CONCEPTS IN BIOLOGY
PART II. CORNERSTONES: CHEMISTRY, CELLS, AND METABOLISM
6. Biochemical Pathways—Cellular Respiration
6.3. The Metabolic Pathways of Aerobic Cellular Respiration
It is a good idea to begin with the simplest description and add layers of understanding as you go to additional levels. Therefore, this discussion of aerobic cellular respiration is divided into two levels:
1. a fundamental description and
2. a detailed description.
Ask your instructor which level is required for your course of study.
Fundamental Description
Glycolysis
Glycolysis is a series of enzyme-controlled reactions that takes place in the cytoplasm. During glycolysis, a 6-carbon sugar molecule (glucose) has energy added to it from two ATP molecules. Adding this energy makes some of the bonds of the glucose molecule unstable, and the glucose molecule is more easily broken down. After passing through several more enzyme-controlled reactions, the 6-carbon glucose is broken down to two 3-carbon molecules known as glyceraldehyde-3-phosphate (also known as PGA, or phosphoglyceraldehyde), which undergo additional reactions to form pyruvic acid (CH3COCOOH).
Enough energy is released by this series of reactions to produce four ATP molecules. Because two ATP molecules were used to start the reaction and four were produced, there is a net gain of two ATPs from the glycolytic pathway (figure 6.4). During the process of glycolysis, some hydrogens and their electrons are removed from the organic molecules being processed and picked up by the electron-transfer molecule NAD+ to form NADH. Enough hydrogens are released during glycolysis to form 2 NADHs. The NADH with its extra electrons contains a large amount of potential energy, which can be used to make ATP in the electron-transport system. The job of the coenzyme NAD+ is to transport these energy-containing electrons and protons safely to the electron-transport system. Once they have dropped off their electrons, the oxidized NAD+ s are available to pick up more electrons and repeat the job.
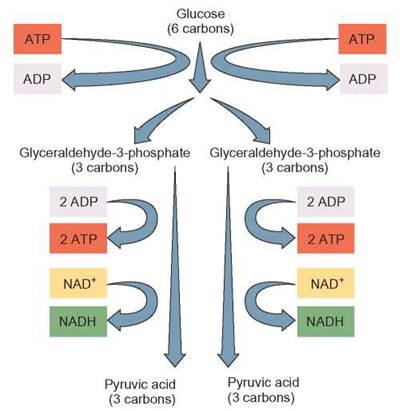
FIGURE 6.4. Glycolysis: Fundamental Description
Glycolysis is the biochemical pathway many organisms use to oxidize glucose. During this sequence of chemical reactions, the 6-carbon molecule of glucose is oxidized. As a result, pyruvic acid is produced, electrons are picked up by NAD+, and ATP is produced.
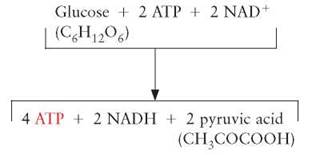
Fundamental Summary of One Turn of Glycolysis
The Krebs Cycle
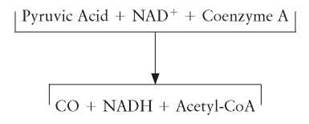
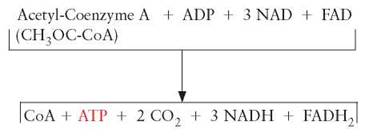
The series of reactions known as the Krebs cycle takes place within the mitochondria of cells. It gets its name from its discoverer, Hans Krebs, and the fact that the series of reactions begins and ends with the same molecule; it cycles. The Krebs cycle is also known as the citric acid cycle and the TriCarboxylic Acid cycle (TCA). The 3-carbon pyruvic acid molecules released from glycolysis enter the mitochondria. These are acted upon by specific enzymes made using genetic information found on DNA located within the mitochondria (mDNA). One of these carbons is stripped off and the remaining 2-carbon fragment is attached to a molecule of coenzyme A (CoA), becoming a compound called acetyl-CoA. Coenzyme A is made from pantethine (pantothenic acid), a form of vitamin B5. Acetyl-CoA is the molecule that proceeds through the Krebs cycle. At the time the acetyl-CoA is produced, 2 hydrogens are attached to NAD+ to form NADH. The carbon atom that was removed is released as carbon dioxide.
