CONCEPTS IN BIOLOGY
PART II. CORNERSTONES: CHEMISTRY, CELLS, AND METABOLISM
6. Biochemical Pathways—Cellular Respiration
6.6. Metabolic Processing of Molecules Other Than Carbohydrates
Up to this point, we have discussed only the methods and pathways that allow organisms to release the energy tied up in carbohydrates (sugars). Frequently, cells lack sufficient carbohydrates for their energetic needs but have other materials from which energy can be removed. Fats and proteins, in addition to carbohydrates, make up the diet of many organisms. These three foods provide the building blocks for the cells, and all can provide energy. Carbohydrates can be digested to simple sugars, proteins can be digested to amino acids, and fats can be digested to glycerol and fatty acids. The basic pathways organisms use to extract energy from fat and protein are the same as for carbohydrates: glycolysis, the Krebs cycle, and the electron-transport system. However, there are some additional steps necessary to get fats and proteins ready to enter these pathways at several points in glycolysis and the Krebs cycle where fats and proteins enter to be respired.
Fat Respiration
A triglyceride (also known as a neutral fat) is a large molecule that consists of a molecule of glycerol with 3 fatty acids attached to it. Before these fats can be broken down to release energy, they must be converted to smaller units by digestive processes. Several enzymes are involved in these steps. The first step is to break the bonds between the glycerol and the fatty acids. Glycerol is a 3-carbon molecule that is converted into glyceraldehyde-3-phosphate. Because glyceraldehyde-3-phosphate is involved in one of the steps in glycolysis, it can enter the glycolysis pathway (figure 6.12). The remaining fatty acids are often long molecules (typically 14 to 20 carbons long), which also must be processed before they can be further metabolized. First, they need to enter the mitochondrion, where subsequent reactions take place. Once inside the mitochondrion, each long chain of carbons that makes up the carbon skeleton is hydrolyzed (split by the addition of a water molecule) into 2-carbon fragments. Next, each of the 2-carbon fragments is carried into the Krebs cycle by coenzyme A molecules. Once in the Krebs cycle, they proceed through the Krebs cycle just like the acetyl-CoAs from glucose (Outlooks 6.3).
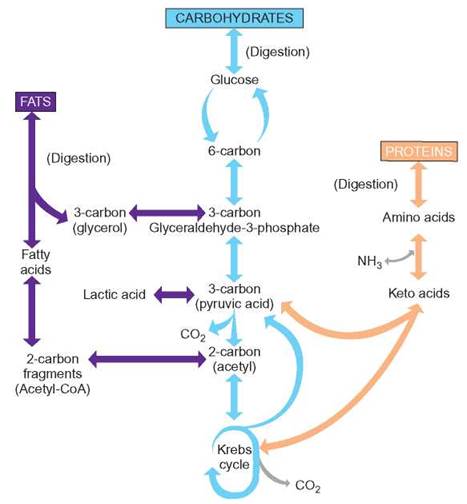
FIGURE 6.12. The Interconversion of Fats, Carbohydrates, and Proteins
Cells do not necessarily use all food as energy. One type of food can be changed into another type to be used as raw materials for the construction of needed molecules or for storage. Notice that many of the reaction arrows have two heads (i.e., these reactions can go in either direction). For example, glycerol can be converted into glyceraldehyde-3-phosphate and glyceraldehyde-3-phosphate can become glycerol.
By following the glycerol and each 2-carbon fragment through the cycle, you can see that each molecule of fat has the potential to release several times as much ATP as does a molecule of glucose. Each glucose molecule has 6 pairs of hydrogen, whereas a typical molecule of fat has up to 10 times that number. This is why fat makes such a good long-term energy storage material. It is also why it takes so long for people on a weight-reducing diet to remove fat. It takes time to use all the energy contained in the fatty acids. On a weight basis, there are twice as many calories in a gram of fat as there are in a gram of carbohydrate.
Fats are an excellent source of energy and the storage of fat is an important process. Furthermore, other kinds of molecules can be converted to fat. You already know that people can get fat from eating sugar. Notice in figure 6.12 that both carbohydrates and fats can enter the Krebs cycle and release energy. Although people require both fats and carbohydrates in their diets, they need not be in precise ratios; the body can make some interconversions. This means that people who eat excessive amounts of carbohydrates will deposit body fat. It also means that people who starve can generate glucose by breaking down fats and using the glycerol to synthesize glucose.
Summary of Fat Respiration
1. Fats are broken down into
a. glycerol.
b. fatty acids.
2. Glycerol
a. is converted to glyceraldehyde-3-phosphate.
b. enters glycolysis.
3. Fatty acids
a. are converted to acetyl-CoA.
b. enter the Kreb’s cycle.
4. Each molecule of fat fuels the formation of many more ATP than glucose.
a. This makes it a good energy-storage molecule.
