Biology of Humans
18. Development throughout Life
In Chapter 17a we learned that some sexually transmitted diseases cause infertility, sterility, and ectopic pregnancy (a pregnancy in which the embryo develops in a location other than the uterus). In this chapter we describe the major milestones of human development, from the beginning of pregnancy (fertilization) to the transition into old age. We also consider treatments for infertility and sterility.
Periods of Development in Human Life
A newborn enters the world outside his mother's womb and rests (Figure 18.1). How did this tiny human develop? Consider that he—like you—began as a fertilized egg, no bigger than the period at the end of this sentence. The miracle of human development begins with fertilization, the union of a sperm and an egg, and the early stages take place within the female reproductive system (see Chapter 17). Birth occurs about 266 days after fertilization, marking the transition to development outside the mother's body.
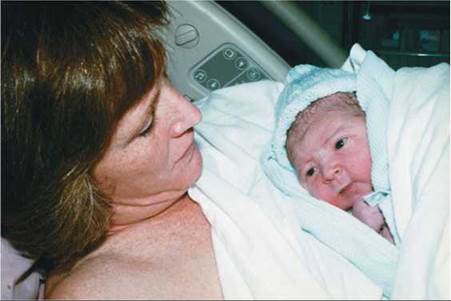
FIGURE 18.1. A newborn and his mother. Birth is the transition from prenatal to postnatal development.
The period of development before birth is the prenatal period. The period after birth is the postnatal period. Relative to prenatal development, postnatal development is a lengthy process, taking from 20 to 25 years to reach adulthood. Bodily changes do not cease once adulthood is reached, however. Our bodies continue to change throughout our lives as part of the growth and aging process.
Prenatal Period
We start our discussion of human development at the moment it all begins—when sperm meets egg. In this section, which presents the major events of prenatal development, we consider what normally happens and what can happen when things go wrong. The major events that occur during prenatal development are summarized by developmental period in Table 18.1.
· Human development begins with fertilization and continues until death. This couple is viewing ultrasound images of their fetus, who is a few months into a life that may last 70 to 80 years.
TABLE 18.1. Review of Major Events during Prenatal Development
Period |
Major Events |
Pre-embryonic (fertilization-week 2) |
Fertilization Cleavage Formation and implantation of the blastocyst Beginning of formation of extraembryonic membranes and placenta |
Embryonic (week 3-week 8) |
Gastrulation Formation of tissues, organs, and organ systems |
Fetal (week 9-birth) |
Continued differentiation and growth of tissues and organs Increase in length as measured from head to rump Increase in weight |
The human prenatal period is divided into three periods: (1) the pre-embryonic period, from fertilization through the second week (during which the developing human is called a pre-embryo); (2) the embryonic period, from week 3 through week 8 (during which the developing human is called an embryo); and (3) the fetal period, from week 9 until birth (during which the developing human is called a fetus). Gestation refers to the time a pre-embryo, embryo, or fetus is carried in the female reproductive tract. Several stages of prenatal human development are shown in Figure 18.2.
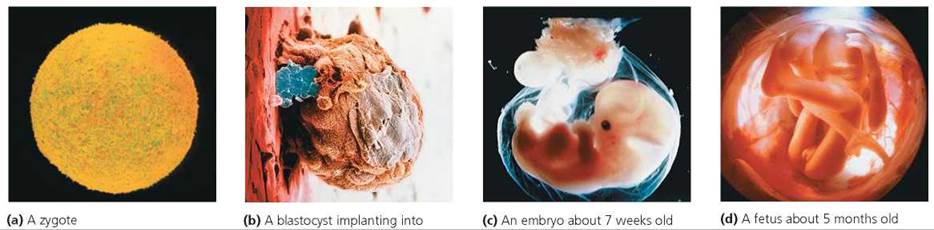
FIGURE 18.2. The developing human
Pre-embryonic Period
The pre-embryonic period begins with fertilization, the union between the nucleus of an egg and the nucleus of a sperm. Fertilization takes about 24 hours and usually occurs in a widened portion of the oviduct, not far from the ovary, as shown in Figure 18.3. Fertilization is a beautifully orchestrated performance by egg and sperm, aided by the actions of the oviducts and uterus.
Fertilization. At ovulation, the ovary releases the egg, still a secondary oocyte (see Chapter 17). The egg is swept into the oviduct by fingerlike projections called fimbriae (singular, fimbria; see Figure 18.3). Cilia and waves of peristalsis move the oocyte slowly along the oviduct toward the uterus (peristalsis is a general term for the rhythmic waves of contraction and relaxation of smooth muscles in the walls of tubular organs, such as oviducts or digestive organs, that push the contents through the tubes).
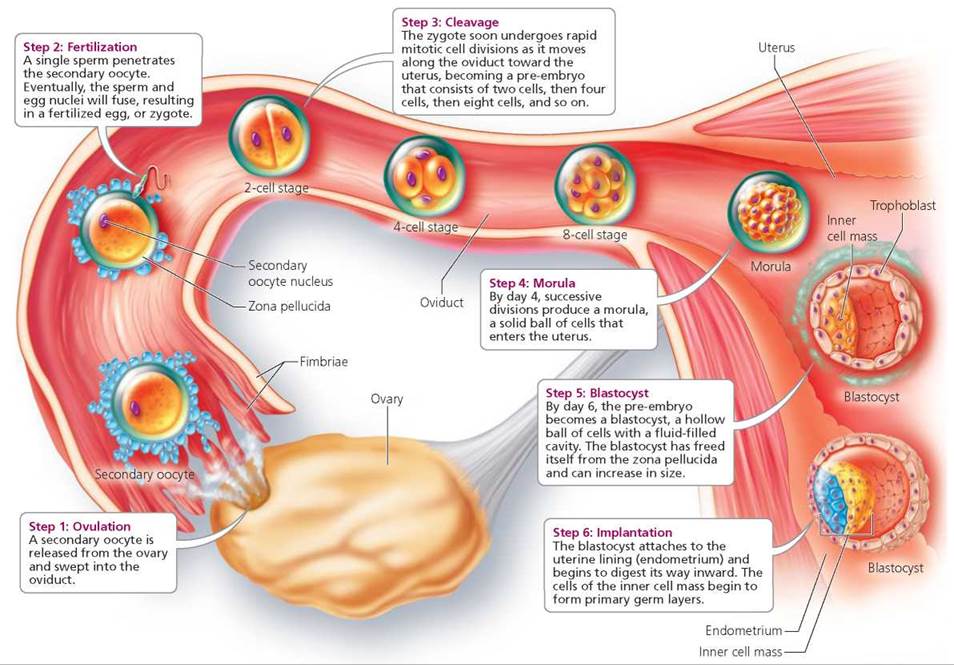
FIGURE 18.3. Early stages in the reproductive process
The trip made by sperm is a considerably more competitive and precarious venture than the movements of the typically lone oocyte. During sexual intercourse, from 200 million to 600 million sperm are deposited in the vagina and on the cervix, the neck of the uterus that extends into the vagina. Many sperm become trapped at the boundary of the vagina and cervix. Indeed, less than 1% of deposited sperm actually enter the uterus. Sperm that escape entrapment use their whiplike tails (recall from Chapters 3 and 17 that each sperm has a flagellum) to move into the uterus and eventually the oviduct. Along the way, the sperm are aided by small uterine contractions stimulated by chemicals (prostaglandins) in the semen. Of the vast number of sperm originally deposited in the female reproductive tract, only about 200 reach the site of fertilization in the widened portion of the oviduct.
Typically, an egg lives only 12 to 24 hours after its release from the ovary. For most eggs that get fertilized, the encounter with the sperm occurs within 12 hours of the egg's release. In addition, most sperm survive no more than 2 days in the female reproductive tract, though some may survive 5 days.
Two critical processes must precede fertilization (Figure 18.4). First, secretions from the uterus or oviducts must alter the surface of the acrosome, the enzyme-containing cap on the head of a sperm. Alteration of the acrosome requires about 6 to 7 hours and appears to involve the removal of cholesterol and possibly other substances from the surface of the sperm. These changes destabilize the sperm's plasma membrane. The second process occurs when sperm with unstable membranes contact the corona radiata, a layer of cells surrounding the secondary oocyte. Once such contact occurs, perforations develop in the sperm's weakened plasma membrane and outer acrosomal membrane. Enzymes (shown as green dots in Figure 18.4) then spill out of the acrosome and digest the attachments between cells of the corona radiata. Digestion of these attachments enables sperm to pass between the cells of the corona radiata to the layer below called the zona pellucida, the thick noncellular layer that immediately surrounds the secondary oocyte. Enzymes released from the acrosome also break apart the zona pellucida, creating a pathway to the oocyte.
