5 Steps to a 5: AP Biology 2017 (2016)
STEP 4
Review the Knowledge You Need to Score High
CHAPTER 7
Respiration
IN THIS CHAPTER
Summary: This chapter covers the basics behind the energy-creation process known as respiration. This chapter also teaches you the difference between aerobic and anaerobic respiration and takes you through the steps that convert a glucose molecule into ATP.

Key Ideas
![]() Aerobic respiration: glycolysis → Krebs cycle → oxidative phosphorylation → 36 ATP.
Aerobic respiration: glycolysis → Krebs cycle → oxidative phosphorylation → 36 ATP.
![]() Anaerobic respiration: glycolysis → regenerate NAD+ → much less ATP.
Anaerobic respiration: glycolysis → regenerate NAD+ → much less ATP.
![]() Oxidative phosphorylation results in the production of large amounts of ATP from NADH and FADH2 .
Oxidative phosphorylation results in the production of large amounts of ATP from NADH and FADH2 .
![]() Chemiosmosis is the coupling of the movement of electrons down the electron transport chain with the formation of ATP using the driving force provided by the proton gradient.
Chemiosmosis is the coupling of the movement of electrons down the electron transport chain with the formation of ATP using the driving force provided by the proton gradient.
Introduction
In this chapter, we explore how cells obtain energy. It is important that you do not get lost or buried in the details. You should finish this chapter with an understanding of the basic process. The AP Biology exam will not ask you to identify by name the enzyme that catalyzes the third step of glycolysis, nor will it require you to name the fourth molecule in the Krebs cycle. But it will ask you questions that require an understanding of the respiration process.
BIG IDEA 2.A.1
All living things require constant input of energy .
There are two major categories of respiration: aerobic and anaerobic. Aerobic respiration occurs in the presence of oxygen, while anaerobic respiration occurs in situations where oxygen is not available. Aerobic respiration involves three stages: glycolysis, the Krebs cycle, and oxidative phosphorylation. Anaerobic respiration, sometimes referred to as fermentation, also begins with glycolysis, and concludes with the formation of NAD+ .
Aerobic Respiration
BIG IDEA 2.A.2
Heterotrophs capture free energy present in the food they eat through cellular respiration .
Glycolysis
Glycolysis occurs in the cytoplasm of cells and is the beginning pathway for both aerobic and anaerobic respiration. During glycolysis, a glucose molecule is broken down through a series of reactions into two molecules of pyruvate. It is important to remember that oxygen plays no role in glycolysis. This reaction can occur in oxygen-rich and oxygen-poor environments. However, when in an environment lacking oxygen, glycolysis slows because the cells run out (become depleted) of NAD+ . For reasons we will discuss later, a lack of oxygen prevents oxidative phosphorylation from occurring, causing a buildup of NADH in the cells. This buildup causes a shortage of NAD+ . This is bad for glycolysis because it requires NAD+ to function. Fermentation is the solution to this problem—it takes the excess NADH that builds up and converts it back to NAD+ so that glycolysis can continue. More to come on fermentation later . . . be patient. ![]()
To reiterate, the AP Biology exam will not require you to memorize the various steps of respiration. Your time is better spent studying the broad explanation of respiration, to understand the basic process, and become comfortable with respiration as a whole. Major concepts are the key. We will explain the specific steps of glycolysis because they will help you understand the big picture—but do not memorize them all. Save the space for other facts you have to know from other chapters of this book.
Examine Figure 7.1 , which illustrates the general layout of glycolysis. The beginning steps of glycolysis require energy input. The first step adds a phosphate to a molecule of glucose with the assistance of an ATP molecule to produce glucose-6-phosphate (G6P). The newly formed G6P rearranges to form a molecule named fructose-6-phosphate (F6P). Another molecule of ATP is required for the next step, which adds another phosphate group to produce fructose 1,6-biphosphate. Already, glycolysis has used two of the ATP molecules that it is trying to produce—seems stupid . . . but be patient . . . the genius has yet to show its face. F6P splits into two 3-carbon-long fragments known as PGAL (glyceraldehyde phosphate). With the formation of PGAL, the energy-producing portion of glycolysis begins. Each PGAL molecule takes on an inorganic phosphate from the cytoplasm to produce 1,3-diphosphoglycerate. During this reaction, each PGAL gives up two electrons and a hydrogen to molecules of NAD+ to form the all-important NADH molecules. The next step is a big one, as it leads to the production of the first ATP molecule in the process of respiration—the 1,3-diphosphoglycerate molecules donate one of their two phosphates to molecules of ADP to produce ATP and 3-phosphoglycerate (3PG). You’ll notice that there are two ATP molecules formed here because before this step, the single molecule of glucose divided into two 3-carbon fragments. After 3PG rearranges to form 2-phosphoglycerate, phosphoenolpyruvate (PEP) is formed, which donates a phosphate group to molecules of ADP to form another pair of ATP molecules and pyruvate. This is the final step of glycolysis. In total, two molecules each of ATP, NADH, and pyruvate are formed during this process. Glycolysis produces the same result under anaerobic conditions as it does under aerobic conditions: two ATP molecules. If oxygen is present, more ATP is later made by oxidative phosphorylation.
