THE LIVING WORLD
Unit Three. The Continuity of Life
13.5. Genetic Engineering and Agriculture
Pest Resistance
An important effort of genetic engineers in agriculture has involved making crops resistant to insect pests without spraying with pesticides, a great saving to the environment. Consider cotton. Its fibers are a major source of raw material for clothing throughout the world, yet the plant itself can hardly survive in a field because many insects attack it. Over 40% of the chemical insecticides used today are employed to kill insects that eat cotton plants. The world’s environment would greatly benefit if these thousands of tons of insecticide were not needed. Biologists are now in the process of producing cotton plants that are resistant to attack by insects.
One successful approach uses a kind of soil bacterium, Bacillus thuringiensis (Bt), that produces a protein that is toxic when eaten by crop pests, such as larvae (caterpillars) of butterflies. When the gene producing the Bt protein is inserted into the chromosomes of tomatoes, the plants begin to manufacture Bt protein. While not harmful to humans, it makes the tomatoes highly toxic to hornworms (one of the most serious pests of commercial tomato crops).
Many important plant pests also attack roots. To combat these pests, genetic engineers are introducing the Bt gene into different kinds of bacteria, ones that colonize the roots of crop plants. Any insects eating such roots consume the bacteria and so are lethally attacked by the Bt protein.
A major advance has been the creation of crop plants that are resistant to the herbicide glyphosate, a powerful biodegradable herbicide that kills most actively growing plants. Glyphosate is used in orchards and agricultural fields to control weeds. Growing plants need to make a lot of protein, and glypho- sate stops them from making protein by destroying an enzyme necessary for the manufacture of so-called aromatic amino acids (that is, amino acids that contain a ring structure, like phenylalanine—see figure 3.5). Humans are unaffected by glyphosate because we don’t make aromatic amino acids—we obtain them from plants we eat! To make crop plants resistant to this powerful plant killer, genetic engineers screened thousands of organisms until they found a species of bacteria that could make aromatic amino acids in the presence of glypho- sate. They then isolated the gene encoding the resistant enzyme and successfully introduced the gene into plants. They inserted the gene into the plants using DNA particle guns, also called gene guns. You can see in figure 13.10 how a DNA particle gun works. Small tungsten or gold pellets are coated with DNA (red in the figure) that contains the gene of interest and placed in the DNA particle gun. The DNA gun literally shoots the gene into plant cells in culture where the gene can be incorporated into the plant genome and then expressed. Plants that have been genetically engineered in this way are shown in figure 13.11. The two plants on top were genetically engineered to be resistant to glyphosate, the herbicide that killed the two plants at the bottom of the photo.
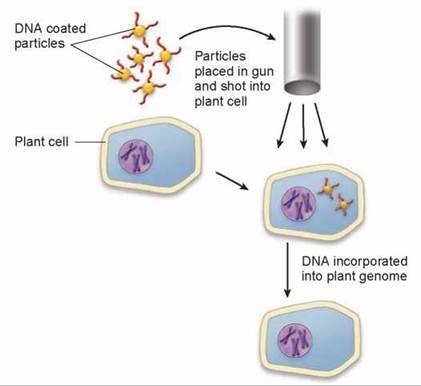
Figure 13.10. Shooting genes into cells.
A DNA particle gun, also called a gene gun, fires tungsten or gold particles coated with DNA into plant cells. The DNA-coated particles pass through the cell wall and into the cell, where the DNA is incorporated into the plant cell's DNA. The gene encoded by the DNA is expressed.

Figure 13.11. Genetically engineered herbicide resistance.
All four of these petunia plants were exposed to equal doses of an herbicide. The two on top were genetically engineered to be resistant to glyphosate, the active ingredient in the herbicide, whereas the two dead ones on the bottom were not.
The creation of glyphosate-tolerant crops is of major benefit to the environment. Glyphosate is quickly broken down in the environment, which makes its use a great improvement over long-lasting chemical herbicides. Also, not having to plow to remove weeds reduces the loss of fertile topsoil to erosion.
