THE LIVING WORLD
Unit two. The Living Cell
3.5. Lipids
For long-term energy storage, organisms usually convert glucose into fats, another kind of storage molecule that contains more energy-rich C-H bonds than carbohydrates. Fats and all other biological molecules that are not soluble in water but soluble in oil are called lipids. Lipids are insoluble in water not because they are long chains like starches but rather because they are nonpolar. In water, fat molecules cluster together because they cannot form hydrogen bonds with water molecules. This is why oil forms into a layer on top of water when the two substances are mixed.
Fats
Fat molecules are lipids composed of two kinds of subunits: fatty acids (the gray boxed structures in figure 3.15a) and glycerol (the orange boxed structure). A fatty acid is a long chain of carbon and hydrogen atoms (called a hydrocarbon) ending in a carboxyl (—COOH) group. The three carbons of glycerol form the backbone to which three fatty acids are attached in the dehydration reaction that forms the fat molecule. That is why the carboxyl groups of the fatty acids in figure 3.15a are not apparent, because they are involved in bonds with glycerol. Because it contains three fatty acids, the resulting fat molecule is sometimes called a triacylglycerol, or triglyceride.
Fatty acids with all internal carbon atoms forming covalent bonds with two hydrogen atoms contain the maximum number of hydrogen atoms. Fats composed of these fatty acids are said to be saturated (figure 3.15b). Saturated fats are solid at room temperature. On the other hand, fats composed of fatty acids that have double bonds between one or more pairs of carbon atoms contain fewer than the maximum number of hydrogen atoms and are called unsaturated (figure 3.15c). The double bonds create kinks in the fatty acid tails, which usually makes the unsaturated fats liquid at room temperature. Many plant fats are unsaturated and occur as oils. Animal fats, in contrast, are often saturated and occur as hard fats. In some cases, unsaturated fats in food products may be artificially hydrogenated (industrial addition of hydrogens) to make them more saturated, extending the shelf life of these products. In some cases, the hydrogenation creates trans fats, a type of unsaturated fat in which some of the double bonds are less kinked than those in naturally occurring unsaturated fats. Eating trans fats and saturated fats may increase the risk of heart disease.
Other Types of Lipids
Organisms also contain other types of lipids that play many roles in cells in addition to energy storage. The male and female sex hormones testosterone and estradiol are lipids called steroids. Unlike the structure of fats shown in figure 3.15, the structure of steroids consists of multiple ring structures and looks somewhat like a section of chicken wire. Other important biological lipids include phospholipids, cholesterol (also a steroid), rubber, waxes, and pigments, such as the chlorophyll that makes plants green and the retinal that your eyes use to detect light (figure 3.16).
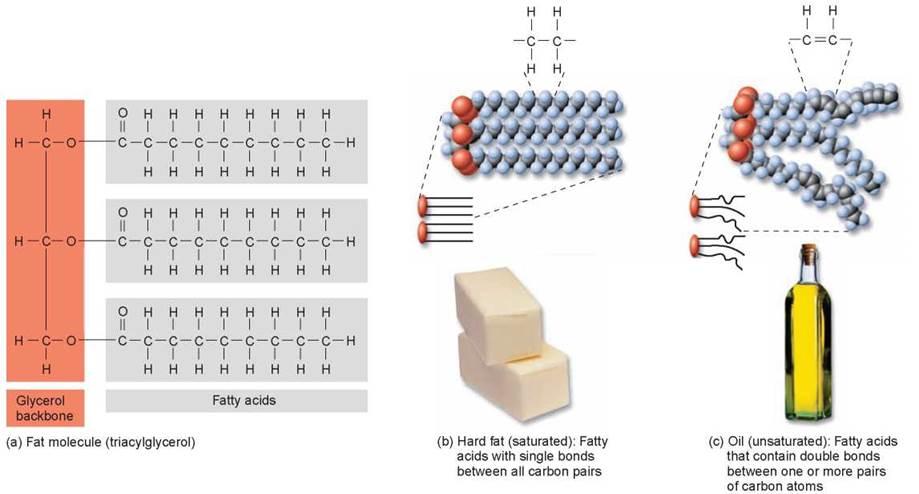
Figure 3.15. Saturated and unsaturated fats.
