Biology For Dummies
Part I Biology Basics
Chapter 3
The Chemistry of Life
In This Chapter
Seeing why matter is so important
Distinguishing among atoms, elements, isotopes, molecules, and compounds
Getting to know acids and bases
Understanding the structure and function of important molecules for life
Everything that has mass and takes up space, including you and the rest of life on Earth, is made of matter. Atoms make up molecules, which make up the substance of living things. Carbohydrates, proteins, nucleic acids, and lipids are four kinds of molecules that are especially important to the structure and function of organisms. In this chapter, we present a bit of the basic chemistry that’s essential for understanding biology.
Exploring Why Matter Matters
Matter is the stuff of life — literally. Every living thing is made of matter. In order to grow, living things must get more matter to build new structures. When living things die, be they plants or animals, microbes such as bacteria and fungi digest the dead matter and recycle it so that other living things can use it again. In fact, pretty much all the matter on Earth has been here since the planet formed 4.5 billion years ago; it has just been recycled since then. So, the stuff that makes up your body may once have been part of Tyrannosaurus rex, a butterfly, or even a bacterium.
Invisible matter
What looks like nothing but is really something? Air! Earth’s atmosphere may seem like nothing, but it’s made of gases such as nitrogen, carbon dioxide, and oxygen. These gases interact with living things in many ways. Plants, for example, take in carbon dioxide to make food and then use that food to build their structures. It’s hard to believe, but the tallest tree in the redwood forest grows and grows from the result of invisible carbon dioxide gas being taken in and incorporated into the body of the tree. Obviously the redwood tree takes up space and has mass, but those invisible carbon dioxide molecules are matter too.
 Following are a few facts you should know about matter:
Following are a few facts you should know about matter:
Matter takes up space. Space is measured in volume, and volume is measured in liters (L).
Matter has mass. Mass is the term for describing the amount of matter that a substance has. It’s measured in grams (g). Earth’s gravity pulls on your mass, so the more mass you have, the more you weigh.
Matter can take several forms. The most familiar forms of matter are solids, liquids, and gases. Solids have a definite shape and size, such as a person or a brick. Liquids have a definite volume. They can fill a container, but they take the shape of the container that they fill. Gasesare easy to compress and expand to fill a container.
 To understand the difference between mass and weight, compare your weight on Earth versus your weight on the Moon. No matter where you are, your body is made of the same amount of stuff, or matter. But the Moon is so much smaller than Earth that it has a lot less gravity to pull on your mass. So, your weight on the Moon would be just one-sixth of your weight on Earth, but your mass would remain the same!
To understand the difference between mass and weight, compare your weight on Earth versus your weight on the Moon. No matter where you are, your body is made of the same amount of stuff, or matter. But the Moon is so much smaller than Earth that it has a lot less gravity to pull on your mass. So, your weight on the Moon would be just one-sixth of your weight on Earth, but your mass would remain the same!
Recognizing the Differences between Atoms, Elements, and Isotopes
All matter is composed of elements. When you break down matter into its smallest components, you’re left with individual elements that themselves break down into atoms consisting of even smaller pieces called subatomic particles. And sometimes the number of those subatomic particles within a particular atom differs, creating isotopes. Whew. That’s a lot to take in, which is why we break the concepts of atoms, elements, and isotopes down for you in the sections that follow.
“Bohr”ing you with atoms
An atom is the smallest whole, stable piece of an element that still has all the properties of that element. It’s the smallest “piece” of matter that can be measured. Every atom actually contains even smaller pieces known collectively as subatomic particles. These include protons, neutrons, and electrons (and even quarks, mesons, leptons, and neutrinos). Subatomic particles can’t be removed from an atom without destroying the atom.
 Here’s the basic breakdown of an atom’s structure (see Figure 3-1):
Here’s the basic breakdown of an atom’s structure (see Figure 3-1):
The core of an atom, called the nucleus, contains two kinds of subatomic particles: protons and neutrons. Both have mass, but only one carries any kind of charge. Protons carry a positive charge, but neutrons have no charge (they’re neutral). Because the protons are positive and the neutrons have no charge, the net charge of an atom’s nucleus is positive.
Clouds of electrons surround the nucleus. Electrons carry a negative charge but have no mass.
 Atoms become ions when they gain or lose electrons. In other words, ions are essentially charged atoms. Positive (+) ions have more protons than electrons; negative (–) ions have more electrons than protons. Positive and negative charges attract one another, allowing atoms to form bonds, as explained in the later “Molecules, Compounds, and Bonds” section.
