MCAT Biology Review
Chapter 10: Homeostasis
Introduction
Have a headache? Pop an ibuprofen. Backache? Works for that, too. Ibuprofen, which has been around for over 50 years, is an inexpensive, over-the-counter, nonsteroidal anti-inflammatory drug (NSAID). Because of its ability to relieve pain, ibuprofen is known as an analgesic. When used in moderation, ibuprofen carries relatively little risk. However, taking multiple doses for many years can profoundly affect the kidneys. Years of analgesic use (usually as self-therapy) can lead to kidney failure, known as analgesic nephropathy. If untreated, kidney failure is universally fatal.
If detected, however, dialysis (or a kidney transplant) could save such an individual’s life. How does dialysis save a patient with kidney failure? Dialyzing fluid has many of the same solutes as blood, in strategic concentrations, and it is separated from blood by a semipermeable membrane. As blood is filtered through the dialysis machine, fluid and solutes diffuse down their concentration gradients, limited only by size (as determined by the membrane). The dialysis machine therefore performs filtration to purify the blood and excrete wastes, a crucial function that the kidneys would normally perform.
In this chapter, we’ll learn more about filtration, as well as reabsorption and secretion. These processes are collectively involved in osmoregulation. Osmoregulation is just one mechanism that the body uses to maintain homeostasis in its fluids and tissues. We’ll also discuss the skin, which plays a significant role in temperature homeostasis (thermoregulation).
10.1 The Excretory System
The excretory system serves many functions, including the regulation of blood pressure, blood osmolarity, acid–base balance, and removal of nitrogenous wastes. The kidneys play an essential role in these functions.
ANATOMY OF THE EXCRETORY SYSTEM
The excretory system consists of the kidneys, ureters, bladder, and urethra, as shown in Figure 10.1. The kidneys are two bean-shaped structures located behind the digestive organs at the level of the bottom rib. The functional unit of the kidney is the nephron; each kidney has approximately 1 million nephrons. All of the nephrons eventually empty into the renal pelvis, which narrows to form the the ureter. Urine travels through the ureter to the bladder. From the bladder, urine is transported through the urethra to exit the body.
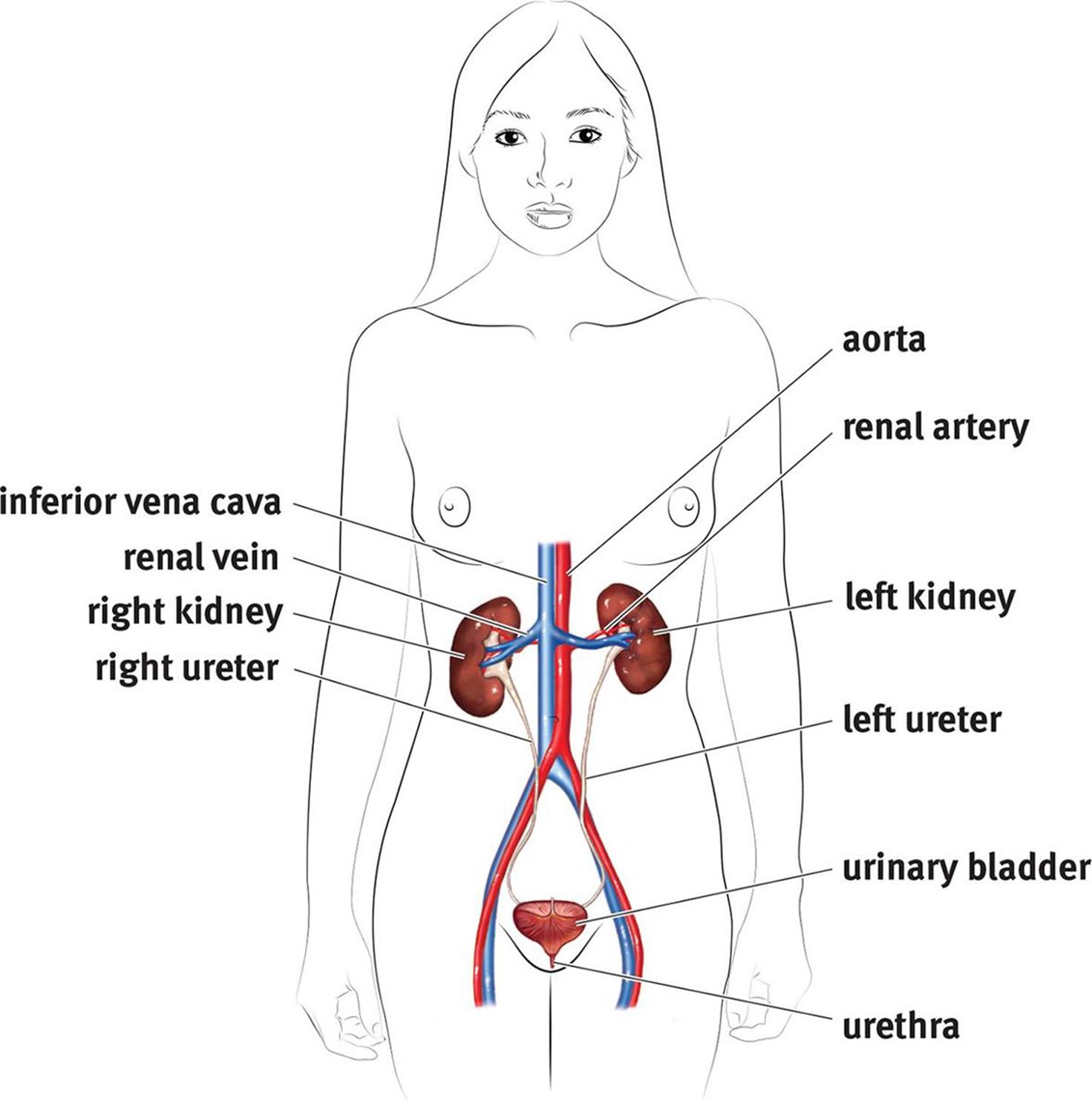 Figure 10.1. The Excretory System Once it leaves the kidneys, urine moves through the ureters to be stored in the urinary bladder until it is excreted through the urethra.
