Biology of Humans
19a. Stem Cells—A Repair Kit for the Body
In the previous chapter, we considered cell division for growth and repair. In this chapter, we learn that, unlike most cells, stem cells are able to divide continuously and without becoming a specialized cell type. We will consider where they are found and the promises they may hold for the future.
Stem Cells: Unspecialized Cells
Imagine growing new heart cells to repair damage from a heart attack. Think about curing Parkinson's disease by restoring the dopamine-producing neurons that are lacking in such patients. What about generating insulin-producing pancreatic islet cells to cure diabetes? Can we find a cure for multiple sclerosis, Alzheimer's, Lou Gehrig's disease, and spinal cord injuries? How? These things may be possible by using a repair kit for the body, stem cells—cells that continually divide and retain the ability to develop into many types of cells—to produce the needed type of cell.
The common thread among these disorders is that they are caused by too little cell division or defective cell function. When there is too little cell division, the body cannot repair damaged tissue, such as neurons to repair spinal cord injury. Parkinson's disease, which is characterized by a lack of neurons that produce the neurotransmitter dopamine, and hemophilia, which is characterized by a lack of production of certain blood clotting factors by liver cells, are caused by defective cell function. If the damaged or defective cells could be replaced by healthy, functional cells, the disorders would be cured. Stem cells may be a source of functional cells to bring about those cures.
Most of the trillions of cells in your body have become specialized to perform a particular job. Muscle cells are specialized to contract, and neurons are specialized to conduct nerve impulses.
The specialization of cells during embryonic development is directed by biological cues in each cell's immediate environment. The cues are usually provided by neighboring cells and include growth factors, surface proteins, salts, and contact with other cells. Because all cells within the same body have the same genetic makeup, the specialization of different cells to perform different functions means that particular genes in each type of cell have been turned on or off. Recall from the previous chapter that specialized cells usually do not divide again.
· Stem cell research is advancing rapidly.
Stem cells, in contrast, are relatively unspecialized cells that divide continually, creating a pool of undifferentiated cells for possible use. If stem cells are given the correct signals—exposure to a particular growth factor or hormone, for example—they can be coaxed into differentiating into a particular specialized cell type. The specialized cell types can then be used to treat diseases or regenerate injured tissues. As we will see, stem cells from different sources differ in the variety of specialized cell types they can develop into.
Stem cells are categorized by the degree of flexibility in their developmental path (Figure 19a.1). A fertilized egg can develop into all the types of cells in the body and is therefore considered to be totipotent. After about a week of development, the embryo is a source of embryonic stem cells, which can develop into nearly every type of cell. Because the developmental path of embryonic stem cells is so flexible, they are described as being pluripotent.1 In an adult, some stem cells remain able to differentiate into several types of cells and are described as being multipotent. Still other adult stem cells are described as unipotent, because they can differentiate into only one type of cell. Thus, embryonic stem cells are more versatile than are adult stem cells.
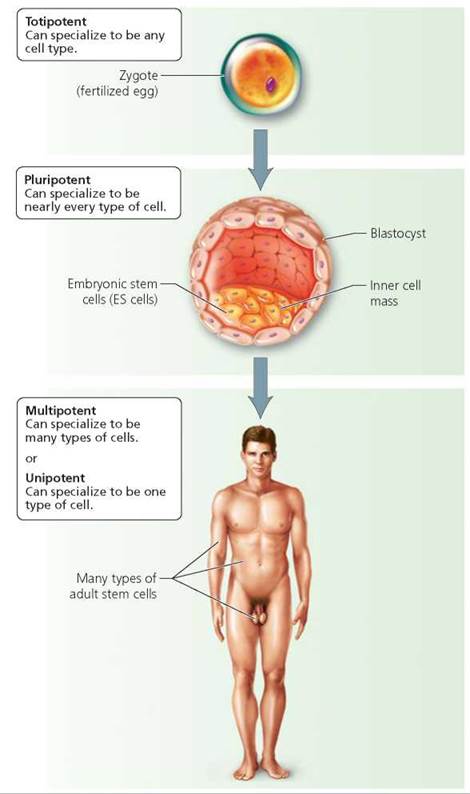
FIGURE 19a.1. Stem cells at different stages in human development
The source of stem cells used for therapeutic purposes influences the kinds of cells they can become as well as the likelihood of the stem cells being rejected in the body of the person being treated. Each person's cells have self markers (MHC markers) identifying the cells as belonging in that body (see Chapter 13). These self markers are genetically determined, so the self markers of relative's cells will be more similar than those of unrelated individuals. Cells that are not recognized as belonging in the body, because of the difference in the self markers, are usually attacked and killed by cells of the body's defense system. The self markers develop during prenatal development and early childhood. Thus, stem cells from an embryo or fetus have less developed self markers and are less likely to be rejected than are adult stem cells. Likewise, adult stem cells from the person being treated or a close relative are less likely to be rejected than are cells from an unrelated person.