Atoms become ions when they gain or lose electrons. In other words, ions are essentially charged atoms. Positive (+) ions have more protons than electrons; negative (–) ions have more electrons than protons. Positive and negative charges attract one another, allowing atoms to form bonds, as explained in the later “Molecules, Compounds, and Bonds” section.
Elements of elements
An element is a substance made of atoms that have the same number of protons. Think of them as “pure” substances all made of the same thing. All the known elements are organized into the periodic table of elements (shown in Figure 3-2), which has the following properties:
Each row of the table is called a period. Moving across the table horizontally, you go from metals to nonmetals, with heavy metals in the middle.
Each column is called a family or group. Elements within the same family/group have similar properties. The size of the atom increases from top to bottom within each column.
Figure 3-1: The Bohr model of anatom’s structure.
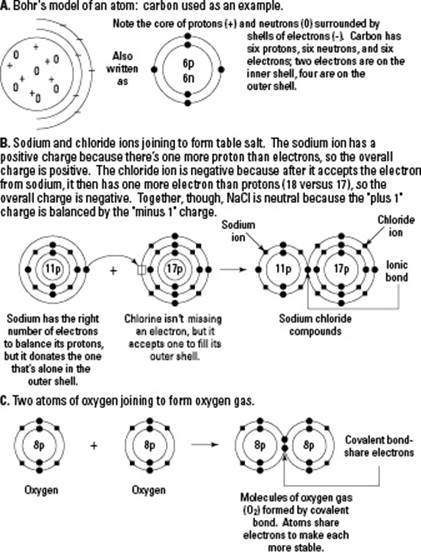
Figure 3-2: The periodic table of elements.

Notice in Figure 3-2 how each element has a number associated with it? That number is the atomic number — the number of protons in the nucleus of an atom of a particular element. For example, carbon (the letter C in the periodic table) has six protons in the nucleus of one atom, so its atomic number is 6. Periodic law states that the properties of elements are a periodic function of their atomic numbers. In other words, when elements are arranged by their atomic number, they form groups with similar properties. The number of electrons in one atom of an element is also equal to the atomic number because atoms are neutral (the positively charged particles are offset by the negatively charged particles one for one).
 Of all the elements in the periodic table, living things use only a handful. The four most common elements found in living things are hydrogen, carbon, nitrogen, and oxygen, all of which are found in air, plants, and water. (Several other elements exist in smaller amounts in organisms, including sodium, magnesium, phosphorus, sulfur, chlorine, potassium, and calcium.)
Of all the elements in the periodic table, living things use only a handful. The four most common elements found in living things are hydrogen, carbon, nitrogen, and oxygen, all of which are found in air, plants, and water. (Several other elements exist in smaller amounts in organisms, including sodium, magnesium, phosphorus, sulfur, chlorine, potassium, and calcium.)
Most often, the elements sodium, magnesium, chlorine, potassium, and calcium circulate in the body as electrolytes, substances that release ions (described in the preceding section) when they break apart in water. For instance, when in the “water” of the body, sodium chloride (NaCl) breaks apart into the ions Na+ and Cl–, which are then used either in organs such as the heart or in cellular processes.
I so dig isotopes
 All atoms of an element have the same number of protons, but the number of neutrons can change. If the number of neutrons is different between two atoms of the same element, the atoms are called isotopes of the element.
All atoms of an element have the same number of protons, but the number of neutrons can change. If the number of neutrons is different between two atoms of the same element, the atoms are called isotopes of the element.
For example, carbon-12 and carbon-14 are two isotopes of the element carbon. Atoms of carbon-12 have 6 protons and 6 neutrons. These carbon atoms have a mass number of 12 because their mass is equal to 12. Atoms of carbon-14 still have 6 protons (because all carbon atoms have 6 protons), but they have 8 neutrons, giving them a mass number of 14.
The atomic mass of an element is the average mass of all the isotopes of that element, taking into account their relative abundance. If you look back at the periodic table in Figure 3-2, you can see that the atomic mass of carbon (written underneath the letter C) is 12.01. This number tells you that if you took the average of the mass of all the carbon atoms on Earth, they’d average out to 12.01. The most stable isotope of carbon is carbon-12, so it’s more abundant than carbon-14. (When you average the mass of lots of atoms of carbon-12 with some of carbon-14, you get a number slightly larger than 12.)
Molecules, Compounds, and Bonds
When you start putting elements together, you get more complex forms of matter, such as molecules and compounds. Molecules are made of two or more atoms, and compounds are molecules that contain at least two different elements.