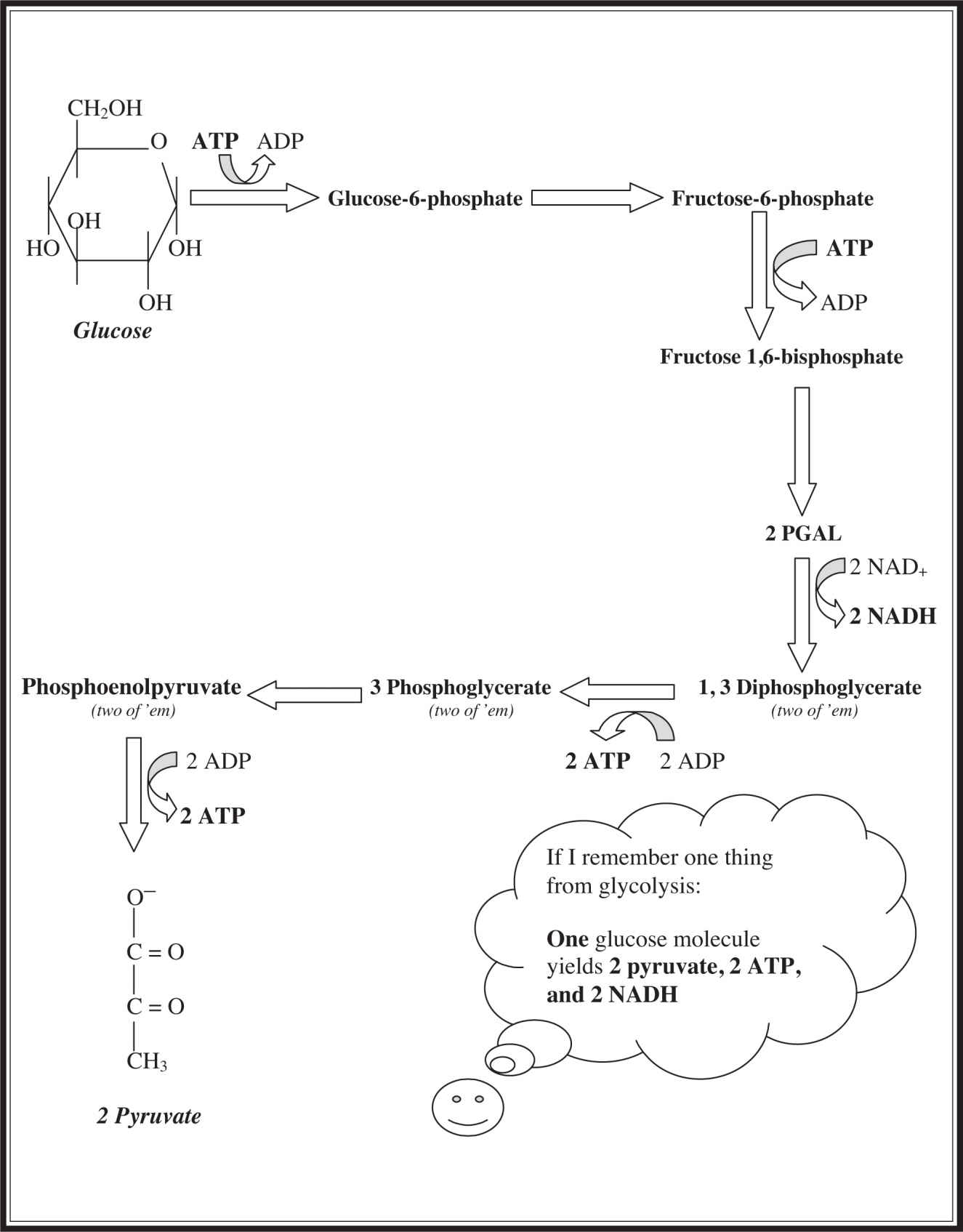
Figure 7.1 Glycolysis.
If you are going to memorize one fact about glycolysis, remember that one glucose molecule produces two pyruvate, two NADH, and two ATP molecules.
One glucose → 2 pyruvate, 2 ATP, 2 NADH
The Krebs Cycle
The pyruvate formed during glycolysis next enters the Krebs cycle, which is also known as the citric acid cycle . The Krebs cycle occurs in the matrix of the mitochondria. The pyruvate enters the mitochondria of the cell and is converted into acetyl coenzyme A (CoA) in a step that produces an NADH. This compound is now ready to enter the eight-step Krebs cycle, in which pyruvate is broken down completely to H2 O and CO2 . You do not need to memorize the eight steps.
As shown in Figure 7.2 , a representation of the Krebs cycle, the 3-carbon pyruvate does not enter the Krebs cycle per se. Rather, it is converted, with the assistance of CoA and NAD+ , into 2-carbon acetyl CoA and NADH. The acetyl CoA dives into the Krebs cycle and reacts with oxaloacetate to form a 6-carbon molecule called citrate . The citrate is converted to a molecule named isocitrate, which then donates electrons and a hydrogen to NAD+ to form 5-carbon α-ketoglutarate, carbon dioxide, and a molecule of NADH. The α-ketoglutarate undergoes a reaction very similar to the one leading to its formation and produces 4-carbon succinyl CoA and another molecule each of NADH and CO2 . The succinyl CoA is converted into succinate in a reaction that produces a molecule of ATP. The succinate then transfers electrons and a hydrogen atom to FAD to form FADH2 and fumarate. The next-to-last step in the Krebs cycle takes fumarate and rearranges it to another 4-carbon molecule: malate. Finally, in the last step of the cycle, the malate donates electrons and a hydrogen atom to a molecule of NAD+ to form the final NADH molecule of the Krebs cycle, at the same time regenerating the molecule of oxaloacetate that helped kick off the cycle. One turn of the Krebs cycle takes a single pyruvate and produces one ATP, four NADH, and one FADH2 .
