Cracking the SAT Chemistry Subject Test
Part II
Subject Review
Chapter 9
Solutions
A solution is a mixture of a solute (the substance of which there is less) and a solvent (that of which there is more). Generally, problems dealing with solutions tend to focus on a few key areas. This chapter will focus on understanding solubility, as well as what factors, both external (temperature and pressure) and internal (nature of the solute and solvent) affect it. It will also deal with some specific reactions between solutions and on how addition of a solute changes the freezing point and melting point of a solution (colligative properties).
MEASURING CONCENTRATIONS
The most commonly used unit for concentration is molarity. Its symbol is M. Molarity is a measure of the number of moles of solute dissolved per liter of solution (volume).
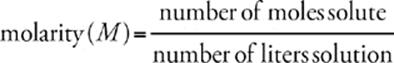
Molality, another fairly common unit for concentration, is a measure of the number of moles of solute dissolved per kilogram of solvent (mass). Its symbol is m.
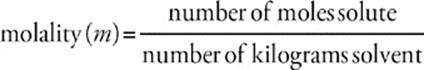
To help you distinguish between the two, think of molarity as moles of dissolved solute per liter of solution and think of molality as moles of dissolved solute per kilogram of solvent.
Solubility and Saturation
Suppose you take a glass of water and add table salt to it. The table salt dissolves. Suppose you keep adding table salt to it. After a while, the table salt doesn't dissolve, it just sits at the bottom of the glass. At that point the water is saturated with table salt. Another way to describe this is to say that the table salt has reached the limit of its solubility in water.
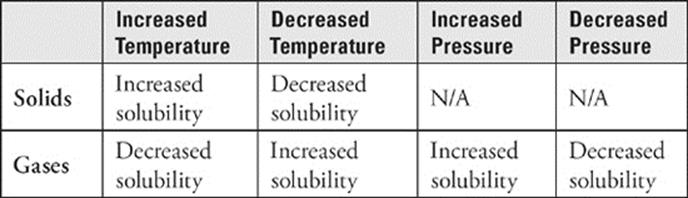
The temperature of the solvent affects solubility. Generally, a solid solute is more soluble in a liquid solvent at higher temperatures and less soluble at lower temperatures. If we took the glass of water and heated it, some of the table salt that hadn't dissolved would dissolve. The increased temperature increases the solubility of table salt in water.
Substances that are held together by ionic bonds (such as table salt, NaCl) are generally soluble in water.
Other solutes are completely insoluble. For instance, if you place a pat of butter in a glass of water, it won't dissolve, ever, even if you heat it. This illustrates an important general principle; polar solutes such as NaCl and HCl dissolve in polar solvents such as water, and nonpolar solutes dissolve in nonpolar solvents. Butter, which is a fat, is nonpolar. Just remember: “Like dissolves like.”
The solubility of gases in water is quite different from that of solids. Think about a bottle of soda; its carbonation is the result of dissolved carbon dioxide gas. Once the bottle has been opened, should you store it where it's warm or cold to prevent it from going flat? You should store it where it's cold, of course. The CO2 gas is more soluble in water at lower temperatures, and flat soda is simply soda after its CO2 has diffused out into the air. This is typical of the solubility of gases in water. One more thing about the solubility of gases in water: the higher the pressure, the more soluble the gas. Again consider soda, bottled under pressure. Once you open the bottle, the pressure over the soda decreases, and CO2 starts to come out of solution.
Dissociation and Electrolytes
When an ionic substance (NaCl, KCl, CaBr2, or CuSO4) dissolves in water, its bonds break, and ions are released into solution. For instance, when KCl dissolves in water, K+ ions and Cl− ions dissociate into solution.
The dissociation of ionic compounds always creates an equal number of positive and negative charges in solution. One mole of NaCl will dissociate into 1 mole of Na+ ions and 1 mole of Cl− ions, and 1 mole of CaBr2 will dissociate into 2 moles of Br− ions and 1 mole of Ca2+ ions. Both of these solutions have equal amounts of positive and negative charges, and both are therefore neutral.
Although these solutions are neutral, the presence of charged particles—;ions—;enables the solution to conduct electricity. This is why ionic solutions are also called electrolytic solutions, and we call the ions electrolytes.
BOILING POINT ELEVATION AND FREEZING POINT DEPRESSION
When a solute is dissolved in a liquid solvent, the solvent's boiling point is raised, its freezing point is lowered, and its vapor pressure is lowered. By how much? The change in boiling point (∆Tb) or freezing point (∆Tf ) is always equal to a constant (k) times the number of moles of dissolved particles of solute per kilogram of solvent.
∆T = kmi
The value k is different for different solvents; however, for all liquid solvents, the extent of boiling point elevation or freezing point depression is directly proportional to the molality of the solute. It is also directly proportional to the number of dissolved particles, which we denote by i. For example, when we dissolve NaCl in water, it dissociates into Na+ ions and Cl− ions. For every mole of NaCl we dissolve, we get 1 mole of Na+ and 1 mole of Cl−, for a total of 2 moles of dissolved particles. For NaCl, then, i = 2. If we dissolve 1 mole of sucrose (table sugar), however, the sugar molecules dissolve but don't dissociate, so we get only 1 mole of dissolved particles, and i = 1.
Now, suppose we take the same amount of water and add 1 mole of KCl. KCl is an ionic substance; it dissociates in solution. Each unit of KCl produces two dissolved particles: 1 K+ ion and 1 Cl− ion. The boiling point elevation and freezing point depression will be twice as great as they would be in the case of the dissolution of sucrose.