 One way to sort out the differences between elements, molecules, and compounds is to think about making chocolate chip cookies. First, you need to mix the wet ingredients: butter, sugar, eggs, and vanilla. Consider each of those ingredients a separate element. You need two sticks of the element butter. When you combine butter plus butter, you get a molecule of butter. Before you add the element of eggs, you need to beat them. So, when you add egg plus egg in a little dish, you get a molecule of eggs. When all the wet ingredients are mixed together, the molecule of butter is combined with the molecule of eggs, and you get a compound called “wet.” Next, you need to mix together the dry ingredients: flour, salt, and baking soda. Think of each of those ingredients as a separate element. When all the dry ingredients are mixed together, you get a compound called “dry.” Only when the wet compound is mixed with the dry compound is the reaction sufficiently ready for the most important element: the chocolate chips.
One way to sort out the differences between elements, molecules, and compounds is to think about making chocolate chip cookies. First, you need to mix the wet ingredients: butter, sugar, eggs, and vanilla. Consider each of those ingredients a separate element. You need two sticks of the element butter. When you combine butter plus butter, you get a molecule of butter. Before you add the element of eggs, you need to beat them. So, when you add egg plus egg in a little dish, you get a molecule of eggs. When all the wet ingredients are mixed together, the molecule of butter is combined with the molecule of eggs, and you get a compound called “wet.” Next, you need to mix together the dry ingredients: flour, salt, and baking soda. Think of each of those ingredients as a separate element. When all the dry ingredients are mixed together, you get a compound called “dry.” Only when the wet compound is mixed with the dry compound is the reaction sufficiently ready for the most important element: the chocolate chips.
So what holds the elements of molecules and compounds together? Bonds, of course. Two important types of bonds exist in living things:
Ionic bonds hold ions joined together by their opposite electrical charges. Ionic reactions occur when atoms combine and lose or gain electrons. When sodium (Na) and chlorine (Cl) combine, for example, sodium loses an electron to chlorine. Sodium becomes the positively charged sodium ion (Na+), and chlorine becomes the negatively charged chloride ion (Cl–). These two oppositely charged ions are attracted to each other, forming an ionic bond.
Covalent bonds are formed when atoms share electrons in a covalent reaction. When two oxygen atoms join together to form oxygen gas, they share two pairs of electrons with each other. Each shared pair of electrons is one covalent bond, so the two pairs of shared electrons in a molecule of oxygen gas have a double bond. Covalent bonds are extremely important in biology because they hold together the backbones of all biological molecules.
Acids and Bases (Not a Heavy Metal Band)
Some substances, such as lemon juice and vinegar, have a real edge when you taste them. Others, such as battery acid and ammonia, are so caustic you don’t even want to get them on your skin. These substances are acids and bases, both of which have the potential to damage cells.
Acids are molecules that can split apart in water and release hydrogen ions (H+). A common example is hydrochloric acid (HCl). When HCl is added to water, it splits apart into H+ and Cl–, increasing the number of hydrogen ions in the water/HCl solution.
Bases are molecules that can split apart in water and release hydroxide ions (OH–). The most common example is sodium hydroxide (NaOH). When NaOH is added to water, it splits apart into Na+ and OH–.
Charged particles, like hydrogen and hydroxide ions, can interfere with the chemical bonds that hold molecules together. Because living things are made of molecules, strong acids and bases can release enough of these ions to cause damage.
 Even water can split apart to create hydrogen and hydroxide ions. Each water molecule (H2O) can separate into one hydrogen ion (H+) and one hydroxide ion (OH–). These ions can easily recombine to reform water molecules and will keep shifting back and forth between their molecular and ionized forms. In pure water, the number of hydrogen ions and hydroxide ions are balanced, so they don’t do any damage to living things.
Even water can split apart to create hydrogen and hydroxide ions. Each water molecule (H2O) can separate into one hydrogen ion (H+) and one hydroxide ion (OH–). These ions can easily recombine to reform water molecules and will keep shifting back and forth between their molecular and ionized forms. In pure water, the number of hydrogen ions and hydroxide ions are balanced, so they don’t do any damage to living things.
The relative concentration of hydrogen to hydroxide ions is represented by the pH scale. The following sections explain the pH scale and how organisms regulate themselves when their pH gets out of balance.
“Ph”iguring out the pH scale
In the early 1900s, scientists came up with the pH scale, a system of classifying how acidic or basic a solution is. The term pH symbolizes the hydrogen ion concentration in a solution (for example, what proportion of a solution contains hydrogen ions). The pH scale goes from 1 to 14. A pH of 7 is neutral, meaning the amount of hydrogen ions and hydroxide ions in a solution with a pH of 7 is equal, just like in pure water.