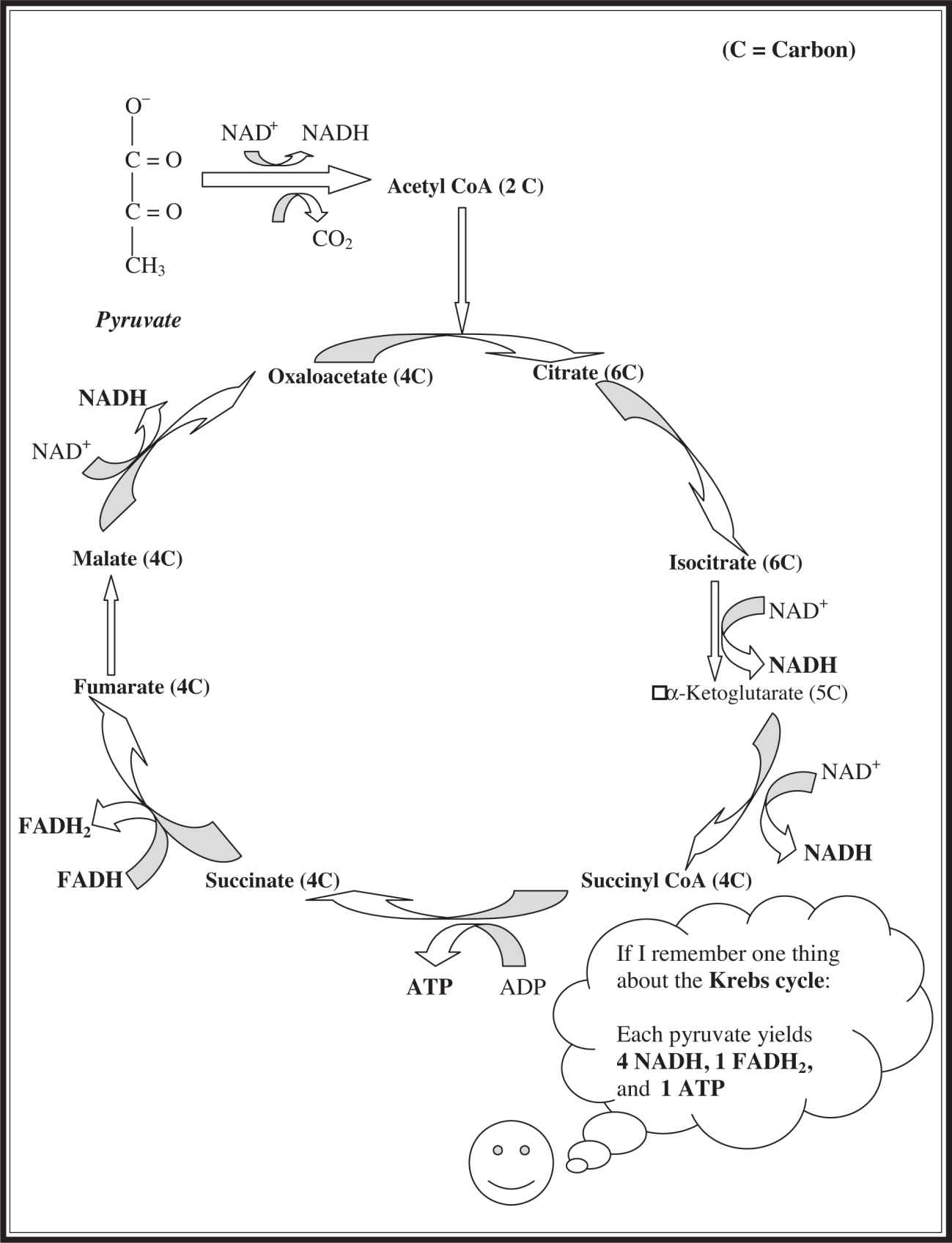
Figure 7.2 The Krebs cycle.

If you are going to memorize one thing about the Krebs cycle, remember that for each glucose dropped into glycolysis, the Krebs cycle occurs twice. Each pyruvate dropped into the Krebs cycle produces
4 NADH, 1 FADH2 , 1 ATP, and 2 CO2
Therefore, the pyruvate obtained from the original glucose molecule produces:
8 NADH, 2 FADH2 , and 2 ATP
Up to this point, having gone through glycolysis and the Krebs cycle, one molecule of glucose has produced the following energy-related compounds: 10 NADH, 2 FADH2 , and 4 ATP. Not bad for an honest day’s work . . . but the body wants more and needs to convert the NADH and FADH2 into ATP. This is where the electron transport chain, chemiosmosis, and oxidative phosphorylation come into play.
Oxidative Phosphorylation
After the Krebs cycle comes the largest energy-producing step of them all: oxidative phosphorylation. During this aerobic process, the NADH and FADH2 produced during the first two stages of respiration are used to create ATP. Each NADH leads to the production of up to three ATP, and each FADH2 will lead to the production of up to two ATP molecules. This is an inexact measurement—those numbers represent the maximum output possible from those two energy components if all goes smoothly. For each molecule of glucose, up to 30 ATP can be produced from the NADH molecules and up to 4 ATP from the FADH2 . Add to this the 4 total ATP formed during glycolysis and the Krebs cycle for a grand total of 38 ATP from each glucose . Two of these ATP are used during aerobic respiration to help move the NADH produced during glycolysis into the mitochondria. All totaled, during aerobic respiration, each molecule of glucose can produce up to 36 ATP.
