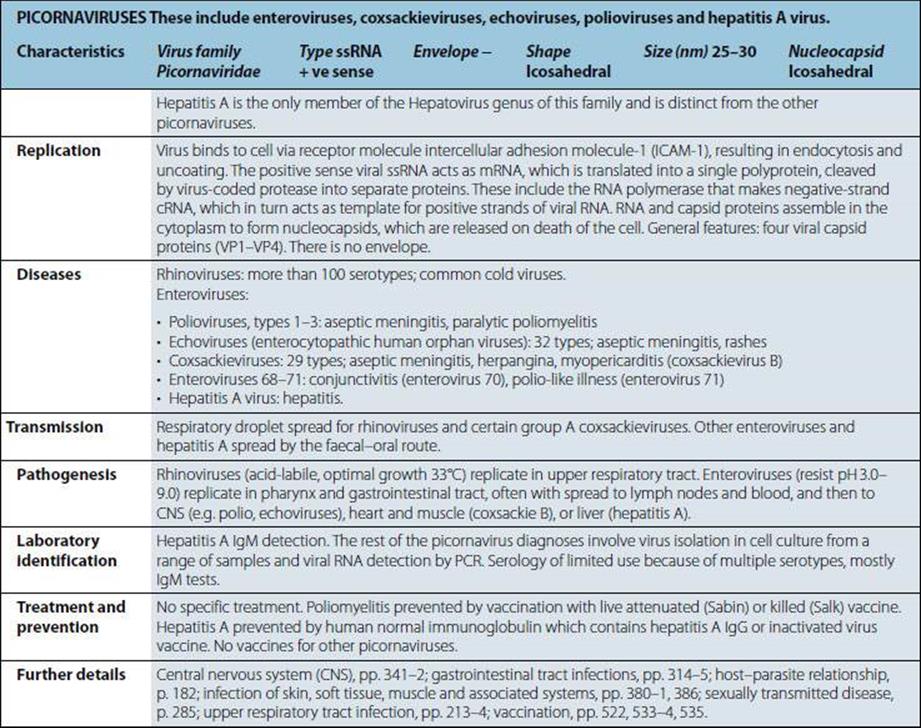
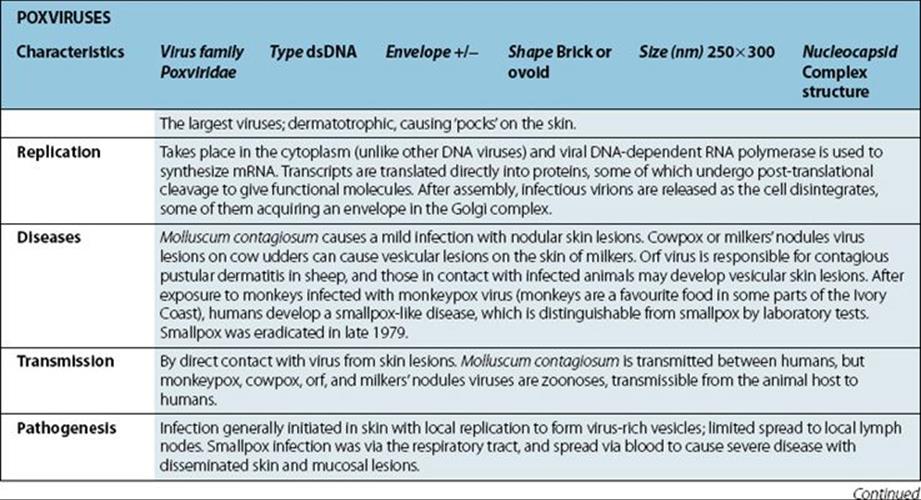
Medical Microbiology
Pathogen parade
Viruses
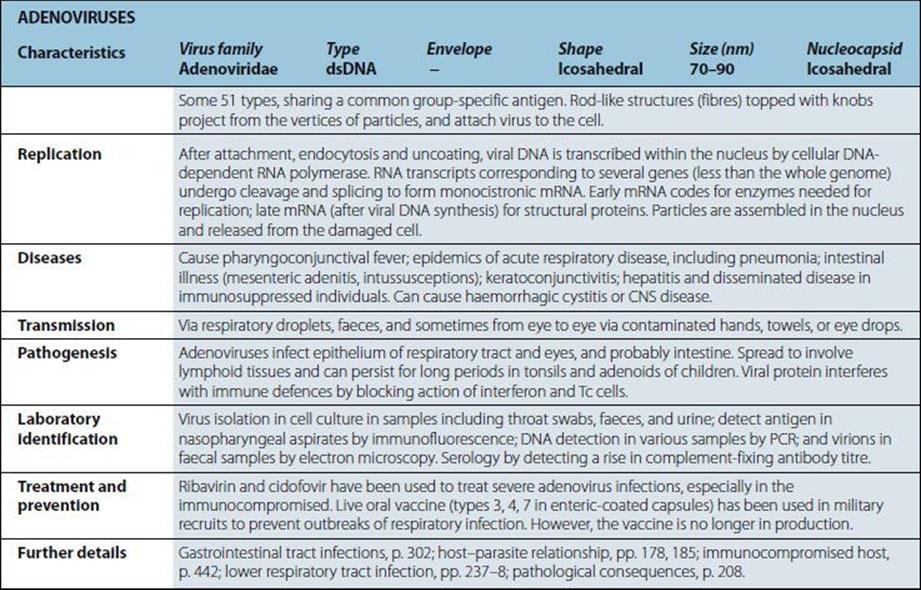
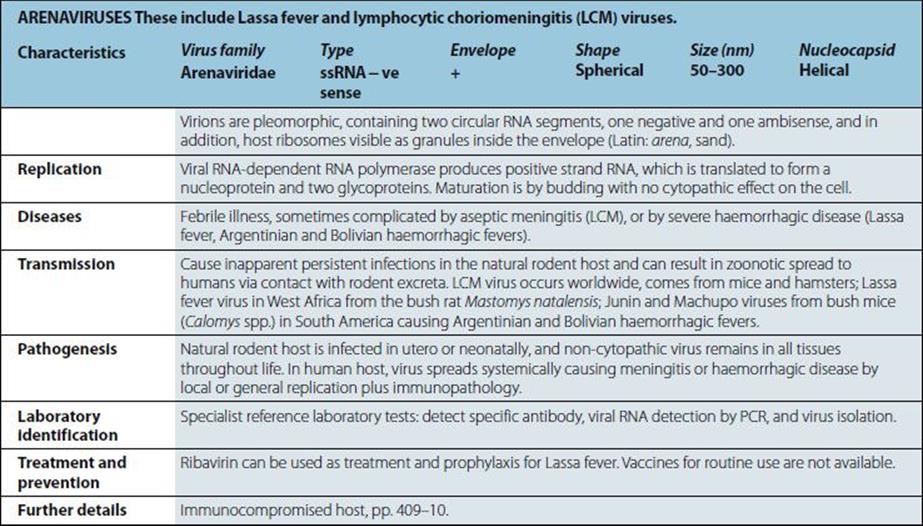
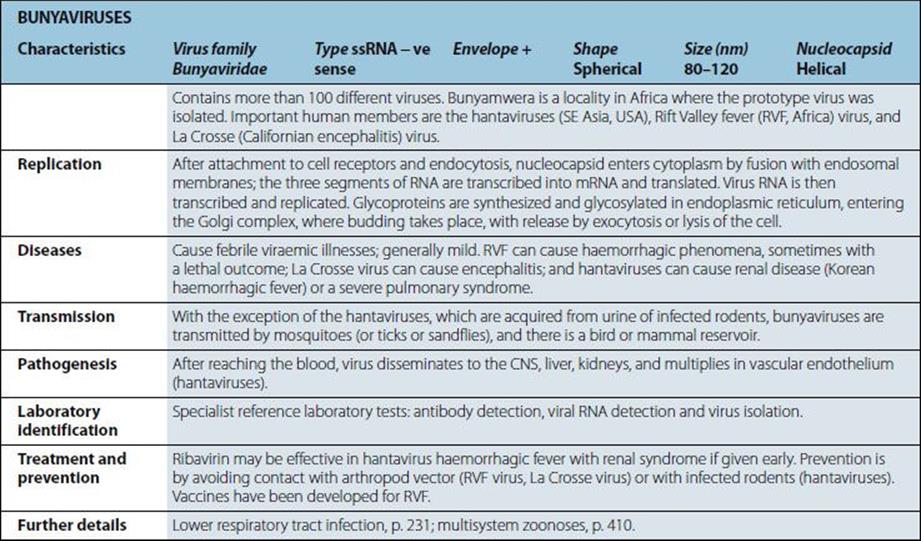
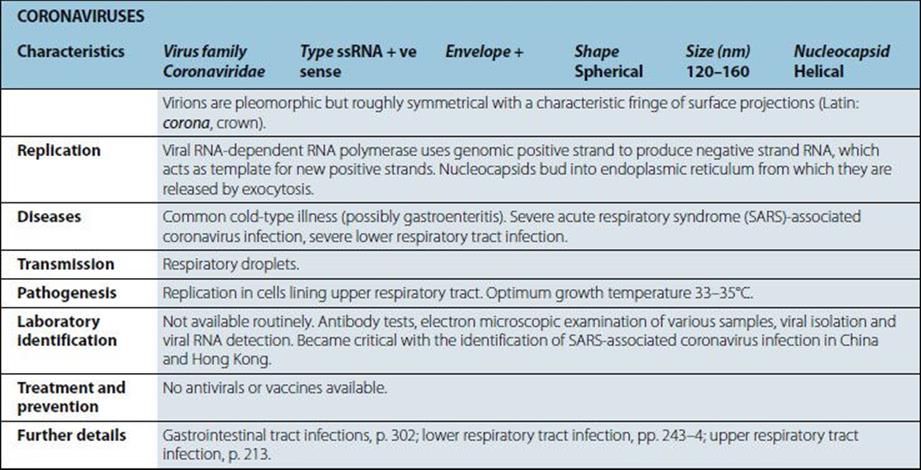

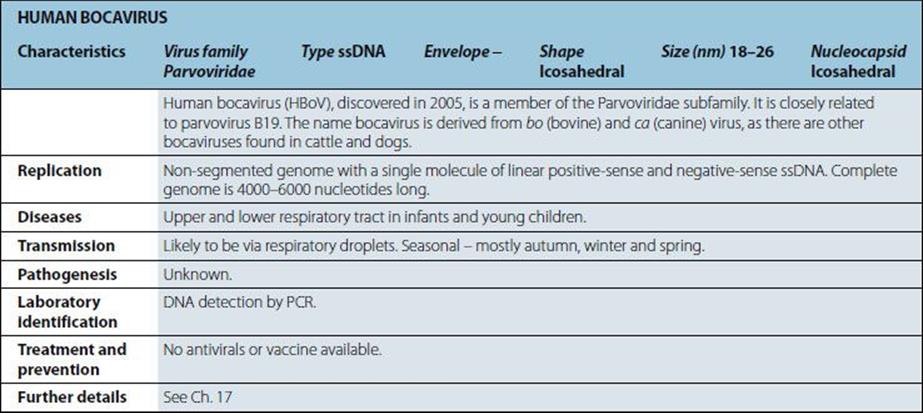

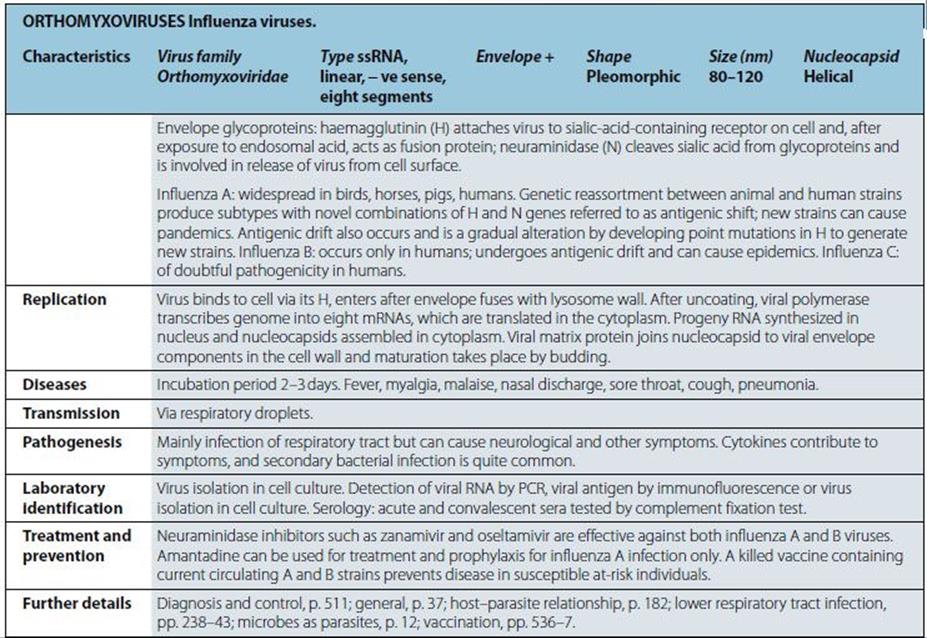
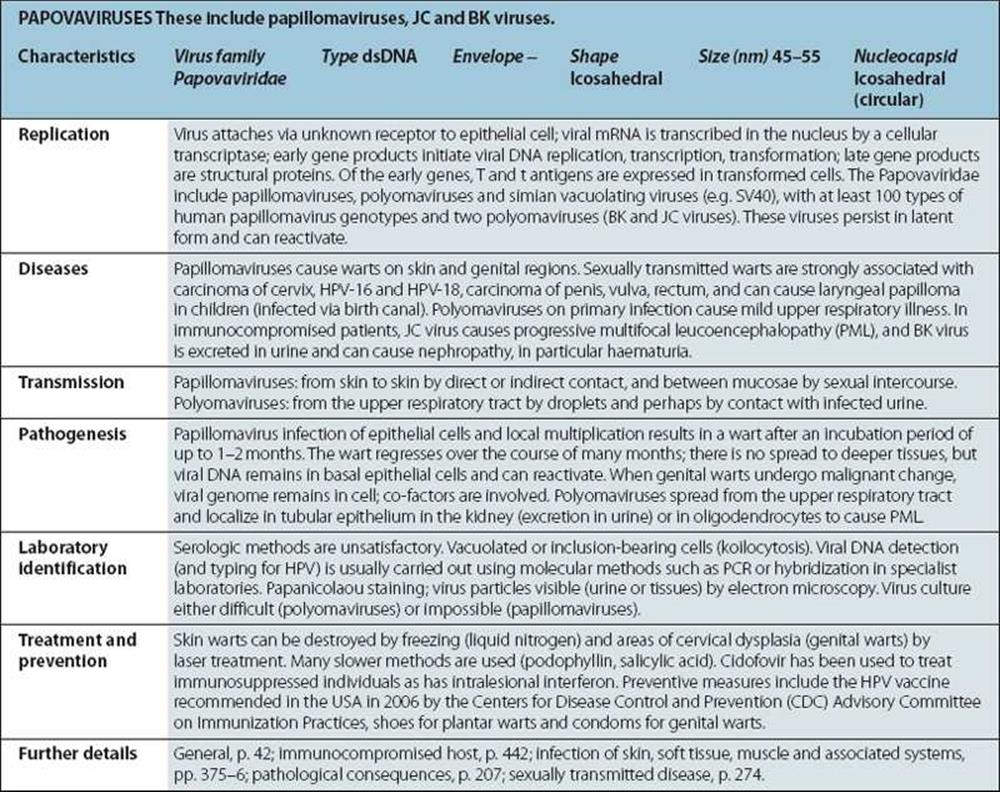





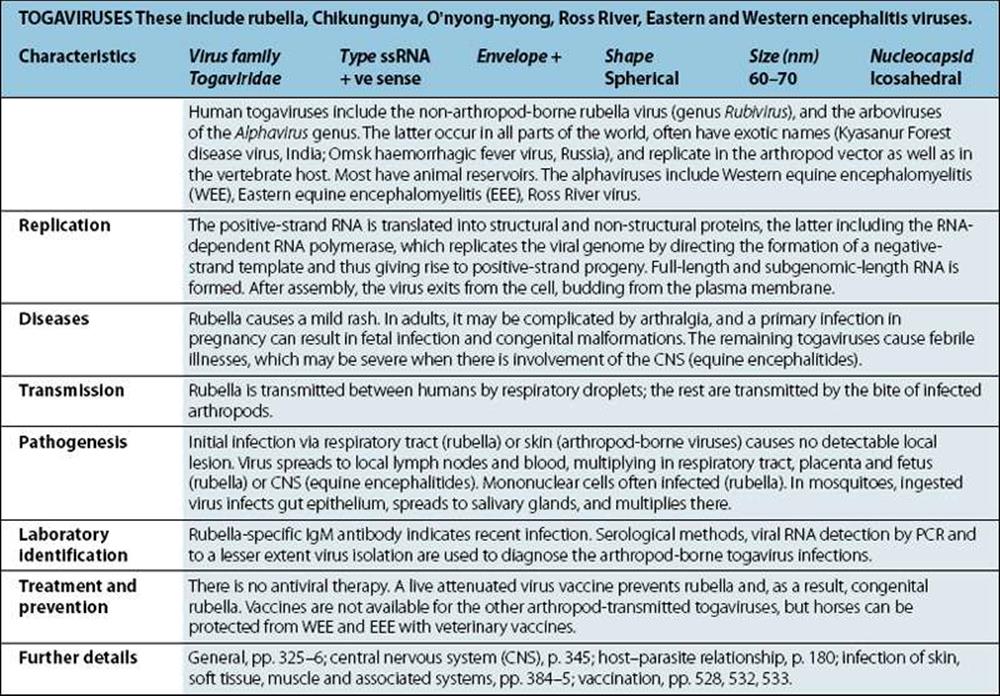
|
Human Prion Diseases |
|
|
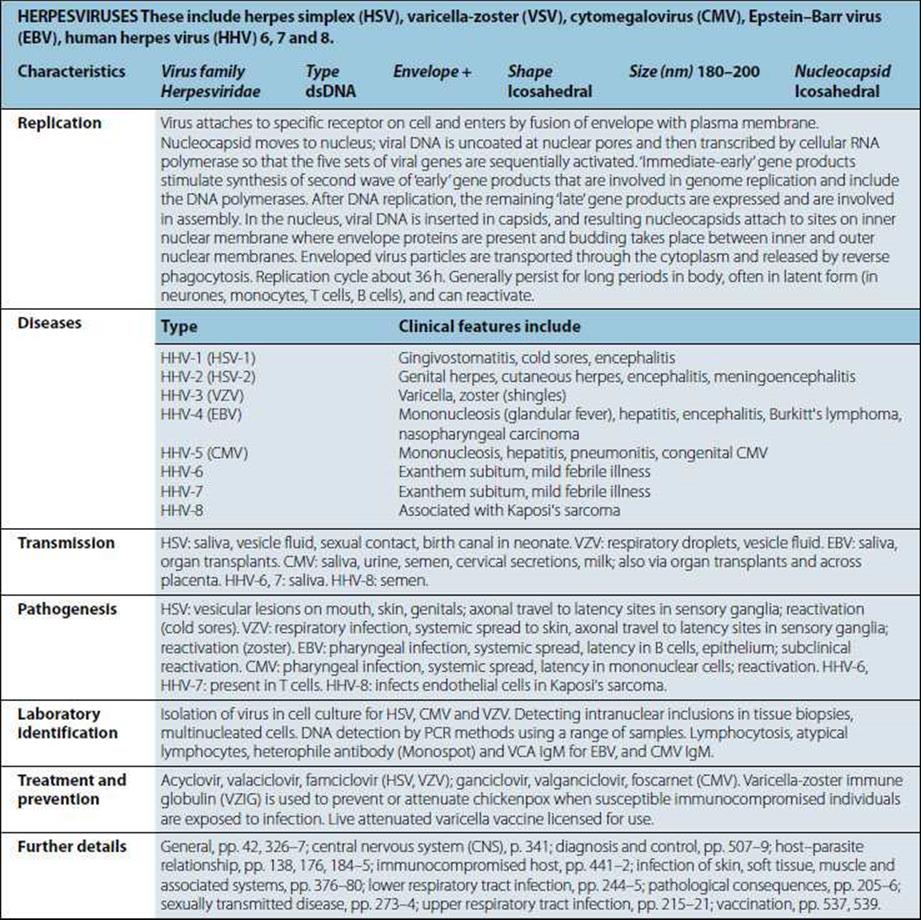
Characteristics |
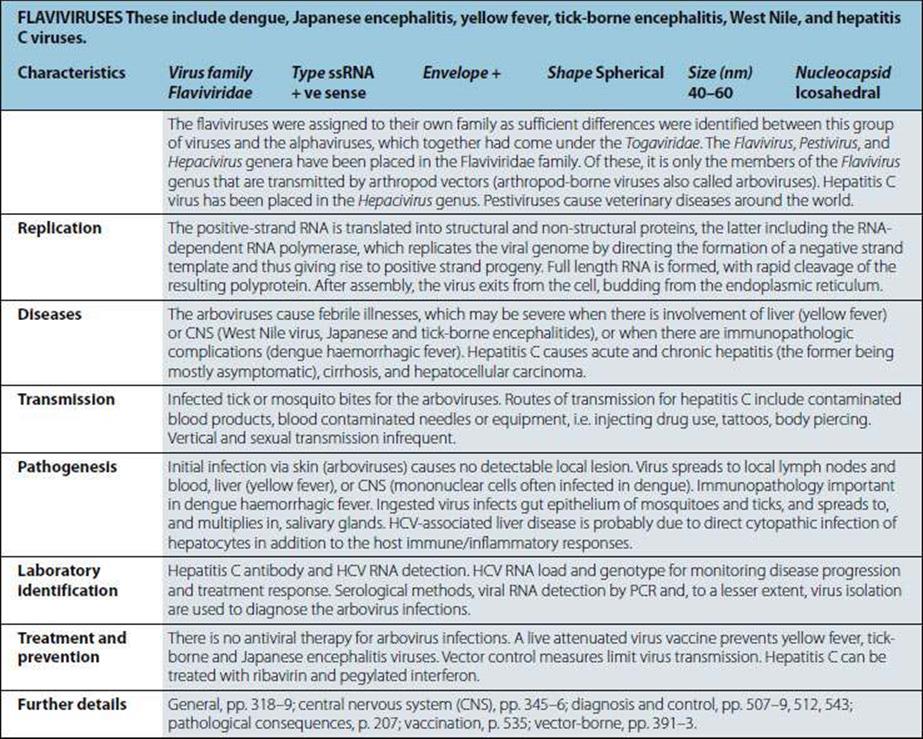
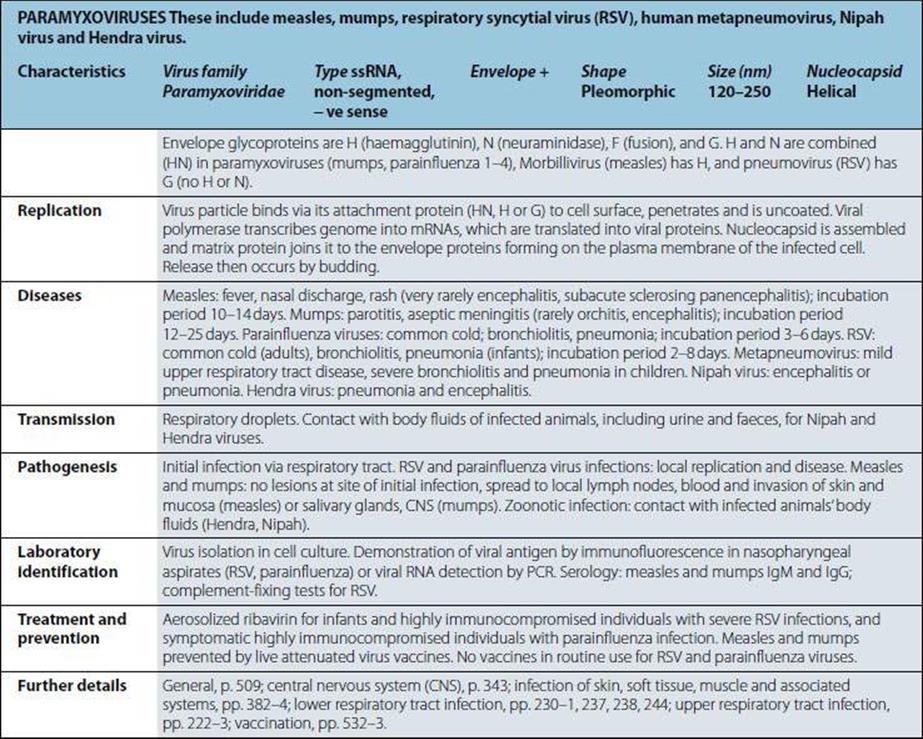
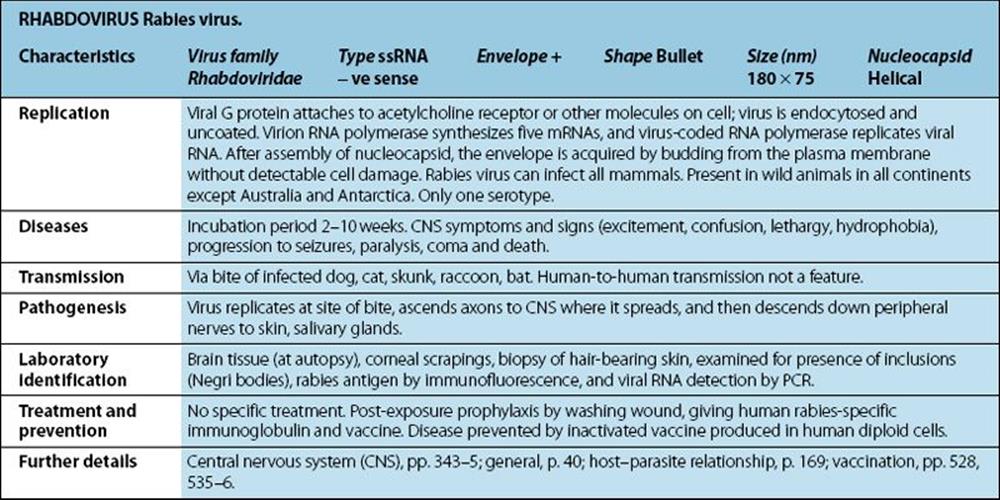
Not viruses. Aetiological agents of the four human prion diseases share the same general features. The diseases are Creutzfeldt–Jakob disease (CJD), kuru, Gerstmann–Straússler–Scheinker syndrome (GSS) and fatal familial insomnia (FFI). The prototype