Biology of Humans
2. Chemistry Comes to Life
Chemistry is the branch of science that is concerned with the composition and properties of substances, including the stuff our bodies are built from. In Chapter 1, we learned that certain large molecules—carbohydrates, lipids, proteins, and nucleic acids—are found in all living organisms. In this chapter, we Major Molecules of Life take a closer look at those molecules. We consider basic chemical concepts, such as the way atoms bond and the way molecules join to form the major molecules of life. By understanding some fundamental concepts of chemistry, we gain important insights into how our bodies work. This chapter lays a foundation for discussions throughout the rest of the book.
The Nature of Atoms
The world around you contains an amazing variety of physical substances: the grass or concrete you walk on, the water you drink, the air you breathe, and even this book you are reading. All of these substances that make up our world are called matter. Fundamentally, matter is anything that takes up space and has mass. The three traditional states of matter are solids, liquids, or gases. All forms of matter are made up of atoms.
Atoms are units of matter that cannot be broken down into simpler substances by ordinary chemical means. Each atom is composed of even smaller, subatomic particles, such as protons, neutrons, and electrons. These subatomic particles are characterized by their location within the atom, their electrical charge, and their mass.
Each atom has a nucleus at its center and a surrounding spherical "cloud" of electrons. As you can see in Figure 2.1, the nucleus contains protons and neutrons. Electrons, on the other hand, move around the nucleus and occur at certain energy levels called shells.
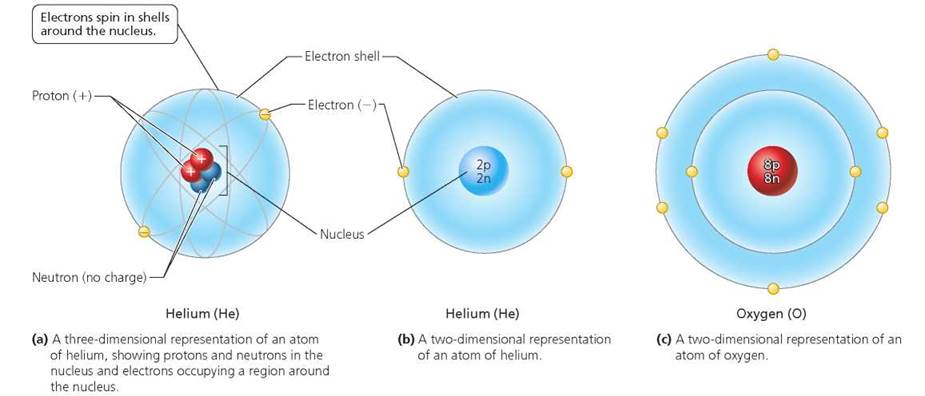
FIGURE 2.1. Atoms can be represented in different ways.
· Understanding basic chemical principles can help us understand how the human body is put together and how it works.
Note that shells are three-dimensional spaces, although they are often depicted in textbook figures as two-dimensional circles for convenience. The shell closest to the nucleus can hold up to 2 electrons. The next shell out can hold up to 8 electrons. Atoms with more than 10 electrons have additional shells. Electrons have different amounts of energy; those with the most energy are found farthest from the nucleus. The chemical properties of an atom are determined by the number of electrons in its outermost shell. As we will see, those atoms whose outermost shells are not full tend to interact with other atoms.
Neutrons, as their name implies, are neutral: they have no electrical charge. In contrast, protons have a positive charge, and electrons have a negative charge. The negatively charged electrons stay near the nucleus because they are attracted to the positively charged protons in it. Most atoms have the same number of positively charged protons and negatively charged electrons. As a result, they are "neutral," having no net charge. Table 2.1 summarizes the basic characteristics of protons, neutrons, and electrons.
TABLE 2.1. Review of Subatomic Particles
Particle |
Location |
Charge |
Mass |
Proton |
Nucleus |
1 positive unit |
1 atomic mass unit |
Neutron |
Nucleus |
None |
1 atomic mass unit |
Electron |
Outside the nucleus |
1 negative unit |
Negligible |
Elements
An element is a pure form of matter containing only one kind of atom. You are probably familiar with many elements, such as gold, silver, iron, and oxygen. Earth and everything on it or in its atmosphere is made up of a little more than 100 elements. Only about 20 elements are found in the human body, which consists mostly of carbon, oxygen, hydrogen, and nitrogen. Each element consists of atoms containing a certain number of protons in the nucleus. For example, all carbon atoms have six protons. The number of protons in the atom's nucleus is called the atomic number.
The periodic table lists the elements and describes many of their characteristics. Figure 2.2 depicts the periodic table. Note that each element has a name and a one- or two-letter symbol. The symbol for the element carbon is C, and that for chlorine is Cl. (The abbreviations are not always as intuitive; for example, the abbreviation for gold is Au, based on the Latin name for the metal.) Besides an atomic number, each atom also has an atomic weight. Each proton and neutron has an approximate mass of 1 atomic mass unit, or amu. The mass of an electron is so small that it is usually considered zero. Because electrons have negligible mass, and protons and neutrons each have an atomic mass of 1, the atomic weight for any atom equals the number of protons plus the number of neutrons. Oxygen has an atomic weight of 16, indicating that it has eight protons (we know this from its atomic number) and eight neutrons in its nucleus.
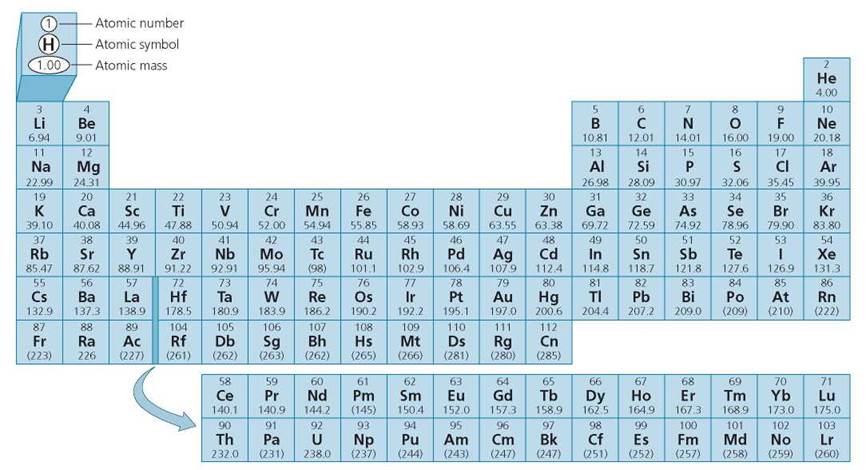
FIGURE 2.2. The periodic table
Isotopes and Radioisotopes
All the atoms of a particular element contain the same number of protons; they can, however, have different numbers of neutrons. Such differences result in atoms of the same element having slightly different atomic weights. Atoms that have the same number of protons but differ in the number of neutrons are called isotopes. More than 300 isotopes occur naturally on Earth. The element carbon, for example, has 3 isotopes. All carbon atoms have six protons in the nucleus. Most carbon atoms also have six neutrons, but some have seven or eight. The isotopes of carbon thus have atomic weights of 12, 13, and 14, respectively, depending on the number of neutrons in the nucleus. These isotopes are written 12C (the most common form in nature), 13C, and 14C.
Radiation is energy moving through space. Examples include radio waves, light, heat, and the excess energy or particles given off by unstable isotopes as they break down. Some elements have both stable and unstable isotopes. Unstable, radiation-emitting isotopes are called radioisotopes. About 60 occur naturally, and many more have been made in laboratories.
Depending on the context, radiation can be dangerous (Figure 2.3) or useful (Figure 2.4). Direct damage to a person absorbing harmful radiation may include a reduction in their white blood cell count, development of some cancers, and damage to organs, such as the skin. In other cases, radiation may not produce any noticeable injury to the person who was exposed; but it may alter the hereditary material in the cells of his or her reproductive system, possibly causing defects in the individual's offspring. For an example of the harmful effects of radiation and how to protect yourself from radiation that occurs naturally in the environment, see the Environmental Issue essay, Radon Gas: A Killer That Can Be Stopped.

FIGURE 2.3. Perhaps the most common burn from radiation, sunburn is caused by overexposure to ultraviolet radiation from the sun’s rays.
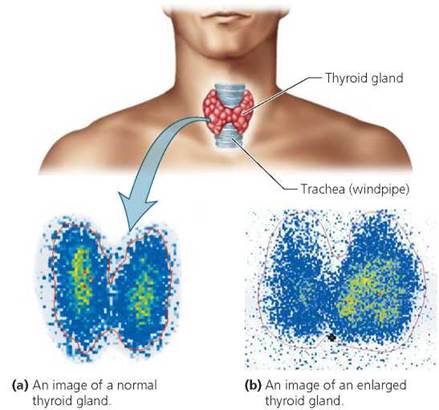
FIGURE 2.4. Radioactive iodine can be used to generate images of the thyroid gland. Such images may be used to diagnose metabolic disorders.
In stark contrast to the harmful effects of radiation are its medical uses. Medical professionals use radiation for diagnosis and therapy. Perhaps the most familiar diagnostic use of radiation is the x-ray. A less common diagnostic procedure is the use of small doses of radiation to generate visual images of internal body parts. Radioactive iodine, for example, is often used to identify disorders of the thyroid gland. This gland, located in the neck, normally accumulates the element iodine, which it uses to regulate growth and metabolism. Small doses of iodine-131 (131I), a radioactive isotope, can be given to a patient suspected of having metabolic problems. The radioactive iodine is taken up by the patient's thyroid gland and detected by medical instruments, as shown in Figure 2.4. The small amount of radioactive iodine used in imaging does not damage the thyroid gland or surrounding structures. However, larger doses can be used to kill thyroid cells when the gland is enlarged and overactive.
Radiation can also be used to kill cancer cells. Cancer cells divide more rapidly and have higher rates of metabolic activity than do most normal cells. For these reasons, cancer cells are more susceptible to the destructive effects of radiation. Still, when medical professionals aim an outside beam of radiation at a tumor to kill the cancer cells inside it, they must also take steps to shield the surrounding healthy tissue. Sometimes a radiation source is placed within the body to treat a cancer. For example, one treatment for prostate cancer (the prostate gland is an accessory reproductive gland in males) involves the placement of radioactive seeds (pellets) directly in the prostate gland (Figure 2.5). Once in place, the seeds emit radiation that damages or kills nearby cancer cells. In most cases, the seeds are left in place, even though they stop emitting radiation within 1 year.
