CCEA GCSE Biology - Denmour Boyd, James Napier 2017
Unit 1
Food and energy

Specification points
This chapter covers specification points 1.3.1 to 1.3.3.
It covers biological molecules (food types), food tests and the energy content of food.
The food we eat, our diet, consists of different biological molecules which provide energy and contain chemicals necessary for growth and the repair or functioning of cells and our body.
There are therefore important links between our diet and our health that are discussed in other chapters of this book, including:
• Obesity (being very overweight) caused by taking in too much energy (Chapter 13).
• Heart disease and strokes caused by the build-up of fatty substances (cholesterol) in the walls of arteries (Chapter 13).
• Diabetes (type 2) caused by poor diet or obesity leading to the body being unable to effectively control blood sugar levels (Chapter 6).
Biological molecules
The foods we eat are made of a range of biological molecules of which there are three important groups: carbohydrates, proteins and fats. The individual molecules of carbohydrates, proteins and fats all contain the elements carbon, hydrogen and oxygen, while all proteins also contain the element nitrogen.
Carbohydrates
These include sugars, such as glucose and lactose (the sugar in milk), and the more complex carbohydrates (cellulose, glycogen and starch). The sugars taste sweet and are soluble in water. The more complex carbohydrates consist of long chains of the sugars joined together (Figure 3.1).
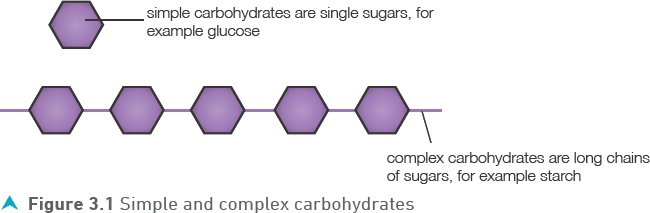
Starch is a very important part of the human diet and it is broken down to glucose during digestion. In our bodies, we store carbohydrate as glycogen and this can be broken down to glucose when our sugar reserves are low.
Cellulose, found in plant cell walls, cannot be digested by humans but still plays an important role in the diet as fibre, adding bulk to our food and helping the muscles of the intestine wall push the food along.
Carbohydrates provide energy — simple sugars are a fast acting energy source and starch is a slow release source. Examples of foods rich in simple sugars include biscuits, cakes, jam and fizzy drinks. Potatoes, rice, pasta and bread are all rich in starch.
Protein
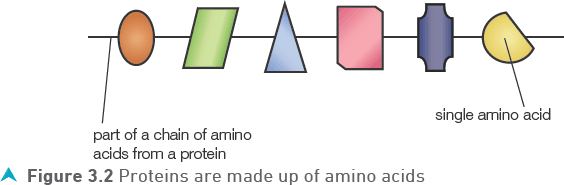
Protein provides the building blocks for the growth and repair of cells but can be used for energy when reserves of carbohydrate and fat are low. Proteins are complex molecules consisting of long chains of amino acids (Figure 3.2). There are 20 different amino acids which can join in different sequences and produce many thousands of different proteins.
Tip
Carbohydrates and fats provide most of the energy in our diet while the proteins are required for growth and repair.
When the proteins we eat are digested, the amino acids are absorbed into the blood, and in our body cells, are reassembled into the different proteins the body needs. Some of the proteins you will learn about are enzymes (Chapter 4), haemoglobin (Chapter 9) and antibodies (Chapter 13).
Good examples of foods rich in protein are lean meat, beans, fish and egg white.
Fat
![]()
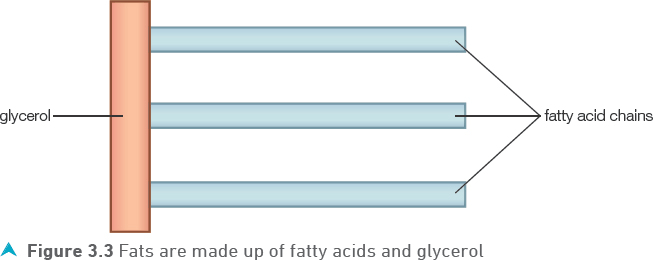
Fats are also known as lipids, their structure is shown in Figure 3.3.
Fat is an excellent energy store, providing double the energy per gram of carbohydrate and protein. Foods high in fat include streaky bacon, cheese, other dairy products and lard.
When fat is digested, it is broken down into fatty acids and glycerol.
Tip
Each molecule of fat breaks down into one molecule of glycerol and three molecules of fatty acids.
Food tests
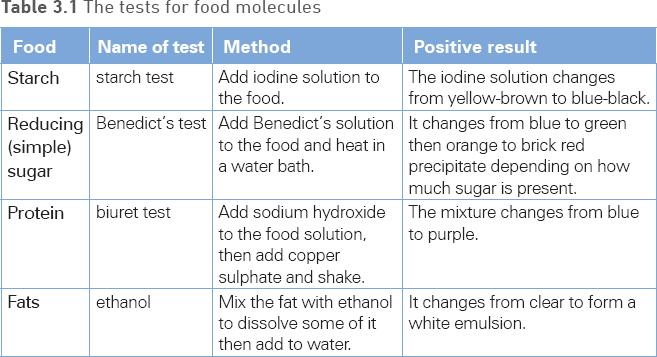
You should carry out food tests for sugars, starch, protein and fats on a range of foods. It is often necessary to break the food up using a pestle and mortar and to add a small quantity of water to make it into a solution before carrying out the test. The tests involve adding a chemical known as a reagent to the food solution. The reagent will change colour if a particular biological molecule is present in the food. Table 3.1 outlines the tests you need to know.
Test yourself
1 Name the three elements found in all biological molecules.
2 Which two types of biological molecule provide most of the energy in our food?
3 Name the reagents used in a biuret test.
4 Apart from the reagent used and the colour change, describe one way the Benedict’s test differs from the other food tests.
Show you can
Plan an experiment using the Benedict’s test to compare how much sugar is present in a potato and an onion. State what you would do to make your results valid (a fair test).
Prescribed practical
Biology Practical 1.3 Double Award Science Practical B2:
Investigating the energy content of food by burning food samples
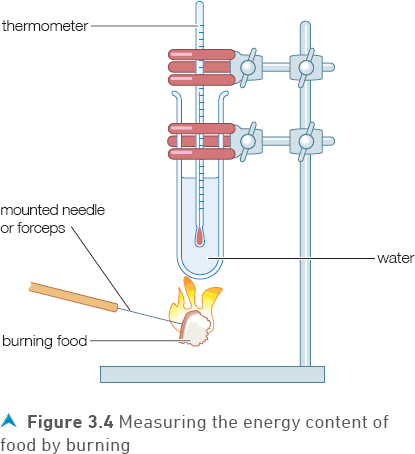
The apparatus in Figure 3.4 can be used to compare energy values in different foods such as crisps, bread and pasta (for safety reasons nuts should not be used). The food is held on the end of a mounted needle, ignited and then placed immediately underneath the test tube. The rise in temperature of the water in the test tube will give an indication of how much energy there is in the food.
The energy released by a food sample is measured in joules (J) and is calculated using the following equation.
![]()
If the same mass of food is burned each time, then the result of this equation can be used to compare foods.
However, it is not always possible to measure the same mass of food each time. Since the more food is burned, the more energy will be released, it is necessary to calculate the energy released by each gram of food burned. This is done by dividing the energy released (J) by the mass (g) of the food burned.
![]()
Although the energy values of foods can be compared in the laboratory as described above, food labels on packaging can also be used to check the accuracy of your calculations.
Tip
The energy in food is often more than 1000 J. In such cases the energy is expressed in kilojoules (kJ): 1000 J = 1 kJ
Procedure
(SAFETY - wear eye protection)
1 Add 20 cm3 of water to a boiling tube, clamped in a retort stand as shown in Figure 3.4.
2 Record the temperature of the water.
3 Weigh approximately half of a potato crisp.
4 Secure the crisp on a mounted needle.
5 Light the crisp in a Bunsen burner flame and quickly hold it under the boiling tube.
6 Keep the burning crisp under the boiling tube until it is completely burned. If necessary, relight and replace under the boiling tube.
7 Record the maximum temperature of the water after the crisp is burned.
8 Repeat the experiment (steps 1—7) with two more samples of the same type of crisp.
9 Record the results in the table.
10 Copy and complete the table by calculating the energy released by each crisp and the average energy released per gram of crisp.