agent, scrapie, causes CNS disease in sheep. Host-coded prion (proteinaceous infectious particle) protein in slightly altered form (some forms protease-resistant) is closely associated with infectivity. Highly resistant to heat (special autoclaving procedures required for destruction), chemical agents and irradiation. Very slow replication, very long incubation period (up to 20 years in humans). Infect a variety of mammals and can be transmitted to cows, mink, cats and mice, for example, when food contains infected material. |
|
Diseases |
“Spongiform encephalopathies”, “prion diseases”. Kuru: fatal neurologic diseases in Papua New Guinea still occur but are very rare. CJD: rare chronic encephalopathy, occurs worldwide; 10% cases familial with mutated prion protein gene. GSS and FFI. |
|
Transmission |
Kuru: from exposure to infected human brain during ritualistic mortuary feasts (may be consumption or due to transmission via lesions in skin). CJD: in most cases unknown; occasionally transmitted from infected human brain but also by blood and by medical and surgical procedures; familial cases genetically transmitted. Variant CJD (vCJD) believed to result from consumption of BSE (bovine spongiform encephalopathy)-infected food. |
|
Pathogenesis |
Infectious agent replicates inexorably in lymphoid tissues, and then in brain cells, where it produces intracellular vacuoles and deposition of altered host prion protein. Uniformly fatal if host lives long enough. |
|
Laboratory identification |
Intracellular vacuoles (spongiform change) visible histologically in brain. Altered prion protein detectable in CNS and LRS. In brain, tests not routinely available but are used for animals. US also uses immunohistochemistry for diagnostics. Isolation of agent requires experimental animals and is lengthy, difficult and not routinely undertaken. No specific immune responses. |