Figure 10.1. The Excretory System Once it leaves the kidneys, urine moves through the ureters to be stored in the urinary bladder until it is excreted through the urethra.
Kidney Structure
Each kidney is subdivided into a cortex and a medulla, as shown in Figure 10.2. The cortex is the kidney’s outermost layer, while the medulla of the kidney sits within the cortex. Each kidney also has a renal hilum, which is a deep slit in the center of its medial surface. The widest part of the ureter, the renal pelvis, spans almost the entire width of the renal hilum. The renal artery, renal vein, and ureter enter and exit through the renal hilum.
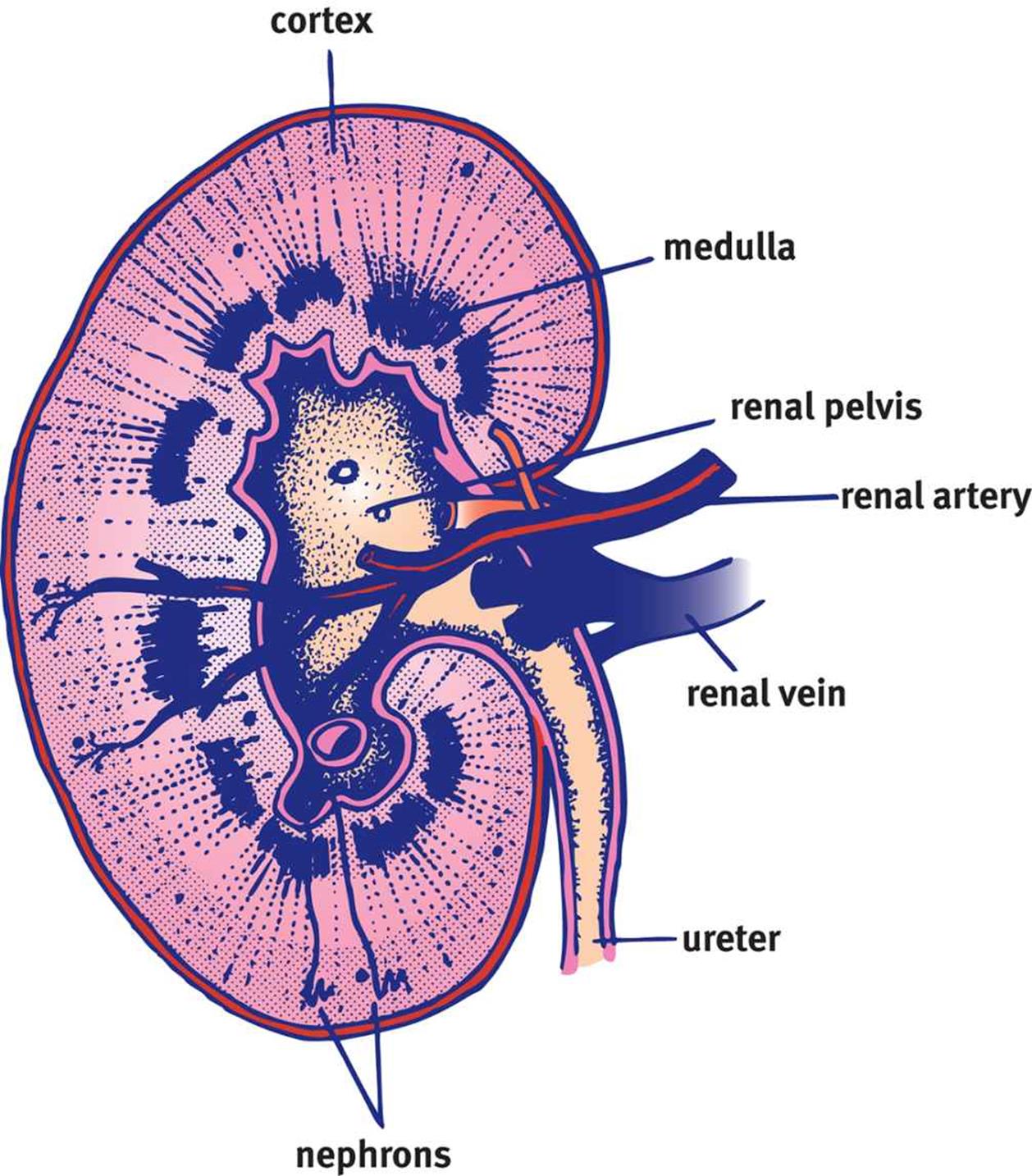 Figure 10.2. Basic Structure of the Kidney
Figure 10.2. Basic Structure of the Kidney
The kidney has one of the few portal systems in the body. A portal system consists of two capillary beds in series through which blood must travel before returning to the heart. The renal artery branches out, passes through the medulla, and enters the cortex as afferent arterioles. The highly convoluted capillary tufts derived from these afferent arterioles are known as glomeruli. After blood passes through a glomerulus, the efferent arterioles lead blood away from afferent arterioles. As is the case in other portal systems, the efferent arterioles then form a second capillary bed. These capillaries surround the loop of Henle and are known as vasa recta. The renal vascular system is shown in Figure 10.3.