A solution that contains more hydrogen ions than hydroxide ions is acidic, and the pH of the solution is less than 7. If a molecule releases hydrogen ions in water, it’s an acid. The more hydrogen ions it releases, the stronger the acid, and the lower the pH value.
A solution that contains more hydroxide ions than hydrogen ions is basic, and its pH is higher than 7. Bases dissociate (break apart) into hydroxide ions (OH–) and a positive ion. The hydroxide ions can combine with H+ to create water. Because the hydrogen ions are used, the number of hydrogen ions in the solution decreases, making the solution less acidic and therefore more basic. So, the more hydroxide ions a molecule releases (or the more hydrogen ions it takes in), the more basic it is.
Table 3-1 shows you the pH of some common substances. Use it to help you visually figure out the pH scale.
|
Table 3-1 The pH of Some Common Substances |
|
|
Increasing pH |
Substances |
|
0 (most acidic) |
Hydrochloric acid (HCl) |
|
1 |
Battery acid |
|
2 |
Lemon juice, vinegar, stomach acid |
|
3 |
Cola, apples |
|
4 |
Beer |
|
4.5 |
Tomatoes |
|
5 |
Black coffee, bananas |
|
5.5 |
Normal rainwater |
|
6 |
Urine |
|
6.5 |
Saliva, milk |
|
7 (neutral) |
Water, tears |
|
7.5 |
Human blood |
|
8 |
Seawater, eggs |
|
9 |
Baking soda, antacids |
|
10 |
Great Salt Lake |
|
11 |
Ammonia |
|
12 |
Bicarbonate of soda, soapy water |
|
13 |
Oven cleaner, bleach |
|
14 (most basic) |
Sodium hydroxide (NaOH), liquid drain cleaner |
Buffing up on buffers
In organisms, blood or cytoplasm are the “solutions” in which the required ions (for example, electrolytes) are floating. That’s why most substances in the body hover around the neutral pH of 7. However, nothing’s perfect, so the human body has a backup system in case things go awry. A system of buffers exists to help neutralize the blood if excess hydrogen or hydroxide ions are produced.
 Buffers keep solutions at a steady pH by combining with excess hydrogen (H+) or hydroxide (OH–) ions. Think of them as sponges for hydrogen and hydroxide ions. If a substance releases these ions into a buffered solution, the buffers will “soak up” the extra ions.
Buffers keep solutions at a steady pH by combining with excess hydrogen (H+) or hydroxide (OH–) ions. Think of them as sponges for hydrogen and hydroxide ions. If a substance releases these ions into a buffered solution, the buffers will “soak up” the extra ions.
The most common buffers in the human body are bicarbonate ion (HCO3–) and carbonic acid (H2CO3). Bicarbonate ion carries carbon dioxide through the bloodstream to the lungs to be exhaled (see Chapter 15 for more on the respiratory system), but it also acts as a buffer. Bicarbonate ion takes up extra hydrogen ions, forming carbonic acid and preventing the pH of the blood from going too low. If the opposite situation occurs and the pH of the blood gets too high, carbonic acid breaks apart to release some hydrogen ions, which brings the pH back into balance.
 If something goes wrong with the buffer system and the pH drops too low, an organism can develop acidosis (meaning the blood becomes too acidic). If the reverse happens and the pH gets too high, an organism can develop alkalosis (meaning the blood becomes too basic).
If something goes wrong with the buffer system and the pH drops too low, an organism can develop acidosis (meaning the blood becomes too acidic). If the reverse happens and the pH gets too high, an organism can develop alkalosis (meaning the blood becomes too basic).
Carbon-Based Molecules: The Basis for All Life
All living things rely pretty heavily on one particular type of molecule: carbon. The little ol’ carbon atom, with its six protons and an outer shell of four electrons, is the central focus of organic chemistry, which is the chemistry of living things. When carbon bonds to hydrogen (which happens frequently in organic molecules), the carbon and hydrogen atoms share a pair of electrons in a covalent bond. Molecules with lots of carbon-hydrogen bonds are called hydrocarbons. Nitrogen, sulfur, and oxygen are also often joined to carbon in organisms.
So where do the carbon-containing molecules come from? The answer’s simple: food. Some living things, like people, need to eat other living things to get their food, but some organisms, like plants, can make their own food. Regardless of the food source, all living things use food as a supply of carbon-containing molecules.
Carbon atoms are central to all organisms because they’re found in carbohydrates, proteins, nucleic acids, and lipids — otherwise known as the structural materials of all living things. The sections that follow describe the roles of these materials.