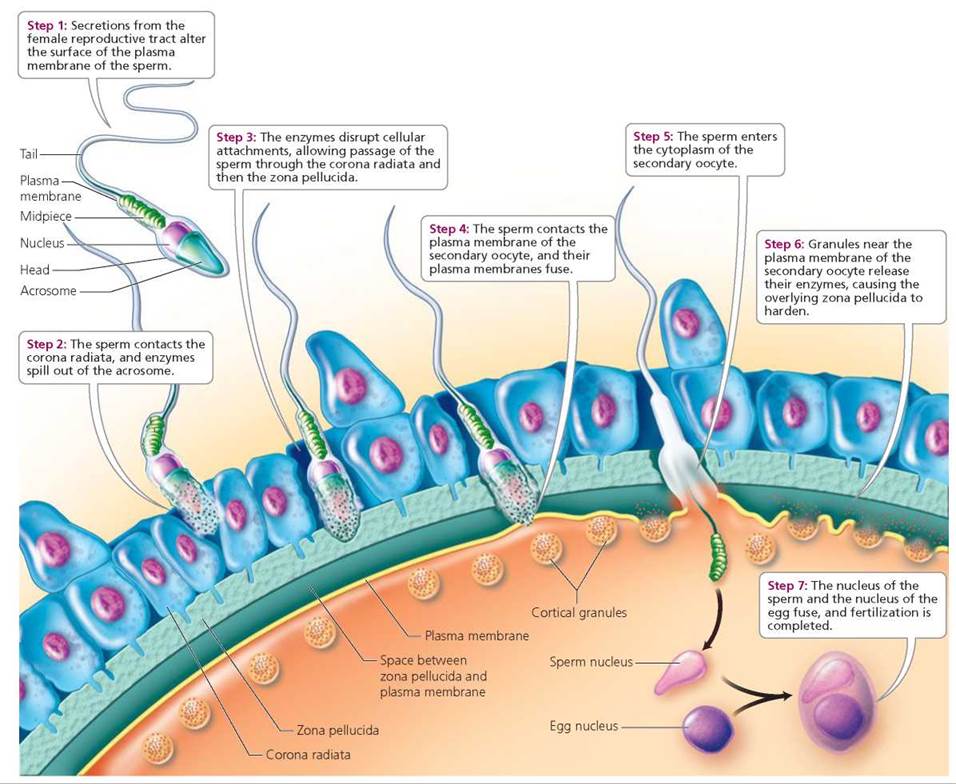
FIGURE 18.4. Fertilization
Several sperm may arrive at the secondary oocyte at about the same time, although usually only one sperm crosses the zona pellucida and reaches the plasma membrane of the secondary oocyte. The plasma membrane of the successful sperm and that of the secondary oocyte fuse, and the sperm enters the egg cytoplasm. Fusion of the plasma membranes triggers two important events. First, enzymes (shown as red dots in Figure 18.4) released by granules near the plasma membrane of the oocyte cause the zona pellucida to quickly harden and thereby prevent passage of other sperm. This block to entry by additional sperm ensures equal genetic contributions from each parent and prevents abnormal numbers of chromosomes in the resulting embryo, which would make it incapable of normal development. Second, the oocyte undergoes its second meiotic division (Chapter 17) and is now considered an ovum. The nucleus of the sperm (located in the sperm's head) and the nucleus of the ovum fuse (Figure 18.4). The remaining parts of the sperm (midpiece and tail) degenerate. The fertilized ovum is called a zygote. Just visible to the unaided eye, the zygote contains genetic material from both the mother (23 chromosomes) and father (23 chromosomes).
Cleavage About 1 day after fertilization, the zygote undergoes cleavage, a rapid series of mitotic cell divisions. During cleavage, the single-celled zygote becomes a pre-embryo, first consisting of two cells, then four cells, then eight cells, and so on. Cleavage occurs as the pre-embryo moves along the oviduct toward the uterus (see Figure 18.3). The cleaving pre-embryo becomes a solid ball of cells by about day 3. By day 4, the preembryo is a morula, a solid ball of 12 or more cells produced by successive divisions of the zygote. These early cell divisions do not result in an overall increase in size; such increases are prevented by the tight-fitting zona pellucida that still covers the morula. Instead, the cells within the ball become progressively smaller as divisions occur.
Sometimes during an early stage of cleavage, the mass of cells splits, and two pre-embryos are formed. Identical twins, also called monozygotic twins ("from one zygote"), develop in this way. Such twins are always the same gender and have nearly identical genetic material (see Chapter 18a for a discussion of twin studies). In rare cases, the splitting of the pre-embryo is incomplete and conjoined twins result. Such twins may be surgically separated after birth once doctors have evaluated which structures are shared and analyzed the likelihood of successful separation. Fraternal twins occur when two secondary oocytes are released from the ovaries and fertilized by different sperm. Such twins also are called dizygotic twins ("from two zygotes"). Fraternal twins may or may not be the same gender and are no more genetically similar than siblings who are not twins. Identical twins (separate and conjoined) and fraternal twins are shown in Figure 18.5.

FIGURE 18.5. Twins
Stop and think
Given what you know about the events necessary to produce identical and fraternal twins, how might a set of triplets form in which two are identical and one fraternal?
The morula enters the uterus about 5 days after fertilization. As cell division continues, a cavity begins to form at the morula's center. Cells lining the cavity flatten and compact as the zona pellucida still prevents increases in overall size. Fluid from the uterine cavity passes through the zona pellucida and accumulates within the forming cavity, and the morula is converted into a blastocyst, a ball of cells with an inner fluid-filled cavity. As shown in Figure 18.3, the blastocyst is made up of two parts: an inner cell mass and a tro- phoblast. The inner cell mass is a group of cells that will become both the embryo proper and some of the extraembryonic membranes that will extend from or surround the embryo. The trophoblast is a thin layer of cells that will give rise to the extraembryonic membrane that is the embryo's contribution to the placenta. The placenta is the organ that delivers oxygen and nutrients to the embryo and carries carbon dioxide and other wastes away. Before the blastocyst becomes implanted in the uterine wall and the placenta develops, the blastocyst floats freely in the uterus. Increases in overall size of the blastocyst become possible when it frees itself from the degenerating zona pellucida.
Implantation. The blastocyst attaches to the endometrium (the lining of the uterus) about 6 days after fertilization. The trophoblast rapidly proliferates and differentiates into two cell layers, one of which begins to invade the endometrium. During this process, called implantation, the blastocyst digests its way inward until it is firmly embedded in the endometrium (Figure 18.6). Implantation normally occurs high up on the back wall of the uterus. Sometimes, however, a blastocyst implants outside the uterus, and an ectopic pregnancy results. The vast majority of implantations outside the uterus occur in the oviducts, usually because passage of the dividing pre-embryo through the oviduct is impaired in some way. Factors that hinder passage of the pre-embryo through the oviduct include structural abnormalities and scar tissue from surgery or pelvic inflammatory disease (see Chapters 17 and 17a). In such instances, the embryo is surgically removed (and hence the pregnancy terminated) because the oviduct cannot support a pregnancy, and because rupture of the oviduct by a growing embryo and the resulting hemorrhage can be fatal to the mother.
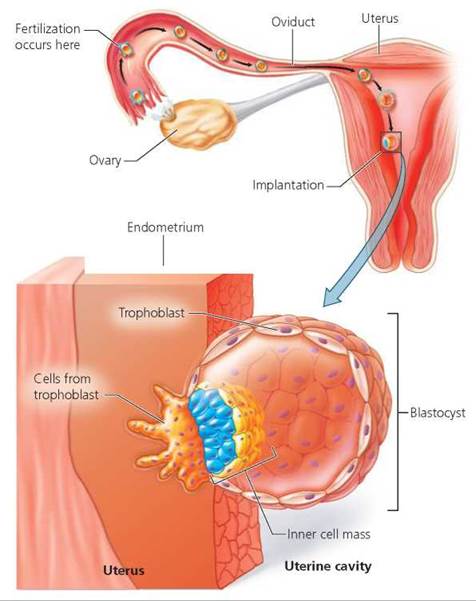
FIGURE 18.6. Implantation. About 6 days after fertilization, the blastocyst attaches to the endometrium of the uterus and begins to digest its way inward.
An estimated one-third to one-half of all zygotes fail to become blastocysts and fail to implant. The failed zygotes and early pre-embryos are either reabsorbed by cells of the endometrium or expelled from the uterus in an early spontaneous abortion (such an end to a pregnancy is described as spontaneous because it was not medically induced). Causes of early spontaneous abortion include chromosomal abnormalities in the zygote and an inhospitable uterine environment for implantation. The latter might result from the presence of an intrauterine device (IUD) or inadequate production of the hormones estrogen and progesterone by the corpus luteum. (Recall from Chapter 17 that the corpus luteum is a glandular structure that forms from the ovarian follicle after ovulation.) These early spontaneous abortions are included in the term miscarriage, which describes pregnancy loss occurring before 20 weeks gestation. (After 20 weeks, the fetus is considered potentially viable, with medical support, and such deliveries are described as preterm or premature births when they occur before 37 weeks gestation; discussed in more detail later in this chapter).
When conditions are right, the blastocyst completes implantation by the end of the second week of the pre-embryonic period. During implantation, cells within the blastocyst produce human chorionic gonadotropin (HCG), a hormone that enters the mother's bloodstream and is excreted in her urine. Many pregnancy tests screen for the presence of this hormone in the mother's urine. Enough HCG is produced by the end of the second week to be detected by the pregnancy test and yield a positive result. The physiological function of HCG is to maintain the corpus luteum and stimulate it to continue producing progesterone. Progesterone is essential for maintenance of the endometrium; without an adequate supply of progesterone, the endometrium would be shed, as during menstruation.
Infertility is the inability of a female to conceive (become pregnant) or of a male to cause conception. Implantation is a major hurdle in the series of steps leading to a successful pregnancy. The Ethical Issue essay Making Babies describes the options available to infertile couples wishing to have children.
Extraembryonic membranes. Toward the end of the preembryonic period, four membranes—the amnion, yolk sac, allantois, and chorion—begin to form around the pre-embryo (Figure 18.7). Formation of these membranes extends into the embryonic period, when the developing human is called an embryo. The membranes lie outside the embryo and are called extraembryonic membranes. These membranes protect and nourish the embryo and later the fetus. The amnion surrounds the entire embryo, enclosing it in a fluid-filled space called the amniotic cavity. Amniotic fluid forms a protective cushion around the embryo that later can be examined as part of prenatal testing in a procedure known as amniocentesis (see Chapter 20). The yolk sac is the primary source of nourishment for embryos in many species of vertebrates. In embryonic humans, however, it remains quite small and does not provide nourishment. Human embryos receive nutrients from the placenta. The yolk sac in humans is a site of early blood cell formation. It also contains primordial germ cells that migrate to the embryo's gonads (testes or ovaries), where they differentiate into immature cells that will eventually become sperm or oocytes. The allantois is a small membrane whose blood vessels become part of the umbilical cord, the ropelike connection between the embryo and the placenta. The umbilical cord consists of blood vessels and supporting connective tissue. Finally, the chorion is the outermost membrane, which rests against the uterine cavity. The chorion develops largely from the trophoblast and becomes the embryo's major contribution to the placenta. The amnion, yolk sac, and allantois develop from the inner cell mass.
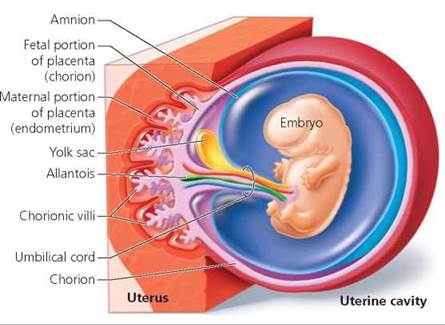
FIGURE 18.7. Extraembryonic membranes. The amnion, yolk sac, chorion, and allantois begin to form during the second to third week after fertilization.
