MCAT Biology Review
Chapter 4: The Nervous System
4.2 Transmission of Neural Impulses
Now that we have discussed the basic anatomy of the neuron, we can turn to the physiology that underlies neuronal signaling.
THE ACTION POTENTIAL
Neurons use all-or-nothing messages called action potentials to relay electrical impulses down the axon to the synaptic bouton. As we will explore in the following section, action potentials ultimately cause the release of neurotransmitters into the synaptic cleft.
Resting Potential
All neurons exhibit a resting membrane potential. This means that there is an electrical potential difference (voltage) between the inside of the neuron and the extracellular space. Usually, this is about –70 mV, with the inside of the neuron being negative relative to the outside. Neurons use selective permeability to ions and the Na+/K+ ATPase to maintain this negative internal environment, as shown in Figure 4.3.
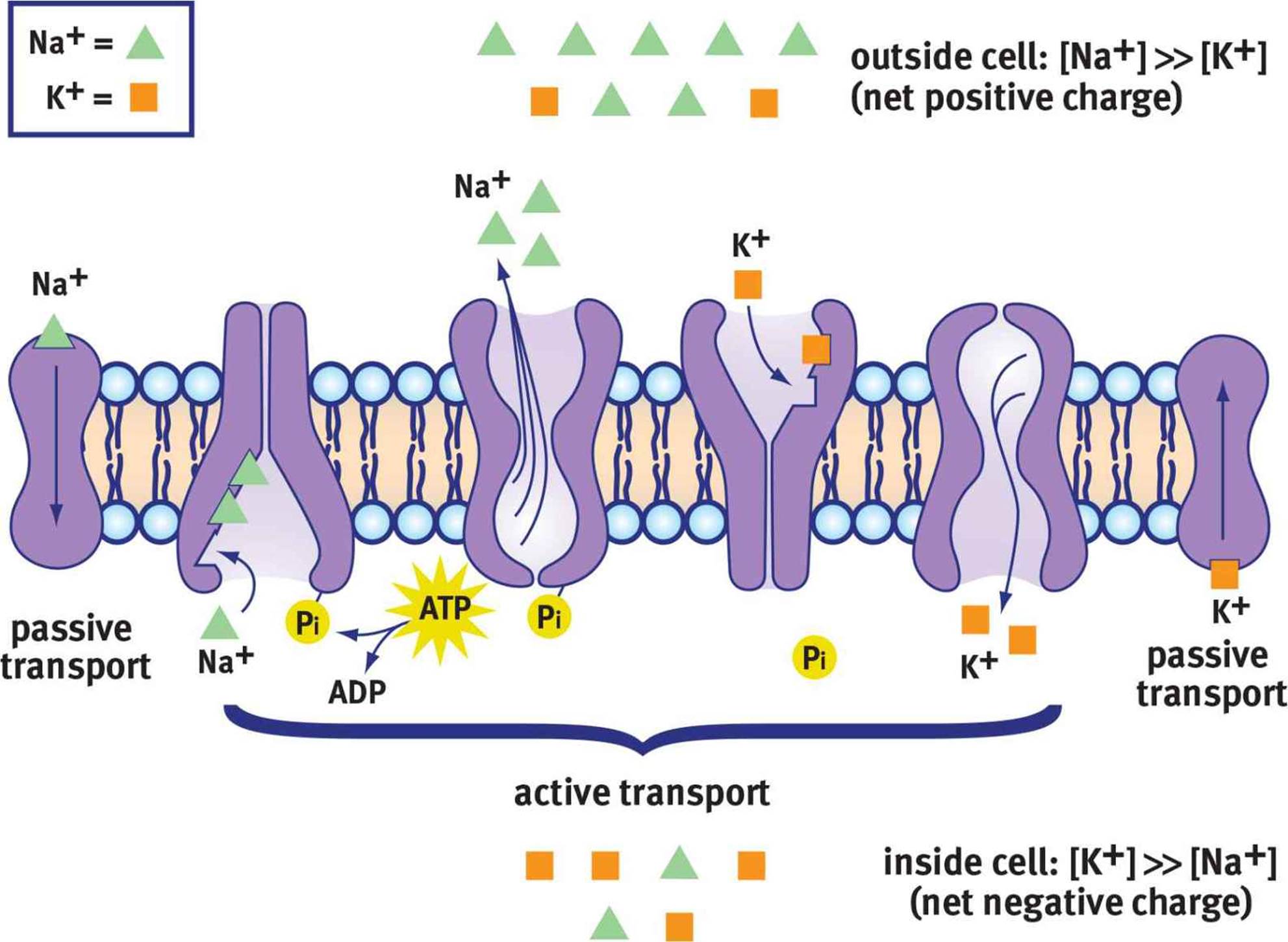 Figure 4.3. Maintenance of Resting Membrane Potential The Na+/K+ ATPase maintains a resting membrane potential of –70 mV by moving 3 Na+ ions out of the cell for every 2 K+ ions moved into the cell.