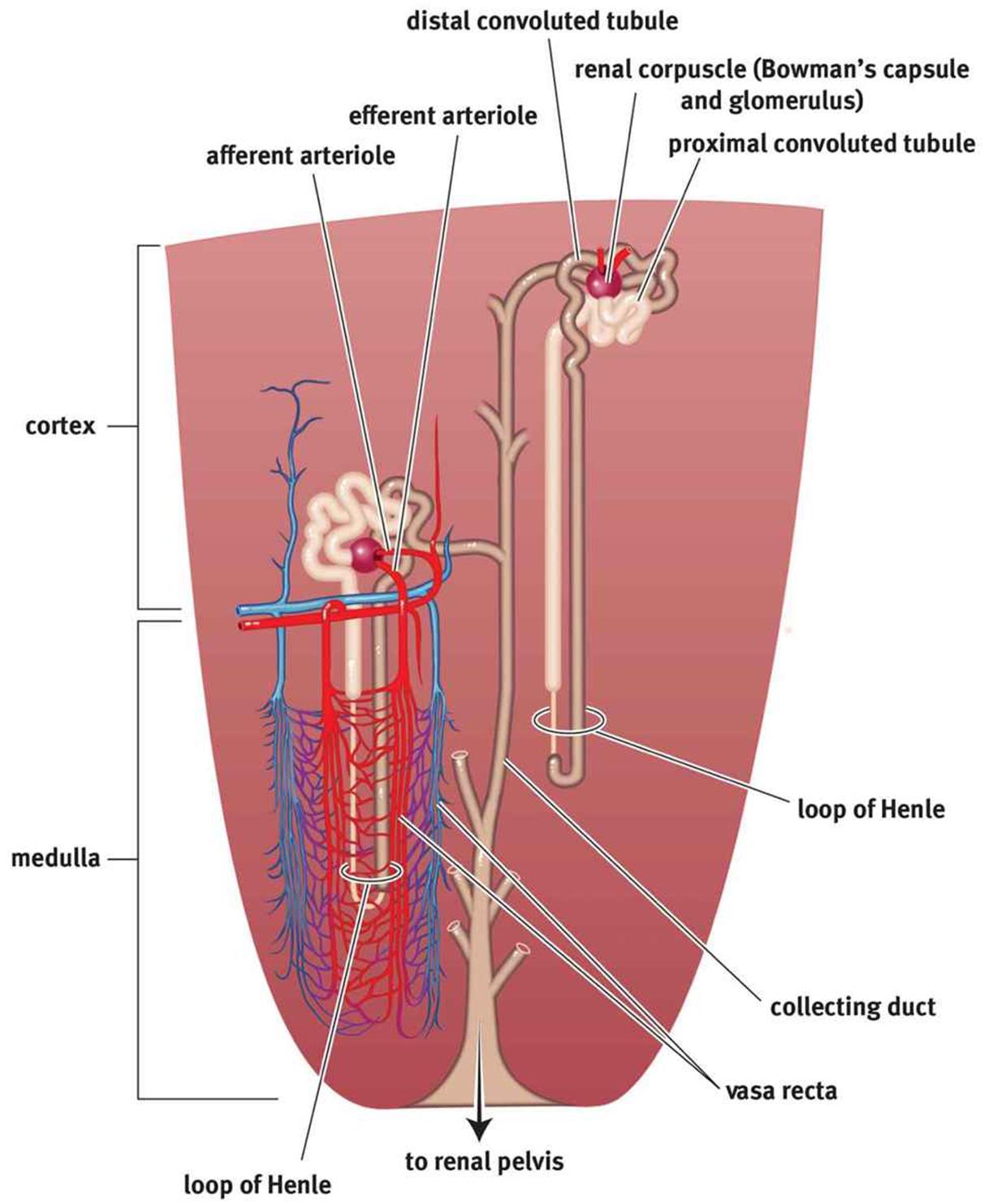 Figure 10.3. Microanatomy of the Renal Vascular System and the Nephron
Figure 10.3. Microanatomy of the Renal Vascular System and the Nephron
BRIDGE
The portal system in the kidney is one of three major portal systems in the body. The other two are the hypophyseal portal system, which connects the hypothalamus and the anterior pituitary (discussed in Chapter 5 of MCAT Biology Review), and the hepatic portal system, which connects the gut tube and the liver (discussed in Chapter 9 of MCAT Biology Review). In each case, blood passes through two capillary beds before returning to the heart.