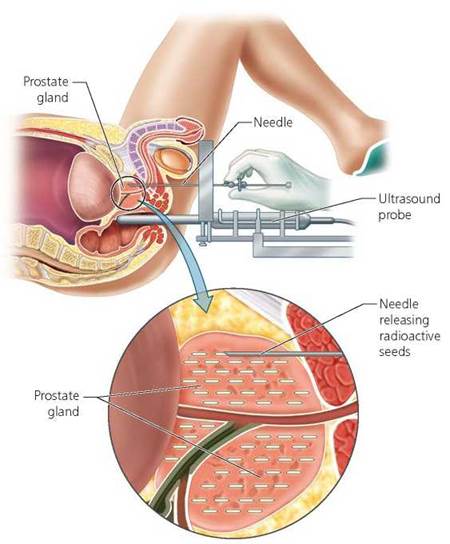
FIGURE 2.5. Prostate cancer can be treated by the implantation of radioactive seeds in the prostate gland. A physician injects the seeds through needles, with guidance from an ultrasound probe placed in the rectum.
What would you do?
Irradiation is the process in which an item is exposed to radiation. Many foods today are intentionally irradiated for various reasons, such as delaying spoilage, increasing shelf life, and removing harmful microorganisms, insect pests, and parasites. The food does not become radioactive as a result. Supporters of the practice note that test animals fed on irradiated food show no adverse effects. Opponents, however, point to the environmental risks of building and operating food irradiation plants. Opponents also note that carefully controlled, long-term experiments are still needed to verify that irradiated food is safe for people of all ages and nutritional states. Several foods, including white potatoes, wheat flour, fresh meat and poultry, and fresh spinach and iceberg lettuce, can be irradiated in the United States. If the entire product is irradiated, then a distinctive logo (Figure 2.6) must appear on its packaging. If an irradiated food is an ingredient in another product, then it must be listed as irradiated in the ingredients statement, but the logo is not required. Would you eat irradiated food? What would you base your decision on?

FIGURE 2.6. Logo for irradiated foods. This logo and words such as “Treated with radiation" must appear on food that has been irradiated in its entirety.
Compounds and Chemical Bonds
Two or more elements may combine to form a new chemical substance called a compound. A compound's characteristics are usually different from those of its elements. Consider what happens when the element sodium (Na) combines with the element chlorine (Cl). Sodium is a silvery metal that explodes when it comes into contact with water. Chlorine is a deadly yellow gas. In combination, however, they form a crystalline solid called sodium chloride (NaCl)—plain table salt (Figure 2.7).

FIGURE 2.7. The characteristics of compounds are usually different from those of their elements.
The atoms (or, as we will soon see, ions) in a compound are held together by chemical bonds. There are two types of chemical bond: covalent and ionic. Recall that atoms have outer shells, which are the regions surrounding the nucleus where the electrons are most likely to be found. Figure 2.8 depicts the first two shells as concentric circles around the nucleus. As discussed earlier, a full innermost shell contains 2 electrons. A full second shell contains 8 electrons. Atoms with a total of more than 10 electrons have additional shells. When atoms form bonds, they lose, gain, or share the electrons in their outermost shell.
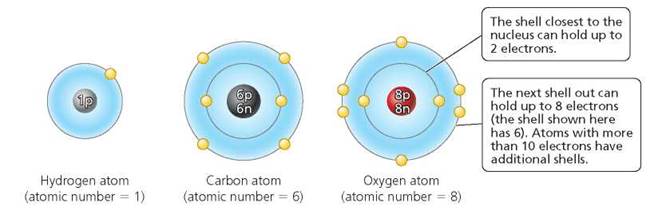
FIGURE 2.8. Atoms of hydrogen, carbon, and oxygen. Each of the concentric circles around the nucleus represents a shell occupied by electrons.
Covalent Bonds
A covalent bond forms when two or more atoms share electrons in their outer shells. Consider the compound methane (CH4). Methane is formed by the sharing of electrons between one atom of carbon and four atoms of hydrogen. Notice in Figure 2.9a that the outer shell of an isolated carbon atom contains only four electrons, even though it can hold as many as eight. Also note that hydrogen atoms only have one electron, although the first shell can hold up to two electrons. A carbon atom can fill its outer shell by joining with four atoms of hydrogen. At the same time, by forming a covalent bond with the carbon atom, each hydrogen atom fills its first shell. We see, then, that the covalent bonds between the carbon atom and hydrogen atoms of methane result in filled outer shells for all five atoms involved.
A molecule is a chemical structure held together by covalent bonds. Recall that compounds are formed by two or more elements, so molecules that contain only one kind of atom are not considered compounds. For example, oxygen gas, formed by the joining of two oxygen atoms, is not a compound, but it is a molecule. Molecules are described by a formula that contains the symbols for all of the elements included in that molecule. If more than one atom of a given element is present in the molecule, a subscript is used to show the precise number of that kind of atom. For example, the molecular formula of sucrose (table sugar) is C12H22O11, showing that one molecule of sucrose contains 12 atoms of carbon, 22 atoms of hydrogen, and 11 atoms of oxygen. Numbers placed in front of the molecular formula indicate more than one molecule. For example, three molecules of sucrose are described by the formula 3C12H22O11.
As shown in the methane molecule in Figure 2.9a, the bond between each of the four hydrogen atoms and the carbon atom consists of a single pair of electrons. A bond in which a single pair of electrons is shared is called a single bond; in this example, the methane molecule contains four single covalent bonds. Sometimes, however, two atoms share two or three pairs of electrons. These bonds are called double and triple covalent bonds, respectively. For example, carbon dioxide, CO2, produced by chemical reactions inside our cells, has double covalent bonds between the carbon atom and each of two oxygen atoms (Figure 2.9b). And the nitrogen atoms in nitrogen gas, N2, are joined by triple covalent bonds (Figure 2.9c).
Covalent bonds in molecules are sometimes depicted by a structural formula. Notice in the box at the right of Figure 2.9a that one straight line is drawn between the carbon atom and each hydrogen atom in the structural formula for the methane molecule. The single line indicates a single covalent bond resulting from a pair of shared electrons. In the box to the right of Figure 2.9b, the double lines between the carbon and oxygen atoms in the carbon dioxide molecule indicate a double covalent bond, or two pairs of shared electrons. In Figure 2.9c, three lines drawn between the two nitrogen atoms in gaseous nitrogen depict a triple covalent bond, or three pairs of shared electrons.
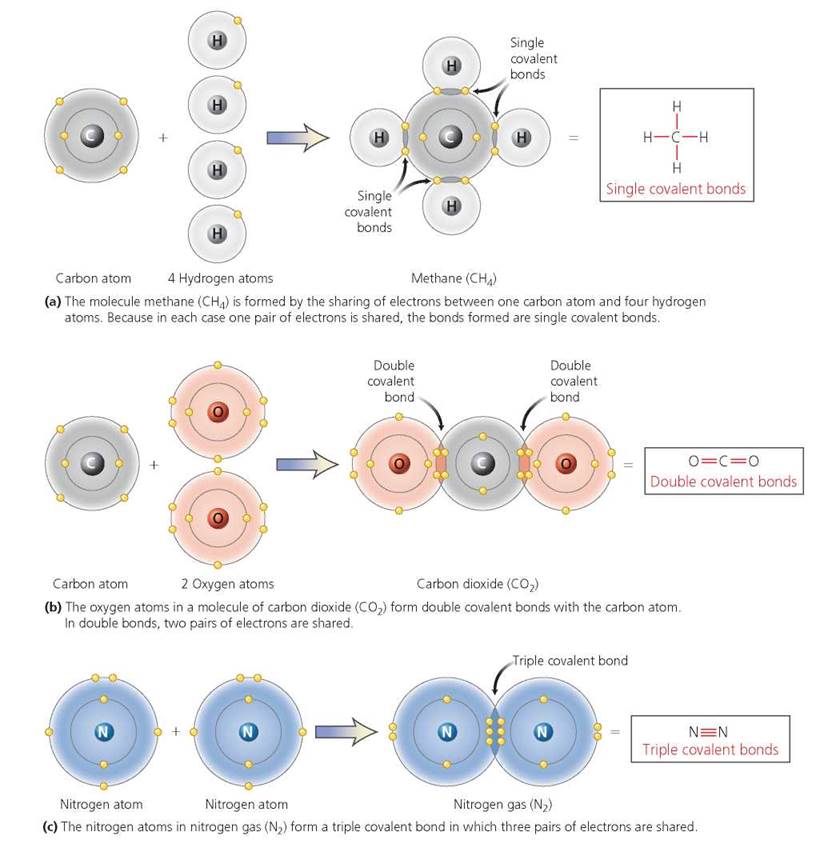
FIGURE 2.9. Covalent bonds form when electrons are shared between atoms. Shown here are examples of single, double, and triple covalent bonds. For each example, the structural formula is shown on the far right.
Ionic Bonds
We have all heard the phrase "opposites attract" in reference to human relationships—and it is no different for ions. An ion is an atom or group of atoms that carries either a positive (+) or a negative (-) electrical charge. Electrical charges result from the transfer (as opposed to sharing) of electrons between atoms. Recall that a neutral atom has the same number of positively charged protons and negatively charged electrons. An atom that loses an electron has one more proton than electrons and therefore has a positive charge. An atom that gains an electron has one more electron than protons and has a negative charge. Oppositely charged ions are attracted to one another. An ionic bond results from the mutual attraction of oppositely charged ions.
Ions form because of the tendency of atoms to attain a complete outermost shell. Consider, again, the atoms of sodium and chlorine that join to form sodium chloride. As shown in Figure 2.10, an atom of sodium has one electron in its outer shell. An atom of chlorine has seven electrons in its outer shell. Sodium chloride is formed when the sodium atom transfers the single electron in its outer shell to the chlorine atom. The sodium atom now has a full outer shell. This comes about because the sodium atom loses its third shell, making the second shell its outermost shell. The sodium atom, having lost an electron, has one more proton than electrons and therefore now has a positive charge (Na+). The chlorine atom, having gained an electron to fill its outer shell, has one more electron than protons and now has a negative charge (Cl-). These oppositely charged ions are attracted to one another, and an ionic bond forms. Because they do not contain shared electrons, ionic bonds are weaker than covalent bonds.
