SAT Physics Subject Test
Chapter 8 Electric Forces and Fields
COULOMB”S LAW
The electric force between two charged particles obeys the same mathematical law as the gravitational force between two masses—that is, it”s an inverse-square law. The electric force between two particles with charges of q1 and q2, separated by a distance r, is given by the equation
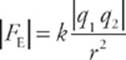
This is Coulomb”s law.
The absolute value symbol is needed to give the magnitude of the force. Some books omit the absolute value symbol around the charges and state that a negative force indicates attraction while a positive force indicates repulsion. This, however, can lead to confusion. Remember that each charge feels this force, equal in magnitude and indirection (Newton”s third laaw).

Since one of these forces points to the left and one points to the right, calling FE negative is problematic. They both can”t feel a negative force.
The value of the proportionality constant, k, depends on the material between the charged particles. In empty space (vacuum)—or air, for all practical purposes—it is called Coulomb”s constant and has the value k = 9 × 109 N × m2/C2.
Remember that the value of the universal gravitational constant, G, is 6.7 × 10–11 N × m2/kg2. The relative sizes of these fundamental constants show the relative strengths of the electric and gravitational forces. The value of k is orders of magnitude larger than G.
![]()
1. Consider two small spheres, one carrying a charge of +4.0 nC and the other a charge of –2.0 nC, separated by a distance of 2 cm. Find the electric force between them. (“n” is the abbreviation for “nano,” which means 10–9.)
Here”s How to Crack It
The electric force between the spheres is given by Coulomb”s law.
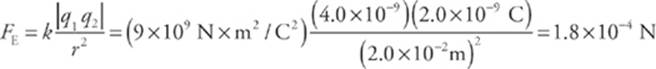
The force between the spheres is attractive and lies along the line that joins the charges, as we”ve illustrated below. The two forces shown form an action/reaction pair.
![]()
Superposition
Consider three point charges: q1, q2, and q3. The total electric force acting on, say, q2 is simply the sum of F1-on-2 (the electric force on q2 due to q1, and F3-on-2 (the electric force on q2 due to q3).
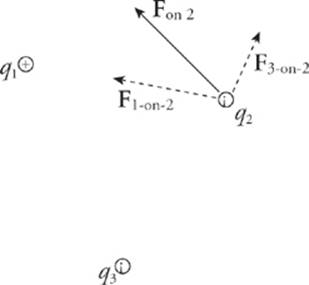
Fon 2 = F1-on-2 + F3-on-2
The fact that electric forces can be added in this way is known as superposition.
Vector Alert
The electric forces are
vectors, so remember to
add them geometrically,
NOT algebraically.
![]()
2. Consider four equal, positive point charges that are situated at the vertices of a square. Find the net electric force on a negative point charge placed at the square”s center.
Here”s How to Crack It
Refer to the diagram below. The attractive forces due to the two charges on each diagonal cancel out F1 + F3 = 0, and F2 + F4 = 0 because the distances between the negative charge and the positive charges are all the same and the positive charges are all equivalent. Therefore, the net force on the center charge is zero.
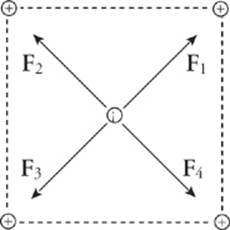
![]()
![]()
3. If the two positive charges on the bottom side of the square in the previous example were removed, what would be the net electric force on the negative charge? Write your answer in terms of the force that each positive charge exerts on the negative charge.
Here”s How to Crack It
If we break down F1 and F2 into horizontal and vertical components, then the two horizontal components cancel each other out, and the two vertical components add.
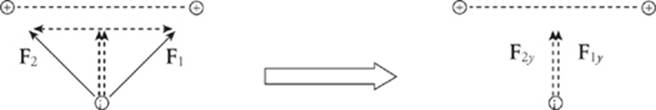
It is clear from the diagram on the left that F1y = F1 sin 45° and F2y = F2 sin 45°. Also, the magnitude of F1 equals that of F2. So the net electric force on the negative charge is F1y + F2y = 2F sin 45°, where F is the strength of the force between the negative charge and each of the positive charges.
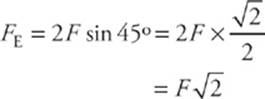
The direction of the net force is straight upward, toward the center of the line that joins the two positive charges.
![]()