Medical Microbiology
Section 4 Clinical manifestation and diagnosis of infections by body system
30 Infections in the compromised host
Introduction
The human body has a complex system of protective mechanisms to prevent infection. This involves both the adaptive (cellular and humoral) immune system and the innate defence system (e.g. skin, mucous membranes). (These have been described in detail in Chapters 910 and 11.) So far, we have concentrated on the common and serious infections occurring in people whose protective mechanisms are largely intact. In these circumstances, the interactions between host and parasite are such that the parasite has to use all its guile to survive and invade the host, and the healthy host is able to combat such an invasion. The focus of this chapter involves the infections that arise when the host defences are compromised, resulting in the host–parasite equation being weighted heavily in favour of the parasite.
The compromised host
Compromised hosts are people with one or more defects in their body’s natural defences against microbial invaders. Consequently they are much more liable to suffer from severe and life-threatening infections. Modern medicine has effective methods for treating many types of cancers, is improving organ transplantation techniques and has developed technology that enables people with otherwise fatal diseases to lead prolonged and productive lives. A consequence of these achievements, however, is an increasing number of compromised people prone to infection. In addition, viral infections including HIV and HTLV result in a compromised immune system referred to as AIDS and adult T-cell leukaemia/lymphoma (ATLL), respectively.
The host can be compromised in many different ways
Compromise can take a variety of forms, falling into two main groups:
• defects, accidental or intentional, in the body’s innate defence mechanisms
• deficiencies in the adaptive immune response.
These disorders of the immune system can be further subclassified as ‘primary’ or ‘secondary’ (Table 30.1):
• Primary immunodeficiency is inherited or occurs by exposure in utero to environmental factors or by other unknown mechanisms. It is rare, and varies in severity depending upon the type of defect.
• Secondary or acquired immunodeficiency is due to an underlying disease state (Table 30.2) or occurs as a result of treatment for a disease.
Table 30.1 Factors that make a host compromised
|
Factors affecting innate systems |
|
|
Primary |
Complement deficiencies, phagocyte cell deficiencies |
|
Secondary |
Burns, trauma, major surgery, catheterization, foreign bodies (e.g. shunts, prostheses), obstruction |
|
Factors affecting adaptive systems |
|
|
Primary |
T-cell defects, B-cell deficiencies, severe combined immunodeficiency |
|
Secondary |
Malnutrition, infectious diseases, neoplasia, irradiation, chemotherapy, splenectomy |
Table 30.2 Infections that cause immunosuppression
|
Viral |
Bacterial |
|
Measles |
Mycobacterium tuberculosis |
|
Mumps |
Mycobacterium leprae |
|
Congenital rubella |
|
Primary defects of innate immunity include congenital defects in phagocytic cells or complement synthesis
Congenital defects in phagocytic cells confer susceptibility to infection, and of these perhaps the best known is chronic granulomatous disease (Fig. 30.1), in which an inherited failure to synthesize cytochrome b245 leads to a failure to produce reactive oxygen intermediates during phagocytosis. As a result, the neutrophils cannot kill invading pathogens.
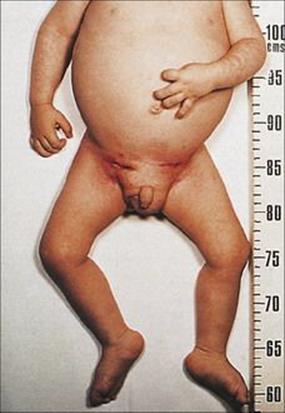
Figure 30.1 Bilateral draining lymph nodes in an 18-month-old boy with chronic granulomatous disease. Abscesses caused by Staphylococcus aureus had developed in both groins and had to be surgically drained.
(Courtesy of A.R. Hayward.)
The central role of complement in the innate defence mechanisms is undisputed, and inability to generate classical C3 convertase (see Ch. 10) through congenital defects in the synthesis of the early components, particularly C4 and C2, is associated with a high frequency of extracellular infections.
Secondary defects of innate defences include disruption of the body’s mechanical barriers
A variety of factors can disrupt the mechanical non-specific barriers to infection. For example, burns, traumatic injury and major surgery destroy the continuity of the skin and may leave poorly vascularized tissue near the body surface, providing a relatively defenceless site for microbes to colonize and invade. In health, the mucosal barriers of the respiratory and alimentary tract are vital to prevent infection. Damage sustained, for example, through endoscopy, surgery or radiotherapy, provides easy access for infecting organisms. Devices such as intravascular and urinary catheters, or procedures such as lumbar puncture or bone marrow aspiration, allow organisms to bypass the normal defences and enter normally sterile parts of the body. Foreign bodies such as prostheses, e.g. hip joints or heart valves, and cerebrospinal fluid (CSF) shunts alter the local non-specific host responses and provide surfaces that microbes can colonize more readily than the natural equivalents.
The adage ‘obstruction leads to infection’ is a valuable reminder that the defences of many body systems work partly through the clearance of undesirable materials, e.g. by urine flow, ciliary action in the respiratory tract, and peristalsis in the gut. Interference with these mechanisms as a result of pathologic obstruction, central nervous system dysfunction or surgical intervention tends to result in infection.
Primary adaptive immunodeficiency results from defects in the primary differentiation environment or in cell differentiation
The major congenital abnormalities arising in the adaptive immune system are depicted in Figure 30.2. A defect in the stromal microenvironment in which lymphocytes differentiate may lead to failure to produce B cells (Bruton-type agammaglobulinaemia) or T cells (DiGeorge syndrome).
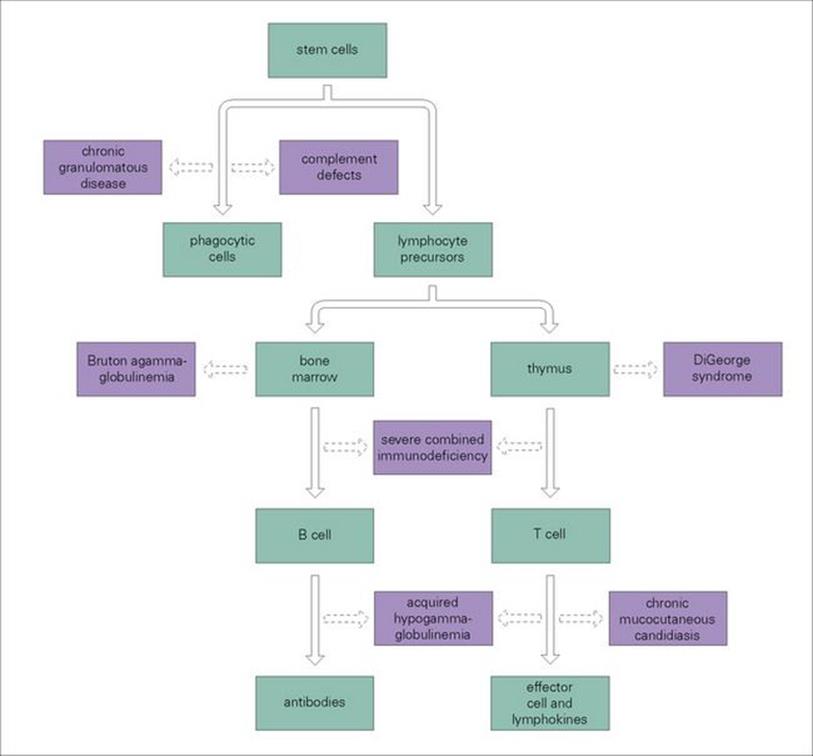
Figure 30.2 The major primary cellular immunodeficiencies. The deficiency states (shown in purple boxes) derive either from defects in the primary differentiation environment (bone marrow or thymus) or during cell differentiation (shown as dashed arrows derived from the differentiation state indicated).