Do not panic when you see the illustration for the electron transport chain (Figure 7.3 ). Once again, the big picture is the most important thing to remember. Do not waste your time memorizing the various cytochrome molecules involved in the steps of the chain. Remember that the 1⁄2 O2 is the final electron acceptor in the chain, and that without the O2 (anaerobic conditions), the production of ATP from NADH and FADH2 will be compromised. Remember that each NADH that goes through the chain can produce three molecules of ATP, and each FADH2 can produce two.
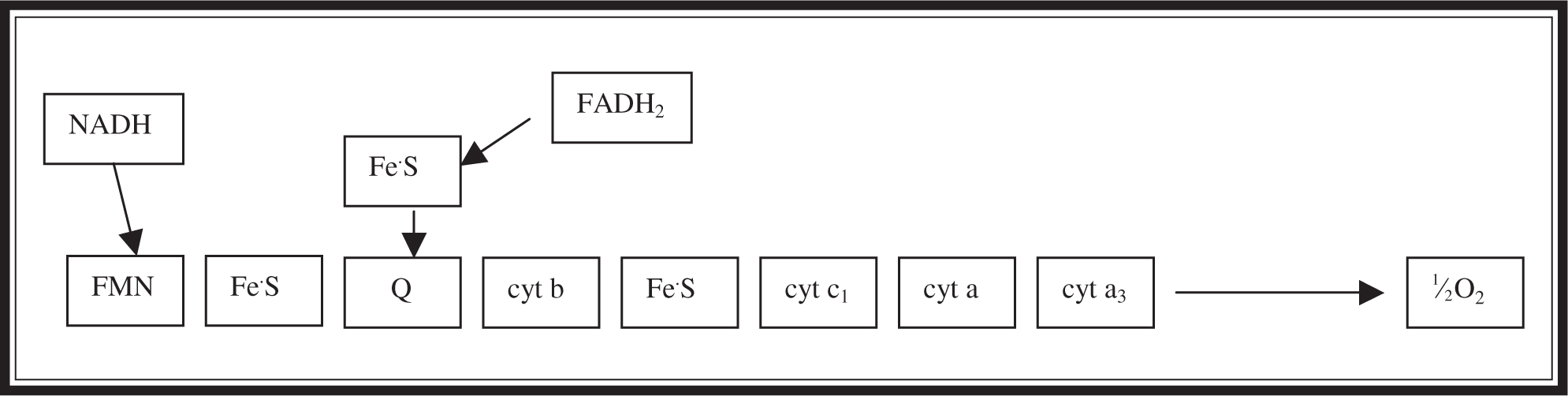
Figure 7.3 Electron transport chain (ETC).
The electron transport chain (ETC) is the chain of enzyme molecules, located in the mitochondria, that passes electrons along during the process of chemiosmosis to regenerate NAD+ to form ATP. Each time an electron passes to another member of the chain, the energy level of the system drops. Do not worry about the individual members of this chain—they are unimportant for this exam. When thinking of the ETC, we are reminded of the passing of a bucket of water from person to person until it arrives at and is tossed onto a fire. In the ETC, the various molecules in the chain are the people passing the buckets; the drop in the energy level with each pass is akin to the water sloshed out as the bucket is hurriedly passed along, and the 1⁄2 O2 represents the fire onto which the water is dumped at the end of the chain. As the 1⁄2 O2 (each oxygen atom, or half of an O2 molecule) accepts a pair of electrons, it actually picks up a pair of hydrogen ions to produce water.

Chemiosmosis is a very important term to understand. It is defined as the coupling of the movement of electrons down the electron transport chain with the formation of ATP using the driving force provided by a proton gradient. So, what does that mean in English? Well, let’s start by first defining what a coupled reaction is. It is a reaction that uses the product of one reaction as part of another reaction. Thinking back to our baseball card collecting days helps us better understand this coupling concept. We needed money to buy baseball cards. We would babysit or do yardwork for our neighbors and use that money to buy cards. We coupled the money-making reaction of hard labor to the money-spending reaction of buying baseball cards.
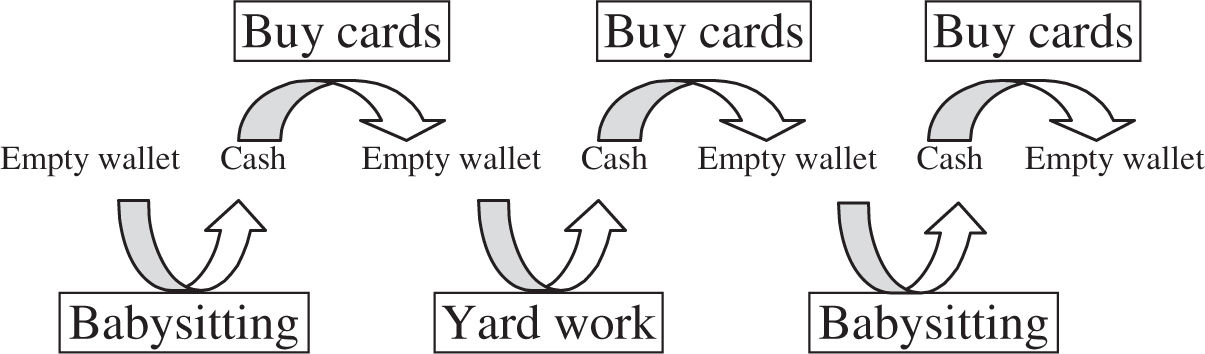
Let’s look more closely at the reactions that are coupled in chemiosmosis. If you look at Figure 7.4 a, a crude representation of a mitochondrion, you will find the ETC embedded within the inner mitochondrial membrane. As some of the molecules in the chain accept and then pass on electrons, they pump hydrogen ions into the space between the inner and outer membranes of the mitochondria (Figure 7.4 b). This creates a proton gradient that drives the production of ATP. The difference in hydrogen concentration on the two sides of the membrane causes the protons to flow back into the matrix of the mitochondria through ATP synthase channels (Figure 7.4 c). ATP synthase is an enzyme that uses the flow of hydrogens to drive the phosphorylation of an ADP molecule to produce ATP. This reaction completes the process of oxidative phosphorylation and chemiosmosis. The proton gradient created by the movement of electrons from molecule to molecule has been used to form the ATP that this process is designed to produce. In other words, the formation of ATP has been coupled to the movement of electrons and protons.