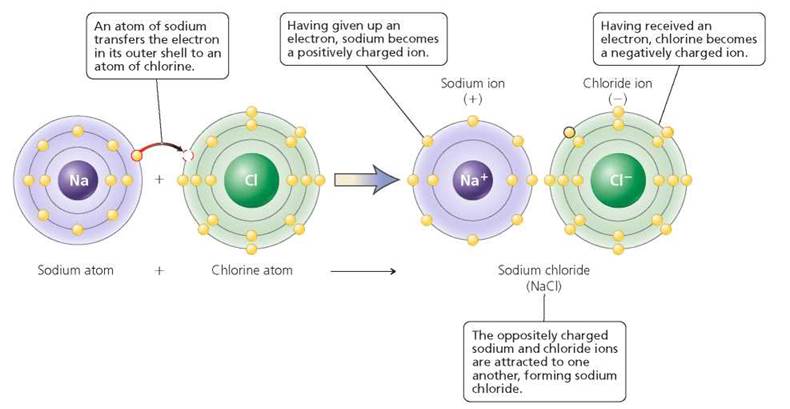
FIGURE 2.10. An ionic bond involves the transfer of electrons between atoms. Such a transfer creates oppositely charged ions that are attracted to one another.
The Role of Water in Life
Water is such a familiar part of our everyday lives that we often overlook its unusual qualities. Unique properties of water include its virtuosity as a dissolving agent, its high heat capacity, and its high heat of vaporization. As it turns out, water's unusual qualities can be traced to its polarity (tendency of its molecules to have positive and negative regions) and the hydrogen bonds between its molecules.
Polarity and Hydrogen Bonds
In covalently bonded molecules, electrons may be shared equally or unequally between atoms. When the sharing of electrons is unequal, different ends of the same molecule can have slight opposite charges. Unequal covalent bonds are called polar, and molecules with unevenly distributed charges are called polar molecules. In water (H2O), for example, the electrons shared by oxygen and hydrogen spend more time near the oxygen atom than near the hydrogen atom. As a result, the oxygen atom has a slight negative charge; each hydrogen atom has a slight positive charge; and water molecules are polar (Figure 2.11a). The hydrogen atoms of one water molecule are attracted to the oxygen atoms of other water molecules. The attraction between a slightly positively charged hydrogen atom and a slightly negatively charged atom nearby is called a hydrogen bond. In the case of water, the hydrogen bonding occurs between hydrogen and oxygen. However, sometimes hydrogen bonds form between hydrogen and atoms of other elements.
Hydrogen bonds are weaker than either ionic or covalent bonds. For this reason, they are generally illustrated by dotted lines rather than solid lines, as shown in Figure 2.11b. Even though individual hydrogen bonds are very weak, collectively they can be significant. Hydrogen bonds maintain the shape of proteins and our hereditary material, DNA, and they account for some of the unique physical properties of water.
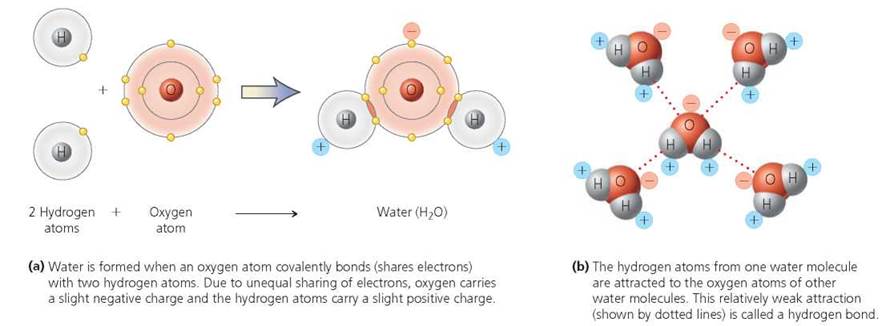
FIGURE 2.11. The hydrogen bonds of water
Covalent bonds, ionic bonds, and hydrogen bonds are summarized in Table 2.2.
TABLE 2.2. Review of Chemical Bonds
Type |
Basis for Attraction |
Strength |
Example |
Covalent |
Sharing of electrons between atoms; the sharing between atoms may be equal or unequal |
Strongest |
CH4 (methane) |
Ionic |
Transfer of electrons between atoms creates oppositely charged ions that are attracted to one another |
Strong |
NaCl (table salt) |
Hydrogen |
Attraction between a hydrogen atom with a slight positive charge and another atom (often oxygen) with a slight negative charge |
Weak |
Between a hydrogen atom on one water molecule and an oxygen atom on another water molecule |
Environmental Issue
Radon Gas: A Killer That Can Be Stopped
Some lethal substances are obvious because you can see, smell, or taste them. Others are not obvious, even when surrounding you in your own home. Such is the case for radon, a radioactive gas produced when uranium breaks down in rock, soil, or water. This invisible, odorless gas moves up through the soil, eventually seeping through cracks in the foundations of buildings. Once inside a building, radon may accumulate, sometimes reaching harmful levels. The same techniques that help to make a building energy efficient also tend to limit the exchange of air from inside a building to outside; and, as a result, modern, energy-efficient buildings can accumulate radon.
Although radon can enter any type of building, the greatest exposure threat usually occurs in homes because people typically spend more time in their home than in their school or workplace. Most radon enters buildings as gas from the soil, but radon can also enter a home through well water. The greatest risk from radon in water occurs when it is released into the air during showers or other household uses of water. The Environmental Protection Agency (EPA) estimates that 1 in 15 homes in the United States has an unacceptable level of radon, defined as 4 or more picocuries of radon per liter of air. (A picocurie is a measure of radioactivity that equals about 2.2 disintegrations of radioactive particles per minute.)

Each year in the United States, radon contributes to the death of about 21,000 people; this number exceeds deaths from drunk driving, drowning, or falls or fires in the home (Figure 2. A). Radon kills by causing lung cancer. Smoking combined with exposure to radon is particularly risky: if 1000 people who smoked were exposed over a lifetime to 4 picocuries of radon per liter of air, 62 will likely get lung cancer as compared to 7 people in the same situation who have never smoked.
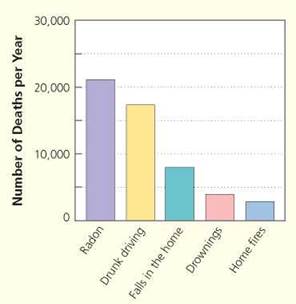
FIGURE 2.A. Radon causes about 21,000 deaths from lung cancer each year. Here, the number of deaths from radon is shown in relation to numbers of deaths from other causes.
EPA's 2003 Assessment of Risks from Radon in Homes (EPA 402-R-03-003); Centers for Disease Control and Prevention’s 1999-2001 National Center for Injury Prevention and Control Report; 2002 National Safety Council Reports.
The EPA recommends that all homes be tested for radon. Residents can test for radon themselves using a do-it-yourself kit available at most hardware stores. Alternatively, residents can hire a certified radon professional to do the testing. If testing reveals high levels of radon in a home, then a contractor trained to fix radon problems can install a venting system that pulls radon from beneath the house and releases it to the outside. New homes can be built with features to reduce radon levels; even so, testing should be performed to make sure that the construction techniques are effective.
Home radon-testing kits are available from the National Safety Council and are sold in many hardware and retail stores.
Questions to Consider
• If you live in a campus dormitory, would you be willing to pay more in fees to fix a newly discovered radon contamination problem in your building? If not, whose responsibility should this be?
• Beginning in 2012, landlords in Maine must test for radon in their buildings every 10 years. Do you think that other states should follow Maine's example, or should radon testing be an individual responsibility?
Properties of Water
Life depends on the properties of water. Let's consider some of the ways that the polarity and hydrogen bonding of water give water the properties that make it so vital to life.
Due to the polarity of its molecules, water interacts with many substances. This interactivity makes it an excellent solvent, easily dissolving both polar and charged substances. Ionic compounds, such as NaCl, dissolve into independent ions in water. The sodium ions and chloride ions separate from one another in water because the sodium ions are attracted to the negative regions of water molecules and the chloride ions are attracted to the positive regions. Because of its excellence as a solvent, water serves as the body's main transport medium. As the liquid component of blood, it carries dissolved nutrients, gases, and wastes through the circulatory system. Metabolic wastes are excreted from the body in urine, another watery medium.
Molecules of water stick together because of hydrogen bonds; this characteristic of water is called cohesion. Water helps prevent dramatic changes in body temperature, and this too is related to its hydrogen bonds. About 67% of the human body is water (thus, if a person weighs 68 kg [150 lb], water makes up about 45 kg [100 lb] of the body weight). Because humans, as well as many other organisms, are made up largely of water, they are well suited to resist changes in body temperature and to keep a relatively stable internal environment. This ability comes from water's high heat capacity, which simply means that a great deal of heat is required to raise its temperature. Hydrogen bonds hold multiple water molecules together, so a large amount of heat is required to break these bonds (a higher temperature corresponds to an increase in the movement of the molecules). Water in blood also helps redistribute heat within our bodies. Our fingers don't usually freeze on a frigid day because heat is carried to them by blood from muscles where the heat is generated.
Another property of water that helps prevent the body from overheating is its high heat of vaporization, which means that a great deal of heat is required to make water evaporate (that is, change from a liquid to a gas). Water's high heat of vaporization is also due to its hydrogen bonds, which must be broken before water molecules can leave the liquid and enter the air. (These bonds, by the way, remain broken as long as water is in the gaseous phase known as water vapor.) Water molecules that evaporate from a surface carry away a lot of heat, cooling the surface. We rely on the evaporation of water in sweat to cool the body surface and prevent overheating (Figure 2.12). By the same token, water vapor in the air can inhibit the evaporation of sweat—which is why we tend to feel hotter on a humid day.

FIGURE 2.12. The evaporation of water in sweat cools the surface of this runner’s body. Water has a high heat of vaporization, so when water molecules in sweat evaporate, they carry away a lot of heat.
Stop and think
Sharp increases in body temperature can cause heat stroke, a condition that may damage the brain. Explain why heat stroke is more likely to occur on a hot, humid day than on an equally hot, dry day.