(a) Fat molecules each contain a three-carbon glycerol to which is attached three fatty acid tails. (b) Most animal fats are "saturated" (every carbon atom carries the maximum load of hydrogens). Their fatty acid chains fit closely together, and these triacylglycerols form immobile arrays called hard fats. (c) Most plant fats are unsaturated, which prevents close association between triacylglycerols and produces oils.
If you look at figure 3.17, you will see two types of l ipids that play key roles in the membranes that encase the cells of your body: phospholipid molecules and cholesterol. Phospholipids are modified triacylglycerol molecules, where one of the carbons in the glycerol backbone bonds to a phosphate group rather than to a third fatty acid (compare figures 3.15a and 3.16a). This gives the molecule an overall shape of a polar head and two nonpolar tails. In water, the nonpolar ends of the phospholipids group together through hydrophobic interactions, leaving the polar heads to interact with water molecules. These interactions result in the formation of two layers of molecules with the nonpolar tails pointed inside—a lipid bilayer. All biological membranes, those that surround the cell and those found inside the cell, have this arrangement. Most animal cell membranes also contain the steroid cholesterol, a lipid composed of four carbon rings (see figure 3.16b). Cholesterol helps membranes stay flexible. However, excess saturated fat intake can cause plugs of cholesterol to form in the blood vessels, which may lead to blockage, high blood pressure, stroke, or heart attack. Membranes are discussed in more detail in chapter 4.
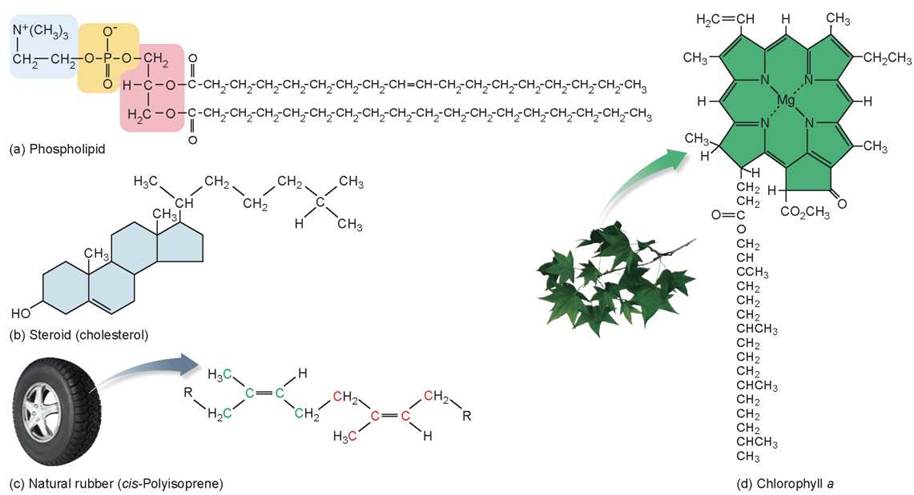
Figure 3.16. Types of lipid molecules.
(a) A phospholipid is similar to a fat molecule with the exception that one of the fatty acid tails is replaced with a polar group linked to the glycerol by a phosphate group. (b) Steroids like cholesterol are lipids with complex ring structures. (c) Natural rubber is a linear polymer of a 5-carbon unit called isoprene (two isoprene units are shown here; one unit has its carbons in green and the other in red). Rubber, harvested from the rubber tree, is used in many products such as the tires on your car. (d) Chlorophyll a has a multiringed area and a long hydrocarbon tail. Chlorophyll is the primary pigment used in photosynthesis, and it is what makes leaves green, as you will see in chapter 6.