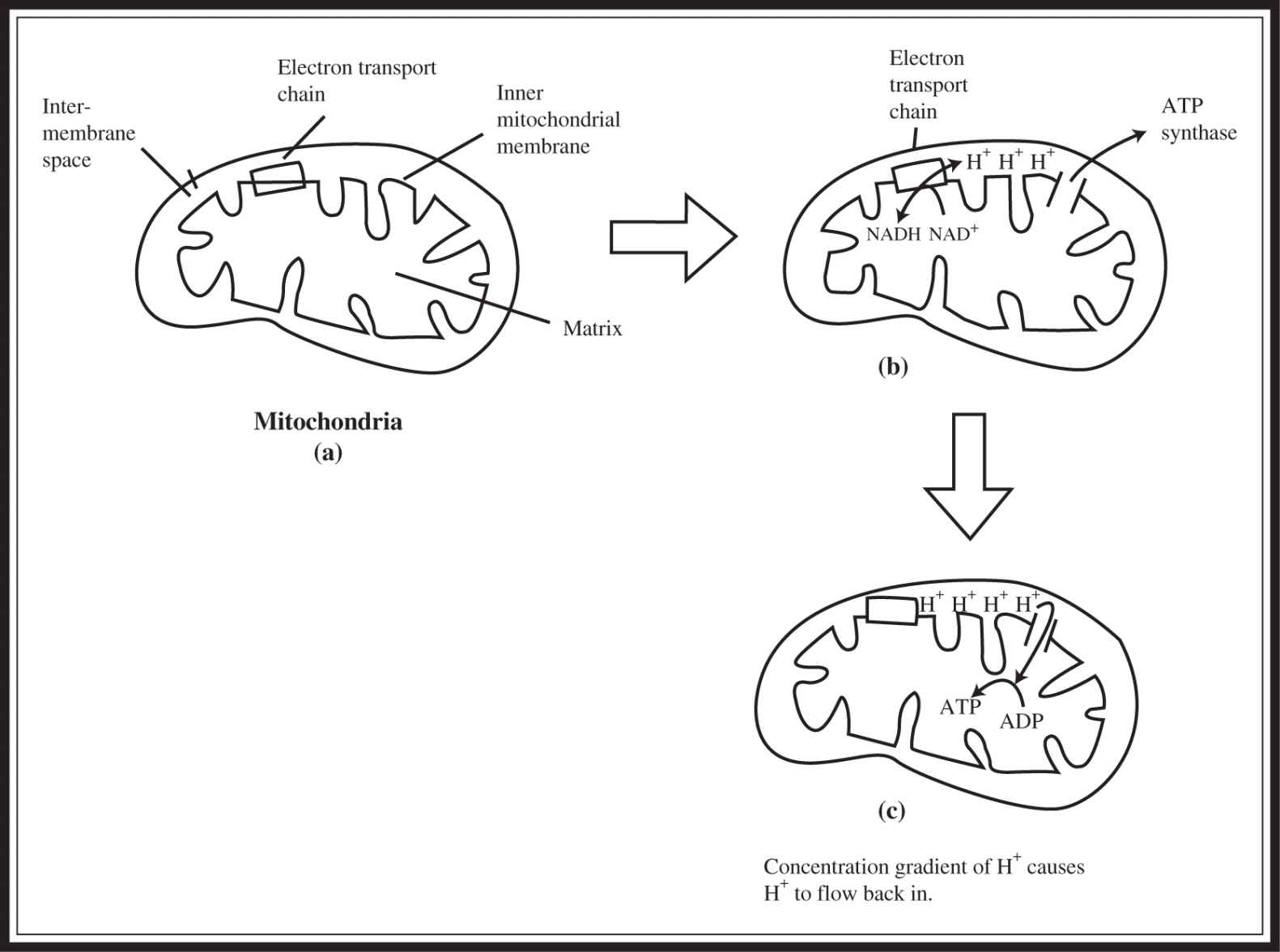
Figure 7.4 Chemiosmosis.
Chemiosmosis is not oxidative phosphorylation per se; rather, it is a major part of oxidative phosphorylation. An important fact we want you to take out of this chapter is that chemiosmosis is not unique to the mitochondria. It is the same process that occurs in the chloroplasts during the ATP-creating steps of photosynthesis (see Chapter 8 ). The difference is that light is driving the electrons along the ETC in plants. Remember that chemiosmosis occurs in both mitochondria and chloroplasts.
Remember the following facts about oxidative phosphorylation (Ox-phos):

1. Each NADH → 3 ATP.
2. Each FADH2 → 2 ATP.
3. 1⁄2 O2 is the final electron acceptor of the electron transport chain, and the chain will not function in the absence of oxygen.