Figure 4.3. Maintenance of Resting Membrane Potential The Na+/K+ ATPase maintains a resting membrane potential of –70 mV by moving 3 Na+ ions out of the cell for every 2 K+ ions moved into the cell.
Like any other cell, the neuronal plasma membrane is fairly impermeable to charged species. Because the plasma membrane contains a thick nonpolar barrier (fatty acid tails), it is not energetically favorable for ions to cross this barrier. Inside the neuron, [K+] is high and [Na+] is low. Outside of the neuron, [Na+] is high, whereas [K+] is low. The negative resting potential is generated by both negatively charged proteins within the cell and the relatively greater permeability of the membrane to K+ compared with Na+. If the cell membrane is more permeable to K+ and the ion’s concentration is higher inside, K+ will diffuse down its gradient out of the cell. What does this mean in terms of charge movement? K+ is positively charged, so its movement out of the cell results in a cell interior that is negative. Put another way, if we assume that the membrane starts at zero, and we take away a positive charge, we end up with a negative charge inside the cell: 0 – (+1) = –1. Na+ cannot readily enter at rest, so the negative potential is maintained.
BRIDGE
The resting membrane potential is dependent on the intra- and extracellular ion concentrations, relative permeability of the membrane to these different ions, and charges of these ions. The Goldman–Hodgkin–Katz voltage equation brings together these different factors into one equation that predicts the resting membrane potential. This equation is discussed in Chapter 8 of MCAT Biochemistry Review.
The Na+/K+ ATPase is important for restoring this gradient after action potentials have been fired. It transports three Na+ out of the cell for every two K+ into the cell at the expense of one ATP. ATP is necessary because both Na+ and K+ are moved against their gradients by this process; thus, this qualifies as primary active transport. Each time the pump works, it results in the inside of the cell becoming relatively more negative, as only two positive charges are moved in for every three that are moved out.
The Axon Hillock
Neurons can receive both excitatory and inhibitory input. Excitatory input causes depolarization (raising the membrane potential, Vm, from its resting potential) and thus makes the neuron more likely to fire an action potential. Inhibitory input causes hyperpolarization (lowering the membrane potential from its resting potential) and thus makes the neuron less likely to fire an action potential. If the axon hillock receives enough excitatory input to be depolarized to the threshold value (usually in the range of –55 to –40 mV), an action potential will be triggered.
This implies that not every stimulus necessarily generates a response. A small excitatory signal may not be sufficient to bring the axon hillock to threshold. Further, a postsynaptic neuron may receive information from several different presynaptic neurons, some of which are excitatory and some of which are inhibitory. The additive effects of multiple signals is known as summation.
There are two types of summation: temporal and spatial. In temporal summation, multiple signals are integrated during a relatively short period of time. A number of small excitatory signals firing at nearly the same moment could bring a postsynaptic cell to threshold, enabling an action potential. In spatial summation, the additive effects are based on the number and location of the incoming signals. A large number of inhibitory signals firing directly on the soma will cause more profound hyperpolarization of the axon hillock than the depolarization caused by a few excitatory signals firing on the dendrites of a neuron.
Ion Channels and Membrane Potential
A graph of membrane potential vs. time during an action potential is shown in Figure 4.4.