More Nutritious Crops
The cultivation of genetically modified (GM) crops of com, cotton, soybeans, and other plants (table 13.3) has become commonplace in the United States. In 2004, 85% of soybeans in the United States were planted with seeds genetically modified to be herbicide resistant. The result has been that less tillage was needed and, as a consequence, soil erosion was greatly lessened. Pest-resistant GM corn in 2004 comprised over 50% of all corn planted in the United States, and pest-resistant GM cotton comprised 81% of all cotton. In both cases, the change greatly lessens the amount of chemical pesticide used on the crops. These benefits of soil preservation and chemical pesticide reduction, while significant, have been largely bestowed upon farmers, making their cultivation of crops cheaper and more efficient.
TABLE 13.3. GENETICALLY MODIFIED CROPS
|
Genes have been added to commercial rice from daffodils for vitamin A, and from beans, fungi, and wild rice to supply dietary iron; transgenic strains that are cold-tolerant are under development. |
|
New strains of wheat, resistant to the herbicide glyphosate, greatly reduce the need for tilling and so reduce loss of topsoil. |
|
A major animal feed crop, soybeans tolerant of the herbicide glyphosate were used in 90% of U.S. soybean acreage in 2010. Varieties are being developed that contain the Bt gene, to protect the crop from insect pests without chemical pesticides. The nutritional value of soybean crops is being improved by genetic engineers in several ways, including transgenic varieties with high tryptophan (soybeans are poor in this essential amino acid), reduced transfatty acids, and enhanced omega-3 (beneficial) fatty acids, common in fish oil but low in plants. |
|
Corn varieties resistant to insect pests (Bt corn) are widely planted (40% of U.S. acreage); varieties also tolerant of the herbicide glyphosate have been recently developed. Varieties that are drought resistant are being developed, as well as nutritionally improved lines with high lysine, vitamin A, and high levels of the unsaturated fat oleic acid, which reduces harmful cholesterol and so prevents clogged arteries. |
|
Cotton crops are attacked by cotton bollworm, budworm, and other lepidopteran insects; more than 40% of all chemical pesticide tonnage worldwide is applied to cotton. A form of the Bt gene toxic to all lepidopterans but harmless to other insects has transformed cotton to a crop that requires few chemical pesticides. 81% of U.S. acreage is Bt cotton. |
|
The lesser cornstalk borer causes serious damage to peanut crops. An insect-resistant variety is under development by gene engineers to control this pest. |
|
Verticillium wilt (a fungal disease) infects the water-conducting tissues of potatoes, reducing crop yields 40%. An antifungal gene from alfalfa reduces infections sixfold. |
|
Canola, a major vegetable oil and animal feed crop, is typically grown in narrow rows with little cultivation, requiring extensive application of chemical herbicides to keep down weeds. New glyphosate-tolerant varieties require far less chemical treatment. 80% of U.S. canola acreage planted is gene-modified canola. |
Like the first act of a play, these developments have served mainly to set the stage for the real action, which is only now beginning to happen. The real promise of plant genetic engineering is to produce genetically modified plants with desirable traits that directly benefit the consumer.
One recent advance, nutritionally improved “golden” rice, gives us a hint of what is to come. In developing countries, large numbers of people live on simple diets that are poor sources of vitamins and minerals (what botanists called “micronutrients”). Worldwide, the two major micronutrient deficiencies are iron, which affects 1.4 billion women, 24% of the world population, and vitamin A, affecting 40 million children, 7% of the world population. The deficiencies are especially severe in developing countries where the major staple food is rice. In recent research, Swiss bioengineer Ingo Potrykus and his team at the Institute of Plant Sciences, Zurich, have gone a long way toward solving this problem. Supported by the Rockefeller Foundation and with results to be made free to developing countries, the work is a model of what plant genetic engineering can achieve.
To solve the problem of dietary iron deficiency among rice eaters, Potrykus first asked why rice is such a poor source of dietary iron. The problem, and the answer, proved to have three parts:
1. Too little iron. The proteins of rice endosperm have unusually low amounts of iron. To solve this problem, a ferritin gene (abbreviated as ![]() in figure 13.12) was transferred into rice from beans. Ferritin is a protein with an extraordinarily high iron content, and so greatly increased the iron content of the rice.
in figure 13.12) was transferred into rice from beans. Ferritin is a protein with an extraordinarily high iron content, and so greatly increased the iron content of the rice.