Sample results and questions
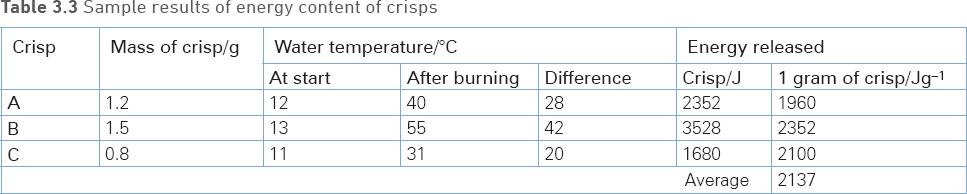
Questions
1 List three factors (variables) you would have to keep the same each time you test a food using the apparatus in Figure 3.4, to give valid results (a fair test).
2 If the piece of food stops burning while under the tube, explain why it should be relit and replaced under the tube until it does not light when placed in a Bunsen burner flame.
3 Explain the importance of calculating the average energy released per gram.
4 The typical energy value of crisps given on the packet is 21 kJ/g. What percentage of the typical energy was measured using this method?
5 Suggest why the energy value you get from burning a food is likely to be an underestimation of the energy in the food.
6 Suggest two ways the accuracy of this method could be improved.
Practice questions
1 Table 3.4 shows the results of food tests carried out on a biscuit.
Use the information in the table to draw conclusions about the types of molecule in the biscuit.

Describe and explain the colour change for each food test.
In this question you will be assessed on your written communication skills, including the use of specialist scientific terms.
(6 marks)