Providing energy: Carbohydrates
Carbohydrates, as the name implies, consist of carbon, hydrogen, and oxygen. The basic formula for carbohydrates is CH2O, meaning the core structure of a carbohydrate is one carbon atom, two hydrogen atoms, and one oxygen atom. This formula can be multiplied; for example, glucose has the formula C6H12O6, which is six times the ratio, but still the same basic formula.
 But what is a carbohydrate? Well, carbohydrates are energy-packed compounds. Living creatures can break carbohydrates down quickly, making them a source of near-immediate energy. However, the energy supplied by carbohydrates doesn’t last long. Therefore, reserves of carbohydrates in the body must be replenished frequently, which is why you find yourself hungry every four hours or so. Although carbohydrates are a source of energy, they also serve as structural elements (such as cell walls in plants).
But what is a carbohydrate? Well, carbohydrates are energy-packed compounds. Living creatures can break carbohydrates down quickly, making them a source of near-immediate energy. However, the energy supplied by carbohydrates doesn’t last long. Therefore, reserves of carbohydrates in the body must be replenished frequently, which is why you find yourself hungry every four hours or so. Although carbohydrates are a source of energy, they also serve as structural elements (such as cell walls in plants).
Carbohydrates come in the following forms:
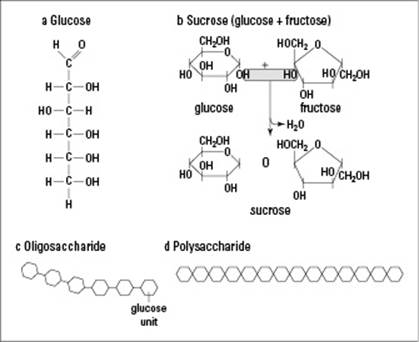
Monosaccharides: Simple sugars consisting of three to seven carbon atoms are monosaccharides (see Figure 3-3a). In living things, monosaccharides form ring-shaped structures and can join together to form longer sugars. The most common monosaccharide is glucose.
Disaccharides: Two monosaccharide molecules joined together form a disaccharide (see Figure 3-3b). Common disaccharides include sucrose (table sugar) and lactose (the sugar found in milk).
Oligosaccharides: More than two but just a few monosaccharides joined together are an oligosaccharide (see Figure 3-3c). Oligosaccharides are important markers on the outsides of your cells (head to Chapter 4 for more on cells), such as the oligosaccharides that determine whether your blood type is A or B (people with type O blood don’t have any of this particular oligosaccharide).
Polysaccharides: Long chains of monosaccharide molecules linked together form a polysaccharide (see Figure 3-3d). Some of these babies are huge, and when we say huge, we mean some of them can have thousands of monosaccharide molecules joined together. Starch and glycogen, which serve as a means of storing carbohydrates in plants and animals, respectively, are examples of polysaccharides.
Figure 3-3: A variety of carbohydrate molecules.
 Note that most of the names of carbohydrates end in -ose. Glucose, fructose, ribose, sucrose, maltose — these are all sugars. A sugar is a carbohydrate that dissolves in water, tastes sweet, and can form crystals. Just like, well, the sugar in your sugar bowl.
Note that most of the names of carbohydrates end in -ose. Glucose, fructose, ribose, sucrose, maltose — these are all sugars. A sugar is a carbohydrate that dissolves in water, tastes sweet, and can form crystals. Just like, well, the sugar in your sugar bowl.
What’s the big deal about “natural” sugars?
Sugars are an important source of energy and carbon for living things. Yet many people these days have a love-hate relationship with sugar. Overconsumption of sugars such as sucrose can lead to obesity, diabetes, and tooth decay, so sometimes people perceive sugar as harmful to the body. This perception has spawned a huge branch of the food industry dedicated to finding alternatives to sucrose. Some products advertise themselves as healthier because they contain “natural” sugars such as fructose (the sugar in fruit). However, fructose really isn’t that different from sucrose — in fact, it’s one of the two monosaccharides that are part of sucrose. Fructose is a six-carbon sugar molecule that digests a little more slowly than glucose and is sweeter to the taste. So, potentially less fructose can be used to achieve the same effect as sucrose or glucose. However, the sweetening power of fructose depends on the food it’s used in. Often there isn’t much nutritional difference between foods sweetened with sucrose and those sweetened with fructose. Plus, much of the commercially available fructose is actually derived from sucrose, not extracted from fruit. So although “natural” fruit sugar may sound better, the differences between sugars may be so slight that they don’t make much difference to the body at all.
The next sections explain how sugars interact with one another and how the human body stores a particular carbohydrate known as glucose.
Making and breaking sugars
Monosaccharides join together in a process known as dehydration synthesis, which involves two molecules bonding and losing a water molecule. Figure 3-3b shows the dehydration synthesis of glucose and fructose to form sucrose.