Sources of Human Stem Cells
There are several sources of stem cells, including certain adult tissues, umbilical cord blood, and early embryos.
Adult Stem Cells: Unipotent and Multipotent
Adults have stem cells, but they are more difficult to locate than those in an embryo are. Waiting in the bone marrow, brain, skin, liver, and other organs, adult stem cells remain ready to generate new cells for repair or replacement of old ones in many parts of the body. Diseased or dying cells provide biological cues that summon stem cells and prompt them to differentiate into the needed type of cell. When a stem cell divides in an adult, one cell usually specializes to be a particular type of cell and the other remains a stem cell (Figure 19a.2).
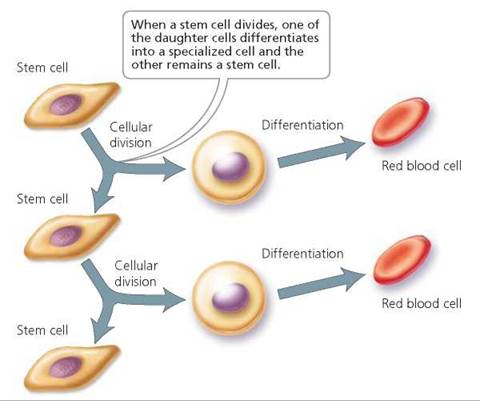
FIGURE 19a.2. Two important characteristics of stem cells are that they divide continually and that their daughter cells can differentiate into one or more specialized stem cells. Here, a blood stem cell divides, producing one daughter cell that becomes specialized to perform a function and another daughter cell that remains a stem cell to replenish the stem cell supply.
Adult stem cells do not seem to be as versatile as stem cells from an embryo. Whereas embryonic stem cells can transform into any cell type in the body, adult stem cells usually form one type of cell or form cells of a particular lineage only. However, we now know that adult stem cells are more versatile than was once believed.
Umbilical Cord Stem Cells: Multipotent
Blood from the umbilical cord is also a good source of stem cells. Stem cells do not settle into an infant's bone marrow until a few days after birth. Before that, during fetal development, they circulate in the bloodstream and therefore travel through vessels in the umbilical cord when circulating to and from the placenta. After the baby is born, it no longer needs its umbilical cord, so the cord is usually discarded. However, an increasing number of parents are choosing to have the blood stem cells from the cord saved in a storage bank.
Most of the stem cells in cord blood are blood stem cells that give rise to red blood cells, white blood cells, and platelets. Within the body, blood stem cells produce about 260 billion new cells (about an ounce of blood) every day. However, umbilical cord blood also contains some stem cells that can turn into other kinds of cells—bone, cartilage, heart muscle, nerve tissue, and liver tissue.
Stem cells from umbilical cord blood have some advantages over adult blood stem cells taken from bone marrow. Blood stem cells can be harvested from bone marrow, but the procedure is done in a hospital under general anesthesia. Stem cells are more easily extracted from umbilical cord blood because the cord is removed from the infant's body. Another advantage of taking stem cells from cord blood is that they are less likely to be rejected by the recipient than are adult stem cells from bone marrow. The self markers on the cord blood stem cells have not yet fully developed, so the match does not have to be as close as with adult stem cells from bone marrow. Finally, stem cells from cord blood are less likely to carry infections.
Because blood stem cells are able to produce an endless supply of blood cells, they hold promise for treating a host of blood conditions. For instance, some patients with the inherited disorders sickle-cell anemia and beta-thalassemia, in which abnormal forms of hemoglobin are produced, have already been treated successfully with stem cell therapy. Stem cells that produce normal hemoglobin can be transplanted into a person with the inherited anemia. Normal red blood cells then begin replacing the abnormal ones. Leukemia patients have also benefitted from umbilical cord stem cells. Traditionally, leukemia has been treated with bone marrow transplants from family members or from donors from the National Marrow Donor Program who have close tissue types. However, most patients in need of a bone marrow transplant do not find donors with matching tissue types. Because the tissue markers on umbilical cord stem cells are immature, the cells are less likely to be rejected by the recipient.