Differentiation pathways themselves may also be affected. For example, a non-functional recombinase enzyme will prevent the recombination of gene fragments that form the B-cell antibody or the T-cell receptor variable regions for antigen recognition, with a resulting severe combined immunodeficiency (SCID).
The most common form of congenital antibody deficiency – common variable immunodeficiency – is characterized by recurrent pyogenic infections and is probably heterogeneous. Although the number of immature B cells in the marrow tends to be normal, the peripheral B cells are either low in number or in some cases absent. Where present, they are unable to differentiate into plasma cells in some cases or to secrete antibody in others.
Transient hypogammaglobulinaemia of infancy, characterized by recurrent respiratory infections, is associated with a low serum IgG concentration, which often normalizes abruptly by 3–4 years of age (Fig. 30.3).
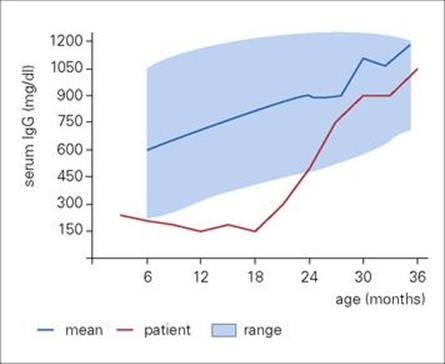
Figure 30.3 Serum immunoglobulin concentrations in a boy with transient hypogammaglobulinaemia compared with the range of normal controls. The patient developed mild paralytic polio when immunized at 4 months of age with live attenuated (Sabin) vaccine.
Immunoglobulin deficiency occurs naturally in human infants as the maternal serum IgG concentration decays. It is a serious problem in very premature babies as, depending on the gestational age, maternal IgG may not have crossed the placental barrier.
Causes of secondary adaptive immunodeficiency include malnutrition, infections, neoplasia, splenectomy and certain medical treatments
Worldwide, malnutrition is common and the most important cause of acquired immunodeficiency. The major form, protein–energy malnutrition (PEM) presents as a wide range of disorders, with kwashiorkor and marasmus at the two poles. It results in:
• drastic effects on the structure of the lymphoid organs (Fig. 30.4)
• gross reductions in the synthesis of complement components
• sluggish chemotactic responses of phagocytes
• lowered concentrations of secretory and mucosal IgA
• reduced affinity of IgG
• in particular, a serious deficit in circulating T-cell numbers (Fig. 30.5), leading to inadequate cell-mediated responses.
Infections themselves are often immunosuppressive (see Table 30.2), and none is more so than HIV infection, which gives rise to AIDS (see Ch. 21). Neoplasia of the lymphoid system frequently induces a state of reduced immunoreactivity, and splenectomy, for whatever reason, results in impaired humoral responses.
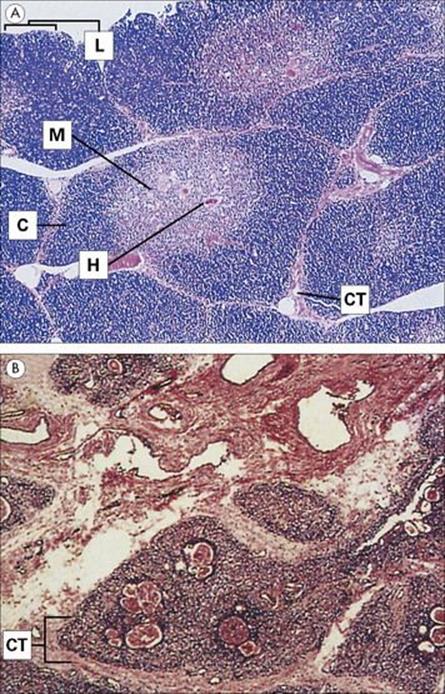
Figure 30.4 Thymic histology in normal children and children with protein–energy malnutrition (PEM). (A) Normal thymus showing a cortex and medullary zones. (B) Acute involution in PEM characterized by lobular atrophy, loss of distinction between cortex and medulla, depletion of lymphocytes and enlarged Hassall’s corpuscles. C, cortex; CT, connective tissue; H, Hassall’s corpuscle; L, lobule; M, medulla.
(Courtesy of R.K. Chandra.)
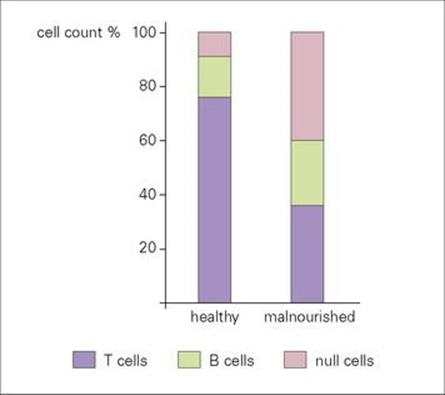
Figure 30.5 The proportion of T cells is decreased in malnourished patients compared with healthy controls. B-cell counts are usually unaltered, and lymphocytes lacking T- and B-cell markers are increased.
Treatment of disease can also cause immunosuppression. For example:
• Cytotoxic agents such as cyclophosphamide and azathioprine cause leukopenia or deranged T- and B-cell function.
• Corticosteroids reduce the number of circulating lymphocytes, monocytes and eosinophils and suppress leukocyte accumulation at sites of inflammation.
• Radiotherapy adversely affects the proliferation of lymphoid cells.
Therefore a patient receiving treatment for neoplastic disease will be immunocompromised as a result of both the disease and the treatment.
It is important to recognize immunodeficiencies and to understand which procedures are likely to compromise the natural defences of a patient. Due to improvements in medical technology, many immune defects, particularly immunosuppression resulting from radiotherapy or cytotoxic drugs, are transient, and patients who survive the period of immunosuppression have a good chance of a complete recovery.
Microbes that infect the compromised host
Immunocompromised people can become infected with any pathogen able to infect immunocompetent individuals as well as those opportunist pathogens that do not cause disease in a healthy person. They may be lethal when the host defences are lowered. Different types of defect predispose to infection with different pathogens depending upon the critical mechanisms operating in the defence against each microorganism (Fig. 30.6). Here, we will concentrate mainly on the opportunist infections and refer to other chapters for information about other pathogens.
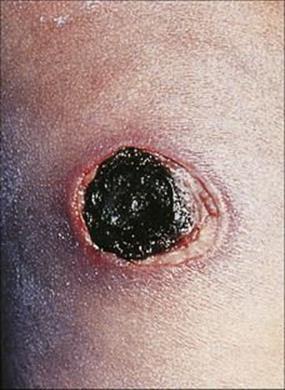
Figure 30.6 Ecthyma gangrenosum in a child with Pseudomonas septicaemia associated with immunodeficiency.