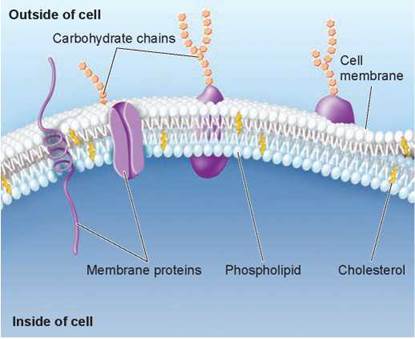
Figure 3.17. Lipids are a key component of biological membranes.
Lipids are one of the most common molecules in the human body, because the membranes of all the body's 10 trillion cells are composed largely of lipids called phospholipids. Membranes also contain cholesterol, another type of lipid.
Key Learning Outcome 3.5. Lipids are not water-soluble. Fats contain chains of fatty acid subunits that store energy. Other lipids include phospholipids, steroids, rubber, and pigment molecules.
Biology and Stavina Health
Anabolic Steroids in Sports
Among the most notorious of lipids in recent years has been the class of synthetic hormones known as anabolic steroids. Since the 1950s some athletes have been taking these chemicals to build muscle and so boost athletic performance. Both because of the intrinsic unfairness of this and because of health risks, the use of anabolic steroids has been banned in sports for decades. Controversy over their use in professional baseball has recently returned anabolic steroids to the nation's front pages.
Anabolic steroids were developed in the 1930s to treat hypogonadism, a condition in which the male testes do not produce sufficient amounts of the hormone testosterone for normal growth and sexual development. Scientists soon discovered that by slightly altering the chemical structure of testosterone, they could produce synthetic versions that facilitated the growth of skeletal muscle in laboratory animals. The word "anabolic” means growing or building.
Further tweaking reduced the added impact of these new chemicals on sexual development. More than 100 different anabolic steroids have been developed, most of which have to be injected to be effective. All require a prescription to be used legally in the United States, and all are banned in professional, college, and high school sports.
Another way to increase the body's level of testosterone is to use a chemical that is not itself anabolic but one that the body converts to testosterone. One such chemical is 4-androstenedione, more commonly called "andro.” It was first developed in the 1970s by East German scientists to try to enhance their athletes' Olympic performances. Because andro does not have the same side effects as anabolic steroids, it was legally available until 2004. It was used by Mark McGwire, but it is now banned in all sports, and possession of andro is a federal crime.
Anabolic steroids work by signalling muscle cells to make more protein. They bind to special "androgenic receptor” proteins within the cells of muscle tissue. Like jabbing these proteins with a poker, the binding prods the receptors into action, causing them to activate genes on the cell's chromosomes that produce muscle tissue proteins, triggering an increase in protein synthesis. At the same time, the anabolic steroid molecules bind to so-called "cortisol receptor” proteins in the cell, preventing these receptors from doing their job of causing protein breakdown, the muscle cell's way of suppressing inflammation and promoting the use of proteins for fuel during exercise. By increasing protein production and inhibiting the breakdown of proteins in muscle cells after workouts, anabolic steroids significantly increase the mass of an athlete's muscle tissue.
If the only effect of anabolic steroids on your body was to enhance your athletic performance by increasing your muscle mass, using them would still be wrong, for one very simple and important reason: fairness. To gain advantage in competition by concealed use of anabolic steroids—"doping”—is simply cheating. That is why these drugs are banned in sports.
The use of anabolic steroids by athletes and others is not only wrong, but also illegal, because increased muscle mass is not the only effect of using these chemicals. Among adolescents, anabolic steroids can also lead to premature termination of the adolescent growth spurt, so that for the rest of their lives, users remain shorter than they would have been without the drugs. Adolescents and adults are also affected by steroids in the following ways. Anabolic steroids can lead to potentially fatal liver cysts and liver cancer (the liver is the organ of the body that attempts to detoxify the blood), cholesterol changes and hypertension (both of which can promote heart attack and stroke), and acne. Other signs of steroid use in men include reduced size of testicles, balding, and development of breasts. In women, signs include the growth of facial hair, lowering of the voice, and cessation of menstruation.