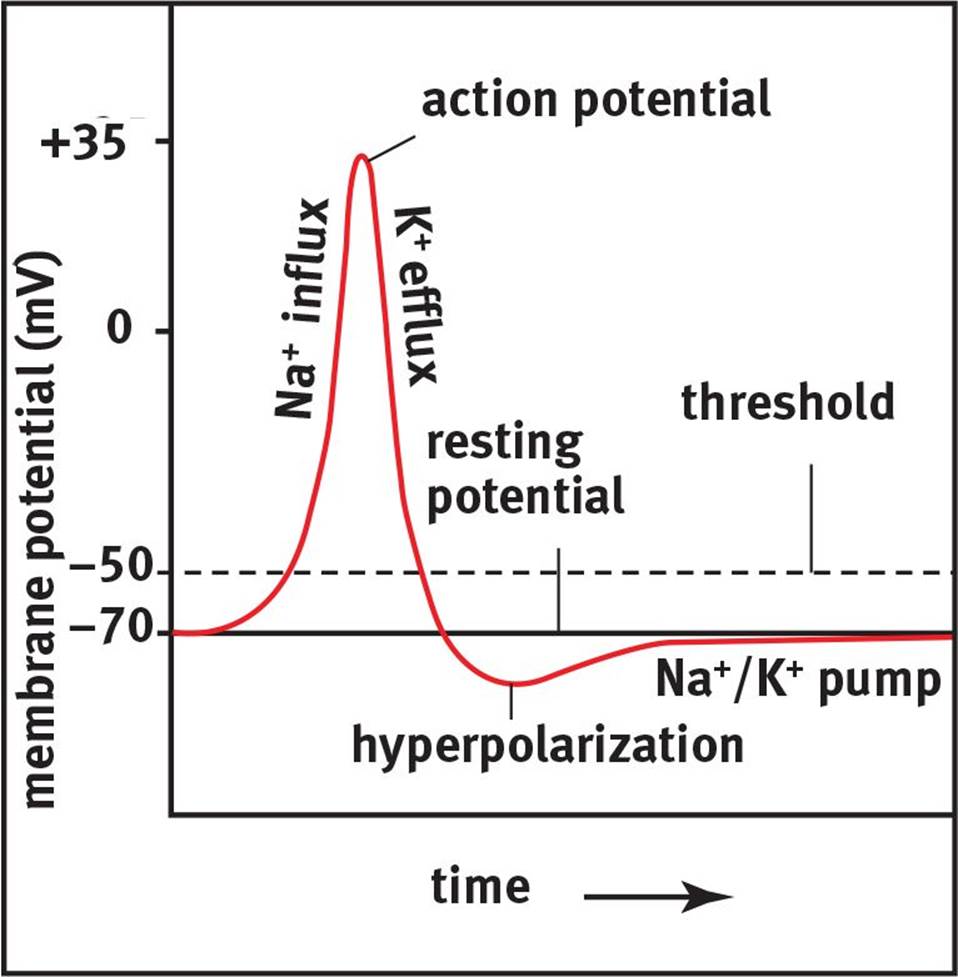 Figure 4.4. Action Potential Generation Sufficient depolarization across the cell membrane to threshold leads to the generation of an action potential, followed by repolarization and hyperpolarization before returning to the resting membrane potential.
Figure 4.4. Action Potential Generation Sufficient depolarization across the cell membrane to threshold leads to the generation of an action potential, followed by repolarization and hyperpolarization before returning to the resting membrane potential.
If the cell is brought to threshold, voltage-gated sodium channels open in the membrane. As the name implies, these ion channels open in response to the change in potential of the membrane (depolarization) and permit the passage of sodium ions. There is a strong electrochemical gradientthat promotes the migration of sodium into the cell. From an electric standpoint, the interior of the cell is more negative than the exterior of the cell, which favors the movement of positively charged sodium cations into the cell. From a chemical standpoint, there is a higher concentration of sodium outside the cell than inside, which also favors the movement of sodium into the cell. As sodium passes through these ion channels, the membrane potential becomes more positive; that is, the cell rapidly depolarizes. Sodium channels not only open in response to changes in membrane potential, but are also inactivated by them. When Vm approaches +35 mV, the sodium channels are inactivated and will have to be brought back near the resting potential to be deinactivated. Thus, these sodium channels can exist in three states: closed (before the cell reaches threshold, and after inactivation has been reversed), open (from threshold to approximately +35 mV), and inactive (from approximately +35 mV to the resting potential).
KEY CONCEPT
Na+ wants to go into the cell because the cell is more negative inside (electrical gradient) and has a lower concentration of Na+ inside (chemical gradient).
The positive potential inside the cell not only triggers the voltage-gated sodium channels to inactivate, but also triggers the voltage-gated potassium channels to open. Once sodium has depolarized the cell, there is an electrochemical gradient favoring the efflux of potassium from the neuron. As positively charged potassium cations are driven out of the cell, there will be a restoration of the negative membrane potential called repolarization. The efflux of K+ causes an overshoot of the resting membrane potential, hyperpolarizing the neuron. This hyperpolarization serves an important function: it makes the neuron refractory to further action potentials. There are two types of refractory periods. During the absolute refractory period, no amount of stimulation can cause another action potential to occur. During the relative refractory period, there must begreater than normal stimulation to cause an action potential because the membrane is starting from a potential that is more negative than its resting value.
The Na+/K+ ATPase acts to restore not only the resting potential, but also the sodium and potassium gradients that have been partially dissipated by the action potential.
KEY CONCEPT
Action potentials rely on both electrical and chemical gradients. The neuron starts at the resting potential, around –70 mV. At the resting potential, potassium is high inside the cell and low outside the cell, while sodium is high outside the cell and low inside the cell. Once the cell reaches threshold, sodium channels open and sodium floods the cell, making it more positive inside (depolarization). Then, sodium channels are inactivated and the potassium channels open. This allows potassium to flow out of the cell, bringing the potential to the negative range (repolarization), and actually overshooting the resting potential (hyperpolarization). The Na+/K+ ATPase then works to restore the resting potential.