(Courtesy of H. Tubbs.)
Infections of the host with deficient innate immunity due to physical factors
Burn wound infections
Burns damage the body’s mechanical barriers, neutrophil function and immune responses
Burn wounds are sterile immediately after the burn is inflicted, but inevitably become colonized within hours with a mixed bacterial flora. Burn injuries cause direct damage to the mechanical barriers of the body and abnormalities in neutrophil function and immune responses. In addition, there is a major physiologic derangement with loss of fluids and electrolytes. The burn provides a highly nutritious surface for organisms to colonize, and the incidence of serious infection varies with the size and depth of the burn and the age of the patient. Topical antimicrobial therapy should prevent infection of burns of < 30% of the total body area, but larger burns are always colonized. Non-invasive infection is confined to the eschar, which is the non-viable skin debris on the surface of deep burns. It is characterized by rapid separation of the eschar from the underlying tissue and a heavy exudate of purulent material from the burn wound. The systemic symptoms are usually relatively mild. However, organisms can invade from heavily colonized burn eschars into viable tissue beneath and rapidly destroy the tissue, converting partial-thickness burns into full skin-thickness destruction. From here, it is a small step to invasion of the lymphatics and thence to the bloodstream or direct invasion of blood vessels, and to septicaemia. Septicaemia in patients with burns is often polymicrobial.
The major pathogens in burns are aerobic and facultatively anaerobic bacteria and fungi
The most important pathogens in burn wounds are:
• Pseudomonas aeruginosa and other Gram-negative rods
• Staphylococcus aureus
• Streptococcus pyogenes
• other streptococci
• enterococci.
Candida spp. and Aspergillus together account for about 5% of infections. Anaerobes are rare in burn wound infections. Herpesvirus infections have been reported and are most likely due to reactivation at a damaged skin site.
P. aeruginosa is a devastating Gram-negative pathogen of burned patients
P. aeruginosa is an opportunist Gram-negative rod that has a long and infamous association with burn infections. It grows well in the moist environment of a burn wound, producing a foul, green-pigmented discharge and necrosis. Invasion is common, and the characteristic skin lesions (ecthyma gangrenosum) that are pathognomonic of P. aeruginosa septicaemia may appear on non-burned areas (see Fig. 30.6). Host factors predisposing to infection include:
• abnormalities in the antibacterial activities of neutrophils
• deficiencies in serum opsonins.
Added to these are the virulence factors of the organism, which include the production of elastase, protease and exotoxin. This combination makes P. aeruginosa the most devastating Gram-negative pathogen of burned patients. Treatment is difficult because of the organism’s innate resistance to many antibacterial agents. A combination of aminoglycoside, usually gentamicin or tobramycin, with one of the beta-lactams such as azlocillin, ceftazidime or imipenem is usually favoured, but several units have reported strains resistant to these agents.
It is virtually impossible to prevent colonization. Prevention of infection depends largely on inhibiting the multiplication of organisms colonizing the burn by applying topical agents such as silver nitrate.
Staph. aureus is the foremost pathogen of burn wounds
The most important predisposing factor to Staph. aureus infection in burns patients appears to be an abnormality of the antibacterial function of neutrophils. Infections follow a more insidious course than streptococcal infections (see below), and it may be several days before the full-blown infection is apparent. The organism is capable of destroying granulation tissue, invading and causing septicaemia. Staph. aureus infections of skin are discussed in detail in Chapter 26. Treatment with antistaphylococcal agents such as cloxacillin or nafcillin (or a glycopeptide if methicillin-resistant Staph. aureus is isolated) should be administered if there is evidence of invasive infection. Every effort should be made to prevent the spread of staphylococci from patient to patient. Although transmissible by both air-borne and contact routes, the contact route is by far the more important.
The high transmissibility of Strep. pyogenes makes it the scourge of burns wards
Strep. pyogenes (group A strep) infections of skin and soft tissue are discussed in some detail in Chapter 23. Strep. pyogenes was the most common cause of burn wound infection in the pre-antibiotic era and is still to be feared in burns wards. The infection usually occurs within the first few days of injury and is characterized by a rapid deterioration in the state of the burn wound and invasion of neighbouring healthy tissue. The patient may become severely toxic and will die within hours unless treated appropriately. Strep. pyogenes rarely infects healthy granulation tissue, but freshly grafted wounds may become infected, resulting in destruction of the graft. Every effort should be made to prevent spread. Penicillin is the drug of choice for treatment, and erythromycin or vancomycin can be used for penicillin-allergic patients.
Beta-haemolytic streptococci of other Lancefield groups (notably groups C and G) and enterococci are also important pathogens of burn wounds.
Traumatic injury and surgical wound infections
Both accidental and intentional trauma destroy the integrity of the body surface and leave it liable to infection. Accidental injury may result in microbes being introduced deep into the wound. The species involved will depend upon the nature of the wound, as discussed in Chapter 26.
Staph. aureus is the most important cause of surgical wound infection
Staph. aureus surgical wound infection (see Ch. 36) may be acquired during surgery or postoperatively and may originate from the patient or from another patient or staff member. The wound is less well defended than normal tissue; it may have a damaged blood supply and there may be foreign bodies such as sutures. Classic studies of wound infections have shown that far fewer staphylococci are needed to initiate infection around a suture than in normal healthy skin. Wound infections can be severe and the organisms can invade the bloodstream, with consequent seeding of other sites such as the heart valves, causing endocarditis (see Ch. 29) or bones, causing osteomyelitis (see Ch. 26), thereby further compromising the patient.
Catheter-associated infection of the urinary tract is common
Urinary catheters disrupt the normal host defences of the urinary tract and allow organisms easy access to the bladder. Such catheter-associated infection of the urinary tract is especially common if catheters are left in place for > 48 h (see Ch. 20). The organisms involved are usually Gram-negative rods from the patient’s own faecal or periurethral flora, but cross-infection also occurs (see Ch. 36).
Staphylococci are the most common cause of intravenous and peritoneal dialysis catheter infections
Intravenous and peritoneal dialysis catheters breach the integrity of the skin barrier and allow organisms from the skin flora of the patient or hands of the carer easy access to deeper sites. Staphylococci are the most common cause of infection, but coryneforms, Gram-negative rods andCandida are also implicated.
Coagulase-negative staphylococci, particularly Staph. epidermidis, account for more than 50% of the infections (Table 30.3). These opportunists are members of the normal skin flora and for many years were considered to be harmless. However, they have a particular propensity for colonizing plastic and can therefore seed sites adjacent to plastic devices and thence cause invasive infections. Their virulence factors are not well understood, but their ability to produce an adhesive slime material and grow as biofilms on plastic surfaces is likely to be important. Infections are characteristically more insidious in onset than those caused by the more virulent Staph. aureus, and recognition is hampered by the difficulty in distinguishing the infecting strain from the normal flora. Treatment is also difficult because many Staph. epidermidis carry multiple antibiotic resistances, and agents such as a glycopeptide (vancomycin or teicoplanin) and rifampicin may be required (see Ch. 33). Whenever possible, the plastic device should be removed.