The placenta. The placenta orchestrates all interactions between the mother and the fetus. The placenta forms from the chorion of the embryo and a portion of the endometrium of the mother (specifically, the endometrium in the area where implantation occurred; Figure 18.8a).
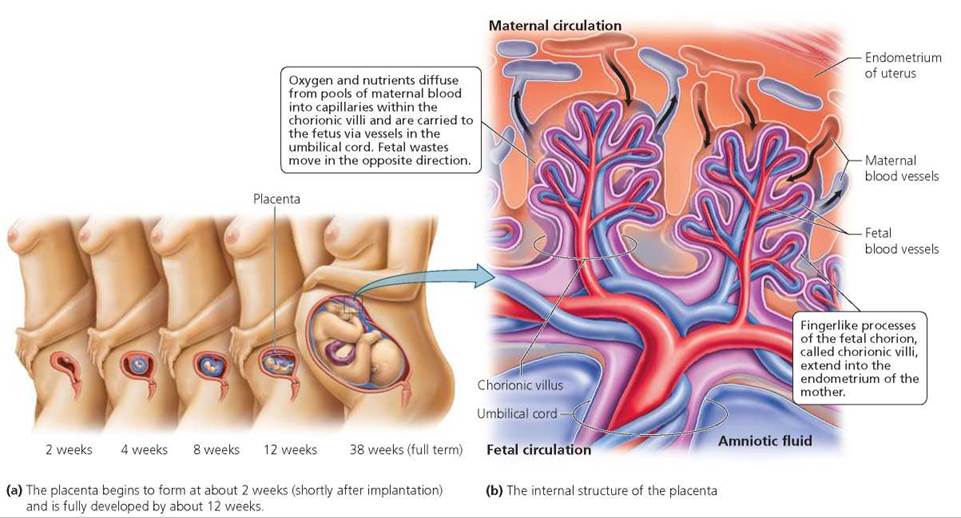
FIGURE 18.8. The placenta is formed from the chorion of the embryo and the endometrium of the mother.
A major function of the placenta is to allow oxygen and nutrients to diffuse from maternal blood into embryonic blood. Wastes such as carbon dioxide and urea diffuse from embryonic blood into maternal blood. The placenta also produces hormones such as HCG, estrogen, and progesterone. Placental hormones, as mentioned earlier, are essential for the continued maintenance of pregnancy. High levels of HCG from the placenta may be responsible for morning sickness, the nausea and vomiting experienced by some women early in pregnancy. (Morning sickness is not restricted to the morning, by the way.)
Formation of the placenta begins shortly after implantation when cells derived from the trophoblast rapidly divide and invade the endometrium. Cavities form that fill with blood from maternal capillaries severed by the invading cells. Soon, fingerlike processes of the chorion, called chorionic villi, grow into the endometrium (Figure 18.8b). These chorionic villi grow, divide, and continue their invasion of maternal tissue, causing ever larger cavities and pools of maternal blood to form. The placenta is fully developed by the third month of pregnancy. At this time, it weighs about 680 g (1.5 lb).
Chorionic villi contain blood vessels connected to the developing embryo. Oxygen and nutrients in the pools of maternal blood diffuse through the capillaries of the villi into the umbilical vein and travel to the embryo. Wastes leave the embryo by the umbilical arteries, move into capillaries within the chorionic villi, and diffuse into maternal blood. Thus, the chorionic villi provide exchange surfaces for diffusion of nutrients, oxygen, and wastes. Some chorionic villi also anchor the embryonic portion of the placenta to maternal tissue.
Under normal circumstances, there is no direct mixing of maternal and fetal blood; all exchanges occur across capillary walls. Nevertheless, the closeness of the maternal and fetal blood vessels within the placenta causes the fetus to share in maternal nutrition, habits, and lifestyle. In addition to beneficial nutrients and oxygen, harmful substances can cross the placenta from the mother to the fetus, including some drugs, alcohol, caffeine, toxins in cigarette smoke, and HIV (human immunodeficiency virus, the virus that causes AIDS).
Usually, implantation occurs high up on the back wall of the uterus, so the placenta forms in the upper portions of the uterus. Sometimes, however, implantation occurs lower in the uterus, and the developing placenta grows to cover the cervix, the neck of the uterus that projects into the vagina; this condition is called placenta previa. Placenta previa may cause premature birth or maternal hemorrhage. Most women with diagnosed placenta previa have their baby delivered by cesarean section before labor begins. Cesarean section, often shortened to C-section, is the procedure by which the fetus and placenta are removed surgically from the uterus through an incision in the abdominal wall and uterus.
Embryonic Period
The embryonic period extends from the third to the eighth week of development. It is a time of great change, beginning with the formation of three distinct germ layers from which all tissues and organs develop. We first describe the general process by which germ layers form, and then we consider development of the central nervous system and reproductive system as examples of tissue and organ formation during the embryonic period. By the end of the embryonic period, all organs have formed, and the embryo has a distinctly human appearance. Three interrelated processes produce this tiny human: cell division (which continues from the pre-embryonic period); cell differentiation (the process by which cells become specialized with respect to structure and function); and morphogenesis (the development of overall body organization and shape).
Gastrulation. Morphogenesis begins during the third week after fertilization. Just before the start of morphogenesis, during implantation, the inner cell mass moves away from the surface of the blastocyst, and the amniotic cavity forms. The amniotic cavity is filled with amniotic fluid and lined by the amnion, one of the four extraembryonic membranes. The inner cell mass, which now becomes a flattened platelike structure called the embryonic disk (Figure 18.9), is destined to become the embryo proper. But first, the cells within the embryonic disk must differentiate and migrate, forming three primary germ layers known as ectoderm, mesoderm, and endoderm. The cell movements that establish the primary germ layers are called gastrulation, and the embryo during this period is called a gastrula (Figure 18.9).
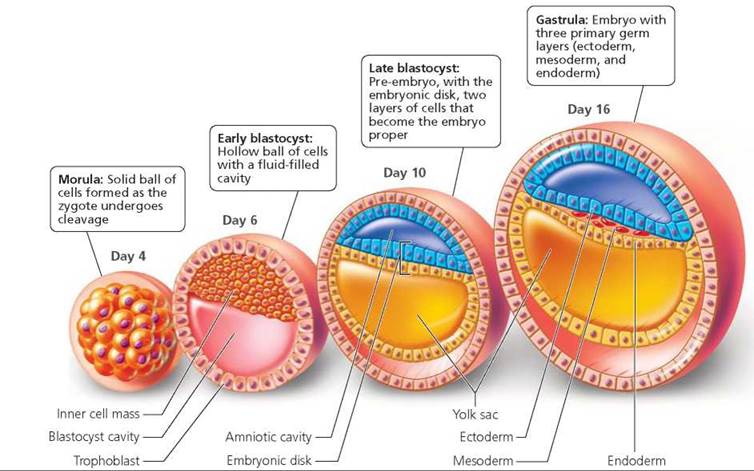
FIGURE 18.9. Early stages of development In cross section
All tissues and organs develop from the primary germ layers. Ectoderm covers the surface of the embryo and forms the outer layer of skin and its derivatives such as hair, nails, oil glands, sweat glands, and mammary glands. Ectoderm also forms the nervous system. Another germ layer, the endoderm, turns inward to form the lining of the digestive tract. Endoderm also forms the lining of the urinary and respiratory tracts, as well as other organs and glands (for example, the pancreas, liver, thyroid gland, and parathyroid glands). Mesoderm fills some of the space between ectoderm and endoderm, and gives rise to muscle; bone and other connective tissue; and various organs, including the heart, kidneys, ovaries, and testes. Because gastrulation initiates the processes by which the developing embryo's body takes shape and becomes organized, it is considered a key part of morphogenesis.
As gastrulation proceeds, a flexible rod of mesoderm tissue called the notochord develops where the vertebral column will form. The notochord defines the long axis of the embryo and gives the embryo some rigidity. The notochord also prompts overlying ectoderm to begin formation of the central nervous system (CNS), as discussed shortly. Vertebrae eventually form around the notochord, and the notochord degenerates. The pulpy, elastic material in the center of intervertebral disks (pads that help cushion the bones of the vertebral column) is all that remains of our notochord.
Development of the central nervous system. A major milestone in embryonic development is the formation of the central nervous system (brain and spinal cord) from ectoderm, as shown in Figure 18.10. The developing notochord induces the overlying ectoderm to thicken. This thickened region of ectoderm above the notochord is called the neural plate. The neural plate folds inward, forming a groove that extends the length of the embryo, along its back surface. The raised sides of the groove, known as neural folds, grow upward and eventually meet and fuse to form the neural tube, a fluid-filled tube that will become the central nervous system. The process by which the neural tube is formed is called neurulation, and the embryo during this period is called a neurula. The anterior portion of the neural tube develops into the brain, and the posterior portion forms the spinal cord. Alongside the neural tube, mesoderm cells organize into blocks called somites. Somites eventually form skeletal muscles of the neck and trunk, connective tissues, and vertebrae.
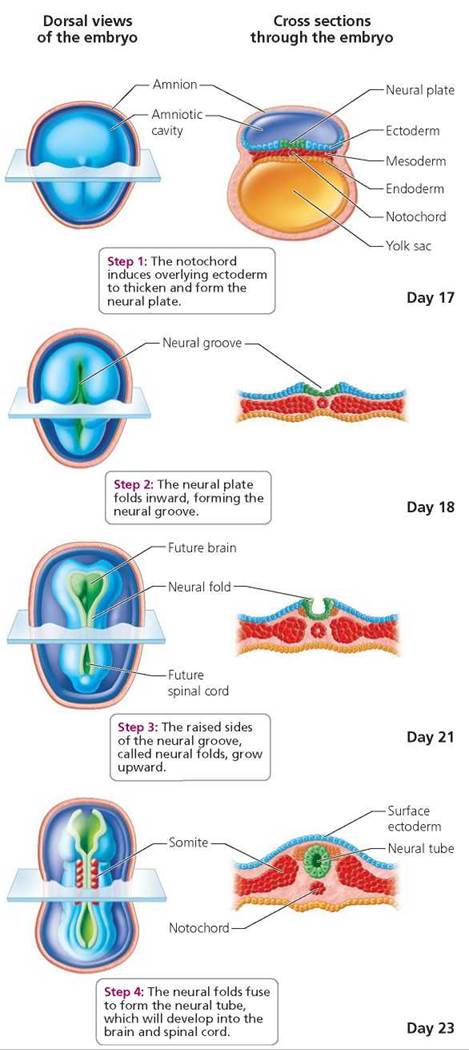
FIGURE 18.10. Formation of the central nervous system from ectoderm. Dorsal views of a human embryo (left) and corresponding crosssectional views or parts of such views (right) are shown at four different days during development of the brain and spinal cord.