2. Inhibition of iron absorption by the intestine. Rice contains an unusually high concentration of a chemical called phytate, which inhibits iron absorption in the intestine—it stops your body from taking up the iron in the rice. To solve this problem, a gene encoding an enzyme called phytase (abbreviated as ![]() ) that destroys phytate was transferred into rice from a fungus.
) that destroys phytate was transferred into rice from a fungus.
3. Too little sulfur for efficient iron absorption. The human body requires sulfur for the uptake of iron, and rice has very little of it. To solve this problem, a gene encoding a sulfur-rich protein (abbreviated as ![]() ) was transferred into rice from wild rice.
) was transferred into rice from wild rice.
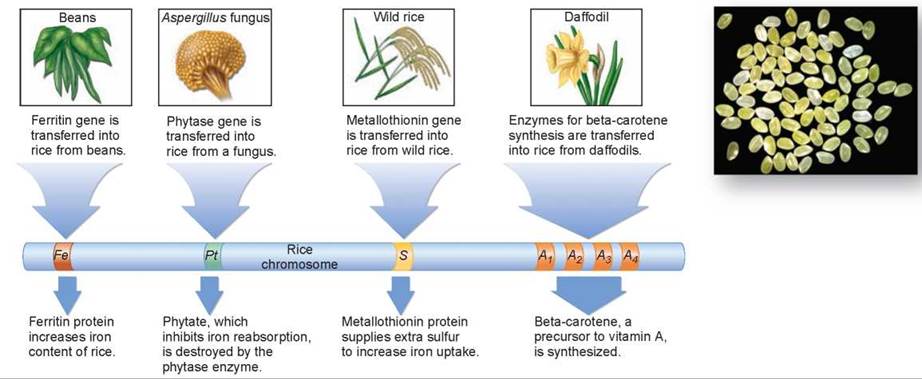
Figure 13.12. Transgenic "golden" rice.
To solve the problem of vitamin A deficiency, the same approach was taken. First, the problem was identified. It turns out rice only goes partway toward making vitamin A; there are no enzymes in rice to catalyze the last four steps. To solve the problem, genes encoding these four enzymes (abbreviated ![]() ) were added to rice from a flower, the daffodil.
) were added to rice from a flower, the daffodil.
The development of transgenic rice is only the first step in the battle to combat dietary deficiencies. The added nutritional value only makes up for half a person’s requirements, and many years will be required to breed the genes into lines adapted to local conditions, but it is a promising start, representative of the very real promise of genetic engineering.
How Do We Measure the Potential Risks of Genetically Modified Crops?
Is Eating Genetically Modified Food Dangerous? Many consumers worry that when bioengineers introduce novel genes into genetically modified (GM) crops, there may be dangerous consequences for the food we eat. The introduction of glyphosate-resistance into soybeans is an example. Could introduced proteins like the enzyme making the GM soybeans glyphosate-tolerant cause a fatal immune reaction in some people? Because the potential danger of allergic reactions is quite real, every time a protein-encoding gene is introduced into a GM crop it is necessary to carry out extensive tests of the introduced protein’s allergen potential. No GM crop currently being produced in the United States contains a protein that acts as an allergen to humans. On this score, then, the risk of genetic engineering to the food supply seems to be slight.
Are GM Crops Harmful to the Environment? Those concerned about the widespread use of GM crops raise three legitimate concerns:
1. Harm to Other Organisms. Might pollen from Bt corn harm non-pest insects that chance to eat it? Studies suggest little possibility of harm.
2. Resistance. All insecticides and herbicides used in agriculture share the problem that pests eventually evolve resistance to them, in much the same way that bacterial populations evolve resistance to antibiotics.
To prevent this, farmers are required to plant at least 20% non-Bt crops alongside Bt crops to provide refuges where insect populations are not under selection pressure and in this way to slow the development of resistance.
As a result, despite the widespread use of Bt crops like corn, soybeans, and cotton since 1996, there are as of yet only a few cases of insects developing resistance to Bt plants in the field. Unfortunately, the same restrictions have not been required for farmers using the herbicide glyphosate, leading to a different result: By the year 2010, glyphosate-resistant weeds have been reported by upset farmers in 22 states.