Impulse Propagation
So far, we have discussed the movements of ions at one small segment of the axon. For a signal to be conveyed to another neuron, the action potential must travel down the axon and initiate neurotransmitter release. This movement is called impulse propagation and is shown in Figure 4.5. As sodium rushes into one segment of the axon, it will cause depolarization in the surrounding regions of the axon. This depolarization will bring subsequent segments of the axon to threshold, opening the sodium channels in those segments. Each of these segments then continues through the rest of the action potential in a wavelike fashion until the action potential reaches the nerve terminal. After the action potential has fired in one segment of axon, that segment becomes momentarily refractory, as described previously. The functional consequence of this is that information can only flow in one direction.
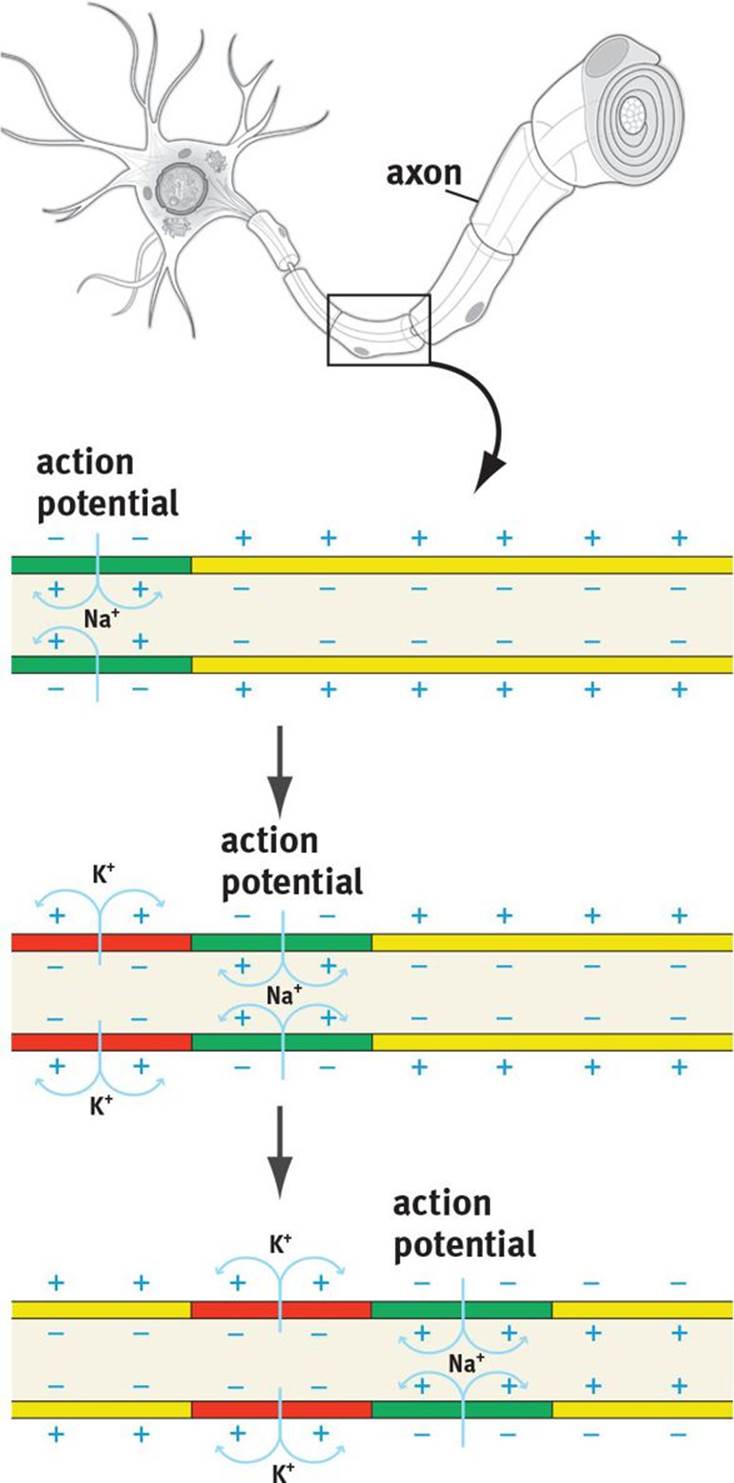 Figure 4.5. Action Potential Propagation Action potentials are propagated down the axon when proximal sodium channels open and depolarize the membrane, inducing neighboring sodium channels to open as well; because of the refractory character of these channels, the action potential can move in only one direction.
Figure 4.5. Action Potential Propagation Action potentials are propagated down the axon when proximal sodium channels open and depolarize the membrane, inducing neighboring sodium channels to open as well; because of the refractory character of these channels, the action potential can move in only one direction.