Failure of the neural tube to develop and close properly results in neural tube defects. Spina bifida ("split spine") is a type of neural tube defect in which part of the spinal cord develops abnormally, as does the adjacent area of the spine. The severity of spina bifida varies greatly, and some cases can be improved through surgery. Anencephaly is a neural tube defect that involves incomplete development of the brain and results in stillbirth or death shortly after birth.
Development of the reproductive system. Among the 23 chromosomes provided by the mother's egg is an X chromosome. Among the 23 chromosomes provided by the father's sperm is an X or a Y chromosome. The X and Y chromosomes are called sex chromosomes, and they determine the gender of human embryos. More specifically, the gender of an embryo is determined at fertilization by the type of sperm that fertilizes the egg. If an X-bearing sperm fertilizes the egg, then the zygote is XX and will normally develop as a female. If a Y-bearing sperm fertilizes the egg, then the zygote is XY and will normally develop as a male.
What would you do?
Methods are available to separate X-bearing sperm from Y-bearing sperm. During the procedure, sperm are treated with a fluorescent dye that binds to DNA. X-bearing and Y-bearing sperm glow differently because there is almost 3% more DNA in X chromosomes than Y chromosomes. Thus X- and Y-bearing sperm can be sorted by an automated machine. Such “sperm sorting” makes possible the selection of a child's gender. Couples could decide whether to use X-bearing sperm to produce a female baby or Y-bearing sperm to produce a male baby. Some couples want to select the gender of their child to avoid X-linked genetic disorders; others want to balance their families with respect to numbers of sons and daughters. The chosen sperm are deposited within the woman's vagina or cervix at a time when pregnancy is likely to occur. Should parents be allowed to choose the gender of their children? What implications would gender selection have for families and for society? If you were given the responsibility of deciding whether such technology should continue to be made available to parents, what would you do?
Male and female embryos have the same external and internal anatomy for the first few weeks of development; their reproductive organs have not yet differentiated. The embryos at this time are described as being "sexually indifferent." However, about 6 weeks after fertilization, a region of the Y chromosome—called SRY for sex-determining region of the Y chromosome—initiates development of testes in XY embryos. The testes soon begin producing testosterone, the hormone that directs development of male reproductive organs. Female embryos lack the Y chromosome; in its absence, ovaries develop. The development of female reproductive structures is not influenced by hormones, and female structures will develop even if ovaries do not fully develop. Thus, it is the absence of the Y chromosome and testosterone that leads to female development. We focus on development of the external genitalia, but keep in mind that internal reproductive organs also are differentiating.
Male and female embryos of about 6 weeks of age have a small bud of tissue between their legs (Figure 18.11). Just below this bud is a shallow depression called the urogenital groove. The urogenital groove is surrounded by urogenital folds, which in turn are surrounded by labioscrotal swellings. In the male embryo, testosterone produced by the newly differentiated testes causes the urogenital groove to elongate and completely close. The bud then becomes the glans penis; the urogenital folds develop into the shaft of the penis; and the labioscrotal swellings become the scrotum (into which the testes will later descend). In the female, the urogenital groove also elongates, but instead of closing, as in the male, it remains open. The bud becomes the clitoris in females, and the urogenital folds develop into the labia minora. The labioscrotal swellings of females become the labia majora. The formation of male and female reproductive structures is typically completed by the end of the third month of gestation.
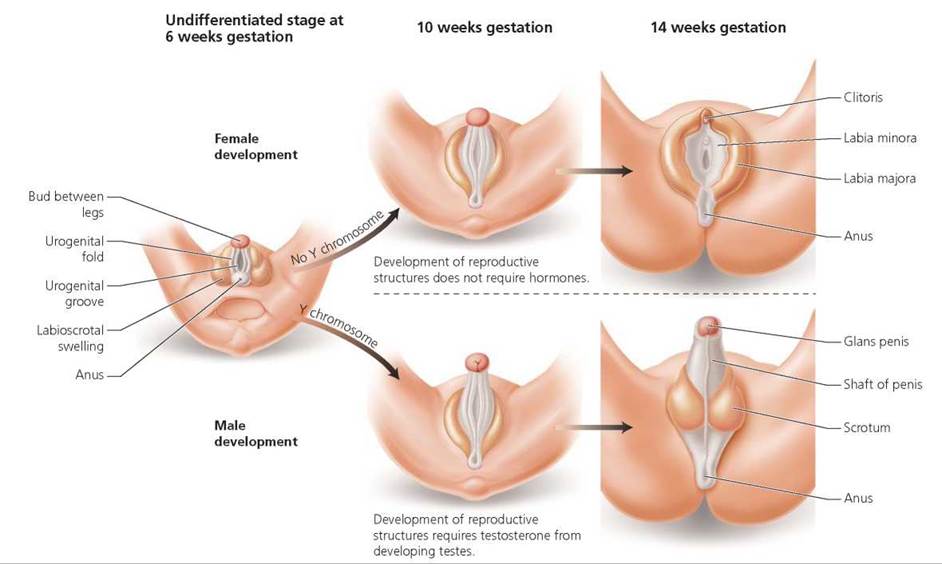
FIGURE 18.11. Development of external genitalia
Fetal Period
The fetal period extends from the ninth week after fertilization until birth. Although tissues and organs continue to differentiate during this period, the most notable change in most body parts is rapid growth, made possible by the placenta. The placenta completes its development early in the fetal period.
Growth. Growth during the fetal period is extremely rapid, as reflected in phenomenal increases in weight (from 8 g to 3400 g, or from about 0.3 oz to 120 oz) and substantial increases in length (the length as measured from the head to the rump changes from 50 to 360 mm, or from about 2 in. to 14 in.), when the pregnancy continues for the full 38 weeks. One striking change during the fetal stage is that the growth rate of the head slows relative to the growth rate of other regions of the body, altering the way the body is proportioned (Figure 18.12). Difference in the relative rates of growth of various parts of the body is called allometric growth. Such growth continues after birth and helps to shape developing humans and other organisms.
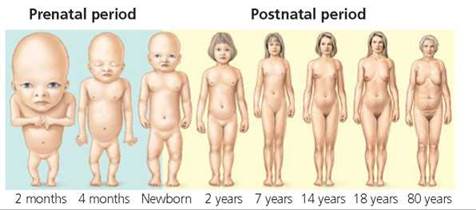
FIGURE 18.12. Allometric growth. Changes in body proportions occur throughout prenatal and postnatal development. For comparison, all stages are drawn to the same total height.
Fetal circulation. The fetal circulatory system differs from circulation after birth because several organs—the lungs, kidneys, and liver, to name a few—do not perform their postnatal functions in the fetus (Figure 18.13). Before birth, most blood is shunted past these organs through temporary vessels or openings. Recall that the fetus receives its oxygen and nutrients from maternal blood and gives up its carbon dioxide and wastes to maternal blood at the placenta.
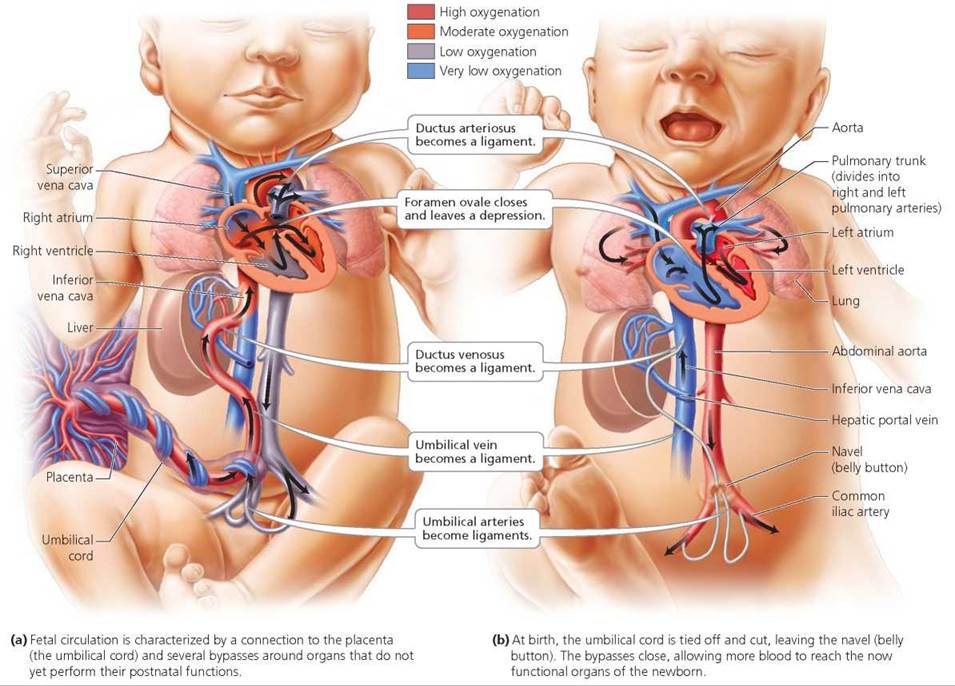
FIGURE 18.13. Fetal circulation and changes at birth
Which two structures in the fetus allow blood to bypass the pulmonary
In the fetus, the foramen ovale (between the right and left atria) and the ductus arteriosus (on the pulmonary trunk) divert blood away from the lungs, which are not yet functional.
Fetal blood must travel to the placenta to rid itself of carbon dioxide and wastes and to pick up oxygen and nutrients. It does so by flowing through umbilical arteries that branch off large arteries in the legs of the fetus. The two umbilical arteries run through the umbilical cord to the placenta. Once exchanges between fetal and maternal blood have occurred at the placenta, blood rich in oxygen and nutrients travels back to the fetus in the umbilical vein (within the umbilical cord; Figure 18.13a). Some of this blood flows to the fetal liver, which produces red blood cells but does not yet function in digestion. Most of the blood from the placenta, however, bypasses the liver by means of a temporary vessel called the ductus venosus and heads to the heart.