Summary of Changes as Pyruvic Acid is Converted to Acetyl-CoA
During the Krebs cycle (figure 6.5), the acetyl-CoA is completely oxidized (i.e., the remaining hydrogens and their electrons are removed). Most of the electrons are picked up by NAD+ to form NADH, but at one point in the process FAD picks up electrons to form FADH2. Regardless of which electron carrier is being used, the electrons are sent to the electron-transport system. The remaining carbon and oxygen atoms are combined to form CO2. As in glycolysis, enough energy is released to generate 2 ATP molecules. At the end of the Krebs cycle, the acetyl portion of the acetyl- CoA has been completely broken down (oxidized) to CO2. The CoA is released and available to be used again. The energy in the molecule has been transferred to ATP, NADH, or FADH2. Also, some of the energy has been released as heat. For each of the acetyl-CoA molecules that enters the Krebs cycle, 1 ATP, 3 NADHs, and 1 FADH2 are produced. If we count the NADH produced during glycolysis, when acetyl-CoA was formed, there are a total of 4 NADHs for each pyruvic acid that enters a mitochondrion.
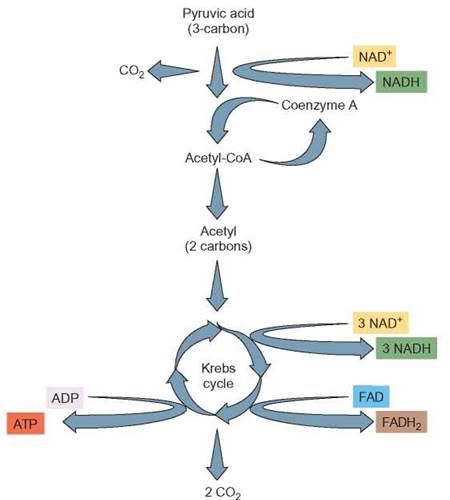
FIGURE 6.5. Krebs Cycle: Fundamental Description
The Krebs cycle takes place in the mitochondria of cells to complete the oxidation of glucose. During this sequence of chemical reactions, a pyruvic acid molecule produced from glycolysis is stripped of its hydrogens. The hydrogens are picked up by NAD+ and FAD for transport to the ETS. The remaining atoms are reorganized into molecules of carbon dioxide. Enough energy is released during the Krebs cycle to form 2 ATPs. Because 2 pyruvic acid molecules were produced from glycolysis, the Krebs cycle must be run twice in order to complete their oxidation (once for each pyruvic acid).
Fundamental Summary of One Turn of the Krebs Cycle
The Electron-Transport System
Of the three steps of aerobic cellular respiration, (glycolysis, Krebs cycle, and electron-transport system) cells generate the greatest amount of ATP from the electron-transport system (figure 6.6). During this stepwise sequence of oxidation- reduction reactions, the energy from the NADH and FADH2 molecules generated in glycolysis and the Krebs cycle is used to produce ATP. Iron-containing cytochrome (cyto = cell; chrom = color) enzyme molecules are located on the membranes of the mitochondrion. The energy-rich electrons are passed (transported) from one cytochrome to another, and the energy is used to pump protons (hydrogen ions) from one side of the membrane to the other. The result of this is a higher concentration of hydrogen ions on one side of the membrane. As the concentration of hydrogen ions increases on one side, a proton gradient builds up. Because of this concentration gradient, when a membrane channel is opened, the protons flow back to the side from which they were pumped. As they pass through the channels, a phosphorylase enzyme (ATP synthetase, also referred to as ATPase) speeds the formation of an ATP molecule by bonding a phosphate to an ADP molecule (phosphorylation). When all the electrons and hydrogen ions are accounted for, a total of 32 ATPs are formed from the electrons and hydrogens removed from the original glucose molecule. The hydrogens are then bonded to oxygen to form water.