 The term dehydration synthesis may sound technical, but it’s not at all if you really think about what the words mean. Dehydration, as we’re sure you know, is what happens when you don’t drink enough water. You dry out because water is removed (but not completely) from some cells, such as those in your tongue, to make sure more important cells, like those in your heart or brain, continue to function. Synthesis means making something. If you really think about it, in dehydration synthesis, something must be made when water is removed. That’s exactly what happens. When glucose and fructose get together, a water molecule is removed from the monosaccharides and given off as a byproduct of the reaction.
The term dehydration synthesis may sound technical, but it’s not at all if you really think about what the words mean. Dehydration, as we’re sure you know, is what happens when you don’t drink enough water. You dry out because water is removed (but not completely) from some cells, such as those in your tongue, to make sure more important cells, like those in your heart or brain, continue to function. Synthesis means making something. If you really think about it, in dehydration synthesis, something must be made when water is removed. That’s exactly what happens. When glucose and fructose get together, a water molecule is removed from the monosaccharides and given off as a byproduct of the reaction.
The opposite of dehydration synthesis is hydrolysis. A hydrolysis reaction breaks down a larger sugar molecule into its original monosaccharides. When something undergoes hydrolysis, a water molecule splits a compound (hydro means “water”; lysis means “break apart”). When sucrose is added to water, it splits apart into glucose and fructose.
Converting glucose for storage purposes
Carbohydrates are found in nearly every food, not just bread and pasta. Fruits, vegetables, and even meats also contain carbohydrates, although meats don’t contain very many. Basically, any food that contains sugar has carbohydrates, and most foods are converted to sugars when they’re digested.
When you digest your food, the carbohydrates from it break down into small sugars such as glucose. Those glucose molecules are then absorbed from your intestinal cells into your bloodstream, which carries the glucose molecules throughout your entire body. The glucose enters each of your body’s cells and is used as a source of carbon and energy.
Because glucose provides a rapid source of energy, organisms often keep some on hand. They store it in various polysaccharides that can be quickly broken down when glucose is needed. Consider the following list your primer on the things glucose can be stored as:
Glycogen: Animals, including people, store a polysaccharide of glucose called glycogen. It has a compact structure, so lots of it can be stored in cells for later use. Your liver, in particular, keeps a large glycogen reserve on hand for when you exercise.
Starch: Plants store glucose as the polysaccharide starch. The leaves of a plant produce sugar during the process of photosynthesis and then store some of that sugar as starch. When the simple sugars need to be retrieved for use, the starch is broken down into its smaller components.
 Plants also make a polysaccharide of glucose called cellulose. Cellulose plays a structural role for plants rather than a storage role by giving rigidity to the plant cells. Most animals, including people, can’t digest cellulose because of the type of bonds between the glucose molecules. Because cellulose passes through your digestive tract virtually untouched, it helps maintain the health of your intestines.
Plants also make a polysaccharide of glucose called cellulose. Cellulose plays a structural role for plants rather than a storage role by giving rigidity to the plant cells. Most animals, including people, can’t digest cellulose because of the type of bonds between the glucose molecules. Because cellulose passes through your digestive tract virtually untouched, it helps maintain the health of your intestines.
Making life possible: Proteins
Without proteins, living things wouldn’t exist. Many proteins provide structure to cells; others bind to and carry important molecules throughout the body. Some proteins are involved in reactions in the body when they serve as enzymes (see Chapter 4 for more on enzymes). Still others are involved in muscle contraction or immune responses. Proteins are so diverse that we can’t possibly tell you about all of them. What we can tell you about, however, are the basics of their structure and their most important functions.
The building blocks of proteins
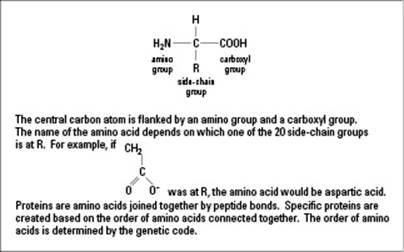
Amino acids, of which there are 20, are the foundation of all proteins. Think of them as train cars that make up an entire train called a protein. Figure 3-4 shows what one amino acid looks like.
The genetic information in cells calls for amino acids to link together in a certain order, forming chains called polypeptide chains. Amino acids link together by dehydration synthesis, just like sugars do (as explained in the earlier “Making and breaking sugars” section), and each polypeptide chain is made up of a unique number and order of amino acids.
Figure 3-4:Amino acid structure.