Like the liver, fetal lungs are not yet functional and need only a small amount of blood for growth and removal of wastes from their cells. Once blood enters the right atrium of the heart, some of it passes to the right ventricle and on to the lungs, as it does in circulation after birth. Most of the blood, however, moves into the left atrium through a small hole in the wall between the atria, called the foramen ovale. The blood in the left atrium moves into the left ventricle and out to the body of the fetus. Another shunt, called the ductus arteriosus, connects the pulmonary trunk to the aorta and also functions to divert blood away from the lungs. Thus, blood flowing out of the right ventricle into the pulmonary trunk is shunted away from the lungs and into the aorta, where it travels to all areas of the fetus. Another glance at Figure 18.13a reveals that much of the blood traveling through the fetus is moderate to low in oxygen. Indeed, the umbilical vein is the only fetal vessel that carries fully oxygenated blood.
At birth—when the lungs, liver, and other organs begin their postnatal functions—fetal circulation converts to the postnatal pattern (Figure 18.13b). Blood flow to the placenta ceases when the umbilical cord is tied and cut off. The scar left by the cord becomes the baby's navel, or belly button. Within the infant, the umbilical arteries, umbilical vein, ductus veno- sus, and ductus arteriosus constrict, shrivel, and form ligaments. The foramen ovale normally closes shortly after birth, leaving a small depression in the wall between the two atria.
Ethical Issue
Making Babies
The inability to become pregnant or to cause a pregnancy is called infertility. Couples are considered infertile if conception does not occur after 1 year of unprotected sexual intercourse. Infertility strikes about one in six American couples. In some of these couples, the woman is infertile. Hormonal imbalances may disrupt ovulation or implantation, or scarring from disease may block the oviducts and prevent passage of gametes. Some women are sterile (they can never conceive and carry a child) because they have had their uterus (and sometimes their ovaries) removed for medical reasons. In other couples, it is the man who is infertile. Male infertility often is caused by production of few or sluggish sperm. Some men are sterile (they can never cause a pregnancy) as a result of conditions such as cryptorchidism (failure of the testes to descend from the abdomen to the scrotum). Also, in a strange twist of fate, otherwise compatible couples may be incompatible at the cellular level—some women produce antibodies that kill their partner's sperm. For some couples the cause of infertility is unknown.
Can anything be done to help those who are infertile? For roughly half of the couples seeking help the answer is yes, although the road to reproduction may be long, expensive, and filled with emotional ups and downs. The procedures to treat infertility are collectively called assisted reproductive techniques (ARTs).
Administering hormones can treat some cases of female infertility. Hormones that trigger ovulation may be given to women whose ovaries fail to release eggs properly. Women who tend to miscarry (spontaneously abort) may be given progesterone to enhance the receptivity of their uterine lining. However, some causes of infertility, such as scarred oviducts in women and low sperm counts in men, do not respond to hormone therapy. What can be done in these cases?
Artificial insemination is one option available to couples whose infertility is caused by a low sperm count. In this procedure, sperm that have been donated and stored at a sperm bank are deposited (usually with a syringe) in the woman's cervix or vagina at about the time of ovulation. Sperm from the male member of the couple may be concentrated and then used. Alternatively, couples may use semen from an anonymous donor. Another possibility for couples in which the man has few sperm, or sperm that lack the strength or enzymes necessary to penetrate an egg, is intracytoplasmic sperm injection (ISI). In ISI, a tiny needle is used to inject a single sperm into an egg. If fertilization occurs, the embryo is transferred to the uterus.
Some cases of infertility can be treated with in vitro fertilization (IVF). IVF involves placing eggs and sperm together in a laboratory dish. First, the woman is treated with hormones to trigger superovulation—the ovulation of many eggs. Next, the eggs are removed from the woman's ovaries and placed into a dish that contains some of the man's sperm. If fertilization occurs, the zygotes are transferred to a solution that will support further development. Eventually one or more of the pre-embryos is transferred to the woman's uterus, which has been primed with hormones to support implantation. Women with blocked oviducts may conceive with IVF because the technique bypasses the oviducts altogether. If the woman lacks a uterus, the couple can hire a surrogate mother to gestate their baby. The egg and sperm may come from the couple or from other individuals.
IVF is sometimes unsuccessful because preembryos fail to implant. The problem seems to be the rather violent squirt of the pre-embryo into a reproductive tract that is already traumatized from hormone treatments and retrieval of eggs. Two other procedures avoid the problem of the pre-embryo's abrupt arrival in the uterus. In gamete intrafallopian transfer (GIFT), eggs and sperm are collected from a couple and inserted into the woman's oviduct, where fertilization may occur. Afterward, any resulting pre-embryos drift naturally (and gently) into the uterus. The second procedure is zygote intrafallopian transfer (ZIFT). In ZIFT, eggs and sperm are collected and brought together in a laboratory dish. If fertilization occurs, the resulting zygotes are inserted into the woman's oviducts, where they continue to develop and travel on their own to the uterus. Normal, healthy oviducts are needed for GIFT and ZIFT to work.
Success rates of ARTs range from about 20% to 28% live births per egg retrieved. Although costs vary somewhat, each attempt typically costs between $8000 and $12,000, and multiple attempts often are needed. What about the couples who do not conceive with ARTs? Is there hope that some day they might also conceive? The answer is yes—advances in reproductive research and technology are rapidly providing potential treatments for infertility.
Questions to Consider
• If you donated eggs or sperm for use by infertile couples, should you have a claim (legal or otherwise) to any offspring produced?
• If you were charged with establishing policies at an infertility clinic, how would you decide what should happen to pre-embryos created at the clinic that turned out to be “extra”? Do they have a right to life? And who gets the pre-embryos when couples divorce or die?
Stop and think
In some newborns, the foramen ovale fails to close. These infants, called blue babies, have a bluish appearance. What might explain their appearance?
Birth
Birth, also called parturition, usually occurs about 38 weeks after fertilization. The process by which the fetus is expelled from the uterus and moved through the vagina to the outside world is called labor. Uterine contractions during true labor occur at regular intervals, are usually painful, and intensify with walking. Recall from Chapter 10 that the hormone oxytocin released from the posterior pituitary gland causes contractions of the smooth muscle of the uterus. These contractions, in turn, stimulate further release of oxytocin in a positive feedback loop. As true labor proceeds, contractions become more intense, and the interval between them decreases. In contrast, false labor consists of irregular contractions that fail to intensify or change with walking; these contractions are sometimes called Braxton Hicks contractions, after the doctor who first described them in 1872. These contractions routinely occur and are most common during the last few months of pregnancy.
True labor can be divided into three stages (Figure 18.14). The first stage, known as the dilation stage, begins with the onset of regular contractions and ends at the point when the cervix has fully dilated to 10 cm (4 in.). This stage usually lasts about 6 to 7 hours, although it can be shorter in women who previously have given birth. During the dilation stage, the amniotic sac usually ruptures, releasing amniotic fluid. Known commonly as breaking water, rupture of the amniotic sac may be done deliberately by a doctor or midwife if it does not happen spontaneously. The second stage, the expulsion stage, begins with full dilation of the cervix and ends with delivery of the baby. This stage may last 1 or more hours. Some physicians make an incision to enlarge the vaginal opening, just before passage of the baby's head. This procedure, an episiotomy, facilitates delivery and avoids ragged tearing, which is generally slower to heal. Once delivery of the baby is complete, the umbilical cord is clamped and cut. The newborn takes his or her first breath, and the conversion of fetal circulation to the postnatal pattern begins. Labor, however, is not over. The third (and final) stage of true labor is the placental stage, which begins with delivery of the newborn and ends when the afterbirth, consisting of the placenta and fetal membranes, is expelled from the mother's body about 15 minutes after the baby. Continuing and powerful uterine contractions aid in expulsion of the placenta and constriction of maternal blood vessels torn during delivery. Any vaginal incision or tearing is sutured once labor has ended.
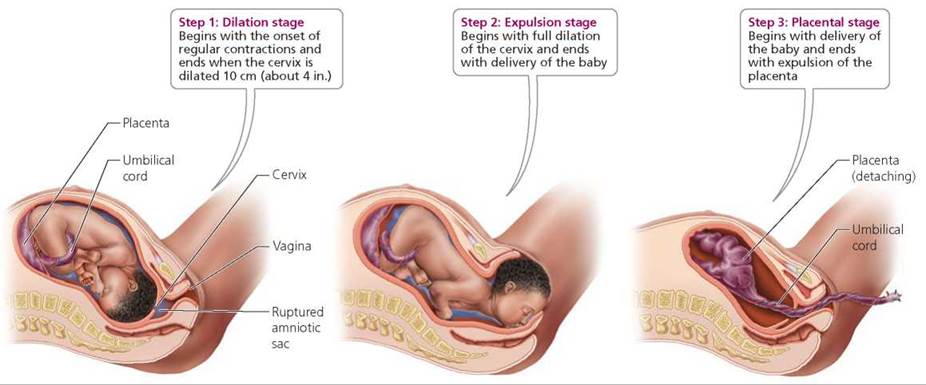
FIGURE 18.14. The stages of true labor
Most babies are born head first, facing the vertebral column of their mother. Some babies, however, are born buttocks first, and their delivery is called breech birth. Breech births are associated with difficult labors and umbilical cord accidents, such as compression or looping of the cord around the baby's neck. Thus, many physicians attempt to turn breech babies into the headfirst position before delivery. The baby may be delivered by cesarean section if turning is not successful.
Babies born at least 38 weeks after fertilization are called full-term infants. These babies weigh, on average, 3400 g (7.5 lb). Some babies are born 1 or more weeks late. Because the placenta begins to deteriorate with time, labor is typically induced if a pregnancy continues for 41 or more weeks. Babies born before 37 weeks of gestation are called premature infants. Their chances for survival increase with length of time spent in the female reproductive tract. Doctors do not usually attempt to save infants born at 22 weeks gestation or less because, even with intensive medical care, major disabilities are almost inevitable. Such babies weigh about 630 g (1.4 lb). Amazingly, infants born at 25 weeks gestation and weighing about 1 kg (2.2 lb) have a 50% to 80% chance of surviving, provided they receive intensive care. Intensive medical care is needed because the organ systems of these infants have not matured sufficiently to take over the functions normally performed by the mother's body. Care for premature infants often includes numerous blood transfusions, tube feeding, and maintenance on a respirator (Figure 18.15). Even with excellent intensive care, health problems may persist well beyond infancy.