Most of the medical benefits that may be achieved using umbilical cord blood have yet to be realized. Nonetheless, there are already umbilical cord blood banks that store cord blood from newborn babies. Some parents pay a setup fee and an annual service fee to store their baby's cord blood just in case the child needs that blood or those stem cells some day. Government-owned banks collect and store thousands of samples of cord blood in hopes of having a tissue match for most individuals in the population. Researchers have developed ways to increase the number of umbilical cord blood stem cells by growing them in the laboratory.
The existence of umbilical cord blood banks raises certain social issues. Should blood stored in private blood banks be reserved only for the possible future need of the donor, or should it be made available for anyone in need whose tissue type matches? Should cord blood banks be privately owned or funded by the government? These are just a few of the many questions remaining to be addressed.
Embryonic Stem Cells: Pluripotent
Embryonic stem cells can divide continually and specialize into nearly any cell type. They come from several sources.
Unused embryos from fertility clinics. Most of the stem cells used for current research come from embryos that were created for reproductive purposes but were not used. Only a few days old, these embryos left in fertility clinics were destined for destruction. When the early embryo is about 6 to 7 days old, it is a rich source of stem cells because the cells it consists of are able to produce all the cells of the new individual. When stem cells are extracted from an early embryo, the embryo is about the size of the head of a pin and is called a blastocyst (see Chapter 18; see also Figure 19a.3). It is a sphere of cells containing a cluster of about 20 to 30 cells, called the inner cell mass, adhering to one side of the sphere. The stem cells make up the inner cell mass. Embryonic stem cells are easy to harvest: the cells of the inner cell mass are extracted and cultured in the laboratory. They will continue to divide for years, creating a stem cell line.

FIGURE 19a.3. Embryonic stem cells come from the inner cell mass, a cluster of 20 to 30 cells, from a 6- to 7-day-old embryo. At this age, the embryo is about the size of a head of a pin.
Some people object to the use of embryonic stem cells because removing the stem cells destroys such an embryo, putting an end to its ability to become a fetus. The controversy has extended into the political realm. In August 2001, President George W. Bush announced that embryonic stem cell research would not receive federal funding under his administration unless the research was limited to using only previously existing stem cell lines. (Use of federal funds under President Bush also prohibited the creation of human embryos to be used in research.) For many years, only 21 embryonic stem cell lines were approved for research, which meant that the genetic diversity of the allowed lines was limited. In 2009, President Obama eliminated the restriction that federally funded stem cell research be only on lines established before August 2001. However, he also created stricter ethical standards for creating new lines—the embryos must be donated with informed consent of the parents. However, in 2010, a court ruling blocked Federal funding of all embryonic stem cell research. Even if this ruling is eventually reversed, it is sure to affect this research.
What would you do?
If you were a parent with an embryo stored in a fertility clinic, would you consent to using that embryo for stem cell research?
Somatic cell nuclear transfer. In somatic cell nuclear transfer (SCNT), the nucleus from a somatic body cell is transferred to an egg from which the egg nucleus has been removed. The newly created cell is then stimulated to begin embryonic development. All the cells of the embryo are genetically identical. Thus, somatic cell nuclear transfer is a form of cloning— producing genetically identical copies of a cell or organism.
Dolly, born on July 5, 1996, was the first sheep—indeed, the first mammal—to be cloned from an adult cell (Figure 19a.4). She was created using somatic cell nuclear transfer, illustrated in Figure 19a.5. In Dolly's case, the nucleus of a mammary cell from an adult sheep's udder was placed into an egg from which the nucleus had been removed. (Dolly was named after Dolly Parton, a country singer known for her mammary "cells" as well as for her voice.) The goal of Dr. Ian Wilmut, the Scotsman whose work resulted in Dolly, was to develop techniques that would eventually lead to the production of animals that could be used as factories for manufacturing proteins, such as hormones, that would be beneficial to humans. Cloning could make genetic engineering (discussed in Chapter 21) more efficient. The ability to clone an adult would mean a scientist would need to engineer an animal with a particular desired trait only once. Then, when the animal was old enough for scientists to be sure it had the desired trait, the animal could be reproduced exactly, in multiple copies. Cloning from an adult instead of from an embryo is advantageous because the scientist can be certain before cloning that the animal has the desired traits. In other words, "What you see is what you get." Since Dolly's birth, scientists have cloned several kinds of animals using the technique of somatic cell nuclear transfer.