Let's Look at That Equation Again
ΔT = kmi
k = constant that depends on solvent
m = molality = 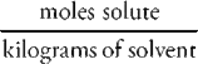
i = whole number equaling the number of particles a substance dissolves into
Again, why is this? Boiling point elevation and freezing point depression are directly proportional to the number of particles dissolved in a solution, but independent of the type of particle (i.e., sucrose and KCl equally affect melting point and freezing point at the same molality). Sucrose doesn't dissociate and KCl does. When 1 mole of sucrose is dissolved in water, it yields 1 mole of dissolved particles. When 1 mole of KCl is dissolved in water, it yields 2 moles of dissolved particles. If the sucrose elevated the water's boiling point by 0.5°C, the KCl would raise it by 1°C. If the sucrose depressed the water's freezing point by 2°C, the KCl would depress it by 4°C. Remember the relationship of proportionality we've just described.
Boiling Point
Elevation and Freezing
Point Depression
These points depend
only on the type of
solvent and the number
of solute particles.
There are many practical applications of boiling point elevation and freezing point depression. For example, spreading calcium chloride onto roadways during snowstorms makes it harder for ice to form on them (since the freezing point of water is made lower).
PRECIPITATION REACTIONS
Okay, so now you know that when ionic solids are dissolved in water, they dissociate. But what happens when soluble ions in separate solutions are mixed together, and they form an insoluble compound? Well, the product of this type of reaction will result in a solid substance that settles out of solution, called a precipitate. One example of this is the reaction between lead nitrate and potassium iodide.
2KI(aq) + Pb(NO3)2(aq) → 2KNO3(aq) + PbI(s)
Each of the reactants in this reaction is an ionic compound that's colorless in solution, but when they're combined, they react to form a product, lead iodide, which precipitates out of solution as a yellow solid. In this reaction, the anions and cations of the reactants are exchanged in a double replacement reaction, which typically results in the formation of a precipitate. But how do you know when a precipitation reaction will proceed? The solubility rules tell you which ionic compounds are soluble in water and which are not and enable you to make predictions about whether certain ions will react with one another to form a precipitate.
The Solubility Rules
• Most silver, lead, and mercury salts are INSOLUBLE except their nitrates and perchlorates.
• Most hydroxides (OH−) are INSOLUBLE except those of alkali metals and barium.
• All nitrates (NO3−) and perchlorates (ClO4−) are SOLUBLE.
• All alkali metal and ammonium (NH4+) compounds are SOLUBLE.
Enough said! Now review what we've said about solutions, and try the following set of questions. Answers can be found in Part III.
Question Type A
Question 9–11 refer to the following.
(A) Nitrogen dioxide, NO2(g)
(B) Iodine, I2(s)
(C) Glucose, C6H12O6(s)
(D) Naphthalene, C10H8(s)
(E) Calcium oxide, CaO(s)
9. Yields an electrolytic solution upon dissolution in water
10. Solubility in water increases as temperature is decreased
11. Produces the greatest boiling point elevation per mole dissolved into 1 L of water
Question Type B
![]()
106. 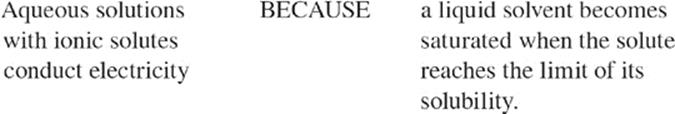
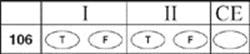
107. 

Question Type C
27. Which of the following will most likely increase the solubility of NaCl in water?
(A) Reducing the temperature of the water
(B) Raising the temperature of the water
(C) Reducing the molality of the solution
(D) Raising the molality of the solution
(E) Raising the molarity of the solution
34. Which of the following would most likely give a sample of water the capacity to conduct electricity?
(A) Reducing the temperature of the water
(B) Raising the temperature of the water
(C) Removing all electrolytes from the water
(D) Dissolving a nonionic substance in the water
(E) Dissolving CaCl2 in the water
45. Aqueous solutions of barium chloride and sodium sulfate react to form -------, an insoluble white solid.
(A) BaSO4(s)
(B) Na2SO4(s)
(C) BaCl2(s)
(D) NaCl(s)
(E) BaNa2SO4(s)
Summary
· Molarity is a measure of concentration and is given by
molarity = M = ![]()
· Molality is another measure of concentration used to determine boiling point elevation and freezing point depression. It is given by
molality = m = 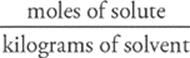
· Solubility refers to the degree to which a given solute will dissolve in a given solvent.
· Solubility of solids in water increases with increasing temperature.
· Solubility of gases in water decreases with increasing temperature.
· Solubility of gases in water increases with increasing pressure.
· When ionic substances dissolve, the ionic bonds are broken and the substance dissociates into free moving positive and negative ions. Such ions are called electrolytes; such a solution is called electrolytic and will conduct electricity.
· Boiling point elevation and freezing point depression are given by
ΔT = kmi
where k is a constant dependent on the solvent, and m is molality.
· Boiling point elevation and freezing point depression depend only on the type of solvent and the number of solute particles, but not the type of solute particles.
· For different solutions with the same molality, the boiling point elevation and freezing point depression will be greatest for the solute that dissociates into the greatest number of particles.
· In a precipitation reaction, a mixture of two solutions of soluble salts results in the precipitation out of solution of an insoluble salt.
· All ammonium (NH+4), alkali (Li+, Na+, K+, Cs+, Rb+), nitrate (NO−3), and perchlorate (ClO−4) salts are soluble.
· Silver, lead, and mercury salts are insoluble, except the perchlorates and nitrates.
· Most hydroxides (OH−) are insoluble, except for the alkalis and barium.