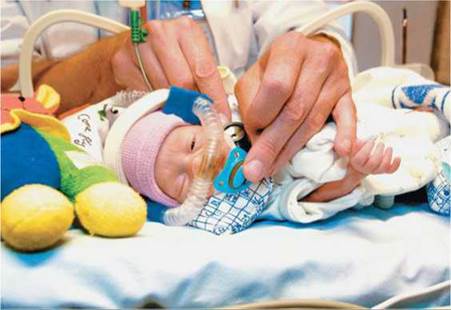
FIGURE 18.15. Many premature infants requireintensive care because their organs are not yet functional.
What would you do?
Babies born before 25 weeks gestation typically have a low chance of survival, even with excellent medical care. In addition, they have a very high probability of suffering from severe disabilities, if they do survive. Should efforts be made to save all such infants? Or should criteria be used to decide which infants the doctors should try to save and which they should let die? If you were given the responsibility of developing such criteria, what would you suggest? More to the point, if your infant were born before 25 weeks, what would you ask the doctor to do?
Birth Defects
The development of a fertilized egg into a baby is an incredibly wondrous yet complex process. In view of its complexity, it is not difficult to imagine that things might go wrong along the way. Indeed, the potential for mistakes seems enormous. Thus, we should not be surprised to learn that not every zygote develops into a baby, and not all babies are born healthy. Developmental defects present at birth are called birth defects. They may involve structure, function, behavior, or metabolism. The study of birth defects is called teratology (teratos, monster), reflecting the unfortunate term used in earlier times to describe severely deformed infants.
The causes of birth defects may be genetic or environmental. Genetic causes include mutant genes or changes in the number or structure of chromosomes. Environmental causes include drugs, chemicals, radiation, deficiencies in maternal nutrition, and certain viruses, such as herpes simplex and rubella (the cause of German measles). Birth defects resulting from genetic causes will be discussed in Chapter 19. Some of the birth defects associated with psychoactive drugs were considered in Chapter 8a.
Environmental agents that disrupt development have their greatest effects during periods of rapid differentiation, when cells and tissues acquire specific functions and form organs. Recall that these events largely occur during the embryonic period (week 3 through week 8). Exposure to disruptive agents during the embryonic period can cause major birth defects (Figure 18.16). Such defects, characterized by major structural abnormalities, occur in 2% to 3% of newborns. Spina bifida is an example of a major birth defect. Disruptive agents produce more minor defects during the fetal period, when most organs are growing rather than differentiating. Minor birth defects, such as having a single horizontal crease across the upper palm of the hand (instead of two creases), occur in about 4% to 5% of newborns.
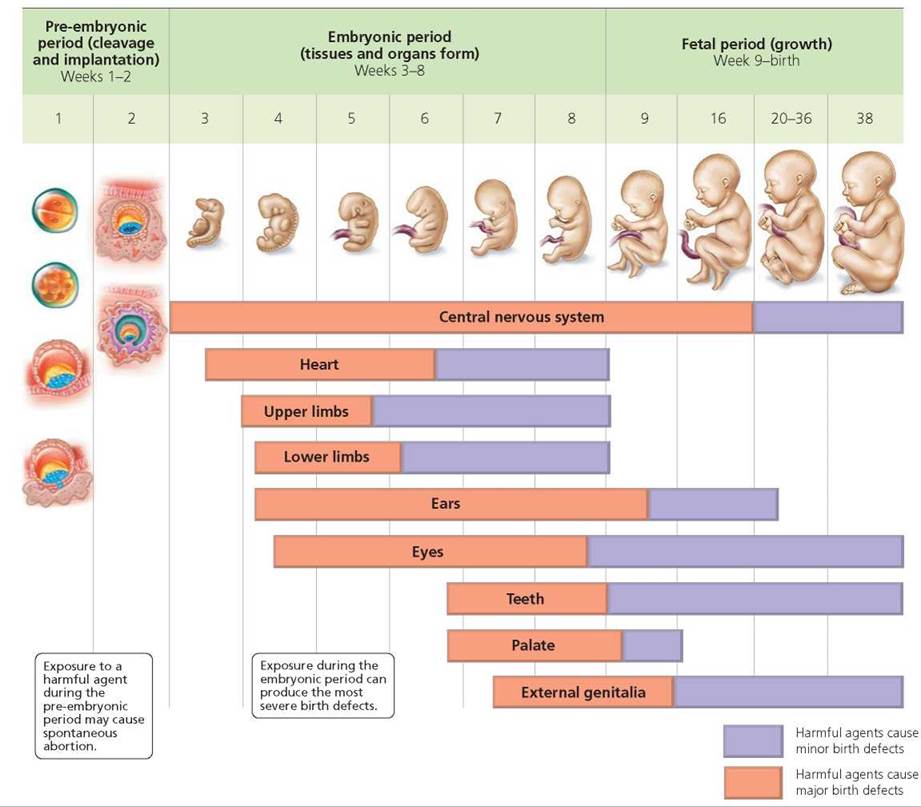
FIGURE 18.16. Critical periods in development
We have said that all organs and organ systems develop primarily during the embryonic period. Nevertheless, different organs develop at somewhat different times and rates. Such variation results in each organ system having a critical period in which it is most susceptible to disruption by environmental agents. The CNS has a critical period extending from week 3 through week 16. During this time, the developing nervous system is highly sensitive, and disruptive agents can have major consequences. By comparison, exposure to such agents after week 16 tends to induce relatively minor defects. The CNS has the longest critical period. It extends beyond the embryonic period and into the fetal period. This long critical period may explain why the brain is the organ in which defects in structure and function are most commonly found. The critical period for development of the upper and lower limbs is relatively short, extending from about week 4 to week 6. It is during this short window of time that certain environmental agents cause major abnormalities such as tiny limbs or complete absence of limbs.
Another look at Figure 18.16 reveals that exposure to harmful agents during the first 2 weeks after fertilization is not known to cause birth defects. Development during this period (the pre-embryonic period) centers on formation of structures outside the embryo, such as the extraembryonic membranes described earlier. However, the developing pre-embryo is not immune to environmental influences. Disruptive agents in the pre-embryonic period may interfere with cleavage or implantation and cause a spontaneous abortion. Chromosomal abnormalities also account for a large proportion of spontaneous abortions at this time.
What would you do?
During the late 1950s and early 1960s, the drug thalidomide was widely used in Europe as a sedative and as relief for morning sickness. Previous animal testing had indicated that thalidomide was safe; although the testing reportedly did not include pregnant animals. Unfortunately, in humans, the drug caused major defects of the limbs such as complete absence of limbs or abnormally small limbs lacking elbows or knees. Thalidomide also caused other abnormalities, some of which caused death shortly after birth. About 12,000 infants whose mothers had taken thalidomide were affected. In later years, thalidomide was found to be an effective treatment for conditions such as leprosy and cancer of the bone marrow as well as appearing to relieve some of the conditions associated with HIV infection. Today, thalidomide is available by prescription in the United States and is sold over the counter in South America, where “thalidomide babies” are still being born. Should a drug known to cause severe birth defects be made available for treatment of seriously ill adults? How can we ensure that pregnant women do not mistakenly take thalidomide? If you were responsible for developing a policy regarding the use of thalidomide, what would you do?
Milk Production by Mammary Glands
Lactation is the production and ejection of milk from the mammary glands. Recall from Chapter 10 that prolactin from the anterior pituitary gland promotes milk production. Recall also that oxytocin from the posterior pituitary gland stimulates milk ejection. The structure of the breast was described in Chapter 17.
Prolactin levels increase during pregnancy and reach their peak at birth. Milk production does not begin during pregnancy, because high levels of estrogen and progesterone inhibit the actions of prolactin. Thus, prolactin cannot initiate milk production until birth, when levels of estrogen and progesterone decline. Milk usually is available about 3 days later. In the interim, infants consume colostrum, a cloudy yellowish fluid produced by the breasts that has a different composition than milk. After birth, the infant's sucking on the breast stimulates the release of prolactin, promoting and maintaining the production of milk. At weaning, when sucking ends, so, too, does milk production.
Which is best—bottle or breast? Most people today believe that breast-feeding is best for infants. Advocates of breastfeeding point out that compared with commercial formulas— many of them based on cow's milk—breast milk is more digestible and much less likely to cause an allergic reaction, constipation, or obesity in infants. In addition, maternal milk and colostrum contain antibodies and special proteins that boost the immune system. Finally, obtaining milk from a breast requires more effort than sucking on a bottle. Health professionals think this greater effort promotes optimum development of the infant's jaws, teeth, and facial muscles.
Breast-feeding also is beneficial to the mother. Nursing helps the mother's uterus return toward its prepregnant size. When oxytocin is released in response to an infant's sucking, it helps the uterus shrink. Also, because milk production is energetically expensive, breast-feeding helps with maternal weight loss in the weeks and months after giving birth. Finally, breast-feeding offers some protection against breast cancer.
Despite its benefits, breast-feeding is not for everyone. Some women simply prefer bottle-feeding, while others have difficulty breast-feeding. Still others are warned against breast-feeding because the medications they take pass into breast milk. Finally, mothers who are HIV positive should not breast-feed because the virus can be transmitted in breast milk.
Postnatal Period
The postnatal period—the period of growth and development after birth—includes several stages all of which involve biological, social, and psychological changes. We consider the following stages: infancy (from birth to 12 months), childhood (from 13 months to 12 or 13 years), adolescence (from puberty to late teens), and adulthood (generally reached by around 20 or 21 years of age).
Infancy is a time of rapid growth, though not as rapid as during prenatal development. Physical milestones reached during the first year include (in approximate chronological order) rolling over, sitting, crawling, standing, and by the end, some infants are walking. Vocalizations also change: cooing is replaced by babbling, which in turn is replaced by single words in some one year olds. An infant's responsiveness to social stimuli increases over the 12 months. Childhood is a time of continued growth during which gross and fine motor skills improve and coping skills develop. With the exception of the reproductive system, organ systems become fully functional.