Table 30.3 Percentage of infections caused by Staph. epidermidis in patients with plastic devices in situ
|
Infection of: |
Infections caused by Staph. epidermidis (%) |
|
Prosthetic heart valve |
|
|
Early (< 2 months postoperatively) |
30–70 |
|
Late (> 2 months postoperatively) |
20–30 |
|
Prosthetic hip |
10–40 |
|
Cerebrospinal fluid shunt |
30–65 |
|
Vascular grafts |
5–20 |
|
Peritoneal dialysis related |
30 |
|
Intravascular catheters |
10–50 |
Data from Gemmell and McCartney.
Infections of plastic devices in situ
The technical developments in plastics and other synthetic materials have enabled many advances in medicine and surgery, but in the process have produced further ways of introducing infectious agents. Staph. epidermidis is an important cause of infection of cardiac pacemakers, vascular grafts and CSF shunts.
Staph. epidermidis is the most common cause of prosthetic valve and joint infections
Patients with prosthetic heart valves or prosthetic joints are compromised by:
• the surgery to implant the prosthesis
• the continued presence of a foreign body.
Staph. epidermidis is again the most common pathogen, gaining access either during surgery or from a subsequent bacteraemia originating from, for example, an intravascular line infection. Endocarditis associated with prosthetic heart valves is discussed in Chapter 29.
The most common complication of joint replacement is loosening of the prosthesis, while infection is the second most common complication and is much more likely to lead to permanent failure of the procedure. The difficulties of treatment have been outlined above, but there is understandably great reluctance to remove a prosthetic device, even though it is sometimes the only way to eradicate an infection.
Infections due to compromised clearance mechanisms
Stasis predisposes to infection, and in health the body functions to prevent stasis. In the respiratory tract, damage to the ciliary escalator predisposes the lungs to invasion, particularly in patients with cystic fibrosis, who are infected with Staph. aureus and Haemophilus influenzae and later with P. aeruginosa (see Ch. 19).
Obstruction and interruption of normal urine flow allows Gram-negative organisms from the periurethral flora to ascend the urethra and to establish themselves in the bladder. Septicaemia is an important complication of urinary tract infection superimposed on obstruction.
Infections associated with secondary adaptive immunodeficiency
The underlying immunodeficiency state determines the nature and severity of any associated infection, and in some cases infection is the presenting clinical feature in a patient with an immunologic deficit. However, septicaemia and related infectious complications of immunodeficiency are most commonly encountered in patients hospitalized for chemotherapy for malignant diseases or organ transplantation. In these patients, infection continues to be a major cause of morbidity and mortality (Table 30.4). Increasingly, these infections are iatrogenic and caused by opportunist pathogens acquired in hospital.
Table 30.4 Examples of opportunistic pathogens in immunocompromised hosts
|
Bacteria Gram-positive Staphylococcus aureus Coagulase-negative staphylococci Streptococci Listeria spp. Nocardia asteroides Mycobacterium tuberculosis Mycobacterium avium-intracellulare Gram-negative Enterobacteriaceae Pseudomonas aeruginosa Legionella spp. Bacteroides spp. |
|
Fungi Candida spp. Aspergillus spp. Cryptococcus neoformans Histoplasma capsulatum Pneumocystis jiroveciia |
|
Parasites Toxoplasma gondii Strongyloides stercoralis |
|
Viruses Herpesviruses, e.g. HSV, CMV, VZV, EBV, HHV-6, HHV-7, HHV-8 Hepatitis B Hepatitis Cb Polyomaviruses, e.g. BKV, JCV Adenoviruses HIVb |
a Formerly P. carinii. BKV, BK virus; JCV, JC virus; CMV, cytomegalovirus; EBV, Epstein–Barr virus; HHV, human herpesvirus; HSV, herpes simplex virus; VZV, varicella-zoster virus.
b HIV and HCV have been transmitted via organ transplantation and unscreened blood.
Haematologic malignancy and bone marrow transplant infections
A lack of circulating neutrophils following bone marrow failure predisposes to infection
Susceptibility to infection of patients with leukaemia is primarily due to the lack of circulating neutrophils that inevitably follows bone marrow failure. Septicaemia may be the presenting feature, but is much more common when the patient has been exposed to chemotherapy to induce a remission of the disease (remission-induction chemotherapy). Neutropenia, defined as a count of < 0.5 × 109 neutrophils/L, may persist for a few days to several weeks. Similarly, prolonged periods of neutropenia occur after bone marrow transplantation.
The length of time over which the patient is neutropenic influences the nature of any associated infection and the frequency with which it occurs. For example, fungal infections are much more common in patients who are neutropenic for more than 21 days. Although Gram-negative rods such as Escherichia coli and P. aeruginosa from the bowel flora have in the past been the most common cause of septicaemia in neutropenic patients, Gram-positive organisms such as staphylococci, streptococci and enterococci are also important. Staph. epidermidis septicaemia associated with intravascular catheters (see above) is common. Infections caused by fungi are also increasing, partly because more patients are surviving the early neutropenic period with the aid of modern antibacterial agents and granulocyte transfusions. Viruses such as CMV infections are an important feature of bone marrow transplantation and are associated with graft-versus-host disease as well as immunosuppressive therapy. In addition, adenovirus, EBV and BK virus infections may be seen, especially in allogeneic bone marrow transplant recipients.
Solid organ transplant infections
Most infections occur within 3–4 months of transplantation
Suppression of a patient’s cell-mediated immunity is necessary to prevent rejection of a grafted organ, and the cytotoxic regimens used usually suppress humoral immunity to some extent as well. In addition, high doses of corticosteroids to suppress inflammatory responses are required. The combination of these factors results in a severely compromised host, and those that have an effect on infection in recipients of solid organ transplants include:
• the underlying medical condition of the patient
• the patient’s previous immune status
• the type of organ transplant
• the immunosuppressive regimen
• the exposure of the patient to pathogens.
The organisms that cause the most common and most severe infections are shown in Table 30.4. Some of the viral infections are latent and reactivate when cell-mediated surveillance is suppressed.
From 3 to 4 months after transplantation, the risk of infection is reduced, but remains for as long as the patient is immunosuppressed.
HIV infection leading to AIDS
The clinical definition of AIDS includes the presence of one or more opportunistic infections
People with AIDS are often infected concomitantly with multiple pathogens, which they fail to eradicate despite prolonged, appropriate and aggressive antimicrobial chemotherapy. Most of the pathogens involved are intracellular microbes that require an intact cell-mediated immune response for effective defence. As the HIV-infected individual progresses to AIDS (see Ch. 21), organisms that are usually controlled by cell-mediated immunity are able to reactivate to cause disseminated infections not seen in the immunologically normal individual. Improved immune surveillance as a result of antiretroviral therapy has reduced the incidence of infections that are the hallmark of AIDS including Candida, Kaposi’s sarcoma and other opportunist pathogens described in more detail below.
Many of the pathogens that cause infections in the immunocompromised host (Table 30.4) are described elsewhere in this book.