OUTLOOKS 6.3
Body Odor and Bacterial Metabolism
In our culture, natural body odor is considered by most to be undesirable. Body odor is the result of bacteria metabolizing chemicals released by glands called aprocrine glands. These glands are associated with hair follicles and are especially numerous within the scalp, underarms, and genitals. They produce fatty acids and other compounds that are secreted onto the skin when people sweat as a result of becoming overheated, exercising, or being stressed. Bacteria metabolize these compounds in perspiration, releasing other compounds responsible for body odor.
A number of factors affect how bacteria metabolize fatty acids and, therefore, the strength and nature of a person's body odor. Hereditary factors can play an important role, as evidenced by the genetic abnormality, hyperhidrosis. People with this condition experience excessive perspiration. Diabetes, low blood sugar, menopause, kidney disease, or liver disease can lead to profuse sweating in some cases. Foods, such as garlic and onions, and spices, such as curry, can lead to stronger body aroma. Caffeine, in coffee, tea, sodas, and chocolate, also affects body odor. People with an imbalance of magnesium and zinc are also more likely to generate more pungent body odors.
These bacteria are usually controlled with commercially available products. Deodorants mask the odors, antiperspirants reduce the flow of perspiration, antiseptics destroy the microorganisms, and soaps remove them. Most antiperspirants work by using aluminum compounds (aluminum chlorhydrate) that reduce the flow of sweat and are moderately antibacterial. If a person is allergic to such compounds, it may be necessary to use deodorant soaps with more powerful antimicrobials, such as chlorhexidine.
Protein Respiration
Proteins can be catabolized and interconverted just as fats and carbohydrates are (review figure 6.12). The first step in using protein for energy is to digest the protein into individual amino acids. Each amino acid then needs to have the amino group (—NH2) removed, a process (deamination) that takes place in the liver. The remaining non-nitrogenous part of the protein is converted to keto acid and enters the respiratory cycle as acetyl-CoA, pyruvic acid, or one of the other types of molecules found in the Krebs cycle. As the acids progress through the Krebs cycle, the electrons are removed and sent to the ETS, where their energy is converted into the chemical- bond energy of ATP. The amino group that was removed from the amino acid is converted into ammonia. Some organisms excrete ammonia directly; others convert ammonia into other nitrogen-containing compounds, such as urea (humans) or uric acid (birds). All of these molecules are toxic, increase the workload of the liver, can damage the kidneys and other organs, and must be eliminated. They are transported in the blood to the kidneys, where they are eliminated. In the case of a high-protein diet, increasing fluid intake will allow the kidneys to remove the urea or uric acid efficiently.
When proteins are eaten, they are digested into their component amino acids. These amino acids are then available to be used to construct other proteins. Proteins cannot be stored; if they or their component amino acids are not needed immediately, they will be converted into fat or carbohydrates or will be metabolized to provide energy. This presents a problem for individuals who do not have ready access to a continuous source of amino acids in their diet (e.g., individuals on a low-protein diet). If they do not have a source of dietary protein, they must break down proteins from important cellular components to supply the amino acids they need. This is why proteins and amino acids are considered an important daily food requirement.
Summary of Protein Respiration
1. Proteins are digested into amino acids.
2. Then amino acids have the amino group removed,
a. generating a keto acid (acetic acid, pyruvic acid, etc.), and
b. entering the Kreb’s cycle at the appropriate place.
One of the most important concepts is that carbohydrates, fats, and proteins can all be used to provide energy. The fate of any type of nutrient in a cell depends on the cell’s momentary needs. An organism whose daily food-energy intake exceeds its daily energy expenditure will convert only the necessary amount of food into energy. The excess food will be interconverted according to the enzymes present and the organism’s needs at that time. In fact, glycolysis and the Krebs cycle allow molecules of the three major food types (carbohydrates, fats, and proteins) to be interchanged.
As long as a person’s diet has a certain minimum of each of the three major types of molecules, a cell’s metabolic machinery can manipulate molecules to satisfy its needs. If a person is on a starvation diet, the cells will use stored carbohydrates first. When the carbohydrates are gone (after about 2 days), the cells begin to metabolize stored fat. When the fat is gone (after a few days to weeks), proteins will be used. A person in this condition is likely to die (How Science Works 6.1).
HOW SCIENCE WORKS 6.1
Applying Knowledge of Biochemical Pathways
As scientists have developed a better understanding of the processes of aerobic cellular respiration and anaerobic cellular respiration, several practical applications of this knowledge have developed:
1. Newborn human infants have a modified respiratory plan that allows them to shut down the ATP production of their mitochondria in certain fatty tissue. Even though ATP production is reduced, it allows them to convert fat directly to heat to keep them warm.
2. Studies have shown that horses metabolize their nutrients 20 times faster during the winter than the summer.
3. Although for centuries people have fermented beverages such as beer and wine, they were often plagued by sour products that were undrinkable. Once people understood that there were yeasts that produced alcohol under anaerobic conditions and bacteria that converted alcohol to acetic acid under aerobic conditions, it was a simple task to prevent acetic acid production by preventing oxygen from getting to the fermenting mixture.