REAL WORLD
A toxin called tetrodotoxin (TTX) is found in the pufferfish, a delicacy in Japan. TTX blocks the voltage-gated Na+ channels, thereby blocking neuronal transmission. This can rapidly cause death because the phrenic nerves innervating the diaphragm can no longer depolarize, leading to paralysis of the muscle and a cessation of breathing. For this reason, chefs who prepare pufferfish must be specially trained and licensed.
REAL WORLD
Local anesthetics work by blocking the voltage-gated Na+ channels. These drugs work particularly well on sensory neurons and therefore block the transmission of pain. They favor pain neurons because these neurons have small axonal diameters and little or no myelin, allowing easy access to the sodium channels. Anesthetic concentrations are kept sufficiently low to block pain neurons without significant effects on other sensory modalities or motor function.
The speed at which action potentials move depends on the length and cross-sectional area of the axon. Increased length of the axon results in higher resistance and slower conduction. Greater cross-sectional areas allow for faster propagation due to decreased resistance. The effect of cross-sectional area is more significant than the effect of length. In order to maximize the speed of transmission, mammals have myelin. Myelin is an extraordinarily good insulator, preventing the dissipation of the electric signal. The insulation is so effective that the membrane is only permeable to ion movement at the nodes of Ranvier. Thus, the signal “hops” from node to node—what is called saltatory conduction.
REAL WORLD
This insulation by myelin is extremely effective. A human spinal cord is about the thickness of a finger. Without this insulation, the cord would have to be almost as wide as a telephone pole to prevent signal loss.
It is important to note that all action potentials within the same type of neuron have the same potential difference during depolarization. Increased intensity of a stimulus does not result in an increased potential difference of the action potential, but rather an increased frequency of firing.
THE SYNAPSE
As discussed previously, neurons are not actually in direct physical contact. There is a small space between neurons called the synaptic cleft into which neurotransmitters are secreted, as shown in Figure 4.6. To clarify the terminology, the neuron preceding the synaptic cleft is called thepresynaptic neuron; the neuron after the synaptic cleft is called the postsynaptic neuron. If a neuron signals to a gland or muscle, rather than another neuron, the postsynaptic cell is termed an effector. Most synapses are chemical in nature; they use small molecules referred to asneurotransmitters to send messages from one cell to the next.