It is clear that water is essential for human life and for the lives of many other organisms we share the planet with. Despite our reliance on water, we continue to pollute both seawater and freshwater. Equally alarming is the global shortage of freshwater, caused largely by the burgeoning human population. For one community's creative response to the water shortage, see the Environmental Issue essay, Toilet to Tap.
Acids and Bases
Sometimes a water molecule dissociates, or breaks up, forming a positively charged hydrogen ion (H+) and a negatively charged hydroxide ion (OH-):
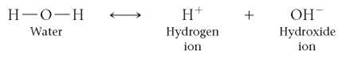
(Note that in equations describing chemical reactions, an arrow should be read as "yields.")
In any sample of water, the fraction of water molecules that are dissociated is extremely small, so water molecules are much more common in the human body than are H+ and OH-. In fact, the amount of H + in the body must be precisely regulated. Substances called acids and bases influence the concentration of H+ in solutions.
Acids and bases are defined by what happens when they are added to water. An acid is anything that releases hydrogen ions (H+) when placed in water. A base is anything that releases hydroxide ions (OH-) when placed in water. Hydrochloric acid (HCl), for example, dissociates in water to produce hydrogen ions (H+) and chloride ions (Cl-). Because HCl increases the concentration of (H+) in solution, it is classified as an acid. Sodium hydroxide (NaOH), on the other hand, dissociates in water to produce sodium ions (Na+) and hydroxide ions (OH-). Because NaOH increases the concentration of OH- in solution, it is classified as a base. The OH- produced when NaOH dissociates reacts with H+ to form water molecules and thus reduces the concentration of H+ in solution. Therefore, acids increase the concentration of H+ in solution, and bases decrease the concentration of H+ in solution.
The pH Scale
We often want to know more than simply whether a substance is an acid or a base. For example, how strong an acid is battery acid? How strong a base is household ammonia? Questions like these can be answered by knowing the pH of these solutions and understanding the pH scale (Figure 2.13). The pH of a solution is a measure of hydrogen ion concentration. The pH scale ranges from 0 to 14, with a pH of 7 being neutral (the substance does not increase H+ or OH-), a pH of less than 7 being acidic, and a pH of greater than 7, basic.
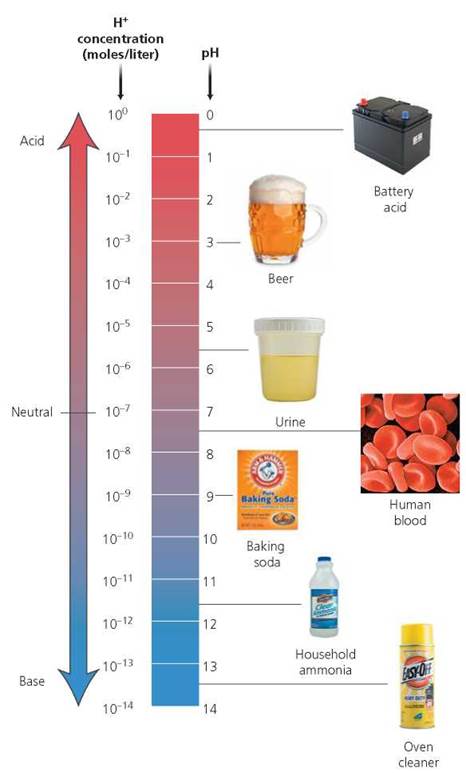
FIGURE 2.13. The pH scale and the pH of some body fluids and other familiar substances
Usually, the amount of H+ in a solution is very small. For example, the concentration of H+ in a solution with a pH of 6 is 1 x 10-6 (or 0.000001) moles per liter (a mole, here, is not a small furry animal with a star-shaped nose but a unit of measurement that indicates a specific number of atoms, molecules, or ions). Similarly, the concentration of H + in a solution with a pH of 5 is 1 x 10-5 (or 0.00001) moles per liter. Technically, pH is the negative logarithm of the concentration of H+ in a solution. According to the pH scale, the lower the pH, the greater the acidity—or concentration of H+—in a solution. Each reduction of pH by one unit represents a tenfold increase in the amount of H+. So a solution with a pH of 5 is 10 times more acidic than a solution with a pH of 6. And a solution with a pH of 4 is 100 times more acidic than one with a pH of 6. Some characteristics of acids and bases, including their values on the pH scale, are summarized in Table 2.3.
TABLE 2.3. Review of the Characteristics of Acids and Bases
Characteristic |
Acid |
Base |
Behavior in water |
Releases H+ |
Releases OH- |
pH |
Less than 7 |
Greater than 7 |
Example |
HCl (hydrochloric acid) |
NaOH (sodium hydroxide) |
Buffers
Most biological systems must keep their fluids within a narrow range of pH values. Substances called buffers keep pH values from changing dramatically. Buffers remove excess H+ from solution when concentrations of H+increase. Buffers add H+ when concentrations of H+ decrease. For example, an important buffering system that keeps the pH of blood at about 7.4 is the carbonic acid-bicarbonate system. When carbon dioxide is added to water it forms carbonic acid (H2CO3), which dissociates into hydrogen ions and bicarbonate ions (HCO3-):
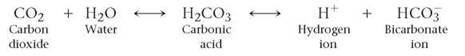
Carbonic acid and bicarbonate have a buffering action because when levels of H+ decrease in the blood, carbonic acid dissociates, adding H+ to solution. When levels of H+ increase in the blood, the H+ combines with bicarbonate and is removed from solution. Such action is essential because even slight changes in the pH of blood—say, a drop from 7.4 to 7.0 or an increase to 7.8—can cause death in a few minutes.
In the human body, almost all biochemical reactions occur around pH 7 and are maintained at that level by powerful buffering systems. An important exception occurs in the stomach, where hydrochloric acid (HCl) produces pH values from about 1 to 3. In the stomach, HCl kills bacteria swallowed with food or drink and promotes the initial breakdown of proteins. These activities require an acid stomach, and the stomach has several ways of protecting itself from the acid (discussed in Chapter 15). However, sometimes stomach acid backs up into the esophagus, and "heartburn" is the uncomfortable result. Taking an antacid can ease the discomfort of heartburn. Antacids consist of weak bases that temporarily relieve the pain of stomach acid in the esophagus by neutralizing some of the hydrochloric acid.
The critical link between pH and life is illustrated by the impact of acid rain on our environment and health. Acid rain is usually defined as rain with a pH lower than 5.6, the pH of natural precipitation. It is caused largely by the burning of fossil fuels in cars, factories, and power plants. The sulfur dioxide and nitrogen oxides produced by these activities react with water in the atmosphere to form sulfuric acid (H2SO4) and nitric acid (HNO3). These acids fall to Earth as rain, snow, or fog.
The effects of acid rain on the environment have been devastating. On land, acid rain has been linked to the decline of forests. Trees, for example, become stressed and more susceptible to disease when their nutrient uptake is disrupted by increased acidity in the soil (Figure 2.14a). In aquatic environments, acid rain has been linked to declines in populations of fish and amphibians. Embryos of spotted salamanders, for example, develop abnormally under somewhat acidic conditions and die when pH values are less than about 5 (Figure 2.14b). Acid rain is harmful to human health, as well. The pollutants that cause acid rain form fine particles of sulfate and nitrate that are easily inhaled. Once inside us, they cause irritation and respiratory illnesses such as asthma and bronchitis.

FIGURE 2.14. Effects of acid rain
Because most acid rain is caused by human activity, it is within our power to reduce, if not eliminate, the problem. Power plants and automobile manufacturers have taken steps to reduce emissions of sulfur dioxide and nitrogen oxides, leading to significant progress in reducing acid rain.
Major Molecules of Life
Most of the molecules we have discussed so far have been small and simple. Many of the molecules of life, however, are enormous by comparison and have complex architecture. Some proteins, for example, are made up of thousands of atoms linked together in a chain that repeatedly coils and folds upon itself. Exceptionally large molecules, including many important biological molecules, are known as macromolecules.
Macromolecules that consist of many small, repeating molecular subunits linked in a chain are called polymers. The small molecular subunits that form the building blocks of the polymer are called monomers. We might think of a polymer as a pearl necklace, with each monomer representing a pearl. As we shall see, a protein is a polymer, or chain, of amino acid monomers linked together. And glycogen, the storage form of carbohydrates in animals, is a polymer of glucose monomers.
Polymers form through dehydration synthesis (sometimes called the condensation reaction). In this process, the reaction that bonds one monomer covalently to another releases a water molecule: one of the monomers donates OH, and the other donates H. The reverse process, called hydrolysis, which the body uses to break many polymers apart, requires the addition of water across the covalent bonds. The H from the water molecule attaches to one monomer, and the OH attaches to the adjoining monomer, thus breaking the covalent bond between the two. Hydrolysis plays a critical role in digestion. Most foods consist of polymers too large to pass from our digestive tract into the bloodstream and on to our cells. Thus, the polymers are hydrolyzed into their component monomers. The monomers can then be absorbed into the bloodstream for transport throughout the body. Dehydration synthesis and hydrolysis are summarized in Figure 2.15.
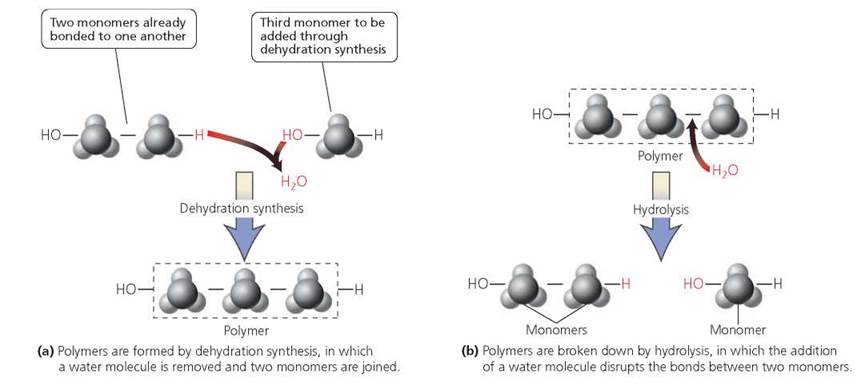
FIGURE 2.15. Formation and breaking apart of polymers
Carbohydrates
The carbohydrates, known commonly as sugars and starches, provide fuel (energy) for the human body. Carbohydrates are compounds made entirely of carbon, hydrogen, and oxygen, with each molecule having twice as many hydrogen atoms as oxygen atoms. Sugars and starches can be classified by size into monosaccharides, oligosaccharides, and polysaccharides.