BRIDGE
Our discussion of neurons in Chapter 4 of MCAT Biology Review used some of the same terms as those that describe the organization of blood vessels in the kidney. Afferent neurons carry sensory information toward the central nervous system much as afferent arterioles carry blood toward the glomeruli. Efferent neurons relay signals away from the central nervous system just as efferent arterioles carry blood away from the glomerulus.
Also visible in Figure 10.3 is the structure of the nephron. Around the glomerulus is a cuplike structure known as Bowman’s capsule. Bowman’s capsule leads to a long tubule with many distinct areas; in order, these are the proximal convoluted tubule, descending and ascending limbs of the Loop of Henle, the distal convoluted tubule, and the collecting duct. The kidney’s ability to excrete waste is intricately tied to the specific placement of these structures and their physiology.
Bladder Structure
The bladder has a muscular lining known as the detrusor muscle. Parasympathetic activity causes the detrusor muscle to contract. However, in order to leave the body, urine must pass through two sphincters—the internal and external urethral sphincters. The internal urethral sphincter, consisting of smooth muscle, is contracted in its normal state. Because the internal sphincter is made of smooth muscle, it is under involuntary control. The external urethral sphincter consists of skeletal muscle and is under voluntary control. When the bladder is full, stretch receptors convey to the nervous system that the bladder requires emptying. This causes parasympathetic neurons to fire, and the detrusor muscle contracts. This contraction also causes the internal sphincter to relax. This reflex is known as the micturition reflex. The next step is up to the individual. The person can choose to relax the external sphincter to urinate, or can maintain the tone of the external sphincter to prevent urination. This can cause a few moments of discomfort, but the reflex usually dissipates in a few minutes. However, if the bladder is not emptied, then the process will begin anew shortly thereafter. Urination itself is facilitated by the contraction of the abdominal musculature, which increases pressure within the abdominal cavity, resulting in compression of the bladder and increased urine flow rate.
OSMOREGULATION
The kidney filters the blood to form urine. The composition and quantity of urine is determined by the present state of the body. For example, if blood volume is low and blood osmolarity is high, then it is most beneficial to the body to maximally retain water. This results in low-volume, highly concentrated urine. Likewise, a patient receiving large amounts of intravenous fluids is likely to produce a larger volume of less concentrated urine. Thus, the primary job of the kidneys is to regulate blood volume and osmolarity. In order to do this, kidney function may be divided into the three different processes: filtration, secretion, and reabsorption.
Filtration
The nephron’s first function is filtration. In the kidneys, approximately 20 percent of the blood that passes through the glomerulus is filtered as fluid into Bowman’s space. The collected fluid is known as the filtrate. The movement of fluid into Bowman’s space is governed by Starling forces, which account for the pressure differentials in both hydrostatic and oncotic pressures between the blood and Bowman’s space, as shown in Figure 10.4. The hydrostatic pressure in the glomerulus is significantly higher than that in Bowman’s space, which causes fluid to move into the nephron. On the other hand, the osmolarity of blood is higher than that of Bowman’s space, resulting in pressure opposing the movement of fluid into the nephron. However, the hydrostatic pressure is much larger than the oncotic pressure, so the net flow is still from blood into the nephron.
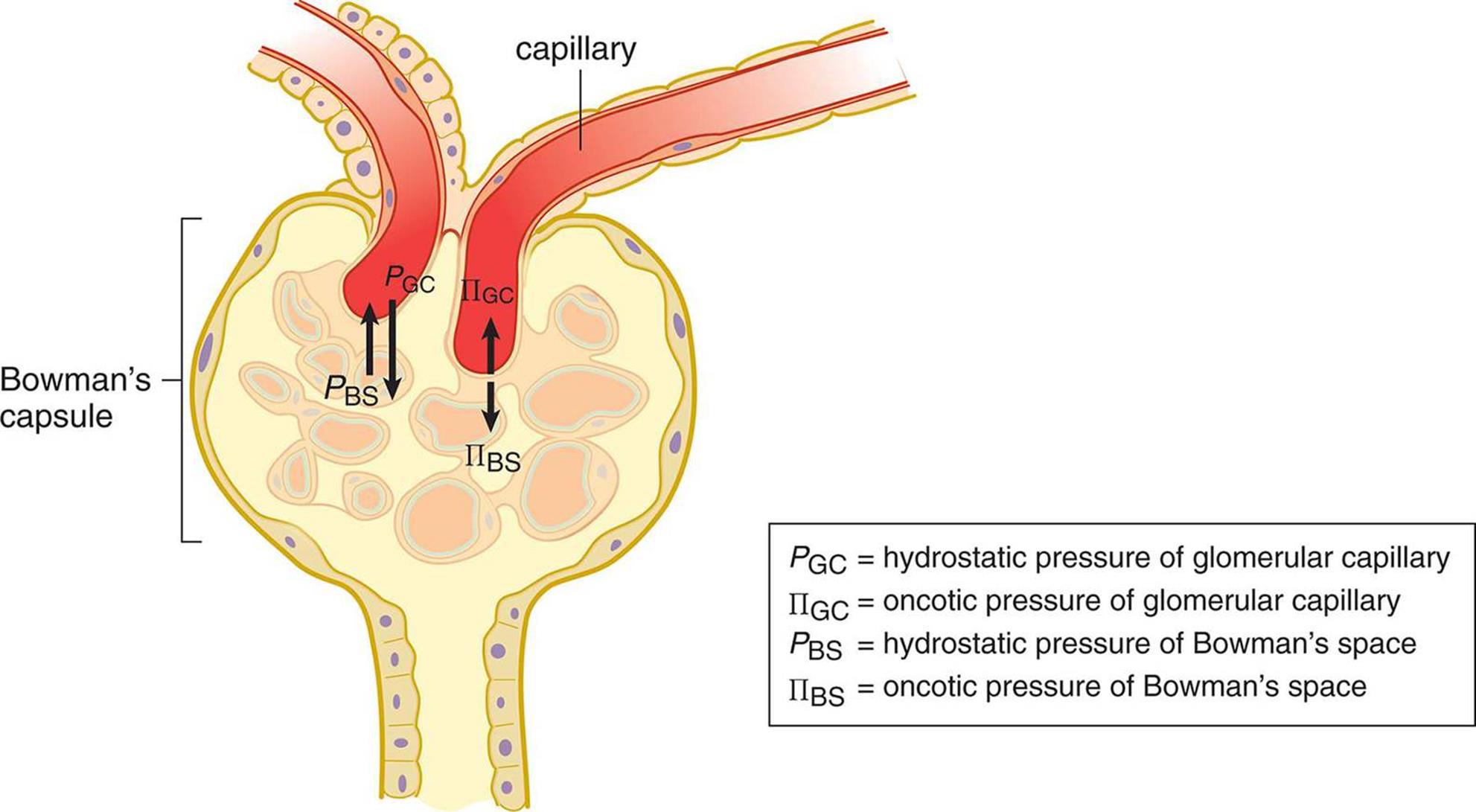 Figure 10.4. Starling Forces The relative hydrostatic and oncotic pressure gradients determine the direction and rate of filtration.