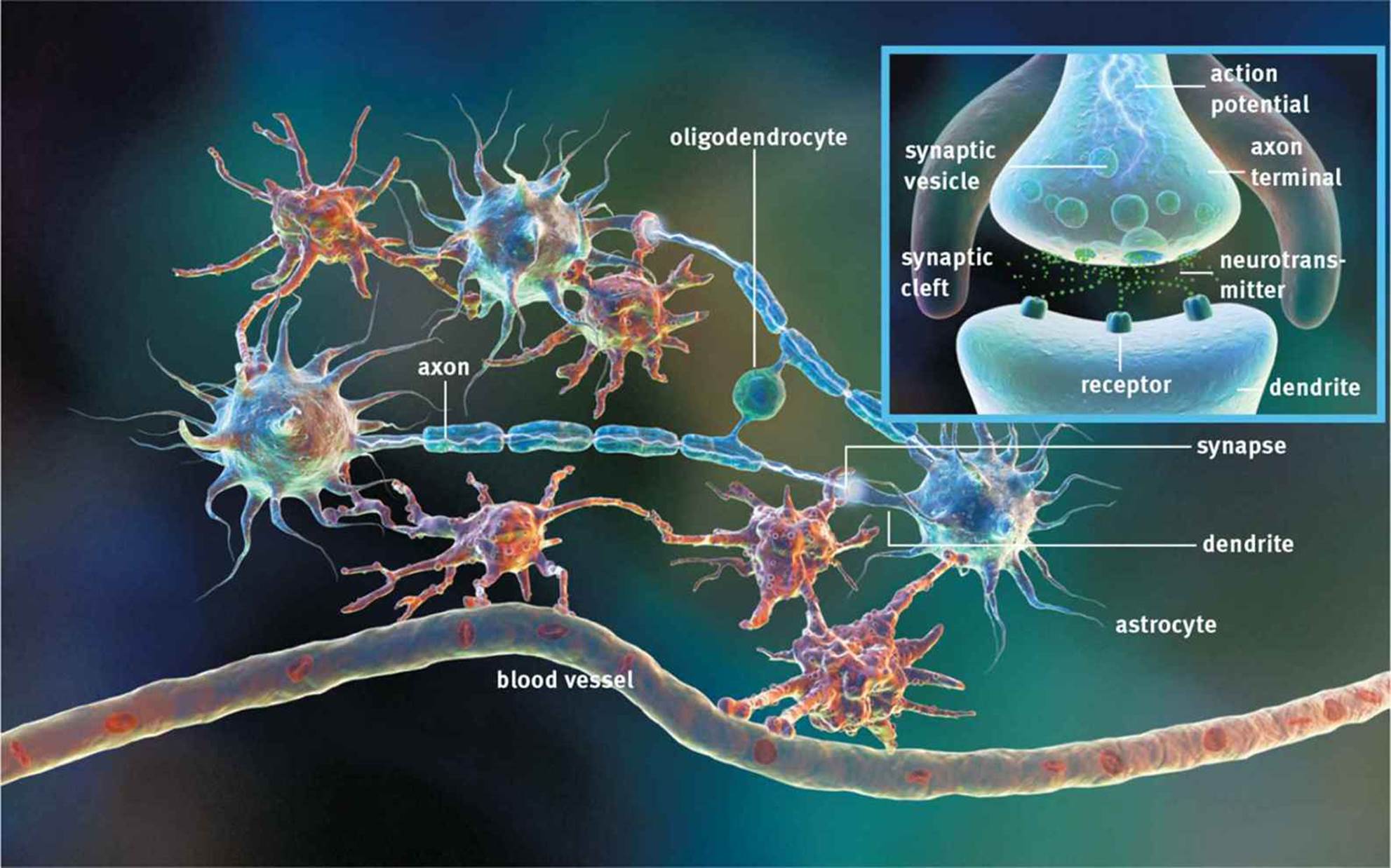 Figure 4.6. The Synapse Synaptic vesicles are released from the presynaptic neuron and diffuse across the synaptic cleft to activate receptors on the postsynaptic neuron (or gland or muscle).
Figure 4.6. The Synapse Synaptic vesicles are released from the presynaptic neuron and diffuse across the synaptic cleft to activate receptors on the postsynaptic neuron (or gland or muscle).
Neurotransmitters
Prior to release, neurotransmitter molecules are stored in membrane-bound vesicles in the nerve terminal. When the action potential reaches the nerve terminal, voltage-gated calcium channels open, allowing calcium to flow into the cell. This sudden increase in intracellular calcium triggers fusion of the membrane-bound vesicles with the cell membrane at the synapse, causing exocytosis of the neurotransmitter.
KEY CONCEPT
It is critical to understand the difference between electrical and chemical transmission. Within a neuron, electricity is used to pass signals down the length of the axon. Between neurons, chemicals (neurotransmitters) are used to pass signals to the subsequent neuron (or gland or muscle).
Once released into the synapse, the neurotransmitter molecules diffuse across the cleft and bind to receptors on the postsynaptic membrane. This allows the message to be passed from one neuron to the next. As we stated earlier, neurons may be either excitatory or inhibitory; this distinction truly comes at the level of the neurotransmitter receptors, which tend to be either ligand-gated ion channels or G protein-coupled receptors. If the receptor is a ligand-gated ion channel, the postsynaptic cell will either be depolarized or hyperpolarized. If it is a G protein-coupled receptor, it will cause either changes in the levels of cyclic AMP (cAMP) or an influx of calcium. Note that the physiology of receptors is further discussed in Chapter 3 of MCAT Biochemistry Review.
Neurotransmission must be regulated—there are almost no circumstances under which constant signaling to the postsynaptic cell would be desirable. Therefore, the neurotransmitter must be removed from the synaptic cleft. There are three main mechanisms to accomplish this goal. First, neurotransmitters can be broken down by enzymatic reactions. The breakdown of acetylcholine (ACh) by acetylcholinesterase (AChE), shown in Figure 4.7, is a classic example.