FIGURE 19a.4. Dolly, the first animal cloned from an adult cell, was created using a technique called somatic cell nuclear transfer.
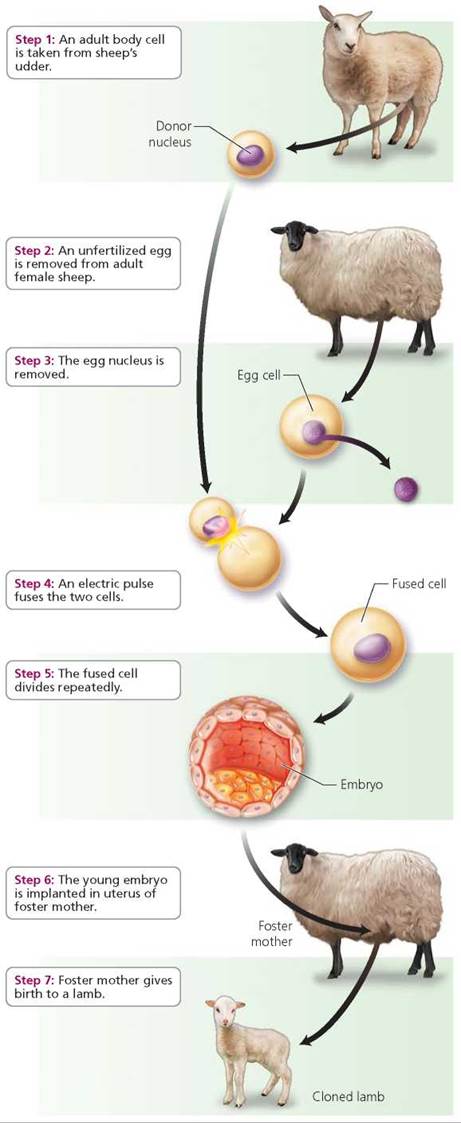
FIGURE 19a.5. Somatic cell nuclear transfer used for reproductive cloning
Discussions about cloning often become heated when the possibility of human cloning is raised. In these discussions, a distinction is often drawn between reproductive cloning and therapeutic cloning. Therapeutic cloning would produce a clone of "replacement cells" having the same genetic makeup as a given patient so that they could be used in the patient's medical therapy without concern of being rejected by the immune system. The cells produced would be embryonic stem cells created using somatic cell nuclear transfer, the technique used to create Dolly. A cell would be taken from the patient, and its nucleus would be put into a donor egg cell from which the original nucleus had been removed. The newly created cell would then begin developing into an embryo (Figure 19a.6). (If the patient's disease were genetic, a normal form of the relevant gene would be substituted for the defective gene before the nucleus was put in the enucleated egg. Replacement of a specific gene has been accomplished in mice but has not yet been done in humans.)
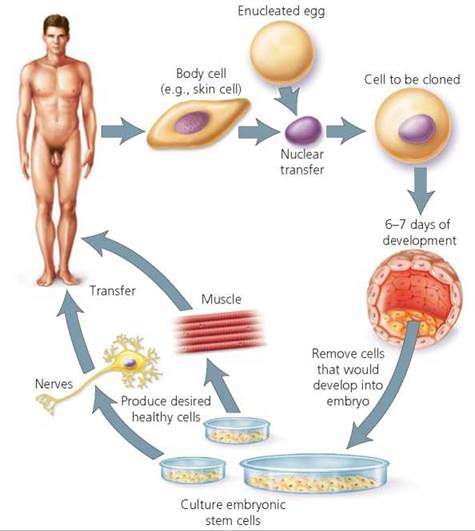
FIGURE 19a.6. Potential method for human therapeutic cloning using somatic cell nuclear transfer
Why is the nucleus removed from the donor egg?
The egg nucleus is removed so the only genetic information directing development is from the adult cell nucleus. The donor nucleus has genes for the desired trait.
Once an embryo is created by somatic cell nuclear transfer, stem cells could be removed and treated with growth factors that make them develop into the type of cells needed for treatment. The stem cells could then be transplanted into the patient to replace faulty cells. For example, neurons might be transplanted to cure Parkinson's disease or to repair spinal cord damage, and insulin-producing cells might be transplanted to cure type 1 diabetes. Because the embryonic stem cells would have the same genetic makeup as the patient, they would not be attacked as foreign by the patient's body-defense responses.