Other important opportunist pathogens
Fungi
Candida is the most common fungal pathogen in compromised patients
This yeast is an opportunist pathogen in a variety of patients and in various body sites. It is the cause of:
• vaginal and oral thrush (see Ch. 21)
• skin infections (see Ch. 26)
• endocarditis, particularly in intravenous drug users (see Ch. 29).
Candida manifests itself in different ways depending upon the nature of the underlying compromise:
• Chronic mucocutaneous candidiasis. This is rare and is a persistent but non-invasive infection of mucous membranes, hair, skin and nails in patients, often children, with a specific T-cell defect rendering them anergic to Candida (Fig. 30.7). It may require repeated or long-term treatment with azole antifungal drugs. Diminished sensitivity to these agents may occur after repeated use.
• Oropharyngeal and oesophageal candidiasis. This is seen in a variety of compromised patients, including HIV-infected individuals (Fig. 30.8), people with ill-fitting dentures, diabetes mellitus or on antibiotics or corticosteroids. Oropharyngeal candidiasis generally responds to treatment with antifungal mouthwashes (nystatin or an azole compound). Those cases which do not respond can be treated with fluconazole. Oesophageal candidiasis requires systemic therapy.
• Gastrointestinal candidiasis. This is seen in patients who have undergone major gastric or abdominal surgery and in those with neoplastic disease. The organism can pass through the intestinal wall and spread from a gastrointestinal focus. Antemortem diagnosis is difficult, and as many as 25% of patients do not have any symptoms in the early stages of disease. If there is dissemination from the gut, blood cultures may become positive and Candida antigens may be detectable in the serum. A high index of suspicion is required to initiate antifungal therapy early in these patients, but disseminated disease is often fatal.
• Disseminated candidiasis. This is probably acquired via the gastrointestinal tract, but also arises from intravascular catheter-related infections. Patients with lymphoma and leukaemia are most at risk. Blood-borne spread to almost any organ can occur. Infections of the eye (endophthalmitis;Fig. 30.9) and the skin (nodular skin lesions; see Ch. 26) are important because they provide diagnostic clues, and without these the non-specific symptoms of fever and septic shock make early diagnosis difficult. Immunocompromised patients are often given antifungal therapy ‘blindly’ if they have a fever and fail to respond to broad-spectrum antibacterial agents (Fig. 30.10).
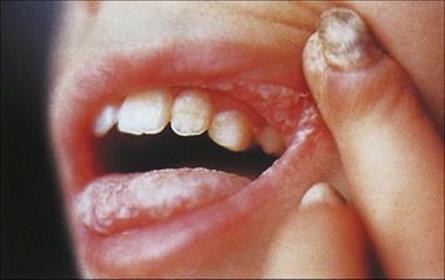
Figure 30.7 Chronic mucocutaneous candidiasis in a child with impaired T-cell response to antigens.
(Courtesy of M.J. Wood.)
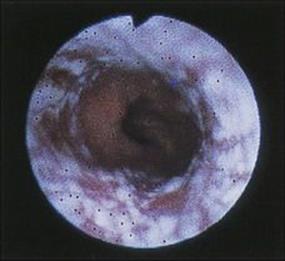
Figure 30.8 Candida oesophagitis. Endoscopic view showing extensive areas of whitish exudate.
(Courtesy of I. Chesner.)
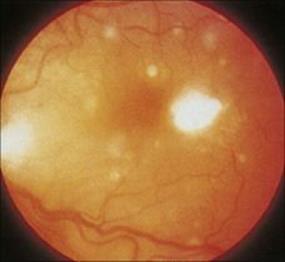
Figure 30.9 Candida endophthalmitis. Fundal photograph showing areas of white exudate.
(Courtesy of A.M. Geddes.)
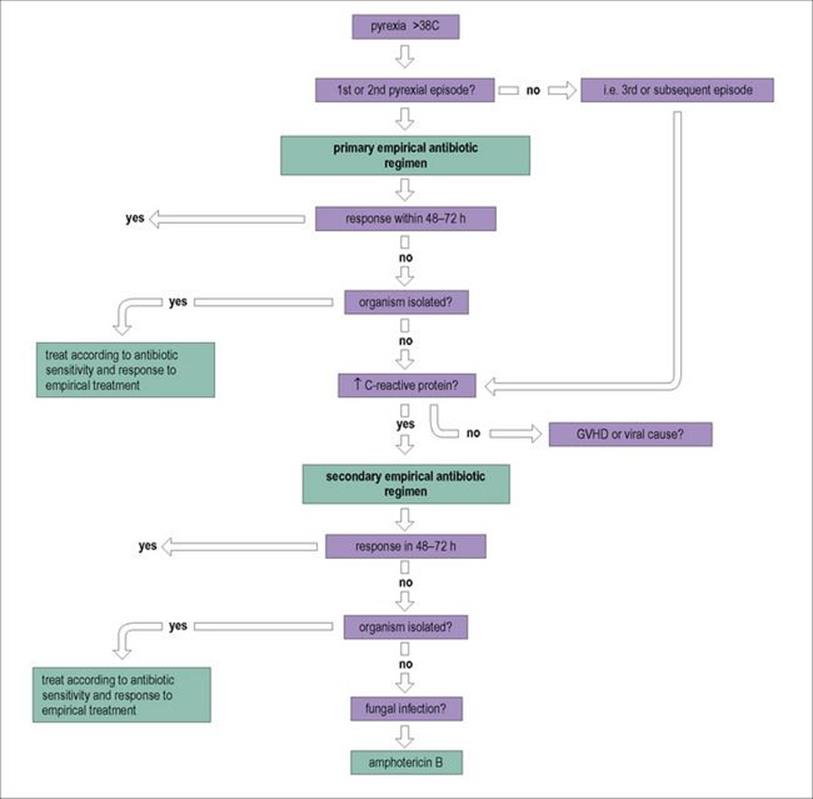
Figure 30.10 Neutropenic patients succumb very rapidly to infections, and decisions to treat have to be made on an empiric basis. This figure shows one example of such a decision-making tree. GVHD, graft-versus-host disease.
(Adapted from: Rogers.)
Cryptococcus neoformans infection is most common in people with impaired cell-mediated immunity
C. neoformans is an opportunistic yeast with a worldwide distribution. It can cause infection in the immunocompetent host, but infection is seen more frequently in people with impaired cell-mediated immunity. The onset of disease may be slow and usually results in lung infection or meningoencephalitis; occasionally other sites such as skin, bone and joints are involved (see Ch. 26).
C. neoformans can be demonstrated in the CSF and is characterized by its large polysaccharide capsule (see Fig. 24.6). Rapid identification can be made by antigen detection in a latex agglutination test using specific antibody-coated latex particles. Treatment involves a combination of amphotericin and flucytosine (see Ch. 33) and can be monitored by detecting a fall in CSF antigen concentration. The prognosis depends largely upon the patient’s underlying disease; in the severely immunocompromised, mortality is approximately 50%. In patients with AIDS it is almost impossible to eradicate the organism even with intensive treatment. Fluconazole can be given as post-treatment prophylaxis.