|
Treatment and prevention |
No treatment or vaccine. Kuru died out when cannibalism ceased. Iatrogenic transfer of CJD preventable (e.g. when genetically engineered growth hormone became available). |
|
Further details |
General, pp. 59–61, 346; host–parasite relationship, p. 70. |
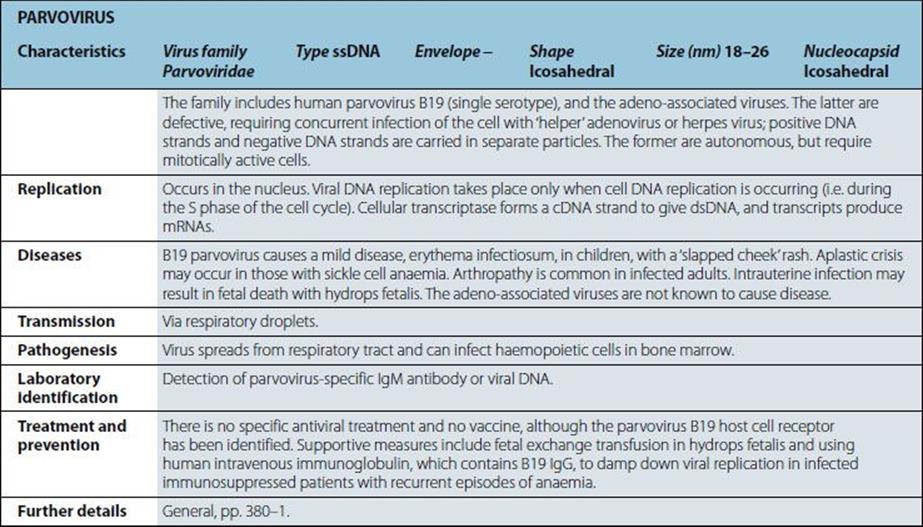
Bacteria
|
Staphylococcus Aureus |
|
|
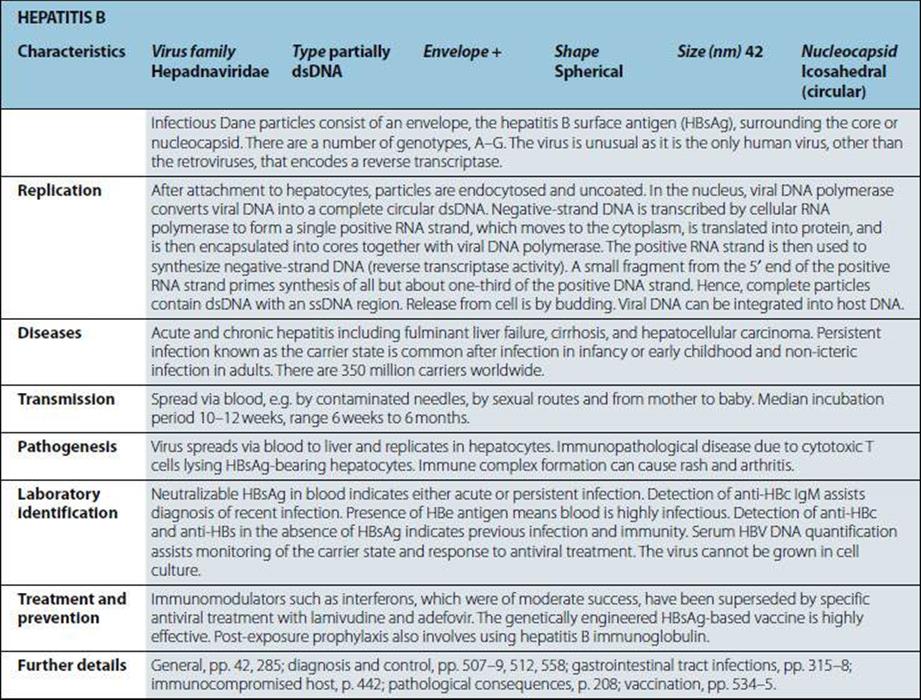
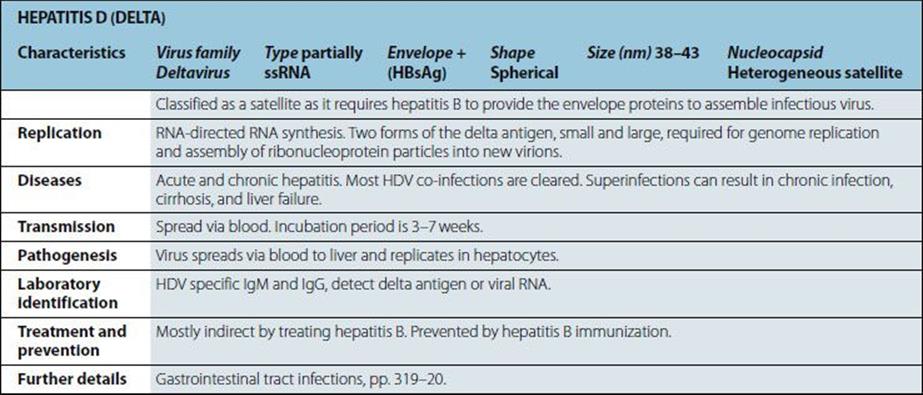
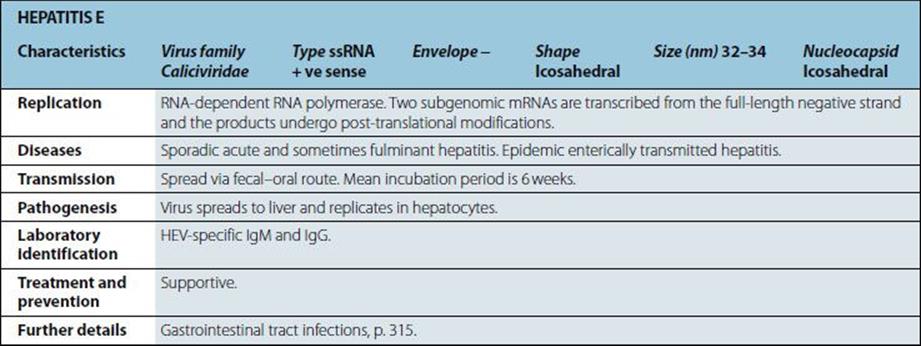
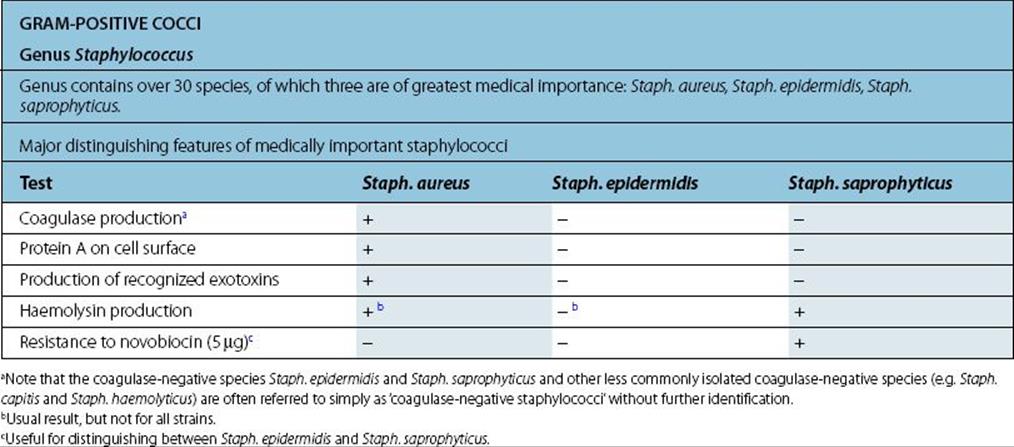
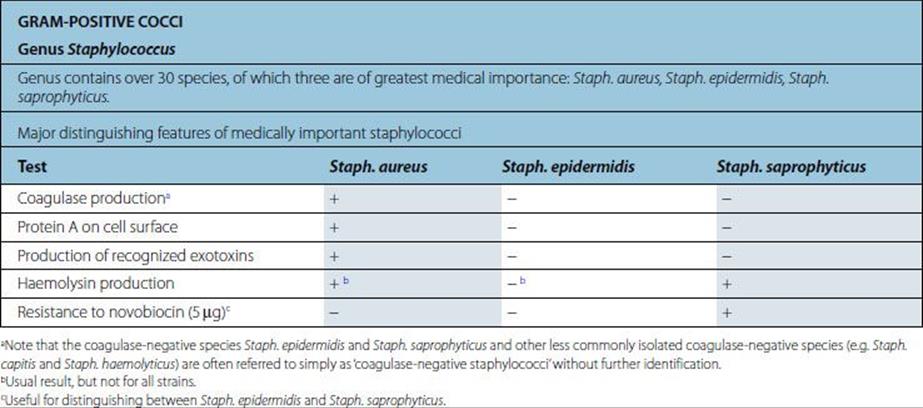
Characteristics |
Gram-positive coccus; cells in clusters (reflecting ability to divide in more than one plane); individual cells approximately 1 mm in diameter. Some strains produce capsules. Non-fastidious; capable of aerobic and anaerobic respiration. |
|
Laboratory identification |
White or golden colonies on blood agar. Catalase positive, coagulase positive; most strains ferment mannitol anaerobically. Kits available for biochemical characterization. |
|
Diseases |
Boils; skin sepsis; postoperative wound infection; scalded skin syndrome; catheter-associated infection; food-borne infection; septicaemia, endocarditis; toxic shock syndrome; osteomyelitis; pneumonia. |
|
Transmission |
Normal habitat: humans (and animals associated with them); skin, especially nose and perineum (carriage rates higher in hospital patients and staff). Spread is by contact and airborne routes. Organism survives drying; tolerant of salt and nitrites. |
|
Epidemiologic analysis |
Pulsed-field gel electrophoresis and other molecular techniques have largely replaced bacteriophage typing. |
|
Pathogenesis |
Virulence multifactorial, and most factors shown below are present in some strains. • mucopeptide • coagulase.