Adolescence begins with puberty, the period of sexual maturation that typically occurs from 12 to 15 years in girls and from 13 to 16 years in boys. A period of rapid growth (the growth spurt) occurs in both sexes. Growth slows for a few years, and adolescence ends with the cessation of growth in the late teens or early twenties. The start of adulthood is imprecise because of the many factors—physical, behavioral, societal—that influence how people define an adult. Aging, the normal and progressive decline in the structure and function of the bodies of adults, begins only a few years after growth has ceased.
Observable characteristics of old age—graying and thinning hair, wrinkles and sagging skin, reduced muscle mass, and stooped posture—are familiar to us. However, we also grow old on the inside. Table 18.2 summarizes some of the changes to organ systems that typically occur with aging. Of course, we do not suddenly become "old." For example, bones weaken and blood vessels stiffen. Nephrons, the functional units of the kidneys, decline in number and efficiency, ultimately challenging efforts by the kidneys to maintain the balance of fluids in our bodies. Aging also strikes our nervous system and sense organs, as evidenced by our deteriorating ability to remember, see, hear, taste, and smell. Changes to body systems occur gradually, many of them beginning in middle age. For example, declines in basal metabolic rate will result in weight gain after about age 35 unless lifestyle changes are made to reduce caloric intake, increase activity, or both. Most people in their forties begin to experience a decreasing ability to focus on close objects, necessitating more frequent visits to the eye doctor and new prescriptions for lenses. Also, around age 50, our ability to hear high-frequency sounds starts to gradually decline, reflecting a slow but continual loss of hair cells in the inner ear that begins in our twenties! Even so, you might wonder why we discuss aging in a textbook geared toward college students. Our reasoning is simple; you can do a lot right now to ensure a high quality of life during old age (Figure 18.17). Also, as described in the Health Issue essay Disparities in Health and Health Care at All Life Stages, significant health and health care disparities exist among populations in the United States, so being aware of these disparities and making your health a key focus when young can lead to a much better old age.

FIGURE 18.17. Wise and informed lifestyle decisions made when young can lead to a healthy old age.
TABLE 18.2. Changes in Organ Systems as We Age
Organ System |
Some Changes That Occur with Aging |
Integumentary |
Wrinkles appear as skin becomes thinner and less elastic. |
Sweat glands decrease in number, making regulation of body temperature more challenging. |
|
Hair thins owing to death of hair follicles and turns gray as pigment-producing cells die. |
|
Skeletal |
Bones become lighter and more brittle, especially in women after menopause. |
About 7.6 cm (3 in.) of height are lost as the intervertebral disks deteriorate and the vertebrae move closer together. |
|
Joints become stiff and painful due to decreased production of synovial fluid. |
|
Muscular |
Muscle mass decreases owing to loss of muscle cells and decreased size of remaining muscle cells. |
Nervous |
Brain mass decreases. |
Movements and reflexes slow as conduction velocity of nerve fibers decreases and release of neurotransmitters slows. |
|
Hearing becomes less acute as hair cells in the inner ear are lost. |
|
Ability of the eye to focus declines as the lens of the eye stiffens. |
|
Smell and taste become less acute. |
|
Endocrine |
In women, production of estrogen and progesterone decreases with menopause. |
In men, production of testosterone decreases. |
|
In both sexes, production of growth hormone decreases. |
|
Cardiovascular |
Cardiac output decreases as walls of the heart stiffen. |
Blood pressure rises as arteries become less elastic and clogged by fatty deposits. |
|
Respiratory |
Lung capacity decreases as alveoli break down and lung tissue becomes less elastic. |
Digestive |
Basal metabolic rate declines. |
|
Ability of the liver to detoxify substances declines. |
Urinary |
In both sexes, kidney mass declines, as does the rate of filtration of the blood by nephrons. |
Particularly in women, the external urethral sphincter weakens, causing incontinence. |
|
In men, the prostate gland (part of the reproductive system) enlarges, causing painful and frequent urination. |
|
Reproductive |
In men, fewer viable sperm are produced. |
In women, ovulation and menstruation cease at menopause. |
Health Issue
Disparities in Health and Health Care at All Life Stages
Developing good health care habits as a young adult will have lasting positive impacts on the quality of your adult life, aging, and longevity. Unfortunately, significant differences exist in access to health care in the United States, and in the effectiveness of the care received, so you need to be proactive. The Centers for Disease Control and Prevention, the Institute of Medicine, and the United States Department of Health and Human Services consistently report differences in health, health care access, and outcomes of care received among certain populations, including those based on age, race, ethnicity, gender, socioeconomic status, sexual orientation, and place of residence (for example, geographic region). Here, we focus on differences related to race and ethnicity.
Some health disparities occur around birth, some in childhood, and others in adulthood. For example, when compared with non-Hispanic whites, African Americans have 2.3 times the infant mortality rate. Asthma is much more prevalent among African American children than among non-Hispanic white children, and within 5 to 24 year olds, African Americans are 4 to 6 times more likely to die from asthma than are whites. Health disparities in mid to late adulthood include chronic diseases, such as diabetes: African American and Hispanic adults are almost twice as likely as non-Hispanic white adults to develop diabetes. Further, the rates of cancer, cardiovascular disease, and HIV/AIDS are higher among minority populations than among non-Hispanic whites.
Racial and ethnic disparities extend to access to health care and the quality of health care received at all life stages. The percentage of live births in which the mother received prenatal care starting in the first trimester (months 1 to 3) was 76.1% for African Americans and 88.1% for nonHispanic whites. Hispanic children between 19 and 35 months of age are less likely than white and African American children to receive all recommended immunizations. Hispanic women over 40 are less likely than white women of this age group to receive recommended screening for breast cancer, and minorities are more likely to have late-stage cancer when the disease is diagnosed. Some health outcomes also are worse for minorities: African Americans and Hispanics with late stage diabetes are more likely than whites to have their lower legs and feet amputated. With few exceptions, the pattern is that racial and ethnic minorities have higher incidences of chronic diseases (such as asthma, diabetes, and heart disease), higher mortality rates, and lower quality health care.
Although racial and ethnic disparities in health and health care are well documented, the causes of the disparities have been challenging to identify, which makes it difficult to solve the problem. A major complication concerns the complex influences of environments—both medical and social—on health problems. How much do disparities reflect unequal treatment by health care professionals? How much do disparities reflect aspects of the social environment, such as income, diet, stress, and other aspects of lifestyle? Do disparities reflect differences in access to education needed for people to make informed choices about their own care? Most people who study health disparities suggest that improvements are necessary both within the health care system and within minority communities (for example, improving the availability of healthy food options and health care providers). Toward this end, Healthy People 2010, an initiative of the United States Department of Health and Human Services, lists eliminating health disparities as one of its two major goals (the other being to increase the quality and years of healthy life). Healthy People 2010 emphasizes prevention and builds on similar initiatives over the last few decades (Healthy People 2020 is now being developed). Backed by the best scientific information available, based on broad consultation with health agencies and organizations, and open to input from the public, the initiatives provide 10-year national health objectives that can be used by states and communities to develop programs to improve health. Also, a major goal of The Patient Protection and Affordable Care Act, which was signed into law in March of 2010, is to increase access to health care for millions of Americans and improve practices of prevention. These comprehensive, prevention-based approaches may help to eliminate disparities in health and health care.
Questions to Consider
• If you were charged with improving minority access to health care, where would you begin? Would you increase insurance coverage, availability of providers, health literacy, or something else?
• How might communication between minority patients and their providers be improved?
Possible Causes of Aging
Scientists do not agree about what causes our bodies to age. Some suggest that aging results from changes in critical body systems. For example, aging might be prompted by a decline in function of the immune system or by changing levels of certain hormones. Recall from Chapter 10 that the production of some hormones, such as growth hormone, declines as we age. Perhaps such declines induce the changes in bodily structure and function that characterize aging. Other scientists seek explanations for the causes of aging at the cellular and molecular levels.
Cessation of cell division. Ongoing cell division is necessary to replace cells that die. Without new cells, most tissues and organs could not continue to function effectively. Yet cell division appears to slow in aging animals. Cells grown in culture in the laboratory do not divide indefinitely, supporting the idea that cessation of cell division is genetically programmed. In fact, they divide only a certain number of times. The number of divisions seems to be correlated with the age of the individual who donated the cells (whether the donor is a roundworm, mouse, or human) and the life span of the particular species. Telomeres, protective pieces of DNA at the tips of chromosomes, may help cells keep track of how many times they have divided (see Chapter 21a). A tiny piece of each telomere is sliced off each time the DNA is copied before cell division. After a certain number of cell divisions, the telomere is gone, and the chromosome is no longer protected from the slicing. Chromosomes that sustain such damage can no longer participate in cell division. Cells grown in culture demonstrate this phenomenon, yet a clear link to human longevity has not been established.
Damage to DNA and other macromolecules. Other researchers postulate that highly reactive molecules known as free radicals disrupt cell processes and lead to aging. Free radicals are by-products of normal cellular activities. They have an unpaired electron and readily combine with and damage DNA, proteins, and lipids. Aging also is associated with a decline in the ability of cells to repair damaged DNA. Such a decline might lead to an accumulation of gene mutations and ultimately to a decline in cell function.
Finally, some scientists have suggested that glucose—our main source of fuel—is the culprit. Glucose changes proteins such as collagen by causing cross-linkages to form between their molecules. This shackling of protein molecules may cause the stiffening of connective tissue and heart muscle associated with aging.
Is there a primary cause of aging? Most scientists believe that aging is not caused by a single factor, but rather by several processes that interact to produce our eventual deterioration and demise.