Disseminated Histoplasma capsulatum infection may occur years after exposure in immunocompromised patients
This is a highly infectious fungus that causes an acute but benign pulmonary infection in healthy people, but can produce chronic progressive disseminated disease in the compromised host. The organism is endemic only in tropical parts of the world and notably in the so-called ‘histoplasmosis belt’ of the central USA, particularly in the Ohio and Mississippi river valleys. The natural habitat of the organism is the soil. It is transmitted by the air-borne route and the fungal spores are deposited in the alveoli, from whence the fungus spreads via the lymphatics to the regional lymph nodes. As disseminated disease may occur many years after the initial exposure in immunocompromised patients it may present in patients who have long since left endemic areas. The infection may occur in HIV-infected individuals who have visited such regions.
Cultures of blood, bone marrow, sputum and CSF may yield Histoplasma, but biopsy and histologic examination of bone marrow, liver or lymph nodes is often required to make the diagnosis (Fig. 30.11). Approximately 50% of cases of progressive disease in the immunocompromised are successfully treated with amphotericin. Itraconazole can be given for post-treatment prophylaxis.
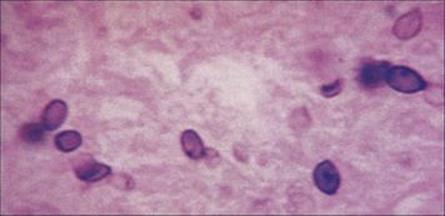
Figure 30.11 Histologic section of the lung showing yeast forms of Histoplasma capsulatum. Methenamine silver stain.
(Courtesy of T.F. Sellers, Jr.)
African histoplasmosis, caused by Histoplasma duboisii, is found in Equatorial Africa. Patients may present with localized cutaneous or disseminated disease.
Invasive aspergillosis has a very high mortality rate in the compromised patient
The role of Aspergillus spp. in diseases of the lung has been outlined in Chapter 19, but this fungus is now increasingly reported as a cause of invasive disease in compromised patients, usually in profoundly neutropenic patients or those receiving high-dose corticosteroids (Fig. 30.12). LikeHistoplasma, aspergilli are found in soil, but have a worldwide distribution. Infection is spread by the air-borne route, and the lung is the site of invasion in almost every case. Dissemination to other sites, particularly the central nervous system (Fig. 30.13) and heart, occurs in about 25% of compromised individuals with lung infection. Diagnosis involves microscopy, culture, antigen detection and PCR on bronchoalveolar lavage specimens. Lung biopsy may be required to make a tissue diagnosis.
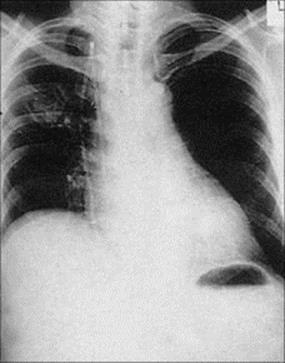
Figure 30.12 Chest radiograph showing invasive aspergillosis in the right lung of a patient with acute myeloblastic leukaemia.
(Courtesy of C. Kibbler.)
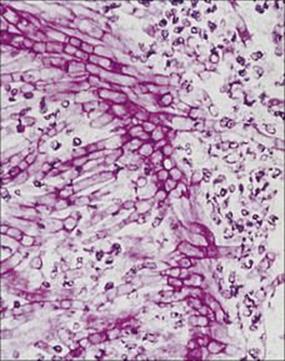
Figure 30.13 Numerous septate hyphae invading a blood vessel wall in cerebral aspergillosis. (Periodic acid-Schiff stain.)
(Courtesy of W.E. Farrar.)
Invasive aspergillosis has a high fatality rate in the compromised patient. Prophylactic antifungal agents such as caspofungin, posaconazole and voriconazole, early diagnosis and institution of treatment using an intravenous lipid formulation of amphotericin B known as liposomal amphotericin B complexes or AmBisome (see Ch. 33), together with a reduction in corticosteroid and cytotoxic therapy wherever possible, appear to improve the prognosis. Outbreaks of hospital-acquired infection have been reported (see Ch. 36), especially in relation to recent building work.
Pneumocystis jirovecii (formerly P. carinii) only causes symptomatic disease in people with deficient cellular immunity
P. jirovecii is an atypical fungus which appears to be widespread; a large proportion of the population has antibodies to the organism, but it only causes symptomatic disease in people whose cellular immune mechanisms are deficient. There is therefore a high incidence of P. jiroveciipneumonia in patients receiving immunosuppressive therapy to prevent transplant rejection and in people with HIV. It is very rare to find Pneumocystis infection in any other site in the body, but the reason for this is unknown.
Diagnosis is not easy and requires a high index of suspicion. The symptoms are non-specific and can mimic a variety of other infectious and non-infectious respiratory diseases. In addition, unlike the other fungi described above, the organism cannot be isolated in expectorated sputum using conventional culture methods, and invasive techniques such as bronchoalveolar lavage or open lung biopsy are required. In samples obtained by these techniques, the organism can be demonstrated by silver or immunofluorescent stains (Fig. 30.14). DNA amplification by the polymerase chain reaction improves the sensitivity of the diagnostic tests.
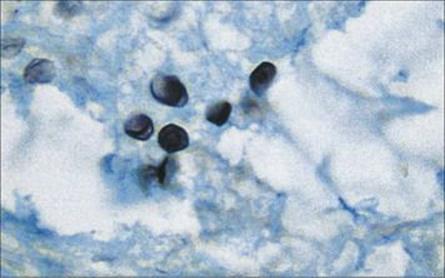
Figure 30.14 Darkly staining cysts of Pneumocystis jirovecii in an open lung biopsy from an AIDS patient with pneumonia. (Grocott silver stain.)
(Courtesy of M. Turner-Warwick.)
Treatment is with high dose co-trimoxazole (trimethoprim- sulfamethoxazole). Pentamidine is an alternative (see Ch. 33). Adjunctive corticosteroid therapy is given in moderate to severe infections in HIV co-infected individuals. Co-trimoxazole is used prophylactically.
Bacteria
Nocardia asteroides is an uncommon opportunist pathogen with a worldwide distribution
The family Actinomycetes, relatives of the mycobacteria, but resembling fungi in that they form branching filaments, contain two pathogenic genera, Actinomyces and Nocardia. N. asteroides infections have been reported in the immunocompromised, especially in renal transplant patients. The lung is usually the primary site, but infection can spread to the skin, kidney or central nervous system (Fig. 30.15). As with Aspergillus, hospital outbreaks of nocardiosis have been described.
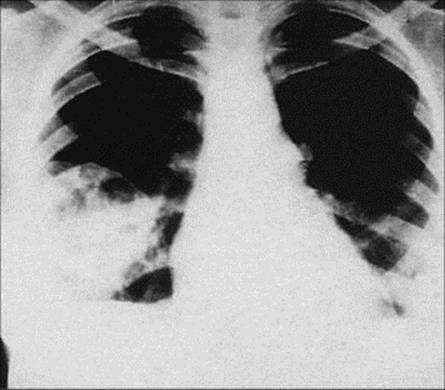
Figure 30.15 Pulmonary nocardiosis. Chest radiograph showing a large rounded lesion in the right lower zone with multiple cavities.
(Courtesy of T.F. Sellers, Jr.)