Figure 10.4. Starling Forces The relative hydrostatic and oncotic pressure gradients determine the direction and rate of filtration.
BRIDGE
Like the electromotive force discussed in Chapter 6 of MCAT Physics and Math Review, Starling forces are really a misnomer. Rather than forces, it is a pressure differential that causes the net movement of fluid from the glomerulus to Bowman’s space. Pressure is discussed in Chapter 4 of MCAT Physics and Math Review.
Under most circumstances, fluid will flow from the glomerulus into Bowman’s space. However, various pathologies can cause derangements of this flow. Consider what might happen if the ureter was obstructed by a kidney stone. An obstruction would result in a buildup of urine behind the stone. Eventually, enough fluid will build up and cause distention of the renal pelvis and the nephrons. What will happen to filtration in this case? The hydrostatic pressure in Bowman’s space would increase to the point that filtration could no longer occur because there would be excessive pressure opposing movement of fluid into the nephron.
The filtrate is similar in composition to blood but does not contain cells or proteins due to the filter’s ability to select based on size. In other words, molecules or cells that are larger than glomerular pores will remain in the blood. As described earlier, the blood remaining in the glomerulus then travels into the efferent arterioles, which empty into the vasa recta. The filtrate is isotonic to blood so that neither the capsule nor the capillaries swell. Our kidneys filter about 180 liters per day, which is approximately 36 times our blood volume. This means that the entire volume of a person’s blood is filtered about every 40 minutes.
KEY CONCEPT
Imagine that the glomerulus is like a sieve or colander. Small molecules dissolved in the blood will pass through the tiny pores (such as glucose, which is later reabsorbed), whereas large molecules such as proteins and blood cells will not. If blood cells or proteins are found in the urine, this indicates a health problem at the level of the glomerulus.
Secretion
In addition to filtering blood, the nephrons are able to secrete salts, acids, bases, and urea directly into the tubule by either active or passive transport. The quantity and identity of the substances secreted into the nephron are directly related to the needs of the body at that time. For example, a diet heavy in meat results in the intake of large amounts of protein, which contains a significant amount of nitrogen. Ammonia (NH3) is a byproduct of the metabolism of nitrogen-containing compounds and, as a base, can disturb the pH of blood and cells. The liver converts the ammonia tourea, a neutral compound, which travels to the kidney and is secreted into the nephron for excretion with the urine. The kidneys are capable of eliminating ions or other substances when present in relative excess in the blood, such as potassium cations, hydrogen ions, or metabolites of medications. Secretion is also a mechanism for excreting wastes that are simply too large to pass through glomerular pores.
Reabsorption
Some compounds that are filtered or secreted may be taken back up for use via reabsorption. Certain substances are almost always reabsorbed, such as glucose, amino acids, and vitamins. In addition, hormones such as antidiuretic hormone (ADH or vasopressin) and aldosterone can alter the quantity of water reabsorbed within the kidney in order to maintain blood pressure.
KEY CONCEPT
· Filtration: movement of solutes from blood to filtrate at Bowman’s capsule
· Secretion: movement of solutes from blood to filtrate anywhere besides Bowman’s capsule
· Reabsorption: movement of solutes from filtrate to blood
NEPHRON FUNCTION
The kidney uses mechanisms such as filtration, secretion, and reabsorption to produce urine and to regulate the blood volume and osmolarity. However, the function of the nephron isn’t quite that simple. In fact, renal physiology is often considered one of the most difficult topics covered in medical school.