Monosaccharides. Monosaccharides, also called simple sugars, are the smallest molecular units of carbohydrates. They contain from three to seven carbon atoms and, in fact, can be classified by the number of carbon atoms they contain. A sugar that contains five carbons is pentose; one with six carbons is hexose; and so on. Glucose, fructose, and galactose are examples of six-carbon sugars. Monosaccharides can be depicted in several ways (Figure 2.16).
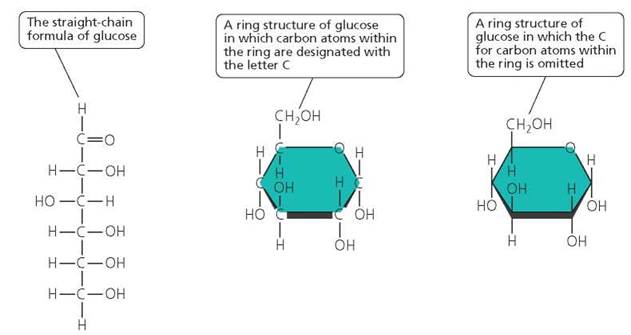
FIGURE 2.16. Monosaccharides are simple sugars, generally having a backbone of three to six carbon atoms. Many of these carbon atoms are also bonded to hydrogen (H) and a hydroxyl group (OH). In the fluid within our cells, the carbon backbone usually forms a ringlike structure. Here, three representations of the monosaccharide glucose (C6H12O6) are shown.
Oligosaccharides. Oligosaccharides (oligo means "few") are chains of a few monosaccharides joined together by dehydration synthesis. Disaccharides, one type of oligosaccharide, are double sugars that form when two monosaccharides covalently bond to each other. The disaccharide sucrose (table sugar) consists of the monosaccharides glucose and fructose (Figure 2.17). Two glucose molecules form the disaccharide maltose, an important ingredient of beer. Another disaccharide is lactose, the principal carbohydrate of milk and milk products. Lactose is formed by the joining of glucose and galactose.

FIGURE 2.17. Disaccharides are built from two monosaccharides. Here, a molecule of glucose and one of fructose combine to form sucrose.
Polysaccharides. Polysaccharides (poly means "many") are complex carbohydrates that form when monosaccharides (most commonly glucose) join together in long chains. Most polysaccharides store energy or provide structure. In plants, the storage polysaccharide is starch;in animals it is glycogen, a short-term energy source that can be broken down to release energy-laden glucose molecules. Humans store glycogen mainly in the cells of the liver and muscles (Figure 2.18a).
Cellulose is a structural polysaccharide found in the cell walls of plants (Figure 2.18b). Humans lack the enzymes necessary to digest cellulose and, as a result, it passes unchanged through our digestive tract. (Enzymes are discussed later in this chapter.) Nevertheless, cellulose is an important form of dietary fiber (roughage) that helps fecal matter move through the large intestines. Including fiber in our diet may reduce the incidence of colon cancer.
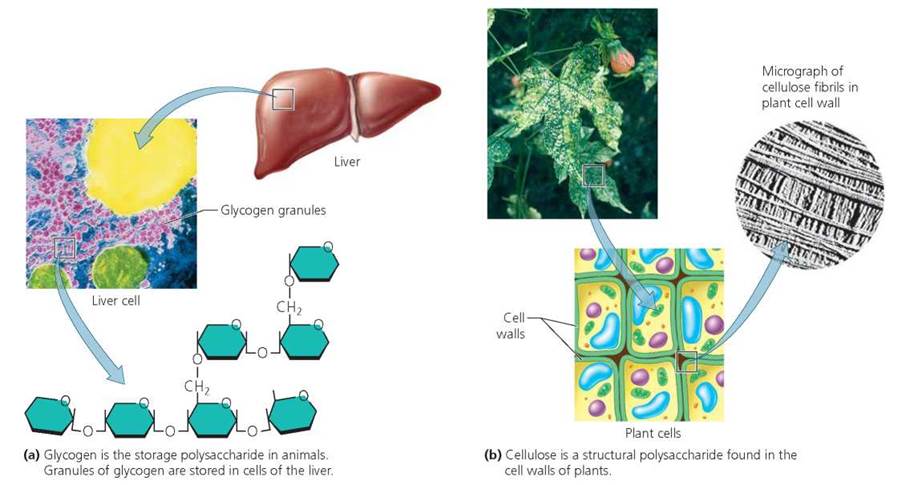
FIGURE 2.18. Polysaccharides may function in storage (as in glycogen) or provide structure (as in cellulose).
Lipids
Lipids, such as fats, are compounds that do not dissolve in water. Lipids are nonpolar (having no electrical charges), while water is polar. Because of this difference, water shows no attraction for lipids and vice versa, so water and lipids do not mix. Three types of lipids that are important to human health are triglycerides, phospholipids, and steroids.
Triglycerides Fats and oils are triglycerides, compounds made of one molecule of glycerol and three fatty acids. Fatty acids are chains of carbon atoms also bonded to hydrogens and having the acidic group COOH at one end. The fatty acids bond to glycerol through dehydration synthesis (Figure 2.19a). Triglycerides are classified as saturated or unsaturated, depending on the presence or absence of double bonds between the carbon atoms in their fatty acids (Figure 2.19b). Saturated fatty acids have only single covalent bonds linking the carbon atoms. They are described as "saturated with hydrogen" because their carbon atoms are bonded to as many hydrogen atoms as possible. Saturated fats are made from saturated fatty acids. Butter, a saturated fat, is solid at room temperature because its fatty acids can pack closely together. Fatty acids with one or more double bonds between carbon atoms are described as unsaturated—that is, not saturated with hydrogen—because they could bond to more hydrogen atoms if the double bonds between their carbon atoms were broken. The double bonds cause "kinks" in the fatty acids and prevent molecules of unsaturated fat from packing tightly into a solid. Thus, unsaturated fats, such as olive oil, are liquid at room temperature. Sometimes hydrogens are added to unsaturated fats to stabilize them, with the goal of lengthening their shelf life, or to solidify them. For example, hydrogens are added to vegetable oil to make margarine. These partially hydrogenated fats are called trans fats and are found in many packaged snacks, such as cookies and potato chips.
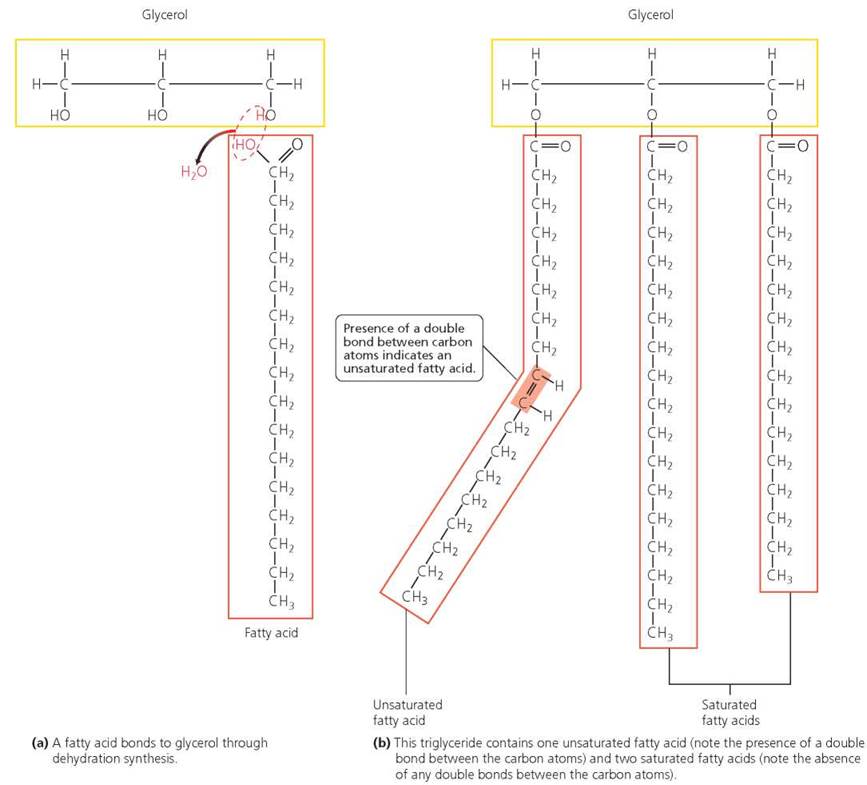
FIGURE 2.19. Triglycerides are composed of a molecule of glycerol joined to three fatty acids.
Would the triglyceride shown in part (b) be a solid or a liquid at room temperature? Why?
A The triglyceride would be a liquid at room temperature because the kink in the unsaturated fatty acid in part (b) would prevent close packing of adjacent molecules.
Stop and think
What would the hydrolysis of a fat yield?
Environmental Issue
Toilet to Tap
We depend on a steady supply of freshwater. Yet, experts tell us that our water supply is dwindling. Most of Earth's water (97.4%) is in the oceans; this water is too salty for us to use without first treating it. About 2% is locked away in glaciers and polar ice. This leaves 0.6% of Earth's water, in the form of freshwater, for humans and other organisms to use. The water available to humans is classified as surface water (rivers, lakes, reservoirs) and ground water (water in porous rock layers below Earth's surface). Over the last century, we have damaged our supplies of freshwater by redirecting the flow of rivers, constructing dams, draining wetlands, and extracting groundwater at rates that exceed its replacement (as part of the water cycle, groundwater is naturally replaced when water that falls as precipitation seeps into the ground). We will have an ever-greater impact on our water supply as the human population continues to grow.
Some communities in California have taken a unique approach to the problem. Like many western states, California has been experiencing prolonged drought. At the same time, it has a steadily growing population, and it faces the threat of seawater moving into its groundwater. Orange County, in southern California, has responded to the impending water crisis by building the Groundwater Replenishment System. This new system, which began operations in 2008, accomplishes two very important things. First, it recharges the groundwater supply with treated wastewater rather than sending the wastewater out to sea. Second, it builds up the county's seawater intrusion barrier—a series of wells into which water is pumped to create an “underground water dam” that blocks seawater from entering the groundwater basin.