4. Ox-phos serves the important function of regenerating NAD+ so that glycolysis and the Krebs cycle can continue.
5. Chemiosmosis occurs in photosynthesis as well as respiration.
Anaerobic Respiration
Anaerobic respiration, or fermentation, occurs when oxygen is unavailable or cannot be used by the organism. As in aerobic respiration, glycolysis occurs and pyruvate is produced. The pyruvate enters the Krebs cycle, producing NADH, FADH2 , and some ATP. The problem arises in the ETC—because there is no oxygen available, the electrons do not pass down the chain to the final electron acceptor, causing a buildup of NADH in the system. This buildup of NADH means that the NAD+ normally regenerated during oxidative phosphorylation is not produced, and this creates an NAD+ shortage. This is a problem, because in order for glycolysis to proceed to the pyruvate stage, it needs NAD+ to help perform the necessary reactions. Fermentation is the process that begins with glycolysis and ends when NAD+ is regenerated. A glucose molecule that enters the fermentation pathway produces two net ATP per molecule of glucose, representing a tremendous decline in the efficiency of ATP production.
Under aerobic conditions, NAD+ is recycled from NADH by the movement of electrons down the electron transport chain. Under anaerobic conditions, NAD+ is recycled from NADH by the movement of electrons to pyruvate, namely, fermentation. The two main types of fermentation are alcohol fermentation and lactic acid fermentation. Refer to Figures 7.5 and 7.6 for the representations of the different forms of fermentation. Alcohol fermentation (Figure 7.5 ) occurs in fungi, yeast, and some bacteria. The first step involves the conversion of pyruvate into two 2-carbon acetaldehyde molecules. Then, in the all-important step of alcohol fermentation, the acetaldehyde molecules are converted to ethanol, regenerating two NAD+ molecules in the process.
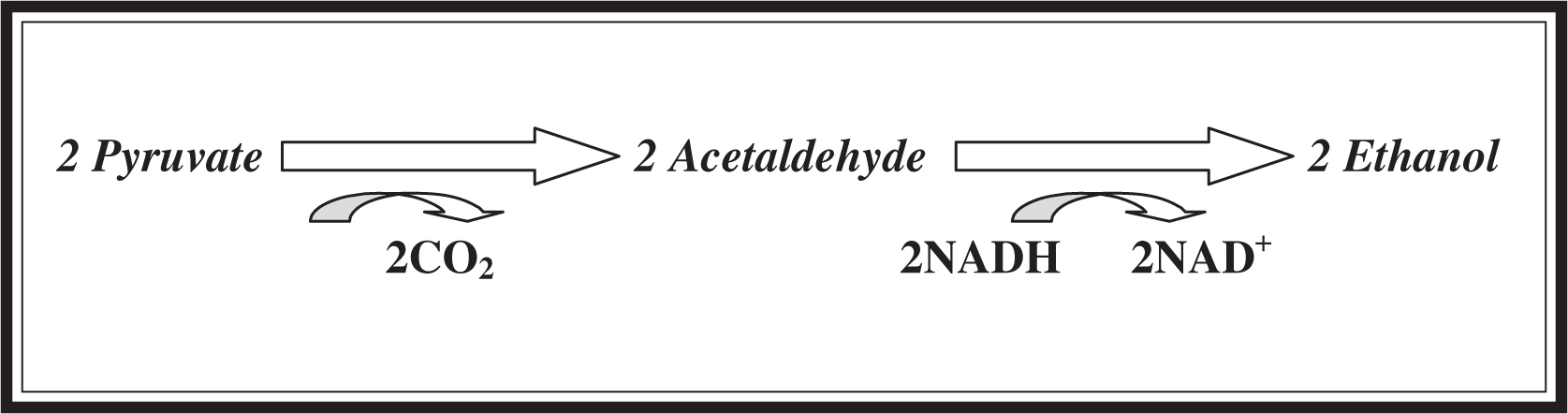
Figure 7.5 Alcohol fermentation.
Lactic acid fermentation (Figure 7.6 ) occurs in human and animal muscle cells when oxygen is not available. This is a simpler process than alcoholic fermentation—the pyruvate is directly reduced to lactate (also known as lactic acid) by NADH to regenerate the NAD+ needed for the resumption of glycolysis. Have you ever had a cramp during exercise? The pain you felt was the result of lactic acid fermentation. Your muscle was deprived of the necessary amount of oxygen to continue glycolysis, and it switched over to fermentation. The pain from the cramp came from the acidity in the muscle.