In order to simplify this topic, it is important to understand that the kidney has two main goals: keep what the body needs and lose what it doesn’t, and concentrate the urine to conserve water. The kidney allows the human body to reabsorb certain materials for reuse, while also selectively eliminating waste. For example, glucose and amino acids are not usually present in the urine because the kidney is able to reabsorb these substances for later use. On the contrary, waste products like hydrogen and potassium ions, ammonia, and urea remain in the filtrate and are excreted. Finally, water is reabsorbed in large quantities in order to maintain blood pressure and adequate hydration.
KEY CONCEPT
Anything that makes it into the filtrate and that is not reabsorbed will be lost from the body in the urine.
In order to understand this complex organ, we will study the nephron piece-by-piece, discussing exactly what is occurring in each segment. Follow along with the nephron diagram shown in Figure 10.5. As a theme, note that segments that are horizontal in the diagram (Bowman’s capsule, the proximal convoluted tubule, and the distal convoluted tubule) are primarily focused on the identity of the particles in the urine (keep what the body needs and lose what it doesn’t). In contrast, the segments that are vertical in the diagram (the loop of Henle and collecting duct) are primarily focused on the volume and concentration of the urine (concentrate the urine to conserve water).
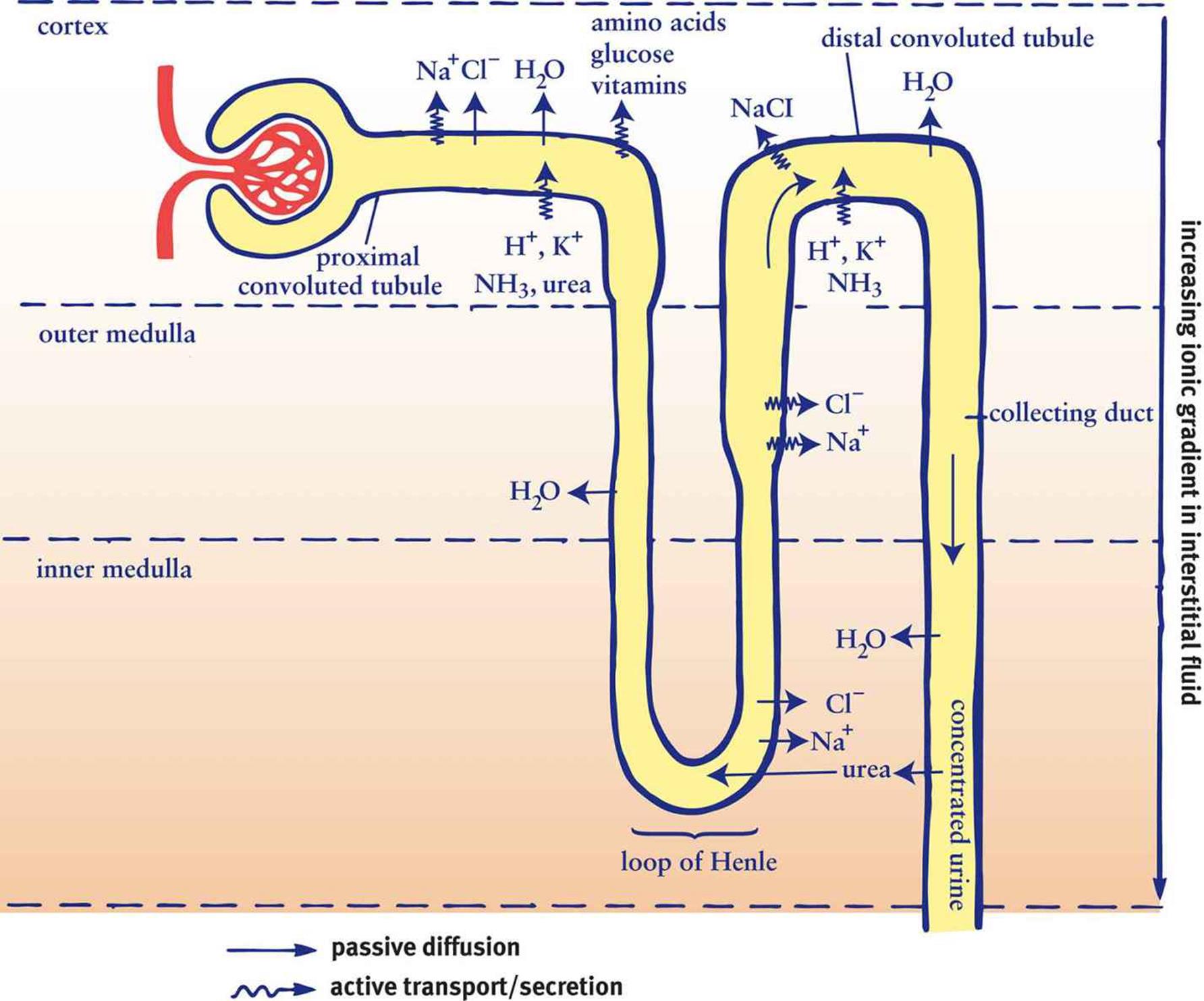 Figure 10.5. Reabsorption and Secretion in the Nephron
Figure 10.5. Reabsorption and Secretion in the Nephron
Proximal Convoluted Tubule
The filtrate first enters the proximal convoluted tubule (PCT). In this region, amino acids, glucose, water-soluble vitamins, and the majority of salts are reabsorbed along with water. Almost 70 percent of filtered sodium will be reabsorbed here, but the filtrate remains isotonic to the interstitium, as other solutes and a large volume of water are also reabsorbed. Solutes that enter the interstitium—the connective tissue surrounding the nephron—are picked up by the vasa recta to be returned to the bloodstream for reuse within the body. The PCT is also the site of secretion for a number of waste products, including hydrogen ions, potassium ions, ammonia and urea.