High-Quality Old Age
The news today is full of stories about antiaging products. Aging, however, is a normal biological process that, at present, cannot be slowed, stopped, or reversed. Today, the maximum documented life span for humans is 122 years, a record recently established by Madame Jeanne Calment of France (Figure 18.18). Most of us will likely never reach her age. In fact, life expectancy for babies born in the United States today is about 77 years. While this news might depress you, especially after reading the long list of declines in organ systems outlined in Table 18.2, the good news is that we can stave off much of the disease and disability associated with aging. The human life span appears to be determined by genes, environment, and lifestyle. Physicians are now able to treat many conditions of old age. For example, worn-out hip or knee joints can be replaced with artificial joints. The clouding of vision caused by cataracts is now treated readily by replacing the old lens of the eye with an artificial lens. Drugs are being tested that may break the cross-links that glucose creates in proteins and thereby restore elasticity to our arteries. In addition, research is under way to determine if antioxidants (substances that inhibit the formation of free radicals) are effective in delaying certain aspects of aging.

FIGURE 18.18. The longest documented human life span is 122years; this record was established by Madame Jeanne Calment, shown here on her last birthday. She died in 1997.
Lifestyle is the factor over which we have the most personal control. Some components of a healthy lifestyle include proper nutrition, plenty of exercise and sleep, no smoking, and routine medical checkups. Indeed, the lifestyle choices we make when young can delay some aspects of aging. For example, we can delay wrinkling and aging of the skin by avoiding excessive exposure to the sun. The importance of a healthy lifestyle continues into our later years, when exercise, social and intellectual stimulation, and good nutrition can help to prevent the physical and mental declines associated with aging. Although it is never too late to change lifestyle habits, the earlier we begin to develop healthy habits, the better. Finally, aging is not only about declining organ systems; it has positive aspects as well. Not the least of these, as the years pass, are the gains we make in experience, perspective, and wisdom.
Looking ahead
In this chapter, we described the major milestones of human development, with a particular focus on the prenatal period. In the next chapter we consider the developmental milestones of early childhood and how careful monitoring of these milestones can be used to diagnose autism spectrum disorders. We also describe the symptoms and possible causes of these lifelong disorders as well as available treatments.
Highlighting the Concepts
Periods of Development in Human Life (p. 362)
• The period of development before birth is the prenatal period. The period of development after birth is the postnatal period.
Prenatal Period (pp. 362-373)
• The prenatal period can be subdivided into the pre-embryonic period (from fertilization through the second week), embryonic period (from week 3 through week 8), and fetal period (from the ninth week until birth).
• The pre-embryonic period is characterized by formation and implantation of the blastocyst. The extraembryonic membranes and placenta also begin to form at this time. The embryonic period is characterized by gastrulation and the formation of organs and organ systems. The fetal period is a period of intense growth.
• Fertilization is the union of an egg and a sperm to form a single cell called the zygote.
• Cleavage is a rapid series of mitotic cell divisions that transforms the zygote into a morula (a solid ball of cells) and then a blastocyst (a hollow ball of cells with a fluid-filled cavity). Within the blastocyst are the inner cell mass, which will become the embryo and some of the extraembryonic membranes, and the trophoblast, which will form part of the placenta.
• Toward the end of the first week, the blastocyst begins implantation, the process by which the embryo becomes embedded in the endometrium of the uterus.
• Extraembryonic membranes—the amnion, yolk sac, chorion, and allantois—lie outside the embryo and begin to form during the second or third week after fertilization.
• The placenta, formed from the chorion of the embryo and the endometrium of the mother, is the organ that delivers nutrients and oxygen to the embryo (and later the fetus) and carries wastes away. The placenta also produces hormones such as estrogen, progesterone, and human chorionic gonadotropin (HCG).
• Gastrulation includes the cell movements by which the primary germ layers—ectoderm, mesoderm, and endoderm—are established.
• Ectoderm forms the nervous system and the epidermis and its derivatives (hair, nails, oil glands, sweat glands, and mammary glands). Mesoderm forms muscle, bone, other connective tissue, and organs such as the heart, ovaries, and testes. Endoderm forms organs such as the liver, some endocrine glands, and the lining of the urinary, respiratory, and digestive tracts.
• Neurulation is the process by which the neural tube forms from ectoderm. The anterior portion of the neural tube forms the brain, and the posterior portion forms the spinal cord.
• Somites, blocks of mesoderm organized alongside the neural tube, eventually form vertebrae, connective tissue, and skeletal muscles of the neck and trunk.
• The gender of a human embryo is determined at fertilization by the type of sperm (X-bearing or Y-bearing) that fertilizes the egg (which is X-bearing). An XX zygote will develop into a female, and an XY zygote into a male. Internal reproductive organs and external anatomy begin to differentiate about 6 weeks after fertilization, when a region on the Y chromosome initiates development of testes in male embryos. Absence of the Y chromosome results in female development.
• Growth during the fetal period is extremely rapid. The growth rate of the head slows relative to that of other parts of the body, producing changes in the way the body is proportioned. Such change in the relative growth rates of different parts of the body is called allometric growth.
• Fetal circulation differs from circulation after birth in that most blood is shunted through temporary vessels or openings past organs such as the lungs and liver, which do not yet perform their postnatal functions. At birth, when organs begin their postnatal functions, the bypasses of fetal circulation begin to close.
Birth (pp. 373-375)
• Labor is the process by which the fetus is expelled from the uterus and moved through the vagina into the outside world. Parturition (birth) usually occurs about 38 weeks after fertilization and marks the transition from the prenatal period of development to the postnatal period.
Birth Defects (pp. 375-376)
• Birth defects are developmental defects present at birth. Such defects may be caused by genetic factors or environmental agents. Disruptive agents have their greatest effects on the developing embryo during periods of rapid differentiation, when organs and organ systems are forming.
Milk Production by Mammary Glands (pp. 376-377)
• Lactation is the production and ejection of milk from the mammary glands. At birth, when levels of estrogen and progesterone drop, prolactin initiates milk production. Oxytocin causes milk ejection.
Postnatal Period (pp. 377-380)
• Stages in postnatal development include infancy, childhood, adolescence, and adulthood. Growth stops in adulthood, and a few years later aging begins.
• Aging is the progressive decline in the structure and function of the body. Changes occur in all organ systems. Possible causes of aging include genetically programmed cessation of cell division; damage to DNA, protein, and lipids caused by free radicals; the inability of cells to repair damaged DNA; alteration of proteins by glucose such that cross-linkages form and produce a stiffening in tissues; and declines in the functioning of key organ systems.
Reviewing the Concepts
1. List the three stages of prenatal development. What ages and major developmental milestones are associated with each stage? pp. 362-373
2. What prompts the destabilization of the acrosome on the head of a sperm? p. 364
3. How is fertilization of an oocyte by more than one sperm prevented? p. 364
4. Describe implantation. Where does it usually occur? What happens if it occurs elsewhere? pp. 366-367
5. What are the functions of the four extraembryonic membranes? p. 367
6. What is the placenta, and how does it form? Explain how nutrients, oxygen, and wastes are exchanged between fetal and maternal blood. pp. 367-368
7. What is gastrulation? What tissues and organs are formed from each of the three primary germ layers? p. 369
8. Describe formation of the central nervous system. pp. 369-370
9. How is the gender of a human embryo determined? p. 370
10. How does fetal circulation differ from circulation after birth? pp. 371-373
11. What are the three stages of labor? p. 374
12. Explain the relationship between the effects of environmental agents that disrupt development and embryonic age. pp. 375-376
13. When does milk production by a mother's breasts begin? What explains the timing? p. 376
14. What are some possible causes of aging? What can you do today to improve the quality of your old age? pp. 378-380
15. Fertilization
a. typically occurs in the uterus.
b. often involves more than one sperm uniting with an egg.
c. is the process by which the zygote becomes embedded in the uterine lining.
d. typically occurs in an oviduct.
16. The yolk sac
a. is a primary source of nourishment for human embryos.
b. is the embryo's major contribution to the placenta.
c. contains primordial germ cells.
d. encloses the embryo in a fluid-filled sac.
17. Which of the following does not characterize the placenta?
a. Produces estrogen, progesterone, and human chorionic gonadotropin
b. Is the site where fetal and maternal blood directly mix
c. Usually forms in upper portions of the uterus
d. Is expelled after birth
18. Neurulation
a. is the process by which the neural tube forms.
b. is the process by which the notochord forms.
c. occurs just before gastrulation.
d. involves endoderm.
19. The fetal period
a. is the time when tissues and organs form.
b. is characterized by rapid growth.
c. runs from week 2 to week 8.
d. is when exposure to harmful environmental agents is most likely to produce major birth defects.
20. Which of the following statements regarding childbirth is true?
a. Most babies are born feet first.
b. Delivery of the baby follows delivery of the placenta.
c. Full-term babies are those born at 32 weeks gestation.
d. Infants born prematurely with very low birth weight require intensive care after birth.
21. Within the blastocyst, the _____ becomes the embryo proper and some extraembryonic membranes, and the _____ becomes part of the placenta.
22. Soon after fertilization, the zygote undergoes _____, a series of rapid mitotic divisions without an increase in overall size.
23. The placenta forms from the _____ of the embryo and a portion of the _____ of the mother.
24. _____ is the process by which primary germ layers form.
25. The hormone _____ stimulates milk production, and the hormone _____ stimulates milk ejection.
26. _____ are highly reactive molecules, produced by normal cellular metabolism, that damage DNA and may lead to aging.
Applying the Concepts
1. Tanya is a few weeks pregnant and is experiencing severe abdominal pain on the right side. She wonders whether the pelvic inflammatory disease she had 2 years ago could have affected her pregnancy. What condition might Tanya have? What is typically done for women with this condition?
2. Juan was described as a "blue baby" at birth and underwent corrective surgery. What did the surgeons correct, and why?
3. Why might exposure to thalidomide, a drug known to disrupt development of the limbs, during weeks 4 to 6 of gestation produce more severe birth defects than would a similar level of exposure toward the end of gestation?
4. Dora is expecting her baby in about a month. She is deciding whether to breast-feed or bottle-feed. You are her physician. First describe how milk is produced and ejected from the mammary glands. Then, describe the advantages of breast-feeding, and explain the circumstances in which you would recommend bottle-feeding to Dora instead.
5. Your grandmother is taking vitamin E and explains to you that it is a potential antioxidant. What is an antioxidant, and what "condition" is she hoping to treat? Is the vitamin E likely to work?
Becoming Information Literate
Human growth hormone (HGH) is often touted as having antiaging effects. Prepare a position paper that recommends the use of HGH for this purpose. Use at least three reliable sources (books, journals, or websites). List each source you considered, and explain why you chose the three sources you used.