3. Gene Flow. How about the possibility that introduced genes will pass from GM crops to their wild relatives? For the major GM crops, there is usually no potential relative around to receive the modified gene from the GM crop. There are no wild relatives of soybeans in Europe, for example. Thus there can be no gene escape from GM soybeans in Europe, any more than genes can flow from you to your pet dog or cat. However—and this is a big however—for secondary crops only now being genetically modified, studies suggest it will be difficult to prevent GM crops from interbreeding with surrounding relatives to create new hybrids.
Key Learning Outcome 13.5. Genetic engineering affords great opportunities for progress in food production. On balance, the risks appear slight, and the potential benefits substantial.
Today’s Biology
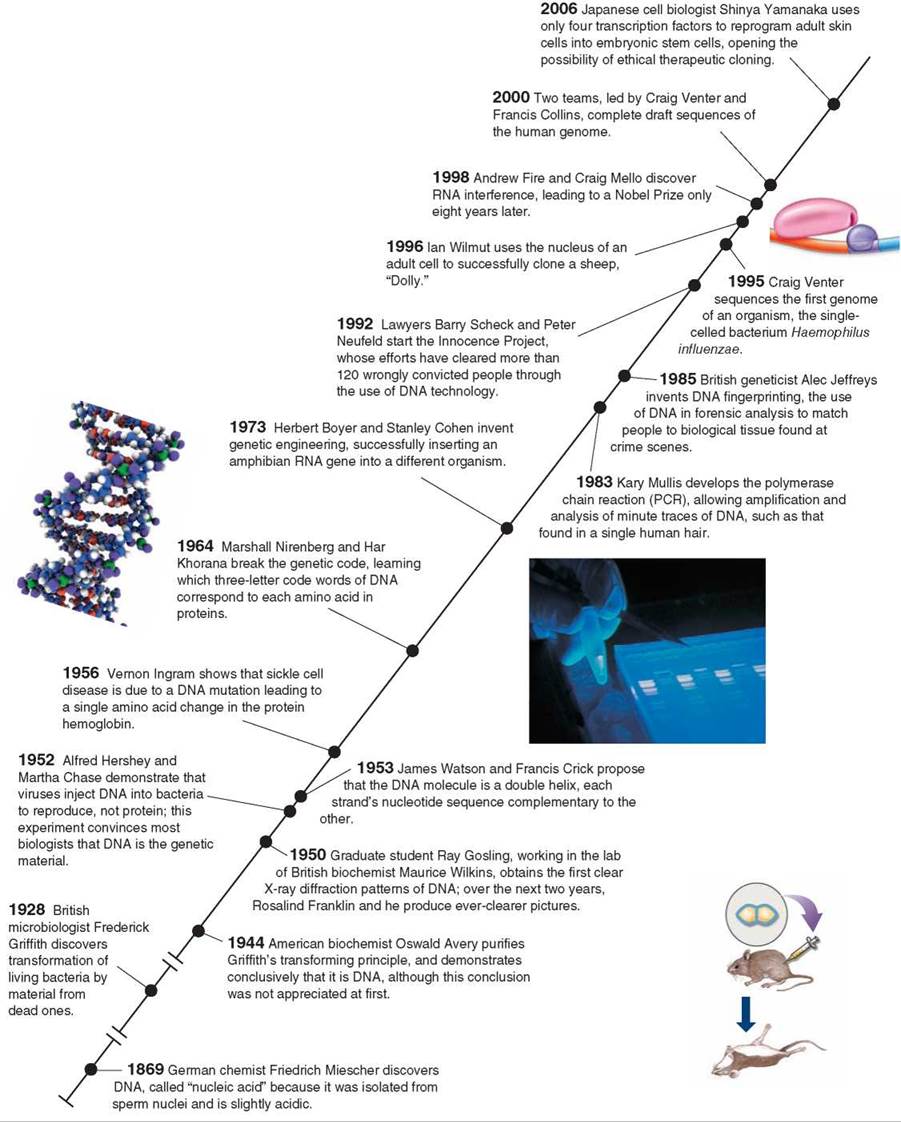
A DNA Timeline

In 2000, Craig Venter of Celera, President Clinton, and Francis Collins of the Human Genome Project announce the human genome.