MNEMONIC
Major waste products excreted in the urine: Dump the HUNK
· H+
· Urea
· NH3
· K+
Loop of Henle
Filtrate from the proximal convoluted tubule then enters the descending limb of the loop of Henle, which dives deep into the medulla before turning around to become the ascending limb of the loop of Henle. The descending limb is permeable only to water, and the medulla has an ever-increasing osmolarity as the descending limb travels deeper into it. Think for a moment how this would affect the flow of water. As the descending limb traverses deeper into the medulla, the increasing interstitial concentration favors the outflow of water from the descending limb, which is reabsorbed into the vasa recta.
The kidney is capable of altering the osmolarity of the interstitium. This creates a gradient that, coupled with selective permeability of the nephron, allows maximal reabsorption and conservation of water. In the normal physiological state, the osmolarity in the cortex is approximately the same as that of the blood and remains at that level. Deeper in the medulla, the osmolarity in the interstitium can range from isotonic with blood (when trying to excrete water) to four times as concentrated (when trying to conserve water). Water will move out of the tubule, into the interstitium, and eventually back into the blood. If the concentration is the same in the tubule and in the interstitium, there is no driving force (gradient), and the water will be lost in urine.
Together, the vasa recta and nephron create a countercurrent multiplier system. What is meant by this is that the flow of filtrate through the loop of Henle is in the opposite direction from the flow of blood through the vasa recta. If the two flowed in the same direction, they would quickly reach equilibrium and the kidney would be unable to reabsorb as much water. By making the two flow in opposite directions, the filtrate is constantly being exposed to hypertonic blood, which allows maximal reabsorption of water.
As the descending limb transitions to become the ascending limb of the loop of Henle, a change in permeability occurs. The ascending limb is only permeable to salts and is impermeable to water. Here the opposite occurs: at the deeper parts of the medulla, salt concentrations are high, but decrease as the ascending limb rises. Thus, increasing amounts of salt are removed from the filtrate as it travels up the loop of Henle.
At the transition from the inner to outer medulla, the loop of Henle becomes thicker in what is termed the diluting segment. This is not because the lumen within the tube has enlarged, but because the cells lining the tube are larger. These cells contain large amounts of mitochondria, which allow the reabsorption of sodium and chloride by active transport. Indeed, because so much salt is reabsorbed while water is stuck in the nephron, the filtrate actually becomes hypotonic compared to the interstitium. While we tend to focus on the concentrating abilities of the nephron, this segment is noteworthy because it is the only portion of the nephron that can produce urine that is more dilute than the blood. This is important during periods of overhydration and provides a mechanism for eliminating excess water.
At the beginning of the loop of Henle, the filtrate is isotonic to the interstitium. Thus, from the beginning of the loop of Henle to the end, there is a slight degree of dilution. Far more important, however, is the fact that the volume of the filtrate has been significantly reduced, demonstrating a net reabsorption of a large volume of water.
Distal Convoluted Tubule
Next, the filtrate enters the distal convoluted tubule (DCT). The DCT responds to aldosterone, which promotes sodium reabsorption. Because sodium ions are osmotically active particles, water will follow the sodium, concentrating the urine and decreasing its volume. The DCT is also the site of waste product secretion, like the PCT.
REAL WORLD
In certain conditions, such as congestive heart failure, the body accumulates excess fluid in the lungs or peripheral tissues (edema). The judicious use of a diuretic drug can help the body get rid of excess fluids. Diuretics typically inhibit the reabsorption of sodium in one or more regions of the nephron, thereby increasing sodium excretion. As an osmotically active particle, sodium will pull water with it, thereby relieving the body of some of its excess fluid.
Collecting Duct
The final concentration of the urine will depend largely on the permeability of the collecting duct, which is responsive to both aldosterone and antidiuretic hormone (ADH or vasopressin). As permeability of the collecting duct increases, so too does water reabsorption, resulting in further concentration of the urine. The reabsorbed water enters the interstitium and makes its way to the vasa recta, where it reenters the bloodstream to once again become part of the plasma. The collecting duct almost always reabsorbs water, but the amount is variable. When the body is very well hydrated, the collecting duct will be fairly impermeable to salt and water. When in conservation mode, ADH and aldosterone will each act to increase reabsorption of water in the collecting duct, allowing for greater water retention and more concentrated urine output.
Ultimately, anything that does not leave the tubule by the end of the collecting duct will be excreted; the collecting duct is the point of no return. After that, there are no further opportunities for reabsorption. As the filtrate leaves the tubule, it collects in the renal pelvis. The fluid, which carries mostly urea, uric acid, and excess ions (sodium, potassium, magnesium, and calcium), flows through the ureter to the bladder where it is stored until voiding.
FUNCTIONS OF THE EXCRETORY SYSTEM
The kidneys use osmolarity gradients and selective permeability to filter, secrete, and reabsorb materials in the process of making urine. However, these processes have larger implications for the human body has a whole. The selective elimination of water and solutes allows the kidneys, in conjunction with the endocrine, cardiovascular, and respiratory systems, to control blood pressure, blood osmolarity, and acid–base balance.