The main functions of proteins
One or more polypeptide chains come together to form functional proteins. Once formed, each protein does a specific job or makes up a specific tissue in the body.
Enzymes are proteins that speed up the rate of chemical reactions. Metabolic processes don’t happen automatically; they require enzymes. For the full scoop on enzymes, head to Chapter 4.
Structural proteins reinforce cells and tissues. Collagen, a structural protein found in connective tissue (the tissue that joins muscles to bones to allow movement), is the most abundant protein in animals with a backbone. Connective tissue includes ligaments, tendons, cartilage, bone tissue, and even the cornea of the eye. It provides support in the body, and it has a great capability to be flexible and resistant to stretching.
Transport proteins move materials around cells and around the body. Hemoglobin is a transport protein found in red blood cells that carries oxygen around the body. A hemoglobin molecule is shaped kind of like a three-dimensional four-leaf clover without a stem. Each leaf of the clover is a separate polypeptide chain. In the center of the clover, but touching each polypeptide chain, is a heme group with an atom of iron at its center. When gas exchange occurs between the lungs and a blood cell (for more on respiration and circulation, see Chapter 15), the iron atom attaches to the oxygen. Then, the iron-oxygen complex releases from the hemoglobin molecule in the red blood cell so the oxygen can cross cell membranes and get inside any cell of the body.
Drawing the cellular road map: Nucleic acids
Until as recently as the 1940s, scientists thought that genetic information was carried in the proteins of the body. They thought nucleic acids, a new discovery at the time, were too small to be significant. That all changed in 1953 when James Watson and Francis Crick figured out the structure of a nucleic acid, proving things were the other way around: Nucleic acids created the proteins!
Nucleic acids are large molecules that carry tons of small details, specifically all the genetic information for an organism. Nucleic acids are found in every living thing — plants, animals, bacteria, and fungi. Just think about that fact for a moment. People may look different than fungi, and plants may behave differently than bacteria, but deep down all living things contain the same chemical “ingredients” making up very similar genetic material.
 Nucleic acids are made up of strands of nucleotides. Each nucleotide has three components of its own:
Nucleic acids are made up of strands of nucleotides. Each nucleotide has three components of its own:
A nitrogen-containing base called a nitrogenous base
A sugar that contains five-carbon molecules
A phosphate group
That’s it. Your entire genetic composition, personality, and maybe even your intelligence hinge on molecules containing a nitrogen compound, some sugar, and a phosphate. The following sections introduce you to the two types of nucleic acids.
Deoxyribonucleic acid (DNA)
You may have heard DNA (short for deoxyribonucleic acid) referred to as “the double helix.” That’s because DNA contains two strands of nucleotides arranged in a way that makes it look like a twisted ladder. See for yourself in Figure 3-5.
Figure 3-5: The twisted-ladder model of a DNA double helix.
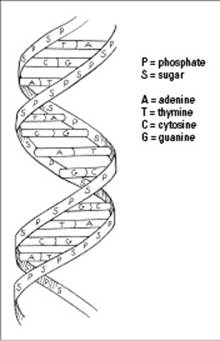
The sides of the ladder are made up of sugar and phosphate molecules, hence the nickname “sugar-phosphate backbone.” (The name of the sugar in DNA is deoxyribose.) The “rungs” on the ladder of DNA are made from pairs of nitrogenous bases from the two strands.
 The nitrogenous bases that DNA builds its double helix upon are adenine (A), guanine (G), cytosine (C), and thymine (T). The order of these chemical letters spells out your genetic code. Oddly enough, the bases always pair in a certain way: Adenine always goes with thymine (A-T), and guanine always links up with cytosine (G-C). These particular base pairs line up just right chemically so that hydrogen bonds can form between them.
The nitrogenous bases that DNA builds its double helix upon are adenine (A), guanine (G), cytosine (C), and thymine (T). The order of these chemical letters spells out your genetic code. Oddly enough, the bases always pair in a certain way: Adenine always goes with thymine (A-T), and guanine always links up with cytosine (G-C). These particular base pairs line up just right chemically so that hydrogen bonds can form between them.
Certain sections of nitrogenous bases along a strand of DNA form a gene. A gene is a unit that contains the genetic information or codes for a particular protein and transmits hereditary information to the next generation. Whenever a new cell is made in an organism, the genetic material is reproduced and put into the new cell. (You can find details about this in Chapter 6.) The new cell can then create proteins and also pass on the genetic information to the next new cell.
But genes aren’t found only in reproductive cells. Every cell in an organism contains DNA (and therefore genes) because every cell needs to make proteins. Proteins control function and provide structure. Therefore, the blueprints of life are stored in each and every cell.