SCNT research has been hampered by a limited supply of human eggs. Some researchers have suggested using enucleated nonhuman animal eggs instead of human eggs. The human nucleus transferred to the egg would direct development. Some people consider the idea of creating an animal-human hybrid embryo to be immoral. The embryos are destroyed at the age of 14 days to ensure that such a hybrid would never fully develop. In 2008, the British Parliament voted to allow the practice of creating animal-human hybrid embryos for use in stem cell research.
In contrast to therapeutic cloning, reproductive cloning produces a new individual with a known genetic makeup. The cloning of nonhuman animals (like Dolly) is reproductive cloning. Currently, reproductive cloning is an extremely inefficient process. Hundreds of cloned embryos must be used to obtain one newborn mammal. When cloned embryos do proceed to develop into a new individual, they grow faster and larger than normal. There is also a suspicion that cloned mammals age prematurely. For example, there are indications that Dolly was genetically older than her birth age: one is that Dolly had arthritis, a painful joint condition usually found only in older animals. For reasons like these, experts agree that scientists are still far from being able to produce a cloned human baby.
If reproductive cloning of humans does become possible someday, what might this technology mean to our society? How would it affect human life? One thing it would not do is give us the power to create an exact copy of a specific person. Environment and genes (also often referred to as nurture and nature) determine personality characteristics. Although cloning could replicate the exact genetic makeup of a specific person, there would be no way to replicate all the experiences in the life of the person being cloned; so the clone would be a different person from the original one. Thus we could not replace a loved one who has died or achieve immortality of our own in the body of a clone. If such technology is ever successful, however, we could produce an identical twin of a person.
Induced Pluripotent Stem Cells
Suppose one could turn back the clock and stimulate an adult cell to revert to a pluripotent stem cell. In 2006, researchers announced that they had done just that—they caused adult mouse skin cells to revert to pluripotent stem cells that could differentiate into any type of tissue. A short time later, in 2007, scientists created induced pluripotent stem cells (iPSCs) using human skin cells. Both groups of researchers used retroviruses to insert four genes into the skin cells. This is an exciting breakthrough—it creates pluripotent stem cells without the need to destroy an embryo, and it could produce patient- specific cell lines without the possibility of rejection. Two concerns with pluripotent stem cells created in this way are that two of the inserted genes are cancer-producing genes and that using retroviruses to deliver the genes carries a risk. Retroviruses integrate themselves directly into the cell's DNA, which allows the genes delivered by the virus to become permanently active in the cell. However, depending on where the virus inserts itself, this incorporation can cause the cell to become cancerous.
Researchers have addressed these concerns. They created pluripotent stem cells from adult skin cells using two genes that are not linked to cancer and then used a cocktail of drugs to revert the skin cells to pluripotent stem cells. Adenoviruses, which normally cause the human cold, have been modified to serve as the gene-delivery system. These viruses persist in the cell for only a short time.
This research brings stem cells created from adult cells closer to clinical use. Researchers have used iPSCs to rescue vision in rats. The rats used in these experiments have a gene mutation that causes blindness by killing the photoreceptor cells. The research team surgically inserted iPSCs of retinal epithelial cells into the retina of rats before the photoreceptors degenerated, and vision was retained. But, don't expect human cures in the near future; it will take years before iPSCs can be put to practical use in humans.
Potential Uses for Stem Cells
The use of stem cells is just beginning to blossom. Let's consider some of their potential uses.
Replacement for Damaged Cells
As of this writing, there are no established stem cell therapies in the United States—except for those that use cells extracted from bone marrow, circulating blood, or umbilical cord blood. There are, however, more than 2000 clinical trials in progress or looking for recruits. For example, clinical trials are underway to determine the effectiveness of umbilical cord stem cells to repair and replace nerve cells in children with cerebral palsy, a condition in which neurons are damaged at birth. Certain other countries, China for example, are offering stem cell treatments. Although these treatments are unproven by U.S. scientific standards, some people are traveling abroad for treatment. To date, most of the studies on stem cells as a source of replacement cells or as factories to produce proteins or hormones that the body is not making have been done on nonhuman animals. Insulin-producing stem cells have eliminated symptoms of type 1 diabetes in mice. In rats, bone marrow stem cells move toward heart muscle with damage similar to that occurring in a heart attack. In the damaged heart muscle, stem cells promote heart muscle strength.