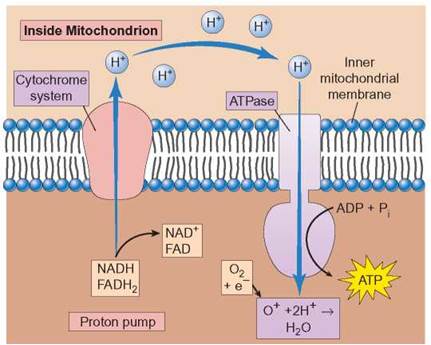
FIGURE 6.6. The Electron-Transport System: Fundamental Description
The electron-transport system (ETS) is also known as the cytochrome system. With the help of enzymes, the electrons are passed through a series of oxidation-reduction reactions. The energy the electrons give up is used to pump protons (H+) across a membrane in the mitochondrion. When protons flow back through the membrane, enzymes in the membrane cause the formation of ATP. The protons eventually combine with the oxygen that has gained electrons, and water is produced.
Fundamental Summary of the Electron-Transport System
Detailed Description
Glycolysis
The first stage of the cellular respiration process takes place in the cytoplasm. This first step, known as glycolysis, consists of the enzymatic breakdown of a glucose molecule without the use of molecular oxygen. Because no oxygen is required, glycolysis is called an anaerobic process. The glycolysis pathway can be divided into two general sets of reactions. The first reactions make the glucose molecule unstable, and later oxidation-reduction reactions are used to synthesize ATP and capture hydrogens.
Because glucose is a very stable molecule and will not automatically break down to release energy, some energy must be added to the glucose molecule in order to start glycolysis. In glycolysis, the initial glucose molecule gains a phosphate to become glucose-6-phosphate, which is converted to fructose-6-phosphate. When a second phosphate is added, fructose-1,6-bisphosphate (P—C6—P) is formed. This 6-carbon molecule is unstable and breaks apart to form two 3-carbon, glyceraldehyde-3-phosphate molecules.
Each of the two glyceraldehyde-3-phosphate molecules acquires a second phosphate from a phosphate supply normally found in the cytoplasm. Each molecule now has 2 phosphates attached to form 1,3-bisphosphoglycerate (P—C3—P). A series of reactions follows, in which energy is released by breaking chemical bonds that hold the phosphates to 1,3-bisphosphoglycerate. The energy and the phosphates are used to produce ATP. Since there are two 1,3-bisphosphoglycerate molecules each with 2 phosphates, a total of 4 ATPs are produced. Because 2 ATPs were used to start the process, a net yield of 2 ATPs results. In addition, 4 hydrogen atoms detach from the carbon skeleton and their electrons are transferred to NAD+ to form NADH, which transfers the electrons to the electron-transport system. The 3-carbon pyruvic acid molecules that remain are the raw material for the Krebs cycle. Because glycolysis occurs in the cytoplasm and the Krebs cycle takes place inside mitochondria, the pyruvic acid must enter the mitochondrion before it can be broken down further (figure 6.7).