Here is how the system works. Industrial and household sewage is treated at the Orange County Sanitation District, as it has been in the past. The sewage, or primary effluent, undergoes several treatments designed to break down organic material and to remove particulate matter. After these treatments, the water, called secondary effluent, would normally be discharged to the ocean. But under the new system, at this point, the treated wastewater enters the Advanced Water Purification Facility (AWPF). At the AWPF, the water undergoes a several-step purification process. About half of the water exiting the AWPF is injected into Orange County's seawater intrusion barrier. The other half of the water exiting the AWPF is piped to a giant percolation pond where it moves through gravel, sand, and clay into the groundwater supply, filtering naturally just as rainwater finds its way into groundwater. Eventually, this water will enter drinking well intakes. Hence, the water has been described as going from “toilet to tap.”
The end result of Orange County's Groundwater Replenishment System is water that meets or exceeds all existing standards for drinking water. While some may cringe at the thought of drinking water that has been recycled from sewage, it is a reality that we may have to get used to as global and regional water supplies continue to tighten.

The toilet-to-tap program replenishes groundwater supplies with reclaimed wastewater.
Questions to Consider
• Would you be hesitant to drink water that has been recycled from sewage, even if it met current drinking water standards? If yes, what is the reason for your hesitation?
• Do you think that human ingenuity will always be able to produce the technology needed to avoid major environmental crises, such as a global shortage of freshwater?
Fats and oils provide about twice the energy per gram that carbohydrates or proteins do. This high energy density makes fat an ideal way for the body to store energy for the long term. Our bulk would be much greater if most of our energy storage consisted of carbohydrates or proteins, given their relatively low energy yield compared with fat.
In preparation for long-term energy storage, excess triglycerides, carbohydrates, and proteins from the foods we consume are converted into small globules of fat that are deposited in the cells of adipose tissue. There, the fat remains until our bodies need extra energy; at that time our cells break down the fat to release the energy needed to keep vital processes going. Besides long-term energy storage, fat serves a protective function in the body. Thin layers of fat surround major organs such as the kidneys, cushioning the organs against physical shock from falls or blows. Fat also serves as insulation and as a means of absorbing lipid-soluble vitamins from the intestines and transporting them to the cells that use them. Despite the importance of fats and oils to human health, in excess they can be dangerous, particularly to our circulatory system (discussed in Chapter 12a).
Phospholipids. A phospholipid is made of a molecule of glycerol bonded to two fatty acids and a negatively charged phosphate group. Another small molecule of some kind— usually polar, called the variable group—is linked to the phosphate group. This general structure provides phospholipids with two regions having very different characteristics. As you will notice in Figure 2.20a, the region made up of fatty acids is nonpolar; it is described as a hydrophobic, or "water-fearing," tail. The other region is polar and it makes up the hydrophilic, or "water-loving," head. The tails, being hydrophobic, do not mix with water. The heads, being hydrophilic, interact readily with water. The hydrophilic heads and hydrophobic tails of phospholipids are responsible for the structure of plasma (cell) membranes. In the membrane surrounding a cell, phospholipids are arranged in a double layer, called a bilayer (Figure 2.20b), with the hydrophilic heads of each layer facing away from each other. That way, each surface of the membrane consists of hydrophilic heads in contact with the watery solutions inside and outside the cell. The hydrophobic tails of the two layers point toward each other and help hold the membrane together.
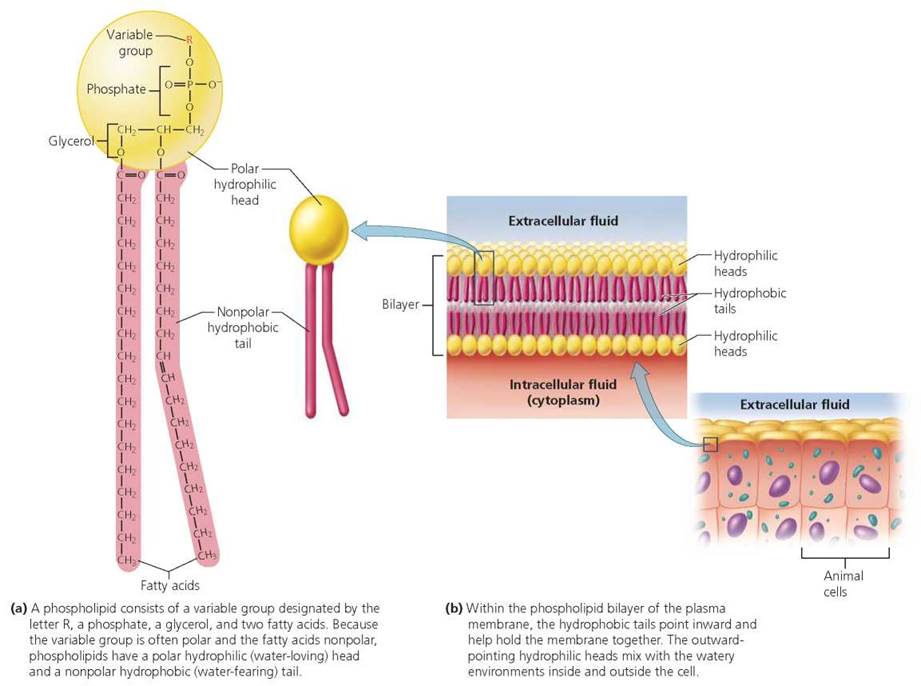
FIGURE 2.20. Structure of a phospholipid. Phospholipids are the main components of the plasma membrane encasing a cell and separating its internal and external watery environments.
Steroids. A steroid is a type of lipid made up of four carbon rings attached to molecules that vary from one steroid to the next. Cholesterol, one of the most familiar steroids, is a component of the plasma membrane and is also the foundation from which steroid hormones, such as estrogen and testosterone, are made. Cholesterol in our blood comes from two sources, our liver and our diet. A high level of cholesterol in the blood is considered a risk factor for heart disease (as described in Chapter 12a). Cholesterol, estrogen, and testosterone are shown in Figure 2.21.
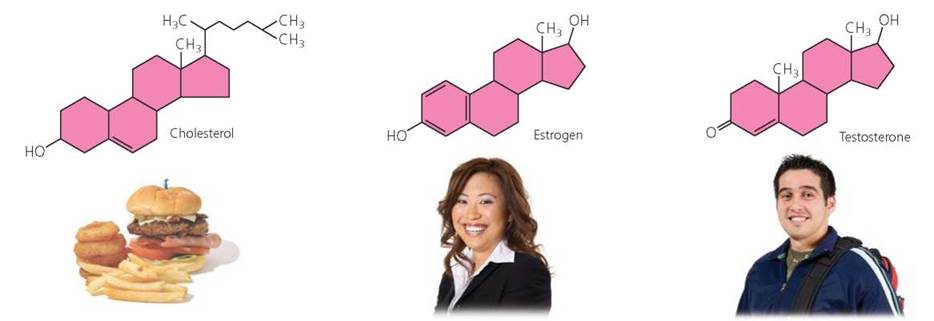
FIGURE 2.21. The steroid cholesterol is a component of cell membranes, and it is the substance used to make steroid hormones such as estrogen and testosterone. All steroids have a structure consisting of four carbon rings. Steroids differ in the groups attached to these rings.
Proteins
A protein is a polymer made of one or more chains of amino acids. In many proteins, the chains are twisted, turned, and folded to produce complicated structures. Thousands upon thousands of different proteins are found in the human body, contributing to structural support, transport, movement, and regulation of chemical reactions. Despite their great diversity in structure and function, all proteins are made from a set of only about 20 kinds of amino acids.
Amino acids. Amino acids are the building blocks of proteins. They consist of a central carbon atom bound to a hydrogen atom (H), an amino group (NH2), an acidic carboxyl group (COOH), and a side chain, often designated by the letter R (Figure 2.22). Amino acids differ from each other only in their side chains. Some amino acids, called nonessential amino acids, can be synthesized by our bodies. Other amino acids, called essential amino acids, cannot be synthesized by our bodies and must be obtained from the foods we eat.
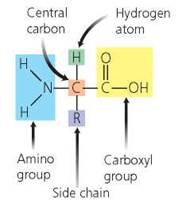
FIGURE 2.22. Structure of an amino acid. Amino acids differ from one another in the type of R group (side chain) they contain.
The amino acids that form proteins are linked by bonds called peptide bonds, which are formed through dehydration synthesis. A peptide bond links the carboxyl group (COOH) of one amino acid to the amino group (NH2) of the adjacent amino acid, as shown in Figure 2.23. Chains containing only a few amino acids are called peptides. Dipeptides contain two amino acids, tn'peptides contain three amino acids, and so on. Chains containing 10 or more amino acids are called polypeptides. The term protein is used for polypeptides with at least 50 amino acids.
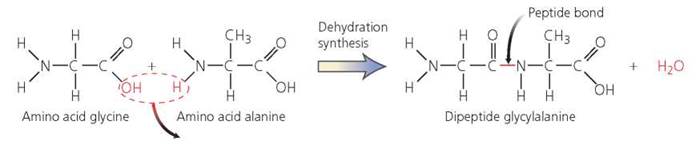
FIGURE 2.23. Formation of a peptide bond between two amino acids through dehydration synthesis. The carboxyl group (COOH) of one amino acid bonds to the amino group (NH2) of the adjacent amino acid, releasing water.
Stop and think
I Which three elements are found in carbohydrates, fats, and proteins? Which additional element do proteins always contain?
Protein structure. Proteins have four levels of structure: primary, secondary, tertiary, and quaternary (see Figure 2.24). The primary structure of a protein is the particular sequence of amino acids. This sequence, determined by the genes, dictates a protein's structure and function. Even slight changes in primary structure can alter a protein's shape and ability to function. The inherited blood disorder sickle-cell anemia provides an example. This disease primarily affects populations in central Africa, but it also occurs in approximately 1 in 500 African Americans. It results from the substitution of one amino acid for another during synthesis of the protein hemoglobin, which carries oxygen in our red blood cells. This single substitution in a molecule that contains hundreds of amino acids creates a misshapen protein that then alters the shape of red blood cells. Death can result when the oddly shaped cells clog the tiny vessels of the brain and heart.