Blood Pressure
In Chapter 5 of MCAT Biology Review, we discussed two hormones that are very important for maintenance of proper blood pressure: aldosterone and antidiuretic hormone (ADH or vasopressin).
Aldosterone is a steroid hormone that is secreted by the adrenal cortex in response to decreased blood pressure. Decreased blood pressure stimulates the release of renin from juxtaglomerular cells in the kidney. Renin then cleaves angiotensinogen, a liver protein, to form angiotensin I. This peptide is then metabolized by angiotensin-converting enzyme in the lungs to form angiotensin II, which promotes the release of aldosterone from the adrenal cortex.
REAL WORLD
Aldosterone increases blood pressure by increasing the reabsorption of sodium. In medicine, we exploit this characteristic by using drugs that actually block angiotensin-converting enzyme or the angiotensin II receptor. In Chapter 5 of MCAT Biology Review, we mentioned the ACE inhibitors, which mostly end with –pril. The angiotensin II receptor blockers mostly end with–sartan (losartan, valsartan, irbesartan) Blocking this receptor limits aldosterone release, which limits salt and water reabsorption, and therefore results in lowered blood pressure.
Aldosterone works by altering the ability of the distal convoluted tubule and collecting duct to reabsorb sodium. Remember that water does not move on its own, but rather travels down an osmolarity gradient. Thus, if we reabsorb more sodium, water will flow with it. This reabsorption of isotonic fluid has the net effect of increasing blood volume and therefore blood pressure. Aldosterone will also increase potassium and hydrogen ion excretion.
Antidiuretic hormone (ADH, also known as vasopressin) is a peptide hormone synthesized by the hypothalamus and released by the posterior pituitary in response to high blood osmolarity. It directly alters the permeability of the collecting duct, allowing more water to be reabsorbed by making the cell junctions of the duct leaky. Increased concentration in the interstitium (hypertonic to the filtrate) will then cause the reabsorption of water from the tubule. Alcohol and caffeine both inhibit ADH release and lead to the frequent excretion of dilute urine.
KEY CONCEPT
ADH only governs water reabsorption and thus results in a lower blood osmolarity. Aldosterone causes both salt and water reabsorption and does not change blood osmolarity.
In addition to the kidneys, the cardiovascular system can also vasoconstrict or vasodilate in order to maintain blood pressure. Constriction of the afferent arteriole will lead to a lower amount of blood reaching the glomeruli, which are adjacent to the juxtaglomerular cells. Therefore, this vasoconstriction will secondarily lead to renin release, which will also help raise blood pressure.
Osmoregulation
The osmolarity of the blood must be tightly controlled to ensure correct oncotic pressures within the vasculature. A note on terminology: osmotic pressure is the “sucking” pressure that draws water into the vasculature caused by all dissolved particles. Oncotic pressure, on the other hand, is the osmotic pressure that is attributable to dissolved proteins specifically. Blood osmolarity is usually maintained at approximately 290 milliosmoles (mOsm) per liter. As described earlier, the kidneys control osmolarity by modulating the reabsorption of water and by filtering and secreting dissolved particles. When blood osmolarity is low, excess water will be excreted, while solutes will be reabsorbed in higher concentrations. In contrast, when blood osmolarity is high, water reabsorption increases and solute excretion increases.
Acid–Base Balance
The bicarbonate buffer system is the major regulator of blood pH. Remind yourself of the buffer equation:
CO2 (g) + H2O (l) ⇋ H2CO3 (aq) ⇋ H+ (aq) + HCO–3 (aq)
In Chapter 6 of MCAT Biology Review, we talked about how the respiratory system can contribute to acid–base balance by increasing or decreasing the respiratory rate. If the blood pH is too low, then increasing the respiratory rate blows off more CO2 and favors the conversion of H+ and HCO–3 to water and carbon dioxide, increasing the pH. If the blood pH is too high, then decreasing the respiratory rate causes the opposite effects. The respiratory system can react to derangements of pH quickly. What can the excretory system do to contribute? The kidneys are able to selectively increase or decrease the secretion of both hydrogen ions and bicarbonate. When blood pH is too low, the kidneys excrete more hydrogen ions and increase reabsorption of bicarbonate, resulting in a higher pH. Likewise, when blood pH is too high, the kidneys can excrete more bicarbonate and increase reabsorption of hydrogen ions. This is slower than the respiratory response, but it is a highly effective way for the body to maintain acid–base balance.
MCAT Concept Check 10.1:
Before you move on, assess your understanding of the material with these questions.
1. List the structures in the excretory pathway, from where filtrate enters the nephron to the excretion of urine from the body.
2. List the vessels in the renal vascular pathway, starting from the renal artery and ending at the renal vein.
3. What arm of the nervous system is responsible for contraction of the detrusor muscle?
4. What are the three processes by which solutes are exchanged between the filtrate and the blood? What happens in each process?
·
·
·
5. For each segment of the nephron listed below, what are its major functions?
· Bowman’s capsule:
· Proximal convoluted tubule:
· Descending limb of the loop of Henle:
· Ascending limb of the loop of Henle:
· Distal convoluted tubule:
· Collecting duct: