constant.
(E) The addition of a catalyst to a reaction at equilibrium has no net effect on the system.
47. Which of the following compounds would be expected to have the greatest lattice binding energy?
(A) LiNO3
(B) LiF
(C) KI
(D) NH4Br
(E) CsNO3
48. The daughter nucleus formed when 18F undergoes positron emission is
(A) 14N
(B) 16O
(C) 18O
(D) 19F
(E) 20Ne
49. Which of the following reactions produces a yellow precipitate?
(A) NaOH(aq) + HCl(aq) → NaCl(s) + H2O
(B) NaOH(aq) + BaCl(aq) → BaOH(s) + NaCl(aq)
(C) Pb(NO3)2(aq) + 2KI(aq) → 2KNO3(aq) + PbI2(s)
(D) CuO(s) + Mg(s) → Cu(s) + MgO(s)
(E) 4Fe + 3O2 → 2Fe2O3
Zn(s)|ZnCl2(aq)|Cl–(aq)|Cl2(g)|C(s)
50. In the electrochemical cell described by the cell diagram above, what reaction occurs at the anode?
(A) Zn → Zn2+ + 2e
(B) Zn2+ + 2e → Zn
(C) Cl2 + 2e → 2Cl−
(D) 2Cl− → Cl2 + 2e
(E) Zn + Cl2 → ZnCl2
51. Given the reaction A → B + C, where ΔHrxn is negative, what effect would increasing the temperature (at constant pressure) have on the system at equilibrium?
(A) No change
(B) Cannot be determined
(C) Shift to the right
(D) Shift to the left for K < 1 and to the right for K >1
(E) Shift to the left
52. An unknown acid solution was presumed to be either HCl or H2SO4. Which one of the following salt solutions would produce a precipitate when added to H2SO4 but not when added to HCl?
(A) LiNO3
(B) NH4NO3
(C) CsNO3
(D) Ba(NO3)2
(E) AgNO3
Ca3(PO4)2(s) ![]() 3 Ca2+(aq) + 2 PO43−(aq)
3 Ca2+(aq) + 2 PO43−(aq)
53. What is the equilibrium expression for the dissolution of Ca3(PO4)2 where the above is true?
(A) Ksp = [Ca2+]3[PO43−]2
(B) Ksp = [Ca2+]2[PO43−]3
(C) Ksp = [Ca2+][PO43−]/[Ca3(PO4)2]
(D) Ksp = [Ca2+]3[PO43−]2/[Ca3(PO4)2]
(E) Ksp = [Ca2+]2[PO43−]3/[Ca3(PO4)2]
54. Which of the following represents a conjugate acid/base pair?
(A) Na+/Cl–
(B) HCl/H+
(C) H2CO3/CO32−
(D) NH3/NH4+
(E) K+/OH–
55. An unknown solution having a pH of 3.5 was titrated with 0.1 M NaOH. Analysis of the resulting titration curve showed a single equivalence point at pH 7. Therefore, which of the following could be the unknown solute in the initial solution?
(A) HF
(B) HCl
(C) LiOH
(D) NH3
(E) H2SO4
56. Acid/base titration experiments could be used to determine all of the following directly EXCEPT
(A) the acid concentration of an acidic solution
(B) the alkalinity of a basic solution
(C) the pKa of an unknown weak acid
(D) whether an unknown acid is monoprotic or polyprotic
(E) the molecular weight of an unknown acid or base
57. What is the correct term for the phase change from gas directly to solid?
(A) Deposition
(B) Sublimation
(C) Liquefaction
(D) Fusion
(E) Vaporization
58. What is the correct name for a straight-chained organic compound with the molecular formula C3H8?
(A) Methane
(B) Ethane
(C) Methylethane
(D) Propane
(E) Isopropane
59. If the pH of a solution is changed from 1 to 3 with the addition of an antacid, what percentage of [H+] was neutralized?
(A) 2%
(B) 10%
(C) 20%
(D) 90%
(E) 99%
60. Which of the following statements is the most accurate with regard to the significance of Avogadro's number, 6.02 × 1023?
(A) It is the conversion factor between grams and atomic mass units.
(B) It is a universal physical constant just as the speed of light.
(C) It is the number of particles that is required to fill a 1-liter container.
(D) It is the inverse diameter of an H atom.
(E) It is the number of electrons in the universe.
Questions 61–64 refer to the following data at standard conditions.
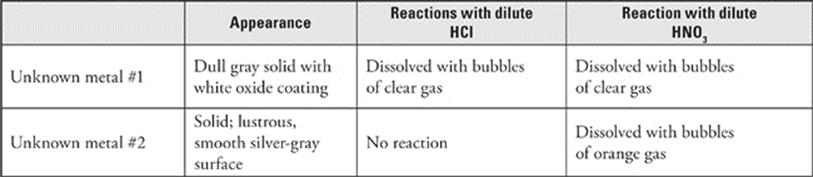
61. Unknown metal #1 could be
(A) mercury
(B) copper
(C) zinc
(D) iron
(E) silver
62. Unknown metal #2 could be
(A) carbon
(B) copper
(C) zinc
(D) sodium
(E) silver
63. The addition of dilute HCl to unknown metal #1 produced a transparent gas. What is the likely identity of this gas?
(A) Cl2
(B) H2
(C) O2
(D) CO2
(E) NO2
64. The addition of dilute HNO3 to unknown metal #2 produced an orange gas. What is the likely identity of this gas?
(A) Cl2
(B) H2
(C) O2
(D) CO2
(E) NO2
65. Which of the following solutions is the product of the neutralization reaction between 10 ml 0.2 M KOH and 10 ml 0.2 M HI?
(A) 0.1 M KI3
(B) 0.1 M KI
(C) 0.2 M KI
(D) 0.4 M KI
(E) 0.4 M HOH
66. Which of the following is true regarding an Ne atom with a mass number of 20 and an O2– ion with a mass number of 16?
(A) They contain the same number of protons.
(B) They contain the same number of neutrons.
(C) They contain the same number of protons plus neutrons.
(D) They are isoelectronic.
(E) They are isomers.
67. Which of the following statements is NOT correct regarding chemical catalysts?
(A) They are not consumed during the chemical reaction.
(B) They cannot make nonspontaneous reactions occur.
(C) They do not have to be the same phase as the reactant molecules.
(D) They shift equilibrated reactions to the product's side.
(E) Enzymes are biological catalysts.
68. Most elements are solids at 25°C and 1 atm pressure, the exception being the 11 elements that are gases and 2 that are liquids. What 2 elements are liquids?
(A) Hg and Br
(B) Hg and I
(C) Ag and Kr
(D) Au and Kr
(E) Pt and Co
69. A student conducted an experiment and obtained three values during three repetitive trials: 1.65, 1.68, 1.71. Later, the student discovered that the true value was 2.37. In contrast to the real value, the experimental results should be characterized as
(A) not accurate and not precise
(B) accurate but not precise
(C) not accurate but precise
(D) accurate and precise
(E) accurate, precise, but unreliable
STOP
If you finish before time is called, you may check your work on this section only.
Do not turn to any other section in the test.
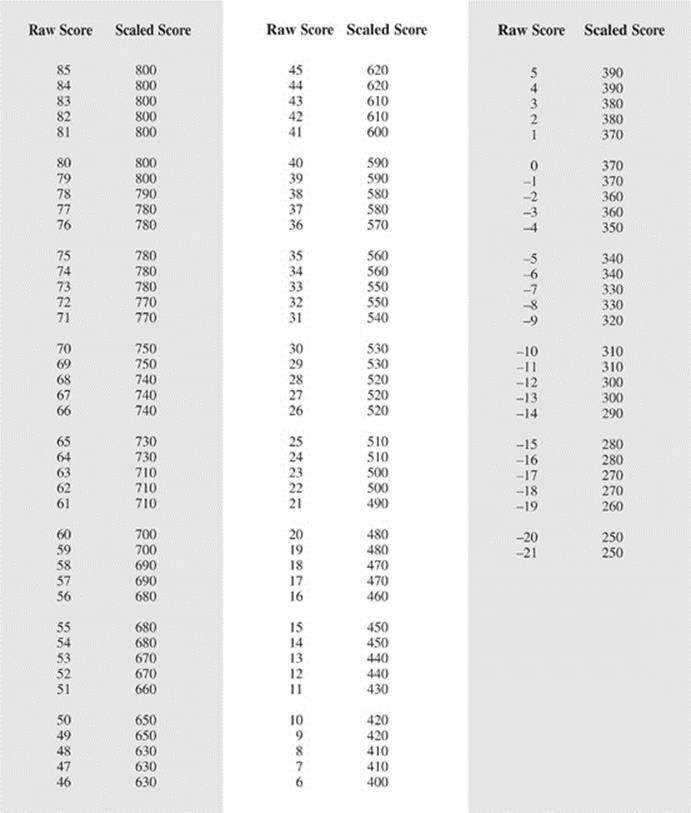
HOW TO SCORE THE PRINCETON REVIEW PRACTICE SAT CHEMISTRY SUBJECT TEST
When you take the real exam, the proctors will collect your test booklet and bubble sheet and send your answer sheet to New Jersey, where a computer looks at the pattern of filled-in ovals on your answer sheet and gives you a score. We couldn't include even a small computer with this book, so we are providing this more primitive way of scoring your exam.
Determining Your Score
STEP 1
Using the answer key on the next page, determine how many questions you got right and how many you got wrong on the test. Remember: Questions that you do not answer don't count as either right or wrong answers.
STEP 2
List the number of right answers here.
(A) ________
STEP 3
List the number of wrong answers here. Now divide that number by 4. (Use a calculator if you're feeling particularly lazy.)
(B) ________ ÷ 4 = (C) ________
STEP 4
Subtract the number of wrong answers divided by 4 from the number of correct answers. Round this score to the nearest whole number. This is your raw score.
A) ________ – (C) ________ = ________
STEP 5
To determine your real score, take the number from Step 4 above, and look it up in the left column of the Score Conversion Table on this page; the corresponding score on the right is your score on the exam.
ANSWERS TO THE PRINCETON REVIEW PRACTICE SAT CHEMISTRY SUBJECT TEST 3
THE PRINCETON REVIEW PRACTICE SAT CHEMISTRY SUBJECT TEST SCORE CONVERSION TABLE