In the fall of 2003, athletic organizations learned that some athletes were using a new performanceenhancing anabolic steroid undetectable by standard antidoping tests, tetrahydrogestrinone (THG). The use of THG was only discovered because an anonymous coach sent a spent syringe to U.S. antidoping officials. THG's chemical structure is similar to gestrinone, a drug used to treat a form of pelvic inflammation, and can be made from it by simply adding four hydrogen atoms, an easy chemical task. THG tends to break down when prepared for analysis by standard means, which explains why antidoping tests had failed to detect it. New urine tests for THG that were developed in 2004 have been used to catch several well-known sports figures. Olympic athlete Marion Jones and baseball sluggers Rafael Palmeiro, Barry Bonds, and Mark McGwire have all been involved in steroid use.

Home run slugger Barry Bonds was involved in a steroid controversy in 2006.
Author’s Corner
My Battle with Cholesterol
By a cruel twist of fate I was born loving steak: a 1 1/2 inch Porterhouse is my idea of culinary perfection. I love French fries, too, and more than anything else, Big Macs. What is cruel about my love affair with Big Macs is that a dozen years ago my doctors informed me I have high levels of cholesterol in my blood, well over the upper recommended level of 200. Because all this cholesterol in my blood tends to deposit itself along the insides of arteries, this puts me at high risk of heart attack if I don't do something about it.
Thus began my decade-long battle with cholesterol. My old friends the Big Mac and French fries pretty much became history, and steak a much more casual acquaintance. My wife and three daughters, knowing my weakness of character when it comes to things bovine, started to do more of the grocery shopping, buying lots of chicken.
Didn't do any good. After two years of Big Mac- less days and steakless nights, my cholesterol levels rose higher than before. When measured in October of 1999, my total cholesterol was 274, the highest it had ever been. When you consider that every 1% increase in level over 200 increases my risk of heart disease 2%, this report was frightening.
I called my brothers to report the bad news, only to find that they too wage the same battle. The problem, it appears, is that I and my brothers inherited a defective steak-hating gene. We suffer from hypercholesterolemia. This mouthful of a word simply means "inherited high cholesterol.” People inheriting a copy of the defective gene from one of their parents have elevated levels of cholesterol in their blood serum that dietary restriction (taking away my steaks) cannot reduce.
My frustrating on-going battle with cholesterol is not unique. Over half a million Americans suffer from hypercholesterolemia, the most frequent of all gene disorders. None of my hundreds of thousands of brothers and sisters in this war, fellow victims of this genetic quirk, are able to reduce cholesterol levels by diet and exercise alone. They, and I, must rely on modern medicine to defeat our Mendelian enemy.
Even a quick look at the biology of cholesterol suggests a likely line of attack. Cholesterol is a special kind of fat that your body uses to control the flexibility of its cell membranes and to insulate nerves. Your liver makes about 80% of the cholesterol in your body, up to 800 mg a day. You take in the rest when you eat steak and other fatty foods.
You can see why my attempting to lower total cholesterol by reducing dietary input of fat (steaks, peanut butter, fried food, chocolate, ice cream—all the good stuff) didn't get the job done. Dietary cholesterol is only 20% of my body's total. Most is manufactured by my liver, and because of hypercholesterolemia, my liver is churning out cholesterol twice as fast as a normal liver does. To solve the problem, I need to put the brakes on my liver's cholesterol-making frenzy.
In the 1980s researchers determined that the rate-limiting step in the liver's manufacture of cholesterol occurs early in the process, when a six-carbon molecule called mevalonate is converted to something called hydroxy methyl glutaryl CoA (HMG-CoA, for short). This reaction is carried out by an enzyme called HMG-CoA reductase. If we want to slow cholesterol production, that's our target.