• cell-associated: capsule, protein A, fibronectin-binding protein, collagen-binding proteins • extracellular products: enterotoxins, epidermolytic toxin, toxic shock syndrome toxin, membrane-damaging toxins (haemolysins), leukocidin, staphylokinase • many strains have protein A bound to the mucopeptide of the cell wall. This protein interacts non-specifically with host IgG antibodies reducing opsonization and causing local activation of complement. |
|
Treatment and prevention |
In susceptible isolates, antibiotics of choice are beta-lactamase-stable penicillins; however, the vast majority of hospital isolates are beta-lactamase producers. Multiple drug resistance (including methicillin and tolerance or resistance to vancomycin) is a worldwide problem. Mupirocin can be used for topical treatment of carriage. Prevention of spread by isolation and/or treatment of carriers in high-risk areas in hospital. No vaccine available. |
|
Further details |
Diagnosis and control, pp. 481, 486, 489, 496, 552, 554, 555, 560; gastrointestinal tract infections, p. 302; immunocompromised host, p. 432; infection of skin, soft tissue, muscle and associated systems, pp. 358–62; microbes as parasites, pp. 23, 25. |
|
Staphylococcus Epidermidis |
|
|
Characteristics |
As for Staph. aureus. |
|
Laboratory identification |
White colonies on blood agar; catalase positive, coagulase negative, mannitol not fermented anaerobically. Kits available for biochemical characterization. |
|
Diseases |
Opportunist pathogen associated with device-related sepsis (e.g. catheter-related sepsis; prosthetic valve endocarditis; infection of artificial joints; shunt infections); urinary tract infection; sternal wound osteomyelitis. |
|
Transmission |
Normal habitat: skin (carriage rate approximately 100%). Spread by contact with self, other patients or hospital personnel. Almost all infections acquired in hospital, but may be endogenous. Survives drying; salt tolerant. |
|
Epidemiologic analysis |
Pulsed-field gel electrophoresis and other molecular techniques have largely replaced bacteriophage typing. |
|
Pathogenesis |
Extracellular slime production may be a marker of virulence and aid in the colonization of plastic implants (e.g. intravenous catheters and prostheses). |
|
Treatment and prevention |
Antibiotic resistance: often multiresistant (including penicillin and methicillin). Prevention of infection: catheter care; no vaccine available. |
|
Further details |
Diagnosis and control, pp. 486, 490; immunocompromised host, p. 433. |
|
Staphylococcus Saprophyticus |
|
|
Characteristics |
As for Staph. aureus. |
|
Laboratory identification |
White colonies on blood agar; catalase positive, coagulase negative, mannitol not fermented anaerobically. Kits available for biochemical characterization. |
|
Diseases |
Urinary tract infection in previously healthy women (associated with intercourse). |
|
Transmission |
Normal habitat: skin and genitourinary mucosa. Endogenous spread to urinary tract in colonized women. |
|
Epidemiologic analysis |
Pulsed-field gel electrophoresis and other molecular techniques. |
|
Pathogenesis |
Virulence factors unknown, but organism has the ability to colonize periurethral skin and mucosa. |
|
Treatment and prevention |
Urination after intercourse helps to wash organisms out of the bladder and prevent infection. |
|
Further details |
Urinary tract infection (UTI), p. 253. |
|
Genus Streptococcus |
|
A large group of Gram-positive cocci distributed widely in humans and animals, mostly forming part of the normal flora, but some species responsible for some major infections. Individual cells 0.5–1 μm diameter and, because they divide in one plane only, occur in pairs and chains. The medically significant streptococci may be divided on the basis of either haemolysis on blood agar (complete haemolysis, beta; partial haemolysis, alpha; no haemolysis, gamma) or by the presence or absence of a group-specific carbohydrate antigen (i.e. the Lancefield Group labelled alphabetically A to V). |
|
Beta-Haemolytic Streptococci |
|
|
Streptococcus Pyogenes (Group A Streptococcus) |
|
|
Characteristics |
Gram-positive cocci in chains, cells less than 1 μm diameter, non-motile, non-spore-forming. |
|
Laboratory identification |
Grown on blood agar. Pronounced haemolytic activity (enhanced anaerobically). Catalase negative. Bacitracin (0.04 units); all strains are susceptible; detection of group-specific carbohydrate (A antigen); detection of L-pyrrolidonyl arylamidase (PYR) |
|
Diseases |
Infections of upper respiratory tract and of skin and soft tissue (e.g. pharyngitis, cellulitis, erysipelas, lymphadenitis). Toxic manifestations include scarlet fever. Non-suppurative sequelae (acute glomerulonephritis and rheumatic fever) are important complications of both skin and throat infections. |
|
Transmission |
Normal habitat is the human upper respiratory tract and skin. Spread by airborne droplets and by contact. Survival in dust may be important. Epidemiologic typing of strains (see below) useful in outbreaks. |
|
Epidemiologic analysis |
Antigen from cell wall reacting with specific antisera (rabbit), either in a grouping precipitin or latex agglutination reaction. In addition to this group-specific polysaccharide, type-specific M and T antigens can be detected for epidemiologic purposes. Pulsed-field gel electrophoresis and other molecular techniques are commonly used for epidemiologic analysis. |
|
Pathogenesis |
Strep. pyogenes elaborates many enzymes and exotoxins, which may play a role in infection: erythrogenic toxin (lysogenic phage mediated); streptolysins; streptokinase A and B (therapeutic applications); deoxyribonuclease; hyaluronidase (“spreading factor”). |
|
Treatment and prevention |
Penicillin is the drug of choice. Vaccines not available. Oral cephalosporin or vancomycin is an alternative for penicillin-allergic patients. |
|
Further details |
Diagnosis and control, pp. 554, 560; immunocompromised host, pp. 433–4; infection of skin, soft tissue, muscle and associated systems, pp. 361–2; upper respiratory tract infection, pp. 221–2. |
|
Streptococcus Agalactiae (Group B Streptococci) |
|
|
Characteristics |
Gram-positive cocci in chains. |
|
Laboratory identification |
Beta-haemolytic on blood agar; colonies larger than Strep. pyogenes frequently pigmented after anaerobic incubation on Columbia agar (Islam”s medium). Grow in the presence of bile on MacConkey agar. Biochemical tests include hippurate hydrolysis (positive), aesculin hydrolysis (negative). Possess Group B Lancefield capsular antigen. Group-specific carbohydrate and commercially available molecular tests for definitive identification. Positive CAMP (Christie, Atkins, Munch–Peterson) test. |
|
Diseases |
Neonatal meningitis and septicaemia. Mastitis in bovines. |
|
Transmission |
Normal habitat; gut and vagina. Babies acquire organism from colonized mother at birth or by contact spread between babies in nursery after birth. |
|
Pathogenesis |
Virulence factors not clearly identified. |
|
Treatment and prevention |
Susceptible to penicillin, but less so than Strep. pyogenes; combination of penicillin and gentamicin for serious infections. Screening pregnant women recommended; prophylactic antibiotics may be given to babies (especially premature) of carriers. |
|
Further details |
Central nervous system (CNS), p. 339. |
|
Other Beta-Hemolytic Streptococci of Medical Importance |
|
Streptococci of Lancefield Groups C and G may sometimes cause pharyngitis. Group D streptococci include the Streptococcus bovis group and organisms now classified in the genus Enterococcus (see below). |
|
Streptococcus Milleri Group |
|
Microaerophilic streptococci that often form small colonies (formerly termed Streptococcus milleri) and carry Lancefield Group A, C, F or G antigens. Have a propensity for abscess formation (especially in liver and brain). |
|
Alpha-Hemolytic Streptococci |
|
|
Streptococcus Pneumoniae |
|
|
Characteristics |
Gram-positive coccus characteristically appearing in pairs (diplococci) in Gram films. Cells approximately 1 μm diameter, often capsulate. Requires blood or serum for growth. Capable of aerobic and anaerobic respiration; growth may be enhanced in CO2. |
|
Laboratory identification |
On blood agar alpha-haemolytic “draughtsman” colonies that may autolyse within 48 h at 35°C. Catalase negative. Susceptible to bile (bile solubility test) and Optochin (ethyl hydrocupreine hydrochloride; available in paper disks). Polysaccharide capsules can be demonstrated by appropriate staining techniques. They are antigenic and in the presence of specific antiserum appear to swell (quellung reaction). |
|
Diseases |
Pneumonia, septicaemia and meningitis. Otitis and related infections in children. Capsular type III frequently associated with pneumonia. |
|
Transmission |
Normal habitat is the human respiratory tract; ca. 5% of population may carry in small numbers. Transmission via droplet spread. |
|
Pathogenesis |