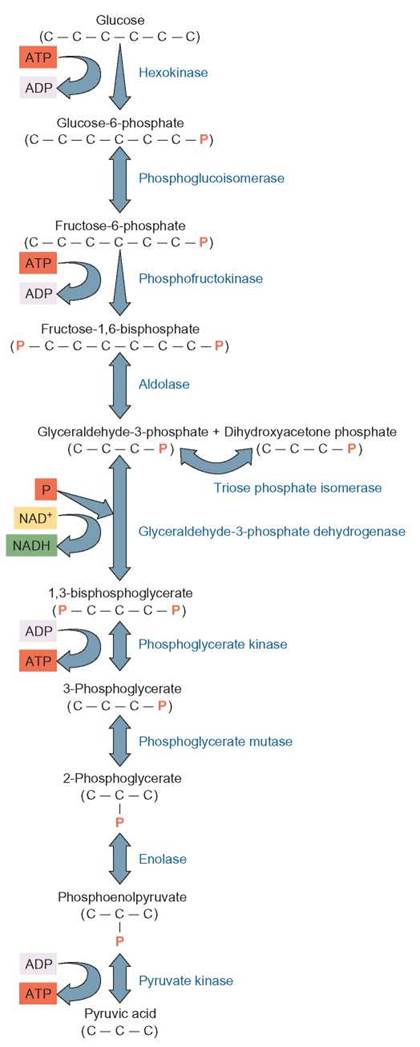
FIGURE 6.7. Glycolysis: Detailed Description
Glycolysis is a process that takes place in the cytoplasm of cells. It does not require the use of oxygen, so it is an anaerobic process. During the first few steps, phosphates are added from ATP and ultimately the 6-carbon sugar is split into two 3-carbon compounds. During the final steps in the process, NAD+ accepts electrons and hydrogen to form NADH. In addition, ATP is produced. Two ATPs form for each of the 3-carbon molecules that are processed in glycolysis. Because there are two 3-carbon compounds, a total of 4 ATPs are formed. However, because 2 ATPs were used to start the process, there is a net gain of 2 ATPs. Pyruvic acid (pyruvate) is left at the end of glycolysis.
Summary of Detailed Description of Glycolysis
The process of glycolysis takes place in the cytoplasm of a cell, where glucose (C6H12O6) enters a series of reactions that:
1. requires the use of 2 ATPs,
2. ultimately results in the formation of 4 ATPs,
3. results in the formation of 2 NADHs, and
4. results in the formation of 2 molecules of pyruvic acid (CH3COCOOH).
Because 2 molecules of ATP are used to start the process and a total of 4 ATPs are generated, each glucose molecule that undergoes glycolysis produces a net yield of 2 ATPs.
The Krebs Cycle
After pyruvate (pyruvic acid) enters the mitochondrion, it is first acted upon by an enzyme, along with a molecule known as coenzyme A (CoA) (figure 6.8). This results in three significant products. Hydrogen atoms are removed and NADH is formed, a carbon is removed and carbon dioxide is formed, and a 2-carbon fragment is formed, which temporarily attaches to coenzyme A to produce acetyl-coenzyme A. (These and subsequent reactions of the Krebs cycle take place in the fluid between the membranes of the mitochondrion.) The acetyl coenzyme A enters the series of reactions known as the Krebs cycle. During the Krebs cycle, the acetyl-CoA is systematically dismantled. Its hydrogen atoms are removed and the remaining carbons are released as carbon dioxide (Outlooks 6.1).
The first step in this process involves the acetyl-CoA. The acetyl portion of the complex is transferred to a 4-carbon compound called oxaloacetate (oxaloacetic acid) and a new 6-carbon citrate molecule (citric acid) is formed. The coenzyme A is released to participate in another reaction with pyruvic acid. This newly formed citrate is broken down in a series of reactions, which ultimately produces oxaloacetate, which was used in the first step of the cycle (hence, the names Krebs cycle, citric acid cycle, and tricarboxylic acid cycle). The compounds formed during this cycle are called keto acids.
In the process, electrons are removed and, along with protons, become attached to the coenzymes NAD+ and FAD. Most become attached to NAD+ but some become attached to FAD. As the molecules move through the Krebs cycle, enough energy is released to allow the synthesis of 1 ATP molecule for each acetyl-CoA that enters the cycle. The ATP is formed from ADP and a phosphate already present in the mitochondria. For each pyruvate molecule that enters a mitochondrion and is processed through the Krebs cycle, 3 carbons are released as 3 carbon dioxide molecules, 5 pairs of hydrogen atoms are removed and become attached to NAD+ or FAD, and 1 ATP molecule is generated. When both pyruvate molecules have been processed through the Krebs cycle, (1) all the original carbons from the glucose have been released into the atmosphere as 6 carbon dioxide molecules; (2) all the hydrogen originally found on the glucose has been transferred to either NAD+ or FAD to form NADH or FADH2; and (3) 2 ATPs have been formed from the addition of phosphates to ADPs (review figure 6.8).