Nocardia can be isolated on routine laboratory media, but is often slow to grow and is consequently easily overgrown by commensal flora. Therefore the laboratory staff should be informed if nocardiosis is suspected clinically, so that appropriate media are inoculated. The organism is a Gram-negative branching rod and weakly acid fast (Fig. 30.16).
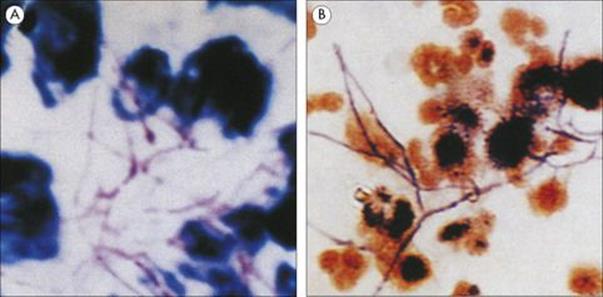
Figure 30.16 Nocardia asteroides in sputum. (A) Acid-fast stain. (Courtesy of T.F. Sellers, Jr.) (B) Gram’s stain.
(Courtesy of H.P. Holley.)
Sulphonamides or co-trimoxazole are the drugs of choice, but treatment can be difficult and various other regimens involving tetracycline, aminoglycosides or imipenem have been described.
Mycobacterium avium-intracellulare disease is often a terminal event in AIDS
Although mycobacterial infections are well documented in immunosuppressed patients, the association between AIDS and mycobacteria includes disseminated infection with Mycobacterium tuberculosis and Mycobacterium avium-intracellulare (Mycobacterium avium complex or MAC). These organisms can be isolated from blood cultures from patients with AIDS. M. tuberculosis has been described in detail in Chapter 19. M. avium-intracellulare belongs to the so-called ‘atypical’ mycobacteria or mycobacteria other than tuberculosis (MOTT). It resembles M. tuberculosisin that it is slow-growing, but it is resistant to the conventional antituberculosis drugs. Multidrug therapy with combinations such as clofazimine or rifamycin derivatives together with macrolides such as azithromycin or clarithromycin, quinolones, isoniazid, ethambutol, cycloserine or pyrazinamide have been recommended.
Protozoa and helminths
Cryptosporidium and Isospora belli infections cause severe diarrhea in AIDS
Cryptosporidium (Fig. 30.17) is a protozoan parasite that causes human disease, and is well known to veterinarians as an animal pathogen. It causes significant but self-limiting diarrhea in healthy normal people (see Ch. 22), but severe and chronic diarrhea in people with AIDS. Highly active antiretroviral therapy in individuals with AIDS infected with Cryptosporidium has been reported to improve the diarrhea symptoms. Paromomycin reduces oocyst output but does not clear infection. Nitazoxanide is effective in HIV-negative patients but is only partially active in those co-infected with HIV. Isospora belli (Fig. 30.18) is a parasite very similar to Cryptosporidium and also produces severe diarrhea in people with AIDS. Unlike Cryptosporidium, however, it is susceptible to co-trimoxazole.
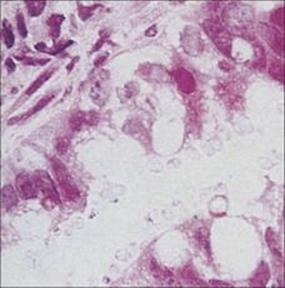
Figure 30.17 Numerous organisms in the brush border of the intestine in cryptosporidiosis.
(Courtesy of J. Newman.)
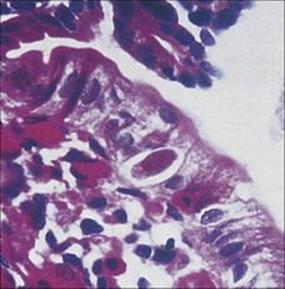
Figure 30.18 Human coccidiosis, with a single Isospora belli organism within an epithelial cell and a chronic inflammatory reaction in the lamina propria.
(Courtesy of G.N. Griffin.)
Cyclospora cayetanensis, also related to Cryptosporidium, likewise produces prolonged and severe diarrhea in immunosuppressed individuals. Co-trimoxazole treatment is effective. Ciprofloxacin is partially effective.
Infections with microsporidia also cause diarrhea in people with AIDS and other immunosuppressed patients. Enterocytozoon bieneusi is the most common cause, although Encephalitozoon intestinalis also occurs. Albendazole treatment is effective against Encephalitozoon intestinalis but has disappointing activity against Enterocytozoon bieneusi. Where feasible, immune reconstitution is the mainstay of treatment.
Immunosuppression may lead to reactivation of dormant Strongyloides stercoralis
Strongyloides stercoralis is a parasitic roundworm that remains dormant for years following initial infection, but may be reactivated to produce massive autoinfection in the immunosuppressed patient. Although rare in the UK and most of the USA, it should be borne in mind in patients who have lived in endemic areas such as the tropics and southern USA, even if this was many years before their immunosuppression. Human T-cell lymphotropic virus type 1 (HTLV-1) infection is associated with disseminated strongyloidiasis due to the modified immune response to this enteric helminth. The lungs, liver and brain are the most common organs affected.
Viruses
Certain virus infections are both more common and more severe in compromised patients, and regular surveillance is critical
The virus infections that are more common or more severe in the compromised patient (see Table 30.4) have been described in detail elsewhere in this book. Many of these represent reactivation of latent infections. Pre-transplantation baseline serology is carried out to determine both the donor and recipient status for a number of virus infections, including HIV, hepatitis B and C, CMV, EBV and HSV.
Suppression of specific virus infections using antiviral agents is part of the management of the recipient in conjunction with regular virological surveillance post-transplantation using viral genome or antigen detection methods.
As part of a pre-emptive treatment strategy, blood samples are collected for early detection of viraemia or antigenaemia which precedes disease. For example, transplant donors and recipients are screened for CMV IgG. CMV causes a broad spectrum of clinical disease in this setting, including pneumonitis, oesophagitis, colitis, hepatitis and encephalitis. If there is a transplant mismatch, i.e. the donor is CMV IgG positive and the recipient CMV IgG negative, the infection may be acquired from the donor organ or bone marrow. If possible, transplant centres try to avoid this situation, as the risk of a primary CMV infection in the first month post-transplantation is extremely high, as is the morbidity and mortality. In this case, CMV DNA monitoring is carried out on blood samples on a regular basis post-transplantation to detect early infection and start antiviral therapy as soon as possible. Some centres offer antiviral therapy in the immediate post-transplant period in this clinical setting to delay the onset of infection to a time when the recipient is less immunosuppressed. CMV IgG positive recipients are at risk of reactivation or reinfection and will also be monitored regularly post-transplantation. A primary CMV infection is usually detected around 4 weeks, compared with reactivation at around 6–8 weeks, post-transplantation, respectively.
Antiviral prophylaxis for HSV reactivation that may occur in the immediate post-transplantation period is often given to bone marrow transplant recipients for prolonged periods post-transplantation. Aciclovir is given at a low dose and is effective in preventing HSV and VZV reactivation. Virus surveillance is therefore not carried out, but if a breakthrough infection occurs it is important to collect material from the lesions for virus isolation or genome sequence analysis to determine the antiviral susceptibility. Herpetic lesions can be persistent and involve the lips, oesophagus and other parts of the gastrointestinal tract, and may cause a pneumonitis, hepatitis or encephalitis.