Capsule protects the organism from phagocytosis. Pneumolysin may have a role as a virulence factor, but to date no known exotoxins. Splenectomy may predispose to infection. Viral infection may be a precursor to pneumonia. |
|
Treatment and prevention |
Penicillin remains the antibiotic of choice, but resistance is increasing rapidly, and susceptibility test results should be used to guide therapy. Vaccine available. |
|
Further details |
Central nervous system (CNS), p. 338; host–parasite relationship, p. 158; lower respiratory tract infection, p. 230; microbes as parasites, p. 16; upper respiratory tract infection, p. 224; vaccination, p. 537. |
|
Oral Streptococci |
|
There are several other species of alpha-haemolytic streptococci that in the past have been lumped together under the colloquial heading “viridans streptococci”. These and some of the non-haemolytic streptococci have now been reclassified. Most species are commensal in the mouth. Strep. mutans is strongly associated with dental caries. Several species are capable of causing bacterial endocarditis. Most strains are susceptible to penicillin; however, moderate to high resistance has also been observed. Moderately resistant isolates may be treated with penicillin plus an aminoglycoside while highly resistant strains require a broad-spectrum cephalosporin or vancomycin. It is important to distinguish these streptococci from Strep. pneumoniae in cultures from the respiratory tract. |
|
Genus Enterococcus (Faecal Streptococci) |
|
|
Formerly classified in the genus Streptococcus, with which they share many characteristics; there are more than 30 species of enterococci. E. faecalis and E. faecium are the most important clinically and are considered together. |
|
|
Characteristics |
Gram-positive cocci, cells often in pairs and chains; more ovate appearance than streptococci. Non-fastidious; capable of aerobic and anaerobic respiration. |
|
Laboratory identification |
On blood agar may produce alpha, beta or no haemolysis. Resistant to 40% bile salts and Optochin; relatively heat tolerant (grow at 45°C), and salt tolerant (grow in 6.5% NaCl); hydrolyze esculin. Kits available for biochemical identification. Carry Lancefield”s Group D antigen, but the antigen is teichoic acid rather than polysaccharide. |
|
Diseases |
Urinary tract infection; endocarditis; infrequent, but severe septicaemia after surgery and in the immunocompromised. |
|
Transmission |
Normal habitat is the gut of humans and animals. Most infections thought to be endogenously acquired, but cross-infection may occur in hospitalized patients. |
|
Pathogenesis |
No toxins or other virulence factors convincingly demonstrated. Plasmid-mediated haemolysin may play a role. |
|
Treatment and prevention |
Penicillins used in combination with aminoglycosides. Resistant to cephalosporins and incidence of resistance to vancomycin (VRE) is a problem. Linezolid, daptomycin, and quinupristin/dalfopristin (only E. faecium) may be used in treatment. Patients with known heart defects should be given prophylactic antibiotics to prevent endocarditis before dentistry or surgery on gut or urinary tract. |
|
Further details |
Diagnosis and control, pp. 488, 497. |
|
Gram-Positive Rods |
|
Genus Corynebacterium |
|
This genus contains many species, is widely distributed in nature. Although cell wall structure has similarities to Mycobacterium and Nocardia, the short-chain mycolic acids present do not confer acid-fast staining. The species of major importance is C. diphtheriae. This and other pathogens within the genus need to be distinguished from commensal corynebacteria. |
|
Corynebacterium Diphtheriae |
|
|
Characteristics |
Gram-positive, non-capsulate, non-sporing, non-motile rods, 2–6 μm in length. In Gram-stained films, cells arranged as “Chinese letters” or palisades and showing irregular staining or granule formation are characteristic. Non-fastidious, but growth enhanced by inspissated serum (Loeffler medium). Capable of aerobic and anaerobic respiration. |
|
Laboratory identification |
Grows on blood agar, but identification aided by a selective medium (e.g. blood tellurite) on which characteristic black colonies form within 48 h at 35°C (but many other organisms may produce black colonies). Clinically, most important biotypes of C. diphtheriae are mitis and gravis, and they have characteristic colony morphology. C. diphtheriae is catalase-positive and reduces nitrate. Species identification is established on the basis of biochemical tests or species-specific sequencing. Toxin production has been traditionally demonstrated by the Elek test. A polymerase chain reaction (PCR) assay for the toxin gene is available. It is important to demonstrate toxigenicity to confirm diphtheria diagnosis, but non-toxigenic strains may also be associated with disease (e.g. septicaemia, endocarditis). |
|
Diseases |
Diphtheria caused by toxigenic strains of C. diphtheriae. Focus of infection may be the throat or the skin. |
|
Transmission |
Normal habitat: usually nasopharynx, occasionally skin of humans. Infection is usually spread by aerosol. Patients may carry toxigenic organisms for up to 2–3 months after infection. |
|
Pathogenesis |
Disease is due to production of diphtheria toxin controlled by the tox gene, which is integrated into the bacterial chromosome on a lysogenic (β) phage. When concentration of exogenous inorganic iron (Fe3 +) is very low, exotoxin production is maximal; the selective advantage to the organism is unknown. The mode of action of the toxin is to block protein synthesis of the host cells by inactivating an elongation factor. |
|
Treatment and prevention |
Urgent supportive therapy to maintain airway essential in throat diphtheria. Antitoxin neutralizes toxin, penicillin kills organisms; antibiotics have little effect since diffusion of toxin is not influenced by inhibition of organisms at local site. In outbreak, carriers treated with erythromycin or penicillin. Immunization effective in prevention of diphtheria; in areas where immunization rates reach 85%, herd immunity sufficient to protect whole population. Circulating antibody after immunization neutralizes test dose of standardized toxin (Schick test). Positive result (i.e. skin reaction) equates with insufficient antibody. Babies acquire immunity from immune mothers for a few months. |
|
Further details |
Microbes as parasites, p. 22; pathological consequences, p. 194; lower respiratory tract infection, pp. 227–8; vaccination, p. 530. |
|
Other Corynebacteria |
|
C. ulcerans has been found in diphtheria-like disease. It produces two toxins, one of which is neutralized by diphtheria antitoxin, the other is similar to that produced by C. pseudotuberculosis. C. jeikeium is isolated from blood cultures and wounds in immunosuppressed patients. It is usually detected by its relative resistance to antibiotics other than glycopeptides such as vancomycin. C. pseudotuberculosis is a significant pathogen of horses and sheep. C. xerosis and C. pseudodiphtheriticum are skin inhabitants, and many other coryneforms may also be found on skin. These, and other related genera such as Brevibacterium and Rhodococcus, are lipophilic and require lipids for optimal growth. |
|
Genus Bacillus |
|
This genus contains more than 70 species, most of which are soil organisms. There are two species of major medical importance: B. anthracis and B. cereus. |
|
Bacillus Anthracis |
|
|
Characteristics |
Large (4–10 μm) Gram-positive spore-forming encapsulated rods. Spores are formed only after the organism is shed from the body. Respires aerobically. |
|
Laboratory identification |
In smears of body fluids, the capsule can be stained with polychrome methylene blue McFadyen reaction or direct fluorescent antibody is diagnostic of B. anthracis. Amplification-based molecular tests (e.g. PCR) are also available. The species is non-fastidious; grows well on simple media. Characteristic colonies (Medusa head) are probably related to chaining of the long rods. Non-haemolytic on horse blood agar (many of the other species are haemolytic). Growth in CO2 encourages the formation of the capsule and smooth colonies. Biochemical reactions are unhelpful except in expert hands. |
|
Diseases |
Anthrax is a significant disease in both domesticated and wild animals. It is a zoonosis and humans are usually infected by contact with infected hides or bones. Intestinal anthrax is rare in humans. Woolsorter”s disease (i.e. respiratory or inhalation anthrax) is also rare. However, the potentially lethal effect of anthrax infections has especially attracted interest as an aspect of biological warfare. |
|
Transmission |
Soil organisms: B. anthracis can survive in competition with other organisms for many years depending on the temperature and humidity. The carcasses of animals dying with anthrax are buried 6 feet deep to prevent organisms being carried to the surface. Humans are accidental hosts, and infection is usually acquired when spores enter abrasions on the skin or are inhaled. |
|
Pathogenesis |