Growing New Organs
Regenerative medicine is still in its infancy, but it is growing rapidly. Its aim is to rebuild or repair damaged organs by coaxing stem cells to grow and fill in tissue scaffolds that were engineered in the laboratory. In 2004, a German man had his jaw removed because of cancer. Doctors created a mesh mold in the shape of the jawbone that had been removed, and they implanted the mold into the muscle below the man's shoulder blade, along with some cow-derived bone mineral, a growth factor to stimulate bone growth, and blood extracted from the man's bone marrow, which contained stem cells (Figure 19a.7). The mold was left in place on the man's shoulder until bone scans showed that new bone had formed around the mold. The new jaw then was removed, along with some muscle and blood vessels, and implanted in the correct location. Four weeks later, the man could chew solid food—a bratwurst sandwich!
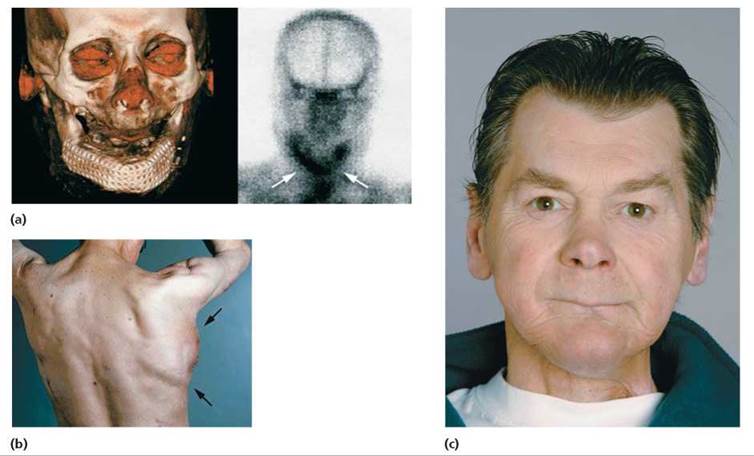
FIGURE 19a.7. Adult stem cells from bone marrow were used to grow a new jaw to replace one that had been removed because of cancer, (a) A mold in the shape of the missing jaw was created out of mesh and seeded with bone marrow cells and bone growth factor, (b) The mold was placed in the muscle of the man’s back until bone filled the mold, (c) The new jaw was then implanted to replace the missing jaw.
Since then other organs have been grown and transplanted. Doctors have grown new urinary bladders for patients using the patients' own cells. In those cases, doctors extracted muscle and bladder cells from the patient and grew them in a petri dish. As the cells formed layers of tissue, the tissue was shaped into new bladders, which were then implanted into the patients. Within a few weeks, the bladders grew to normal size and performed the required functions. An adult woman and a 10-year-old boy have each received a new windpipe (trachea) built from their own stem cells. In each case, doctors removed cells from a cadaver's trachea, a procedure that reduces the risk of rejection by the recipient. This cell-less trachea served as a scaffold on which a new trachea would be built. Researchers then seeded the trachea with two types of stem cells—epithelial cells and cartilage cells—from the patient. The new trachea grew from the patient's cells and was, therefore, not rejected.
An exciting application of regenerative medicine is "regrowing" limbs of wounded soldiers returning from Afghanistan (Figure 19a.8). Many soldiers are so badly mutilated by explosives that limbs must be amputated. Now there is hope that some limbs can be saved. The secret is an application of a powder created from pig bladders, which is nick-named magic "pixie dust." The pig bladders contain an extracellular matrix that contains the protein collagen. Researchers think that the powder works by attracting stem cells in the body and giving these cells the chemical signals to make new tissues. The new limbs have all the appropriate tissue types, including skin, muscle, and nerve.

FIGURE 19a.8. Corporal Isais Hernandez' leg was severely injured by a mortar round. New tissue was grown in the wound using a powder created from pulverized pig bladders.
Stop and think
Why would chemical signals be necessary for the development of new tissues?
Testing New Drugs
Before a new drug can go to clinical trials, it must be shown to be safe for animals and human cells cultured in petri dishes. The problem is that not all tissues will survive in tissue culture. For example, heart cells, brain cells, and liver cells do not. Stems cells may be a way to test new drugs for safety and effectiveness before testing them on humans.
Researchers are also using iPSCs to create cells that display the characteristics of cells with a particular disease. Cell cultures of these cells could then be used to study the nature of the disease or to develop drugs to treat the disease. Skin samples from Alzheimer's patients are being collected with an interest in converting them into pluripotent stem cells for such purposes.
___________________________________________________
1 Totipotent cells can give rise to all cells of the body and those of all four extraembryonic membranes (see Chapter 18). Pluripotent cells can give rise to all cells of the body but cannot give rise to cells of the extraembryonic membrane that will contribute to the placenta.