4. When it was discovered that the bacterium that causes gas gangrene is anaerobic and is, in fact, poisoned by the presence of oxygen, various oxygen therapies were developed to help cure patients with gangrene. Some persons with gangrene are placed in hyperbaric chambers, with high oxygen levels under pressure. In other patients, only the affected part of the body is enclosed. Under such conditions, the gangrene-causing bacteria die or are inhibited (see figure 4.22).
5. When physicians recognized that the breakdown of fats releases ketone bodies, they were able to diagnose diseases such as diabetes and anorexia more easily, because people typically have low amounts of carbohydrates and therefore metabolize fats. The ketones produced by excess breakdown of fats results in foulsmelling breath.

6.6. CONCEPT REVIEW
10. What are the differences between fat and protein metabolism biochemical pathways?
11. Describe how carbohydrates, fats, and proteins can be interconverted from one to another.
Summary
In aerobic cellular respiration, organisms convert foods into energy (ATP) and waste materials (carbon dioxide and water). Three distinct metabolic pathways are involved in aerobic cellular respiration: glycolysis, the Krebs cycle, and the electron-transport system. Glycolysis takes place in the cytoplasm of the cell, and the Krebs cycle and electron-transport system take place in mitochondria. Organisms that have oxygen can perform aerobic cellular respiration. Organisms and cells that do not use oxygen perform anaerobic cellular respiration (fermentation) and can use only the glycolysis pathway. Aerobic cellular respiration yields much more ATP than anaerobic cellular respiration. Glycolysis and the Krebs cycle serve as a molecular interconversion system: Fats, proteins, and carbohydrates are interconverted according to the cell’s needs.
Basic Review
1. Organisms that are able to use basic energy sources, such as sunlight, to make energy-containing organic molecules from inorganic raw materials are called
a. autotrophs.
b. heterotrophs.
c. aerobic.
d. anaerobic.
2. Cellular respiration processes that do not use molecular oxygen are called
a. heterotrophic.
b. anaerobic.
c. aerobic.
d. anabolic.
3. The chemical activities that remove electrons from glucose result in the glucose being
a. reduced.
b. oxidized.
c. phosphorylated.
d. hydrolysed.
4. The positively charged hydrogen ions that are released from the glucose during cellular respiration eventually combine with _____ ion to form _____.
a. another hydrogen, a gas
b. a carbon, carbon dioxide
c. an oxygen, water
d. a pyruvic acid, lactic acid
5. The Krebs cycle and ETS are biochemical pathways performed in which eukaryotic organelle?
a. nucleus
b. ribosome
c. chloroplast
d. mitochondria
6. In a complete accounting of all the ATPs produced in aerobic cellular respiration in eukaryotic cells, there are a total of _____ ATPs: _____ from the ETS, _____ from glycolysis, and _____ from the Krebs cycle.
a. 36, 32, 2, 2
b. 38, 34, 2, 2
c. 36, 30, 2, 4
d. 38, 30, 4, 4
7. Anaerobic pathways that oxidize glucose to generate ATP energy by using an organic molecule as the ultimate hydrogen acceptor are called
a. fermentation.
b. reduction.
c. Krebs.
d. electron pumps.
8. When skeletal muscle cells function anaerobically, they accumulate the compound _____, which causes muscle soreness.
a. pyruvic acid
b. malic acid
c. carbon dioxide
d. lactic acid
9. Each molecule of fat can release _____ of ATP, compared with a molecule of glucose.
a. smaller amounts
b. the same amount
c. larger amounts
d. only twice the amount
10. Some organisms excrete ammonia directly; others convert ammonia into other nitrogen-containing compounds, such as
a. urea or uric acid.
b. carbon dioxide.
c. sweat.
d. fat.
11. The ATP generating process in mitochondria works by using which of the following?
a. proton pump
b. DNA
c. oxygen pump
d. chlorophyll
12. Which best explains the need to reduce pyruvic acid in fermentation?
a. Fermenting cells cannot produce water.
b. Not enough energy would be produced to keep them alive.
c. There is no oxygen available to accept the electrons.
d. NAD+ needs to be regenerated for continued use in glycolysis.
13. Why don’t human muscle cells produce alcohol and CO2 during anaerobic respiration?
a. They only carry out aerobic respiration.
b. We do not have the genes to produce the enzymes needed to generate alcohol and CO2.
c. The cells would blow up with the gas produced.
d. There is no way to destroy the alcohol.
14. What is the ultimate destination of hydrogen electrons in aerobic cellular respiration?
a. pyruvic acid
b. lactic acid
c. oxygen
d. water
15. Which electron carrier releases the most potential during the ETS?
a. NADH
b. FAD
c. oxygen
d. NAD+
Answers
1. a 2. b 3. b 4. c 5. d 6. a 7. a 8. d 9. c 10. a 11. a 12. d 13. b 14. c 15. A
Thinking Critically
Personalizing Your Pathway
Picture yourself as an atom of hydrogen tied up in a molecule of fat. You are present in the stored fat of a person who is starving. Trace the biochemical pathways you would be part of as you moved through the process of aerobic cellular respiration. Be as specific as you can in describing your location and how you got there, as well as the molecules of which you are a part. Of what molecule would you be a part at the end of this process?