In the last 20 years a series of drugs called statins have been developed that reduce levels of cholesterol by inhibiting HMG-CoA reductase. Among them are fluvastatin (Lescol), lovastatin (Mevacor), simvastatin (Zocor), and pravastatin (Pravachol).
Atorvastatin (Lipitor), introduced in 1997, is a particularly potent inhibitor of HMG-CoA reductase with fewer side effects than other statins. Lipitor is a blockbuster drug. It generated $4.4 billion in sales its first year, and became the top-selling global drug by 2005, with sales in excess of $10 billion.
It is upon Lipitor that I have banked my future.
I started taking it in 1999, a little white pill every morning before breakfast. It is likely I shall be taking it the rest of my life.
How well did it work? Very well indeed. Within three months of starting daily Lipitor tablets, my cholesterol level has fallen to below 200. In the years since then, it has never risen above 200. Not once. When last measured at the start of 2010, my cholesterol level was 158.
In The Naming of Cats, the T.S. Eliot poem on which the Broadway musical Cats was based, "a cat must have three different names.” I think of my anticholesterol drug as being like T.S. Eliot's cat. Like his cat, my drug has a "sensible everyday name”, Lipitor, a name that anyone can use. Then Eliot says a cat needs a second name, "a name that's particular,” "a name that never belonged to more than one cat.” Atorvastatin is such a name, one that drug manufacturers and scientists use.
According to T.S. Eliot, every cat also needs a third very private name, a "deep and inscrutable singular name.” For Lipitor, this name, its chemical soul, is [R-(R*,R*)]-2-(4-fluorophenyl)-β,δ-dihydroxy-5-(1-methylethyl)-3-phenyl-4-[(phenylamino)carbonyl]-1H-pyrrole-1-heptanoic acid.
To someone looking at a cholesterol level 116 points below 274, that's beautiful.

Inquiry & Analysis
How Does pH Affect a Protein's Function?
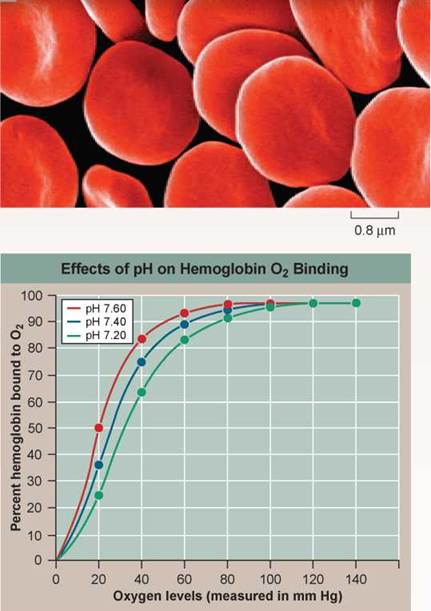
The red blood cells you see to the right carry oxygen to all parts of your body. These cells are red because they are chock full of a large iron-rich protein called hemoglobin. The iron atoms in each hemoglobin molecule provide a place for oxygen gas molecules to stick to the protein. When oxygen levels are highest (in the lungs), oxygen atoms bind to hemoglobin tightly, and a large percent of the hemoglobin molecules in a cell possess bound oxygen atoms. When oxygen levels are lower (in the tissues of the body), hemoglobin doesn't bind oxygen atoms as tightly, and as a consequence hemoglobin releases its oxygen to the tissues. What causes this difference between lungs and tissues in how hemoglobin loads and unloads oxygen? Oxygen concentration is not the only factor that might be responsible. Blood pH, for example, also differs between lungs and body tissues (pH is a measure of how many H+ ions a solution contains). Tissues are slightly more acid (that is, they have more H+ions and a lower pH) because their metabolic activities release CO2 into the blood, which you will recall from chapter 2, quickly becomes converted to carbonic acid.
The graph to the right displays so-called "oxygen loading curves" that reveal the effectiveness with which hemoglobin binds oxygen. The more effective the binding, the less oxygen required before hemoglobin becomes fully loaded, and the farther to the left a loading curve is shifted. To assess the impact of pH on this process, O2 loading curves were carried out at three different blood pH values.