The order of the nitrogenous bases on a strand of DNA (or in a section of the DNA that makes up a gene) determines the order in which amino acids are strung together to make a protein. Which protein is produced determines which structural element is produced within your body (such as muscle tissue, skin, or hair) or what function can be performed (such as the transportation of oxygen to all the cells).
 Every cellular process and every aspect of metabolism is based on genetic information stored in DNA and thus the production of the proper proteins. If the wrong protein is produced (as in the case of cancer), then disease occurs.
Every cellular process and every aspect of metabolism is based on genetic information stored in DNA and thus the production of the proper proteins. If the wrong protein is produced (as in the case of cancer), then disease occurs.
Ribonucleic acid (RNA)
RNA, short for ribonucleic acid, is a chain of nucleotides that serves as an important information molecule. It plays an important role in the creation of new proteins (which we cover in Chapter 8). The structure of RNA is slightly different from that of DNA.
RNA molecules have only one strand of nucleotides.
The nitrogenous bases used are adenine, guanine, cytosine, and uracil (rather than thymine).
The sugar in RNA is ribose (not deoxyribose).
Supplying structure, energy, and more: Lipids
In addition to carbohydrates, proteins, and nucleic acids, your body needs one more type of large molecule to survive. Yet, if you’re like most people, you try to avoid too much of it in your diet. We’re talking about fats, which can be both a blessing and a curse because of their incredibleenergy density (the ability to store lots of calories in a small space). The energy density of fats makes them a highly efficient way for living things to store energy — very useful when food isn’t always available. But that same energy density makes it really easy to pack in the calories when you eat fatty foods!
Fats are an example of a type of molecule called lipids. Lipids are hydrophobic molecules, meaning they don’t mix well with water. You’ve probably heard the saying that “oil and water don’t mix.” Well, oil is a liquid lipid, so the old saying is true; it really doesn’t mix with water. Butter and lard are examples of solid lipids, as are waxes, which are valued for their water-repellent properties on snowboards, skis, and automobiles.
 Three major types of lipid molecules exist:
Three major types of lipid molecules exist:
Phospholipids: These lipids, made up of two fatty acids and a phosphate group, have an important structural function for living things because they’re part of the membranes of cells (see Chapter 4 for more on cell membranes). Phospholipids aren’t the type of lipid floating around the bloodstream clogging arteries.
Steroids: These lipid compounds, consisting of four connecting carbon rings and a functional group that determines the steroid, generally create hormones. Cholesterol is a steroid molecule used to make testosterone and estrogen; it’s also found in the membranes of cells. The downside to cholesterol is that it’s transported around the body by other lipids. If you have too much cholesterol floating in your bloodstream, then you have an excess of fats carrying it through your bloodstream. This situation is troubling because the fats and cholesterol molecules can get stuck in your blood vessels, leading to blockages that cause heart attacks or strokes.
Triglycerides: These fats and oils, which are made up of three fatty acid molecules and a glycerol molecule, are important for energy storage and insulation. In people, fats form from an excess of glucose. After the liver stores all the glucose it can as glycogen, whatever remains is turned into triglycerides. (Both sugars and fats are made of carbon, hydrogen, and oxygen, so your cells just rearrange the atoms to convert from one to another.) The triglycerides float through your bloodstream on their way to be deposited into adipose tissue — the soft, squishy fat you can see on your body. Adipose tissue is made up of many, many molecules of fat. The more fat molecules that are added to the adipose tissue, the bigger the adipose tissue (and the place on your body that contains it) gets.
Whether a triglyceride is a fat or an oil depends on the bonds between the carbon and hydrogen atoms.
• Fats contain lots of single bonds between their carbon atoms. These saturated bonds pack tightly (see Figure 3-6), so fats are solid at room temperature.
• Oils contain lots of double bonds between their carbon atoms. These unsaturated bonds don’t pack tightly (see Figure 3-6), so oils are liquid at room temperature.
Figure 3-6:Saturated and unsaturated bonds in a typical triglyceride.

Fat provides an energy reserve to your body. When you use up all of your stored glucose (which doesn’t take long because sugars “burn” quickly in aerobic conditions), your body starts breaking down glycogen, which is stored primarily in the liver and muscle. Liver glycogen stores can typically last 12 or more hours. After that, your body starts breaking down adipose tissue to retrieve some stored energy. That’s why aerobic exercise, so long as it’s enough to use up more calories than you took in that day, is the best way to lose fat. (Notice we didn’t say “pounds” here. Pounds measure everything in your body’s composition: fat tissue, muscle tissue, and bone, along with water, your organs, skin, and some incidental stuff.)