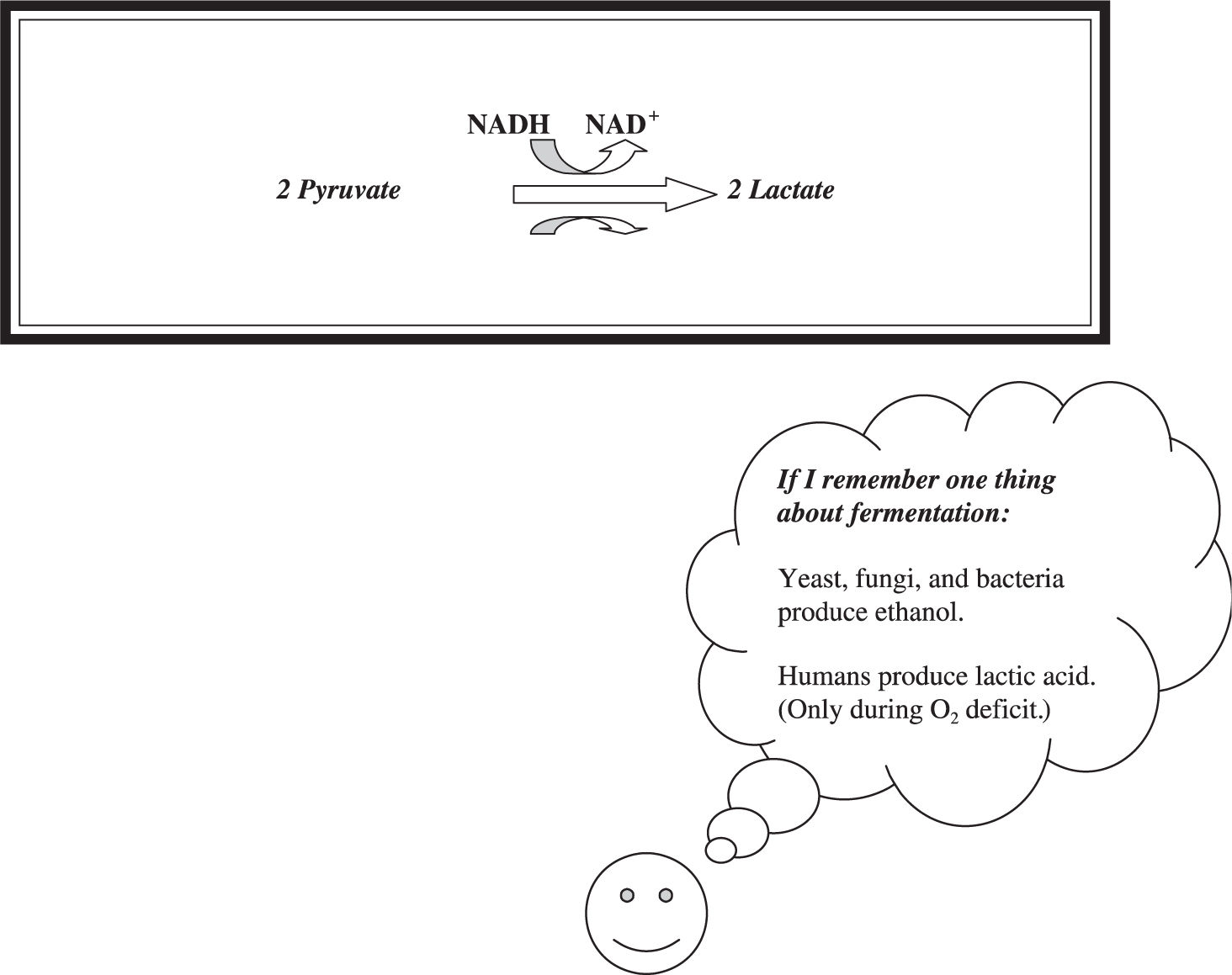
Figure 7.6 Lactic acid fermentation.
![]() Review Questions
Review Questions
1 . Most of the ATP creation during respiration occurs as a result of what driving force?
A. Electrons moving down a concentration gradient
B. Electrons moving down the electron transport chain
C. Protons moving down a concentration gradient
D. Sodium ions moving down a concentration gradient
E. Movement of pyruvate from the cytoplasm into the mitochondria
2 . Which of the following processes occurs in both respiration and photosynthesis?
A. Calvin cycle
B. Chemiosmosis
C. Citric acid cycle
D. Krebs cycle
E. Glycolysis
3 . What is the cause of the cramps you feel in your muscles during strenuous exercise?
A. Lactic acid fermentation
B. Alcohol fermentation
C. Chemiosmotic coupling
D. Too much oxygen delivery to the muscles
E. Oxidative phosphorylation
4 . Which of the following statements is in correct?
A. Glycolysis can occur with or without oxygen.
B. Glycolysis occurs in the mitochondria.
C. Glycolysis is the first step of both anaerobic and aerobic respiration.
D. Glycolysis of one molecule of glucose leads to the production of 2 ATP, 2 NADH, and 2 pyruvate.
For questions 5–8, use the following answer choices:
A. Glycolysis
B. Krebs cycle
C. Oxidative phosphorylation
D. Lactic acid fermentation
E. Chemiosmosis
5 . This reaction occurs in the matrix of the mitochondria and includes FADH2 among its products.
6 . This reaction is performed to recycle NAD+ needed for efficient respiration.
7 . This process uses the proton gradient created by the movement of electrons to form ATP.
8 . This process includes the reactions that use NADH and FADH2 to produce ATP.
9 . Which of the following molecules can give rise to the most ATP?
A. NADH
B. FADH2
C. Pyruvate
D. Glucose
10 . Which of the following is a proper representation of the products of a single glucose molecule after it has completed the Krebs cycle?
A. 10 ATP, 4 NADH, 2 FADH2
B. 10 NADH, 4 FADH2 , 2 ATP
C. 10 ATP, 4 FADH2 , 2 NADH
D. 10 NADH, 4 ATP, 2 FADH2
E. 10 NADH, 4 FADH2 , 2 ATP
![]() Answers and Explanations
Answers and Explanations
1 . C —This is the concept of chemiosmosis: the coupling of the movement of electrons down the electron transport chain and the formation of ATP via the creation of a proton gradient. The protons are pushed out of the matrix during the passage of electrons down the chain. They soon build up on the other side of the membrane, and are driven back inside because of the difference in concentration. ATP synthase uses the movement of protons to produce ATP.
2 . B —This is an important concept to understand. The AP examiners love this topic!
3 . A —Lactic acid fermentation occurs in human muscle cells when oxygen is not available. Answer choice B would be incorrect because alcohol fermentation occurs in yeast, fungi, and some bacteria. During exercise, if your muscle becomes starved for oxygen, glycolysis will switch over to fermentation. The pain from the cramp is due to the acidity in the muscle caused by the increased concentration of lactate.