In the graph, oxygen levels in the blood are presented on the x axis, and for each data point the corresponding % hemoglobin saturation (a %, or percent, is the numerator [top part] of a fraction whose denominator [bottom part] is 100—in this case, a measure of the fraction of the hemoglobin that is bound to oxygen) is presented on the y axis. The oxygen-loading curve was repeated at pH values of 7.6, 7.4, and 7.2, corresponding to the blood pH that might be expected in resting, exercising, and very active muscle tissue, respectively.
1. Applying Concepts
a. Variable. In the graph, what is the dependent variable?
b. Concentration. Which of the three pH values represents the highest concentration of hydrogen ions? (The concentration of a substance is the amount of that substance present in a given volume.) Is this value more acidic or more basic than the other two?
2. Interpreting Data
a. What is the percent hemoglobin bound to O2 for each of the three pH concentrations at saturation? At an oxygen level of 20 mm Hg? At 40 mm Hg? at 60 mm Hg?
b. What general statement can be made regarding the effect of the oxygen levels in the blood (as measured by partial pressure of oxygen, measured in mm Hg) on the binding of oxygen to hemoglobin?
c. Are there any significant differences in the hemoglobin saturation values for the three pHs at high oxygen levels?
3. Making Inferences At an oxygen level of 40 mm Hg, would hemoglobin bind oxygen more tightly at a pH of 7.8 or 7.0?
4. Drawing Conclusions How does pH affect the release of oxygen from hemoglobin?
5. Further Analysis
a. Carbon dioxide acts to lower the pH of the blood. Predict what would happen to hemoglobin loading when carbon dioxide enters the blood as the blood circulates through the tissues of the body.
b. In the lungs, oxygen levels are high and carbon dioxide leaves the blood and is exhaled, leading to lower carbon dioxide levels in the blood. Predict what happens to hemoglobin's oxygen loading under these conditions.
Test Your Understanding
1. The four kinds of organic macromolecules are
a. hydroxyls, carboxyls, aminos, and phosphates.
b. proteins, carbohydrates, lipids, and nucleic acids.
c. DNA, RNA, simple sugars, and amino acids.
d. carbon, hydrogen, oxygen, and nitrogen.
2. Organic molecules are made up of monomers. Which of the following is not considered a monomer of organic molecules?
a. amino acids
b. simple sugars
c. polypeptides
d. nucleotides
3. Your body is filled with many types of proteins. Each type has a distinctive sequence of amino acids that determines both its specialized _____ and its specialized _____.
a. number, weight
b. length, mass
c. structure, function
d. charge, pH
4. A peptide bond forms
a. by the removal of a water molecule.
b. by a dehydration reaction.
c. between two amino acids.
d. All of the above.
5. Nucleic acids
a. are the energy source for our bodies.
b. act on other molecules, breaking them apart or building new ones to help us function.
c. are only found in a few, specialized locations within the body.
d. are information storage devices found in body cells.
6. The two strands of a DNA molecule are held together through hydrogen bonds between nucleotide bases. Which of the following best describes this base pairing in DNA?
a. Adenine forms hydrogen bonds with thymine.
b. Adenine forms hydrogen bonds with cytosine.
c. Cytosine forms hydrogen bonds with thymine.
d. Guanine forms hydrogen bonds with adenine.
7. Carbohydrates are used for
a. structure and energy.
b. information storage.
c. fat storage and hair.
d. hormones and enzymes.
8. Which of the following carbohydrates is not found in plants?
a. glycogen
b. cellulose
c. starch
d. All are found in plants.
9. A characteristic common to fat molecules is
a. that they contain long chains of C—H bonds.
b. that they are insoluble in water.
c. that they have a glycerol backbone.
d. All of these are characteristics of fats.
10. Lipids are used for
a. motion and defense.
b. information storage.
c. energy storage and for some hormones.
d. enzymes and for some hormones.