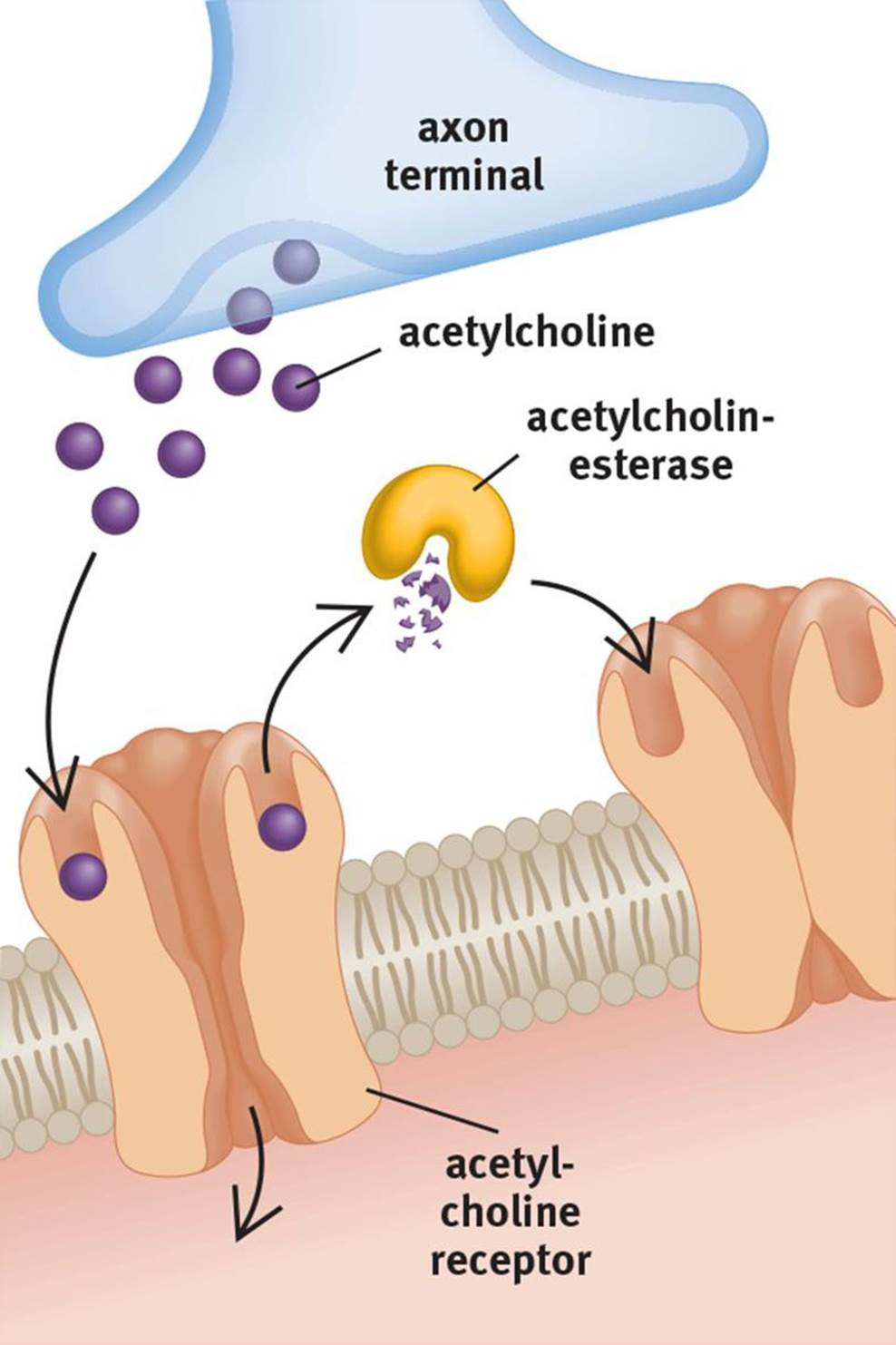 Figure 4.7. Breakdown of a Neurotransmitter by an Enzyme Acetylcholine (ACh) can be broken down by acetylcholinesterase (AChE).
Figure 4.7. Breakdown of a Neurotransmitter by an Enzyme Acetylcholine (ACh) can be broken down by acetylcholinesterase (AChE).
Second, neurotransmitters can be brought back into the presynaptic neuron using reuptake carriers. The reuptake of serotonin (5-HT), shown in Figure 4.8, is a classic example of this mechanism. Dopamine (DA) and norepinephrine (NE) also use reuptake carriers.