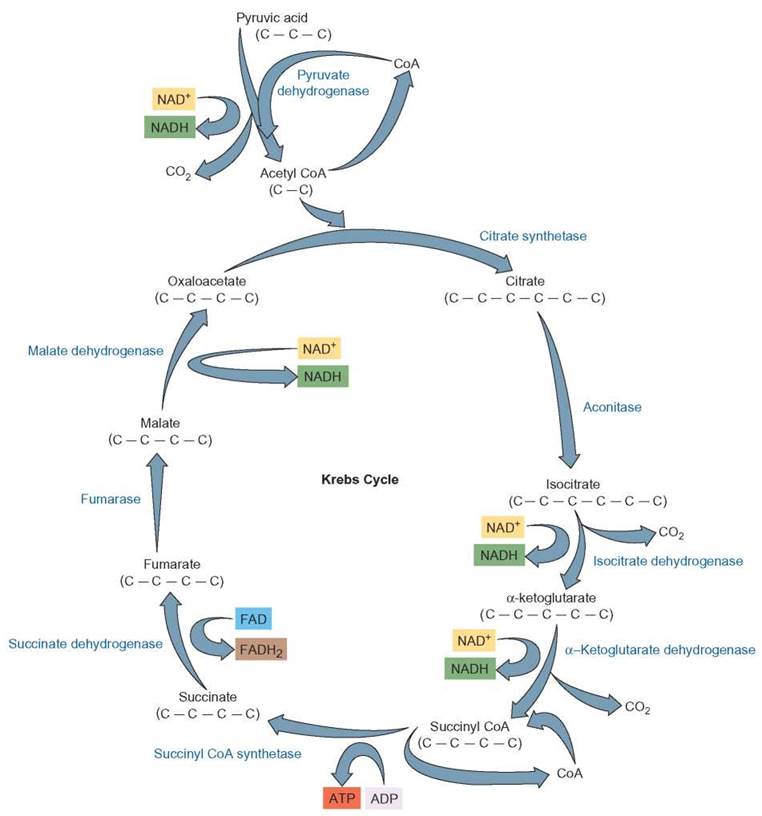
FIGURE 6.8. Krebs Cycle: Detailed Descriptions
The Krebs cycle occurs within the mitochondrion. Pyruvate enters the mitochondrion from glycolysis and is converted to a 2-carbon fragment, which becomes attached to coenzyme A to from acetyl-CoA. With the help of CoA, the 2-carbon fragment (acetyl) combines with 4-carbon oxaloacetate to form a 6-carbon citrate molecule. Through a series of reactions in the Krebs cycle, electrons are removed and picked up by NAD+ and FAD to form NADH and FADH2, which will be shuttled to the electron-transport system. Carbons are removed as carbon dioxide. Enough energy is released that 1 ATP is formed for each acetyl-CoA that enters the cycle.
Summary of Detailed Description of the Eukaryotic Krebs Cycle
The Krebs cycle takes place within the mitochondria. For each acetyl-CoA molecule that enters the Krebs cycle:
1. The three carbons from a pyruvate are converted to acetyl-CoA and released as carbon dioxide (CO2). One CO2 is actually released before acetyl-CoA is formed.
2. Five pairs of hydrogens become attached to hydrogen carriers to become 4 NADHs and 1 FADH2. One of the NADHs is released before acetyl-CoA enters the Krebs cycle.
3. One ATP is generated.
The Electron-Transport System
The series of reactions in which energy is transferred from the electrons and protons carried by NADH and FADH2 is known as the electron-transport system (ETS) (figure 6.9). This is the final stage of aerobic cellular respiration and is dedicated to generating ATP. The reactions that make up the electron-transport system are a series of oxidation-reduction reactions in which the electrons are passed from one electron carrier molecule to another until, ultimately, they are accepted by oxygen atoms. The negatively charged oxygen combines with the hydrogen ions to form water. It is this step that makes the process aerobic. Keep in mind that potential energy increases whenever things experiencing a repelling force are pushed together, such as adding the third phosphate to an ADP molecule. Potential energy also increases whenever things that attract each other are pulled apart, as in the separation of the protons from the electrons.