Herpes zoster, a reactivation of VZV infection, may occur within a few months post-transplantation, affecting the skin dermatome supplied by the involved nerve. Sometimes the distribution may be multidermatomal and dissemination can occur to other sites.
HHV-6 and HHV-7 infection, reinfection or reactivation has been reported in transplant recipients, in particular with neurologic conditions including encephalitis. HHV-8 has been associated with the development of Kaposi’s sarcoma (KS) in individuals with AIDS as well as classic and endemic KS in HIV-uninfected individuals.
EBV infection can lead to tumour development
EBV infection has been associated with the development of Hodgkin’s disease, non-Hodgkin’s lymphomas in individuals with HIV infection, post-transplantation lymphoproliferative disease and smooth-muscle tumours in immunosuppressed children. EBV-associated post-transplant lymphoproliferative disorder (PTLD) has a broad spectrum of clinical syndromes ranging from infectious mononucleosis to malignancies containing clonal chromosomal abnormalities with a high mortality rate, especially with the monoclonal tumours. The risk factors recognized for PTLD development in solid organ transplant recipients include post-transplantation primary EBV infection, mismatched donor and recipient CMV status, CMV disease, and intensity and type of immunosuppressive therapy. With respect to EBV infection, EBV-susceptible recipients have a 10–76-fold higher risk of PTLD compared with recipients with previous EBV exposure.
As the two peaks of primary EBV infection are in children and adolescents, the incidence of PTLD is higher in paediatric transplant recipients. In addition, without an effective cytotoxic T-cell response due to post-transplant immunosuppression to prevent graft rejection, the EBV-infected B lymphocytes may proliferate in an uncontrolled fashion. This results in B-cell hyperplasia with CD20-positive lymphocytes that ranges from polyclonal and benign to developing a monoclonal or oligoclonal B-cell lymphoma. The prevalence of PTLD in paediatric liver transplant recipients ranges from 4% to 14%, depending on the immunosuppressive regimen. Retrospective studies have shown that up to 50% of paediatric transplant recipients with primary EBV infections are at risk of developing PTLD. The infection may be acquired in the community or, in the transplant setting, from the donor organ or blood products. The natural history of EBV infection and pathophysiology of post-transplant EBV-driven lymphoproliferation is not well understood.
Diagnostic criteria for EBV-associated PTLD have been developed. However, in the absence of randomized, placebo-controlled trials, there is little information on the efficacy of specific treatment protocols. The treatment of PTLD includes reducing immunosuppression to allow a better host response to control the infection, although there is a risk of rejecting the graft, using rituximab, an anti-CD20 monoclonal antibody that targets the B cells with the EBV receptor, and chemotherapy. Treatment of post-transplant lymphomas by adoptive transfer of EBV-specific cytotoxic T lymphocytes has been reported.
Respiratory virus infections
Immunocompromised patients, especially transplant recipients, are at increased risk of pneumonia and death if they develop respiratory tract infections with viruses such as RSV, influenza, parainfluenza and adenoviruses. Preventive measures include influenza immunization, prophylaxis with palivizumab, an RSV-specific monoclonal antibody that is used in specific clinical settings, and early diagnosis of an upper respiratory tract infection using sensitive tests such as viral genome detection. There are some specific antiviral treatments that include oseltamivir for influenza and ribavirin for RSV infections.
Adenovirus infection has a high mortality rate
Primary and reactivated adenovirus infections can result in disseminated disease in immunocompromised hosts, in particular paediatric and adult bone marrow transplant recipients. Hepatitis and pneumonia are most frequently reported. Again, adenovirus surveillance is often carried out in centres by collecting blood samples post-transplantation which are tested for adenovirus DNA in order to detect early viraemia. Where adenovirus viraemia is detected, management options include reducing immunosuppression and treating with an antiviral agent such as ribavirin or cidofovir. However, there are few reports of successful outcomes in patients with disseminated infections.
Hepatitis B and C infection in transplant recipients
Hepatitis B virus infection has an immunopathologic pathogenesis, with jaundice occurring after cytotoxic T cells have lysed the hepatitis B surface-antigen-bearing hepatocytes. The virus is integrated in the hepatocytes after acute hepatitis B. Bone marrow transplant recipients with evidence of previous, not current, hepatitis B infection are likely to suffer a hepatitis B reactivation post-transplantation. They will be asymptomatic as they are immunosuppressed and will not mount a cytotoxic T-cell response until they have engrafted. It is at this stage they will become symptomatic, develop jaundice and the morbidity and mortality can be high. Antiviral prophylaxis with antiviral agents such as lamivudine or tenofovir is given to prevent reactivation, together with HBV DNA monitoring. Antiviral treatment will be given pre- and post-transplant if a transplant recipient has a current HBV infection, i.e. is hepatitis B surface antigen positive.
The most common reason for liver transplantation in the USA and Europe is hepatitis C-related end-stage liver disease. HCV reinfection of the graft post-transplantation is common and within 5 years nearly one-third of these transplant recipients may develop cirrhosis and subsequently suffer liver decompensation and lose their graft.
Hepatitis C virus infection is also associated with veno-occlusive disease in bone marrow transplant recipients. Venous congestion occurs in the liver due to a non-specific vasculitis and results in liver necrosis. Multiorgan failure can be precipitated due to increased capillary permeability throughout the body.
Polyomaviruses can cause haemorrhagic cystitis and progressive multifocal leukoencephalopathy
BK or JC viruses are polyomaviruses acquired via the respiratory tract that lie latent in the kidney, and may be detected in the urine of bone marrow transplant recipients (see Ch. 20). BK viruria is associated with haemorrhagic cystitis.
JC virus can reactivate and disseminate to cause central nervous system infections such as progressive multifocal leucoencephalopathy (PML) in individuals with AIDS. However, since the advent of highly active antiretroviral therapy resulting in higher CD4 counts and suppressed HIV load, PML is seen less often.
![]()
 Key Facts
Key Facts
• A compromised person is one whose normal defences against infection are defective. Immunodeficiencies may involve the innate or adaptive immune systems and may be primary or secondary.
• Compromised patients can be infected with any of the pathogens capable of infecting immunocompetent individuals. In addition, they suffer many infections caused by opportunist pathogens. The type of infection is related to the nature of the compromise.
• Effective antimicrobial therapy is often difficult to achieve in the absence of a functional immune response, even when the pathogen is susceptible to the drug in vitro.
• Important bacterial opportunists include P. aeruginosa, especially in neutropenic patients and those with major burns, and Staph. epidermidis in patients with plastic devices in situ. In AIDS, the predominant bacterial opportunists are intracellular pathogens benefiting from the lack of cell-mediated immunity.
• AIDS, and neutropenia (particularly following cytotoxic therapy), predispose to fungal infections (e.g. Candida, Aspergillus and Cryptococcus) especially when the patient has received previous antibacterial therapy.
• Viral infections are more common and severe in immunodeficient patients than in immunocompetent patients, particularly reactivation of latent infections (e.g. herpes simplex virus, CMV, JC virus).
![]()