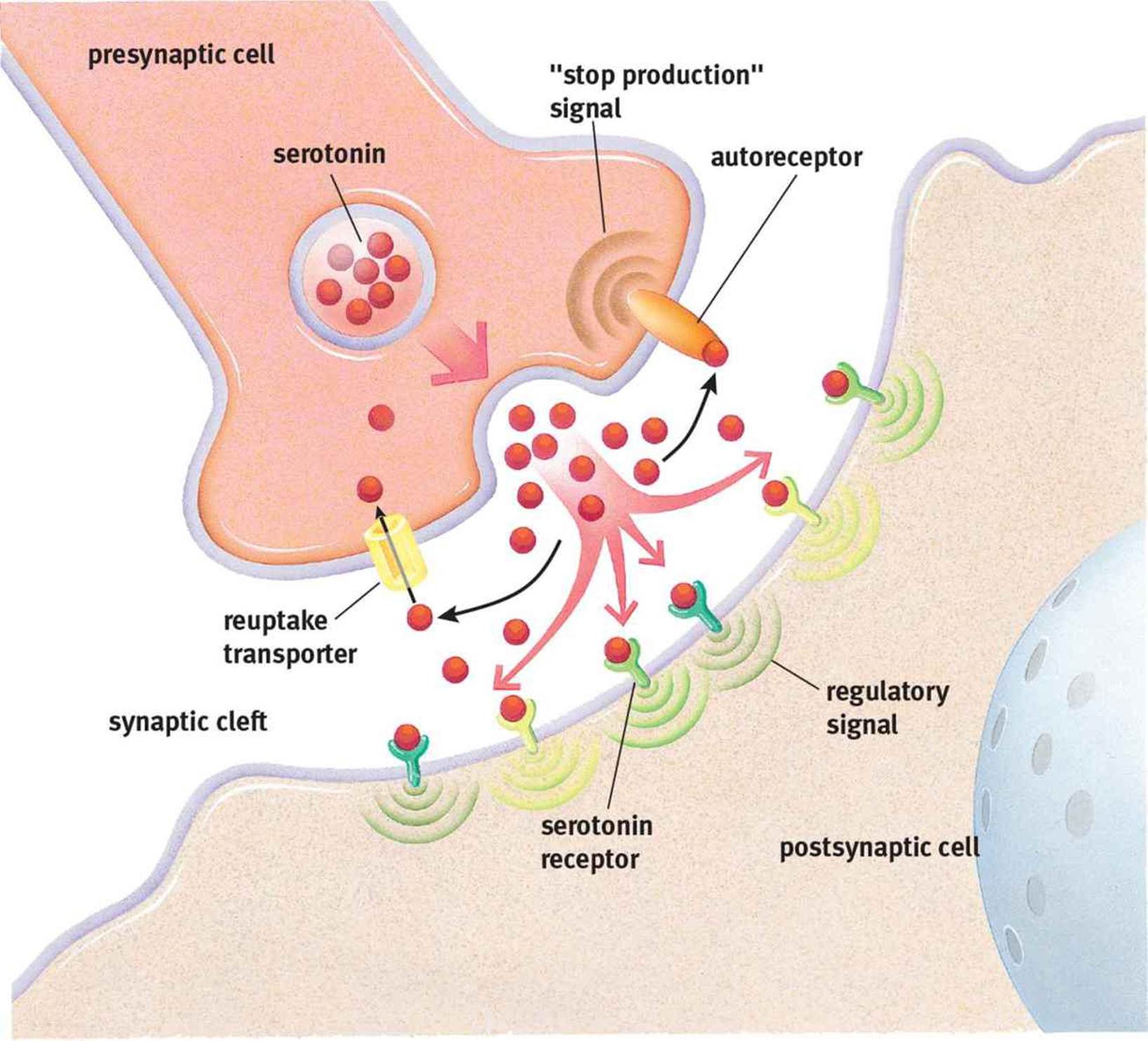 Figure 4.8. Reuptake of a Neurotransmitter Serotonin (5-HT) can be taken back up by the presynaptic cell; an autoreceptor will signal the presynaptic cell to stop releasing serotonin and start the reuptake process.
Figure 4.8. Reuptake of a Neurotransmitter Serotonin (5-HT) can be taken back up by the presynaptic cell; an autoreceptor will signal the presynaptic cell to stop releasing serotonin and start the reuptake process.
Third, neurotransmitters may simply diffuse out of the synaptic cleft. Nitric oxide (NO), a gaseous signaling molecule, fits into this category.
REAL WORLD
Many common drugs (either in clinical use or street drugs) modify processes that occur in the synapse. For instance, cocaine acts by blocking neuronal reuptake carriers, thus prolonging the action of neurotransmitters in the synapse. There are clinically useful drugs (some of which are used to treat Alzheimer’s disease, glaucoma, and myasthenia gravis) that inhibit acetylcholinesterase, thereby elevating synaptic levels of acetylcholine. Nerve gases, which have been used in warfare and terrorism, are extremely potent acetylcholinesterase inhibitors. Nerve gas causes rapid death by preventing the relaxation of skeletal muscle (most importantly, the diaphragm), leading to respiratory arrest.
MCAT Concept Check 4.2:
Before you move on, assess your understanding of the material with these questions.
1. What neural structure initiates the action potential?
2. What entity maintains the resting membrane potential? What is the approximate voltage of the resting membrane potential?
3. What is the difference between temporal and spatial summation?
· Temporal summation:
· Spatial summation:
4. During the action potential, which ion channel opens first? How is this ion channel regulated? What effect does the opening of this channel have on the polarization of the cell?
· Ion channel:
· Regulation:
· Effect on polarization:
5. During the action potential, which ion channel opens second? How is this ion channel regulated? What effect does the opening of this channel have on the polarization of the cell?
· Ion channel:
· Regulation:
· Effect on polarization:
6. What is the difference between the absolute and relative refractory period?
· Absolute refractory period:
· Relative refractory period:
7. What ion is primarily responsible for the fusion of neurotransmitter-containing vesicles with the nerve terminal membrane?
8. What are the three main methods by which a neurotransmitter’s action can be stopped?
·
·
·