Let’s now look in just a bit more detail at what happens to the electrons and protons that are carried to the electron- transport systems by NADH and FADH2 and how these activities are used to produce ATP. The mitochondrion consists of two membranes—an outer, enclosing membrane and an inner, folded membrane. The reactions of the ETS are associated with this inner membrane. Within the structure of the membrane are several enzyme complexes, which perform particular parts of the ETS reactions (review figure 6.9). The production of ATPs involves two separate but connected processes. Electrons carried by NADH enter reactions in enzyme complex I, where they lose some energy and are eventually picked up by a coenzyme (coenzyme Q). Electrons from FADH2 enter enzyme complex II and also are eventually transferred to coenzyme Q. Coenzyme Q transfers the electrons to enzyme complex III. In complex III, the electrons lose additional energy and are transferred to cytochrome c, which transfers electrons to enzyme complex IV. In complex IV, the electrons are eventually transferred to oxygen. As the electrons lose energy in complex I, complex III, and complex IV, additional protons are pumped into the intermembrane space. When these protons flow down the concentration gradient through channels in the membrane, phosphorylase enzymes (ATPase) in the membrane are able to use the energy to generate ATP.
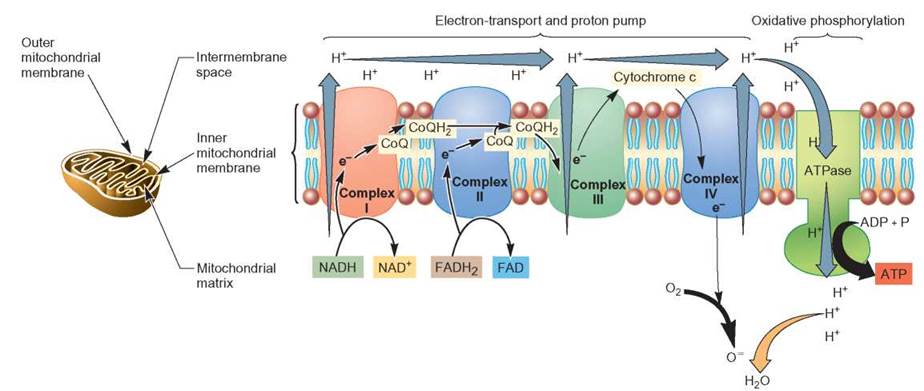
FIGURE 6.9. The Electron-Transport System: Detailed Description
Most of the ATP produced by aerobic cellular respiration comes from the ETS. NADH and FADH2 deliver electrons to the enzymes responsible for the ETS. There are several protein complexes in the inner membrane of the mitochondrion, each of which is responsible for a portion of the reactions that yield ATP. The energy of electrons is given up in small amounts and used to pump protons into the intermembrane space. When these protons flow back through pores in the membrane, ATPase produces ATP. The electrons eventually are transferred to oxygen and the negatively charged oxygen ions accept protons to form water.
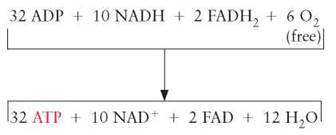
A total of 12 pairs of electrons and hydrogens are transported to the ETS from glycolysis and the Krebs cycle for each glucose that enters the process. In eukaryotic organisms, the pairs of electrons can be accounted for as follows: 2 pairs are carried by NADH and were generated during glycolysis outside the mitochondrion, 8 pairs are carried as NADH and were generated within the mitochondrion, and 2 pairs are carried by FADH2 and were generated within the mitochondrion.
• For each of the 8 NADHs generated within the mitochondrion, enough energy is released to produce 3 ATP molecules. Therefore, 24 ATPs are released from these electrons carried by NADH.