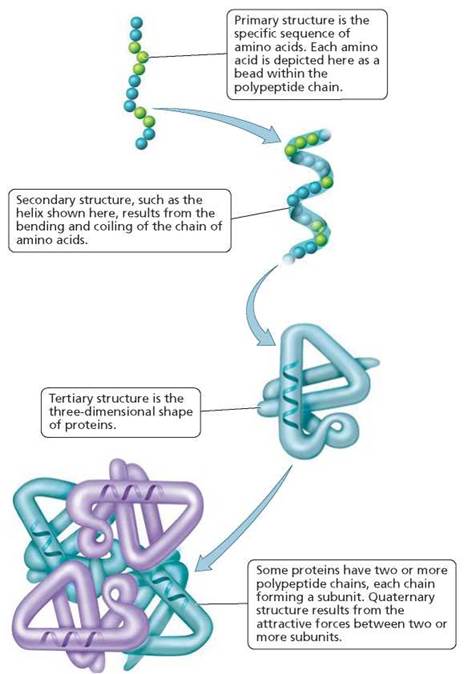
FIGURE 2.24. Levels of protein structure
The secondary structure of proteins consists of patterns known as pleated sheets and helices, which are formed by certain kinds of bends and coils in the chain, as a result of hydrogen bonding. Alterations in the secondary structure of a protein normally found on the surface of nerve cells can transform the protein into an infectious agent known as a prion. Prions have been implicated in several diseases, including Creutzfeldt-Jakob disease in humans and mad cow disease in cattle (see Chapter 13a).
The tertiary structure is the overall three-dimensional shape of the protein. Hydrogen, ionic, and covalent bonds between different side chains may all contribute to tertiary structure. Changes in the environment of a protein, such as increased heat or changes in pH, can cause the molecule to unravel and lose its three-dimensional shape. This process is called denaturation. Even a minor change in the shape of a protein can result in loss of function.
Finally, some proteins consist of two or more polypeptide chains. Each chain, in this case, is called a subunit. Quaternary structure is the structure that results from the assembled subunits. The forces that hold the subunits in place are largely the attractions between oppositely charged side chains.
Enzymes Life is possible because of enzymes. Without them, most chemical reactions within our cells would occur far too slowly to sustain life. Enzymes are substances—almost always proteins—that speed up chemical reactions without being consumed in the process. Typically, reactions with enzymes proceed 10,000 to 1,000,000 times faster than the same reactions without enzymes.
The basic process by which an enzyme speeds up a chemical reaction can be summarized by the following equation:

The particular substance that an enzyme works on is called its substrate. For example, the enzyme sucrase speeds up the reaction in which sucrose is broken down into glucose and fructose. In this reaction, sucrose is the substrate, and glucose and fructose are products. Similarly, the enzyme mal- tase speeds up the breakdown of maltose (the substrate) into molecules of glucose (the product). From these examples you can see that an enzyme's name may resemble the name of its substrate. These particular examples are decomposition reactions, in which a substance is broken down into its component parts. Enzymes also increase the speed of many synthesis reactions.
During reactions promoted by enzymes, the substrate binds to a specific location, called the active site, on the enzyme, to form an enzyme-substrate complex. This binding orients the substrate molecules so they can react. The substrate is converted to one or more products that then leave the active site, allowing the enzyme to bind to another substrate molecule. The entire process occurs very rapidly. One estimate suggests that a typical enzyme can convert about 1000 molecules of substrate into product every second. Figure 2.25 summarizes the steps of enzymatic reactions.
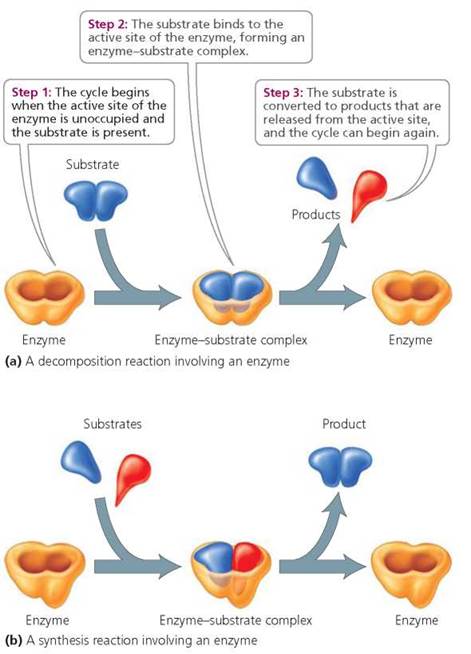
FIGURE 2.25. The working cycle of an enzyme
Enzymes are very specific in their activity; each is capable of binding to and acting on only one or at most a few particular substrates. This specificity is due to the unique shape of each enzyme's active site. The enzyme's active site and the substrate fit together like pieces of a jigsaw puzzle.
Sometimes enzymes need cofactors, nonprotein substances that help them convert substrate to product. Some cofactors permanently reside at the enzyme's active site, while others bind to the active site at the same time as the substrate. Some cofactors are the organic (carbon-containing) substances we know as vitamins. Organic cofactors are called coenzymes. Other cofactors are inorganic (non-carbon-containing) substances such as zinc or iron.
Enzyme deficiencies can affect our health. Lactase deficiency is a relatively common example. Lactase is the enzyme needed to digest the lactose in milk products, breaking it down to glucose and galactose. Infants and young children usually produce enough lactase, but many adults do not. For these adults, consumption of milk and milk products can lead to diarrhea, cramps, and bloating, caused by undigested lactose passing into the large intestine where it feeds resident bacteria. The bacteria, in turn, produce gas and lactic acid that irritate the bowels. The milk industry has responded to this problem (often called lactose intolerance) by marketing lactose-reduced milk, and tablets and caplets that contain the enzyme lactase are also available.
Nucleic Acids and Nucleotides
In our discussion of protein structure we mentioned that genes determine the protein's primary structure, which is the sequence of amino acids. Genes, our units of inheritance, are segments of long polymers called deoxyribonucleic acid (DNA). DNA is one of the two types of nucleic acids.
Nucleotides. The two nucleic acids in our cells are DNA and ribonucleic acid (RNA). Both are polymers of smaller units called nucleotides, joined into chains through dehydration synthesis. Every nucleotide monomer consists of a five-carbon (pentose) sugar bonded to one of five nitrogen-containing bases and at least one phosphate group (Figure 2.26). The five nitrogen-containing bases are adenine, guanine, cytosine, thymine, and uracil. The bases cytosine, thymine, and uracil have a single ring made of carbon and nitrogen atoms; adenine and guanine have two such rings. The sequence of bases in DNA and RNA determines the sequence of amino acids in a protein. DNA, as we said earlier, is the nucleic acid found in genes. RNA, in various forms, converts the genetic information found in DNA into proteins.
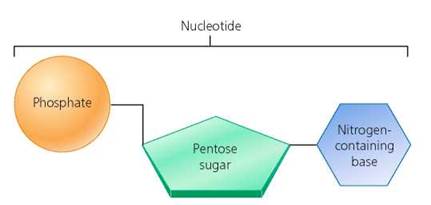
FIGURE 2.26. Structure of a nucleotide. Nucleotides consist of a five-carbon (pentose) sugar bonded to a phosphate molecule and one of five nitrogen-containing bases. Nucleotides are the building blocks of nucleic acids.
DNA and RNA. Key differences in the structures of RNA and DNA are summarized in Table 2.4. RNA is a single strand of nucleotides. The five-carbon sugar in RNA is ribose. The nitrogen-containing bases in RNA are cytosine (C), adenine (A), guanine (G), and uracil (U). See Figure 2.27. In contrast, DNA is a double-stranded chain. Its two parallel strands, held together by hydrogen bonds between the nitrogen-containing bases, twist around one another to form a double helix. The five-carbon sugar in DNA is deoxyribose. The nitrogen-containing bases in DNA are adenine (A), thymine (T), cytosine (C), and guanine (G). See Figure 2.28.
TABLE 2.4. Review of the Structural Differences between RNA and DNA
Characteristic |
RNA |
DNA |
Sugar |
Ribose |
Deoxyribose |
Bases |
Adenine, guanine, cytosine, uracil |
Adenine, guanine, cytosine, thymine |
Number of strands |
One |
Two; twisted to form double helix |
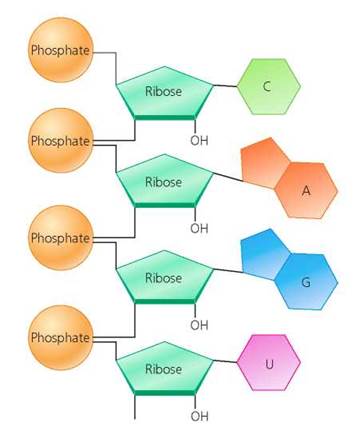
FIGURE 2.27. RNA is a single-stranded nucleic acid. It is formed by the linking together of nucleotides composed of the sugar ribose, a phosphate group, and the nitrogen-containing bases cytosine (C), adenine (A), guanine (G), and uracil (U).
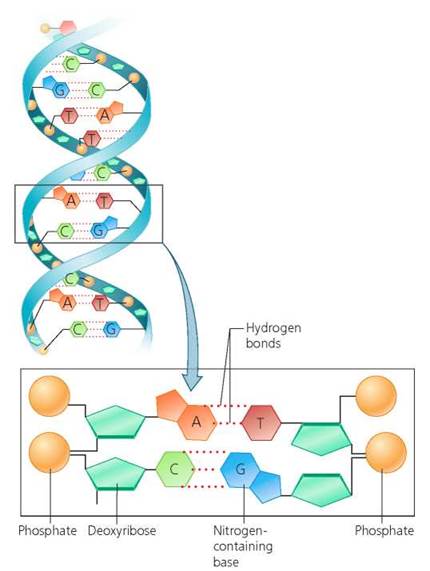
FIGURE 2.28. DNA is a nucleic acid in which two chains of nucleotides twist around one another to form a double helix. The two chains are held together by hydrogen bonds between the nitrogen-containing bases. Each nucleotide of DNA contains the pentose sugar deoxyribose, a phosphate group, and one of the following four nitrogen-containing bases: adenine (A), thymine (T), cytosine (C), and guanine (G).
