Homework Helpers: Physics
5 Electric Charges, Forces, and Fields
Lesson 5–2: Electric Forces
Charged objects exert forces at a distance on other charged objects. If you rub a rubber balloon on your hair, and then hold the balloon over some small bits of paper or salt crystals, you will see the balloon attract the objects from a distance. The paper bits or salt crystals will appear to leap off the table and stick to the balloon. This electrostatic force is another example of a field force. Similar to the force of gravity, it can act over a distance. Also, as with the case of gravitational attraction, electrostatic forces decrease with the square of the distance between the charged objects in question.

Figure 5.3
You will notice that the formula for Coulomb”s law, which is used to calculate the force between charged objects, looks similar to Newton”s universal law of gravitation. There are, however, some significant differences between the two laws, as summarized by the table on page 165.
By comparing the proportionality constants required for the two laws in the table, you can gain an understanding of why gravity is considered a relatively weak force.
Comparing and Contrasting Coulomb”s Law and Newton”s Universal Law of Gravitation
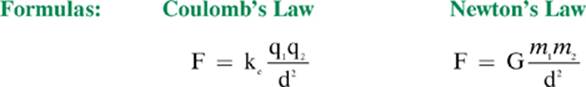
Newton”s law: The gravitational force of attraction between two objects is proportional to the product of the two masses and inversely proportional to the square of the distance between them.
Coulomb”s law: The electrostatic force of attraction or repulsion between two charged objects is proportional to the product of the two charges and inversely proportional to the square of the distance between them.
Similarities:
![]() Both laws follow what is called the inverse square law for distance.
Both laws follow what is called the inverse square law for distance.
![]() Both formulas contain a proportionality constant.
Both formulas contain a proportionality constant.
Differences:
![]() The force calculated with Newton”s law can only be positive, but the force calculated with Coulomb”s law may be positive or negative.
The force calculated with Newton”s law can only be positive, but the force calculated with Coulomb”s law may be positive or negative.
![]() The proportionality constant used in Newton”s law (G) is a very small number, 6.67 × 10–11 N · m2/kg2, but the proportionality constant (kc) used in Coulomb”s law is a very large number, 9.0 × 109 N · m2/C2.
The proportionality constant used in Newton”s law (G) is a very small number, 6.67 × 10–11 N · m2/kg2, but the proportionality constant (kc) used in Coulomb”s law is a very large number, 9.0 × 109 N · m2/C2.
Example 1
Calculate both the electrostatic force (Fe) and the gravitational force (Fg) between a proton (mp = 1.67 × 10–27 kg) and an electron (me = 9.11 × 10–31 kg) separated by a distance of 3.0 × 10–3 m.
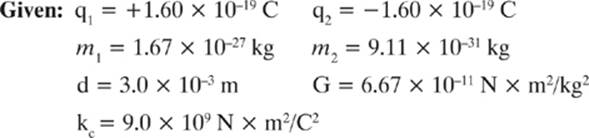
Find: Fe and Fg
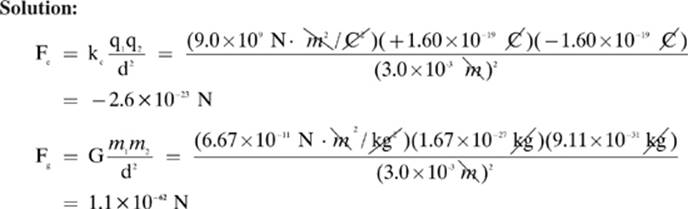
Note: The answer for the electrostatic force (Fe) is negative, indicating that unlike charges were involved, so the particles will attract each other. Also notice that the force of gravity (Fg) between the two particles is much less than the electrostatic force. How much stronger is the electrostatic force than the force of gravity in this example? Simply divide the smaller number into the larger:
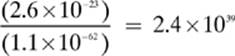
So, the electrostatic force of attraction between the two particles in this problem is about 2 400 000 000 000 000 000 000 000 000 000 000 000 000 times as strong as the force of gravity between them! They aren”t kidding when they say that gravity is a relatively weak force!
Even more interesting than the implications of the weakness of gravity are the implications of the strength of the electrostatic force. The bottom line is that the electrostatic force is so strong that we never actually touch anything, at least not in the way that most people think we do. Objects never really come in contact with each other. Matter is made up of atoms, and each atom is surrounded by an electron cloud. All electrons are like charges, which repel each other. The electrostatic force of repulsion between neutral atoms is so great that they can only come so close together without going through a chemical reaction and bonding. So, when we shake hands with someone, the atoms of our individual hands never touch. When we walk across the floor, the atoms of our shoes never actually touch the atoms of the floor. When we lie in bed, our atoms never actually touch the atoms of our sheets! You may want to think about this for a while before moving on to the next practice problem.
Example 2
A calcium ion (Ca2+) and an aluminum ion (Al3+) exert an electrostatic force of repulsion on each other with a magnitude of 3.0 × 10–28 N. How far apart are these ions?
Convert: First, let”s calculate the charge on each ion. To do this, all we need to do is multiply the number of excess protons given by the superscript in each ion by the elementary charge (charge on each proton).

As a final point of interest, notice again that the electrostatic force between two charged objects is inversely proportional to the square of the distance between them. As the distance increases, the force between them decreases. Although the force gets quite small as the distance increases, it doesn”t approach zero until the distance between the objects approaches infinity. So objects don”t really “escape” these fields, they just get to a point where the force exerted on them is not significant.
Lesson 5–2 Review
1. According to ________________, the electrostatic force between two charged objects is directly proportional to the product of their charges and inversely proportional to the square of the distance between them.
2. What would happen to the electrostatic force between two objects if the charge on each of them was doubled?
3. What would happen to the electrostatic force between two objects if the distance between them was doubled?