• In eukaryotic cells, the electrons released during glycolysis are carried by NADH and converted to 2 FADH2 in order to shuttle them into the mitochondria. Once they are inside the mitochondria, they follow the same pathway as the other 2 FADH2s from the Krebs cycle.
The electrons carried by FADH2 are lower in energy. When these electrons go through the series of oxidation- reduction reactions, they release enough energy to produce a total of 8 ATPs. Therefore, a total of 32 ATPs are produced from the hydrogen electrons that enter the ETS.
Finally, a complete accounting of all the ATPs produced during all three parts of aerobic cellular respiration results in a total of 36 ATPs: 32 from the ETS, 2 from glycolysis, and 2 from the Krebs cycle.
Summary of Detailed Description of the Eukaryotic Electron-Transport System
The electron-transport system takes place within the mitochondrion, where:
1. Oxygen is used up as the oxygen atoms accept hydrogens from NADH and FADH2 forming water (H2O).
2. NAD+ and FAD are released, to be used over again.
3. Thirty-two ATPs are produced.
OUTLOOKS 6.1
What Happens When You Drink Alcohol
Ethyl alcohol (CH3CH2OH) is a 2-carbon organic compound with a single alcoholic functional group. Because it is soluble in water, it is easily absorbed into the bloodstream. After an alcoholic beverage enters the body, it is spread by the circulatory system rapidly throughout the body and enters the brain. The majority of the alcohol is absorbed from the stomach (20%) and small intestine (80%). The more a person drinks, the higher the blood alcohol level. How fast alcohol is absorbed depends on several factors.
1. Food in the stomach slows absorption.
2. Strenuous physical exercise decreases absorption.
3. Drugs (e.g., nicotine, marijuana, and ginseng) increase absorption.
Ninety percent of ethyl alcohol is oxidized in mitochondria to acetate (CH3CH2OH + NAD+ → CH3CHO + NADH). The acetate is then converted to acetyl-CoA that enters the Krebs cycle where ATP is produced. Alcohol is high in calories (1g = 7,000 calories, or 7 food calories). A standard glass of wine has about 15 g of alcohol and about 100 kilocalories. The 10% not metabolized is eliminated in sweat or urine, or given off in breath. It takes the liver one hour to deal with one unit of alcohol. A unit of alcohol is:
• 250 ml (1/2 pint) of ordinary strength beer/lager.
• One glass (125 ml/4 fl oz) of wine.
• 47 ml/1.5 oz of sherry/vermouth.
• 47 ml/1.5 oz of liquor.
If alcohol is consumed at a rate faster than the liver can break it down, the blood alcohol level rises. This causes an initial feeling of warmth and light-headedness. However, alcohol is a depressant, that is, it decreases the activity of the nervous system. At first, it may inhibit circuits in the brain that normally inhibit a person's actions. This usually results in a person becoming more talkative and active—uninhibited. However, as the alcohol's effect continues, other changes can take place. These include increased aggression, loss of memory, and loss of motor control.

Long-term, excessive use of alcohol can cause damage to the liver, resulting in the development of a fatty liver, alcoholic hepatitis, and alcoholic cirrhosis. It can also interfere with the kidneys' regulation of water, sodium, potassium, calcium, and phosphate and with the kidney's ability to maintain a proper acid-base balance, and produce hormones. It also causes low blood sugar levels, dehydration, high blood pressure, strokes, heart disease, birth defects, osteoporosis, and certain cancers.
Drinking alcohol in moderation does have some health benefits if the beverage contains antioxidants (for example, red wines and dark beers). The antioxidants in red wine (polyphenols) appear to counteract the negative effect of chemicals called free radicals released during metabolism. Free radicals are known to destroy cell components and cause mutations, damage which can lead to heart disease and cancers. Antioxidants protect against this kind of harm by capturing free radicals.
6.3. CONCEPT REVIEW
6. For glycolysis, the Krebs cycle, and the electron- transport system, list two molecules that enter and two that leave each pathway.
7. How is each of the following involved in aerobic cellular respiration: NAD+, pyruvic acid, oxygen, and ATP?