4 . B —Glycolysis occurs in the cytoplasm. All the other statements are correct.
5 . B
6 . D
7 . E
8 . C
9 . D —A glucose molecule can net 36 ATP, an NADH molecule can net 3, an FADH2 molecule can net 2, and a pyruvate molecule can net 15.
10 . D —During glycolysis, a glucose molecule produces 2 ATP, 2 NADH, and 2 pyruvate. The 2 pyruvate then go on to produce 8 NADH, 2 FADH2 , and 2 ATP during the Krebs cycle to give the total listed in answer choice D.
![]() Rapid Review
Rapid Review
Try to rapidly review the material presented below.
There are two main categories of respiration: aerobic and anaerobic.
Aerobic respiration: glycolysis → Krebs cycle → oxidative phosphorylation → 36 ATP per glucose molecule
Anaerobic respiration (fermentation ): glycolysis → regenerate NAD+ → 2 ATP per glucose molecule
Glycolysis: conversion of 1 glucose molecule into 2 pyruvate, 2 ATP, and 2 NADH; occurs in the cytoplasm, and in both aerobic and anaerobic respiration; must have NAD+ to proceed.
Total energy production to this point → 2 ATP + 2NADH
Krebs cycle: conversion of 1 pyruvate molecule into 4 NADH, 1 FADH2 , 1 ATP, H2 O, and CO2 ; occurs twice for each glucose to yield 8 NADH, 2 FADH2 , and 2 ATP; occurs in mitochondria.
Total energy production per glucose molecule to this point → 4 ATP + 10 NADH + 2 FADH2
Oxidative phosphorylation: production of large amounts of ATP from NADH and FADH2 .
• Occurs in the mitochondria; requires presence of oxygen to proceed.
• NADH and FADH2 pass their electrons down the electron transport chain to produce ATP.
• Each NADH can produce up to 3 ATP; each FADH2 up to 2 ATP.
• 1⁄2 O2 is the final acceptor in the electron transport chain.
• Movement of electrons down the chain leads to movement of H+ out of matrix.
• Ox-phos regenerates NAD + so that glycolysis and the Krebs cycle can continue!
Chemiosmosis: coupling of the movement of electrons down the ETC with the formation of ATP using the driving force provided by the proton gradient; occurs in both cell respiration and photosynthesis to produce ATP.
ATP synthase: enzyme responsible for using protons to actually produce ATP from ADP.
Total energy production per glucose molecule to this point → 38 ATP (use 2 in process) → 36 ATP total
Fermentation (general ): process that regenerates NAD+ so glycolysis can begin again.
• Occurs in the absence of oxygen.
• Begins with glycolysis: 2 ATP, 2 pyruvate, and 2 NADH are produced from 1 glucose molecule.
• Because there is no oxygen to accept the electron energy on the chain, there is a shortage of NAD+ , which prevents glycolysis from continuing.
Fermentation (alcohol ): occurs in fungi, yeast, and bacteria; causes conversion of pyruvate to ethanol.
Fermentation (lactic acid ): occurs in human and animal muscle cells; causes conversion of pyruvate → lactate; causes cramping sensation when oxygen runs low in muscle cells.
CHAPTER 7
Respiration
1 . What is the ratio of ATP produced via the chemiosmotic principle for a molecule of NADH relative to a molecule of FADH2 as they transfer high-energy electrons to the electron transport chain?
(A) 3:2
(B) 3:1
(C) 1:3
(D) 2:3
2 . The absence of which of the following substrates would directly block the progression of oxidative phosphorylation?
(A) FADH
(B) NAD+
(C) FADH2
(D) RuBP
3 . Which process occurs in the cytoplasm of cells and is the beginning pathway for both aerobic and anaerobic respiration?
(A) Oxidative phosphorylation
(B) Chemiosmosis
(C) Carbon fixation
(D) Glycolysis
4 . Each pyruvate dumped into the Krebs cycle yields
(A) 4 NADH, 1 FADH2 , and 1 ATP.
(B) 8 NADH, 2 FADH2 , and 2 ATP.
(C) 2 NADH, 1 FADH2 , and 2 ATP.
(D) 4 NADH, 2 FADH2 , and 2 ATP.
![]() Answers and Explanations
Answers and Explanations
1 . A —Three molecules of ATP are produced for every molecule of NADH that transfers high-energy electrons to the electron transport chain (ETC). Two molecules of ATP are produced for every molecule of FADH2 that transfers high-energy electrons to the ETC. So the ratio would be 3:2.
2 . C —FADH2 is the only choice listed that is an input to the OX-PHOS process.
3 . D —Oxidative phosphorylation is the aerobic process in which NADH and FADH2 pass their electrons down the electron transport chain (ETC) to produce ATP. Chemiosmosis represents the coupling of the movement of electrons down the ETC with the formation of ATP, using the driving force provided by a proton gradient. Carbon fixation is the attachment of the carbon from CO2 to a molecule that is able to enter the Calvin cycle. This process occurs in photosynthesis rather than respiration. Glycolysis occurs in the cytoplasm of cells and is, in fact, the first step of both aerobic and anaerobic respiration.
4 . A —Choice B is sneaky because the Krebs cycle occurs twice for each glucose dropped into glycolysis. Each pyruvate produces what is listed in choice A (which is what the question is asking). But if the question were to ask about the yield from the pyruvate produced from each glucose molecule, it would be choice B.