ATP. At this moment within your cells, many molecules of the nucleotide adenosine triphosphate (ATP) are each losing a phosphate group. As the phosphate group is lost, energy that the molecule stored by holding on to the phosphate group is released. Your cells trap that energy and use it to perform work. It is because of this activity that you are able to sit up and read this book. ATP consists of the sugar ribose, the base adenine, and three phosphate groups, attached to the molecule by phosphate bonds. It is formed from adenosine diphosphate (ADP) by covalent bonding of a phosphate group to the ADP in an energy-requiring reaction. The energy absorbed during the reaction is stored in the new phosphate bond. This high- energy phosphate bond is easily broken when the cell requires energy (Figure 2.29).
ATP is often described as the energy currency of cells. All energy from the breakdown of molecules such as glucose must be channeled through ATP before it can be used by the body.
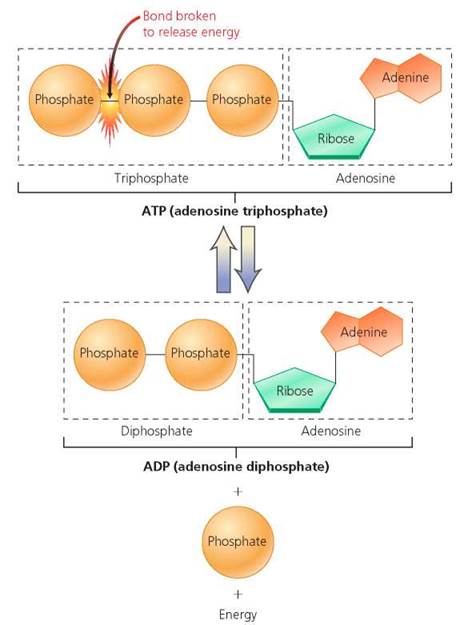
FIGURE 2.29 Structure and function of adenosine triphosphate (ATP). This nucleotide consists of the sugar ribose, the base adenine, and three phosphate groups. The phosphate bonds of ATP are unstable. When cells need energy, the last phosphate bond is broken, yielding adenosine diphosphate (ADP), a phosphate molecule, and energy.
Looking ahead
In Chapter 2 we learned about the important roles that water plays in the human body. Recall that water serves as the body's main transport medium and helps to prevent dramatic changes in body temperature. Foodborne illnesses are caused by the ingestion of contaminated food or water, and they wreak havoc on the body's water balance. These illnesses are usually associated with severe diarrhea and vomiting, which rob the body of its water. In Chapter 2a we will consider the causes, treatments, and prevention of foodborne illness.
Highlighting the Concepts
The Nature of Atoms (pp. 14-17)
• Atoms consist of subatomic particles called protons, neutrons, and electrons. Protons have a positive charge. Neutrons have no charge. Both are found in the nucleus and have atomic masses equal to 1. Electrons have a negative charge, weigh almost nothing, and are found around the nucleus in shells.
• Each element is made of atoms containing a certain number of protons. The atomic number of an element is the number of protons in one of its atoms. The atomic weight for any atom equals the number of protons plus the number of neutrons.
• Isotopes are atoms that have the same number of protons but different numbers of neutrons. Some isotopes, called radioisotopes, emit radiation.
Compounds and Chemical Bonds (pp. 17-20)
• Elements combine to form compounds. The characteristics of compounds are usually different from those of the elements that form them.
• When atoms join to form compounds, chemical bonds form between them. Covalent bonds form when atoms share electrons. Atoms that have lost or gained electrons have an electric charge and are called ions. The attraction between oppositely charged ions is an ionic bond.
The Role of Water in Life (pp. 20-24)
• Sometimes the sharing of electrons in a covalent bond is unequal, resulting in a polar covalent bond and sometimes a polar molecule. Hydrogen bonds are weak attractive forces between the charged regions of polar molecules.
• Water is a polar molecule, and the polarity (the tendency to have positive and negative regions) results in hydrogen bonding. Both make water an important component of the human body. Water is an excellent solvent because of its polarity. Its hydrogen bonds give water a high heat capacity, which helps the body's internal temperature remain constant, and a high heat of vaporization, so that evaporation of perspiration from the body surface prevents overheating.
• Acids increase the concentration of H + in solution. Bases decrease the concentration of H + in solution. The strengths of acids and bases are measured on the pH scale. Biological fluids must remain within a narrow pH range. Buffers prevent dramatic changes in pH.
Major Molecules of Life (pp. 24-33)
• A polymer is a large molecule made of many smaller molecules, called monomers. Polymers form through dehydration synthesis (removal of a water molecule) and are broken apart by hydrolysis (addition of a water molecule).
• Carbohydrates are sugars and starches that provide fuel for the human body. Monosaccharides are the smallest monomers of carbohydrates. Oligosaccharides are chains of a few monosaccharides joined together by dehydration synthesis. Disaccharides consist of two monosaccharides and are one type of oligosaccharide. Polysaccharides are complex carbohydrates formed when large numbers of monosaccharides join through dehydration synthesis.
• Lipids, such as triglycerides, phospholipids, and steroids, are nonpolar molecules that do not dissolve in water. Triglycerides (fats and oils) are made of glycerol and three fatty acids and function in long-term energy storage. Phospholipids are important components of plasma membranes. Steroids include cholesterol, a component of the plasma membrane that also serves as the foundation for steroid hormones.
• Proteins are polymers made from a set of about 20 amino acids linked together through dehydration synthesis to form chains. There are four levels of protein structure. Primary structure is the specific sequence of amino acids in a protein. Secondary structure results from the bending and coiling of the amino acid chain into pleated sheets or helices. Tertiary structure is the threedimensional shape of the protein. Some proteins consist of two or more polypeptide chains, each comprising a subunit. Attractive forces between the subunits of these proteins produce quaternary structure.
• Enzymes are proteins that speed up chemical reactions without being consumed in the process. The substrate binds to the enzyme at the active site, forming an enzyme-substrate complex, and is then converted to products that leave the active site.
• Deoxyribonucleic acid (DNA) and ribonucleic acid (RNA) are polymers of nucleotides. A nucleotide consists of a five-carbon sugar bonded to one of five nitrogen-containing bases and a phosphate group. Genes are stretches of DNA that determine the sequence of amino acids during protein synthesis. DNA is double stranded, its sugar component is deoxyribose, and its nitrogen-containing bases are adenine, guanine, cytosine, and thymine. RNA is a single-stranded molecule that also plays a major role in protein synthesis. The sugar in RNA is ribose, and the nitrogen-containing bases are adenine, guanine, cytosine, and uracil.
• Adenosine triphosphate (ATP), the energy currency of cells, is a nucleotide made of the sugar ribose, the base adenine, and three phosphate groups. When cells require energy, one of the high-energy phosphate bonds is broken and energy is released.
Reviewing the Concepts
1. What is an atom? p. 14
2. Compare protons, neutrons, and electrons with respect to their charge, mass, and location within an atom. pp. 14-15
3. What is an isotope? p. 15
4. How is radiation used to diagnose or cure illness? pp. 16-17
5. Describe covalent, ionic, and hydrogen bonds. Give an example of each. pp. 18-21
6. What characteristics of water make it a critical component of the body? pp. 21-22
7. Define acids and bases according to what happens when they are added to water. pp. 22-23
8. How are polymers formed and broken? Give three examples of polymers and their component monomers. pp. 24-33
9. Name two important energy-storage polysaccharides and one important structural polysaccharide. pp. 25-26
10. Describe the structure of a phospholipid. What important roles do phospholipids play in the human body? pp. 28-29
11. Describe the four levels of protein structure. pp. 30-31
12. Compare the structures of RNA and DNA. pp. 32-33
13. Describe the structure and function of ATP. p. 33
14. Compare and contrast the ways in which proteins and nucleic acids are used in the body. pp. 29-33
15. Hydrogen bonds
a. are stronger than either ionic or covalent bonds.
b. form between a slightly positively charged hydrogen atom and a slightly negatively charged atom nearby.
c. maintain the shape of proteins and DNA.
d. b and c
16. Water
a. is a nonpolar molecule and therefore an excellent solvent.
b. has a high heat capacity and therefore helps maintain a constant body temperature.
c. has a low heat of vaporization and therefore helps prevent overheating of the body.
d. makes up about 25% of the human body.
17. Carbohydrates
a. consist of chains of amino acids.
b. supply our cells with energy.
c. contain glycerol.
d. function as enzymes.
18. Triglycerides
a. have one molecule of glycerol and three fatty acids.
b. are poor sources of energy.
c. are saturated when there are two or more double bonds linking carbon atoms.
d. are major components of plasma membranes.
19. Enzymes
a. speed up chemical reactions and are consumed in the process
b. only function in decomposition reactions.
c. are usually nonspecific and therefore capable of binding to many different substrates.
d. have locations, known as active sites, to which the substrate binds.
20. Changes in temperature or pH can cause a protein to lose its three-dimensional shape and become nonfunctional. This process is called _____.
21. DNA and RNA are polymers of smaller units called _____.
22. The _____ structure of a protein is the precise sequence of amino acids.
23. _____ are arranged in a double layer (bilayer) that forms the plasma membrane of cells.
24. In plants, the storage polysaccharide is _____, and in animals it is _____.
25. Buffers prevent dramatic changes in _____.
Applying the Concepts
1. A friend eyes your lunch and begins to lecture you on the perils of high-fat foods. You decide to acknowledge the health risks of eating a high-fat diet but also to point out to her the important roles that lipids play in your body. What will you say?
2. Bill claims that eating fruits and vegetables is overrated because humans lack the enzyme needed to digest cellulose and thus cellulose passes unchanged through our digestive tract. Is Bill correct that we gain nothing from eating food that contains cellulose?
3. For years your mother has been telling you to drink several large glasses of water each day. Having just read this chapter, you now understand the critical roles that water plays in your body. You vow to admit during your next phone call home that she has been right all along. Explain why dehydration might be dangerous.
4. Dental x-rays are taken using small doses of radiation and are used to detect tooth decay, injuries to the roots of teeth, and problems with the bones supporting the teeth. A dental professional covers the patient with a lead apron that runs from the neck to the abdomen and then leaves the room to take the x-ray. Why are such precautions necessary?
Becoming Information Literate
In nutrition class, you and your fellow students are scheduled to debate the pros and cons of irradiating food. You have been assigned the position of supporting food irradiation. Use at least three reliable sources (books, scientific journals, Internet sites) to develop your arguments in favor of food irradiation and to gather information needed to refute potential arguments made by the opposing side.
List the sources you used, and describe why you considered each source reliable.