The polyglutamic acid capsule is antiphagocytic. In addition, an exotoxin encoded on a temperature-sensitive plasmid is produced. Toxin has three components: oedema factor, lethal factor and protective antigen. Individually, the components have no biologic effect, but toxicity is produced by either of the first two factors together with the antigen. The toxin acts locally in the skin and lung. Pasteur used heat attenuation to produce a virulent strain that could be used as an attenuated vaccine. |
|
Treatment and prevention |
Ciprofloxacin is the drug of choice but (depending on susceptibility and especially in the case of inhalation anthrax) may be combined with other antibiotics (e.g., penicillins, doxycycline), Prevention includes control measures such as formalin disinfection of hides, strict control of infected domestic animals, and the immunization of veterinarians and laboratory workers at risk. |
|
Further details |
Multisystem zoonoses, pp. 412–3. |
|
BACILLUS CEREUS |
|
|
Characteristics |
Large Gram-positive spore-forming rod. This and many other Bacillus species are similar to B. anthracis in many respects except most are motile and non-capsulate. Respires aerobically. |
|
Laboratory identification |
Non-fastidious. Produces haemolysis on horse and sheep blood agar. Lecithinase production and inability to utilize mannitol are used as distinguishing features on a specially designed selective medium. |
|
Diseases |
B. cereus causes food poisoning, the commonest association being with reheated cooked rice and pulses. Two different syndromes are recognized, due to different toxins (see below). The organism is also a rare cause of bacteraemia, especially in immunocompromised hosts. |
|
Transmission |
B. cereus spores are found on many foods, especially rice, pulses and vegetables. Infection/symptoms occur following ingestion of organisms or toxin. |
|
Pathogenesis |
Some strains produce heat-stable toxin in food associated with spore germination; this gives rise to a syndrome of vomiting within 1–5 h of ingestion. Others produce a heat-labile enterotoxin after ingestion, which causes diarrhea within 10–15 h. |
|
Treatment and prevention |
The majority of illness is short-lived and self-limiting, and antibiotic treatment is not indicated. Bacteraemia in immunocompromised patients and other B. cereus infections should be treated promptly with gentamicin, vancomycin, ciprofloxacin, or clindamycin. As with other food-borne infections, hygienic preparation of food is paramount. Cooked food should be stored in a refrigerator and reheated thoroughly before serving. |
|
Further details |
General, pp. 298–9. |
|
GENUS LISTERIA |
|
These organisms were included with the genus Corynebacterium in older classifications. They also share antigenic relationships with enterococci and lactobacilli. L. monocytogenes is the species of major medical importance. |
|
Listeria Monocytogenes |
|
|
Characteristics |
Short Gram-positive rods, often coccobacillary in clinical material (must avoid confusion with streptococci in chains); frequently Gram variable. Motile at 25°C with a characteristic “tumbling” movement; non-motile at 37°C. |
|
Laboratory identification |
Haemolytic on sheep or horse blood agar. Selective medium aids recovery of these organisms, especially from food samples (fish, chicken and cheeses). Cold enrichment at + 4°C for several weeks is also an effective selective technique. On translucent, non-blood-containing agar, colonies appear green-blue in oblique light. Catalase-positive, nitrate reduction negative; coupled with motility at room temperature these results are useful identifying features. Biochemical and serological tests provide definitive identification. |
|
Diseases |
Meningitis and sepsis in neonates. Infections in the immunocompromised (particularly meningitis) and in pregnant women. |
|
Transmission |
Widely distributed in nature, survives well in cold. Reaches food chain via silage as well as more directly via for example vegetables. Excreted in large numbers in cows” milk. Humans may carry Listeria in gut as normal flora. Infection may be acquired by ingestion or transplacentally to the baby in utero. While 13 different serotypes exist, pulsed-field gel electrophoresis and other molecular techniques are routinely used to investigate outbreaks. |
|
Pathogenesis |
Virulent strains produce internalins (cell attachment factors), haemolysins, and a motility protein; organism can survive in phagocytes. |
|
Treatment and prevention |
Treatment with penicillin or ampicillin, often in combination with gentamicin. Widespread distribution of organism in nature makes prevention of acquisition difficult. Pregnant women have been advised against eating uncooked food thought to be of particular risk (e.g. coleslaw, pâté, soft cheese, unpasteurized milk). |
|
Further details |
Central nervous system (CNS), p. 338; congenital disease, p. 328; gastrointestinal tract infections, pp. 313–4, host–parasite relationship, p. 73. |
|
Genus Clostridium |
|
This genus contains many species of Gram-positive anaerobic spore-forming rods; a few are aerotolerant. Widely distributed in soil and in the gut of humans and animals. The spores are resistant to environmental conditions. The major diseases associated with species of the genus are gangrene, tetanus, botulism, food poisoning and pseudomembranous colitis. In each of these, the production of potent protein exotoxins is an important cause of pathology, and in several species the genes encoding toxins are carried by plasmids or bacteriophages. |
|
Clostridium Perfringens |
|
|
Characteristics |
Anaerobic Gram-positive rods; spore-forming, but spores rarely seen in infected material. More tolerant of oxygen than other clostridia. |
|
Laboratory identification |
Haemolytic colonies on blood agar incubated anaerobically. Identification confirmed by demonstration of alpha-toxin (lecithinase) production in the Nagler”s test. Germination of heat-resistant spores (with subsequent toxin production) may be responsible for food poisoning. Five types of C. perfringens (A–E) identified on the basis of toxins produced; type A strains can be further divided into several serotypes. |
|
Diseases |
Gas gangrene resulting from infection of dirty ischaemic wounds. Food poisoning following ingestion of food contaminated with enterotoxin-producing strains. |
|
Transmission |
Spores and vegetative organisms widespread in soil and normal flora of humans and animals. Infection acquired by contact; may be endogenous (e.g. wound contaminated from patient”s own faecal flora) or exogenous (e.g. contamination of a wound with soil, ingestion of contaminated food). |
|
Pathogenesis |
In ischaemic wounds, production of numerous toxins and tissue-destroying enzymes allows organism to establish itself and multiply in wound. Local action of toxins produces necrosis thereby further impairing blood supply and keeping conditions anaerobic, and aiding spread of organism into adjacent tissues. Food poisoning results from the ingestion of large numbers of vegetative cells, which sporulate in the gut and release enterotoxin. |
|
Treatment and prevention |
Gangrene requires rapid intervention with extensive debridement of the wound. Penicillin is the antibiotic of choice (alternatively metronidazole, clindamycin or imipenem). Hyperbaric oxygen may also be helpful. Food poisoning does not usually require specific treatment. |
|
Further details |
General, pp. 298–9. |
|
Clostridium Tetani |
|
|
Characteristics |
Gram-positive spore-forming rod with terminal round spore (drumstick). Strict anaerobe. |
|
Laboratory identification |
Grows on blood agar in anaerobic conditions as a fine spreading colony; “ground glass” appearance (hand lens inspection of cultures important). Has very little biochemical activity useful for identification purposes. Demonstration of toxin in a specimen is possible in a two-mouse model in which one animal is protected with antitoxin, the other unprotected (performed in Public Health reference laboratories). |
|
Diseases |
Tetanus (lockjaw). Severe disease characterized by tonic muscle spasms and hyperflexia, trismus, opisthotonos and convulsions. |
|
Transmission |
Organism widespread in soil. Acquired by humans by implantation of contaminated soil into wound. Wound may be major (e.g. in war, in road traffic accident) or minor (e.g. a rose-thorn puncture while gardening). No person-to-person spread. |
|
Pathogenesis |
Tetanus results from neurotoxin (tetanospasmin) produced by organisms in wound. Toxin genes are plasmid-encoded. The organism is non-invasive, but the toxin spreads from site of infection via bloodstream and acts by binding to ganglioside receptors and blocking release of inhibitory neurotransmitters. Causes convulsive contractions of voluntary muscles. |
|
Treatment and prevention |
Antitoxin is available (hyperimmune human gamma globulin; tetanus immune globulin). Metronidazole and spasmolytic drugs indicated. Prevention readily available and effective in form of immunization with toxoid. Usually given in childhood, but if immunization status of injured patient is unknown, toxoid is given in addition to antitoxin. |
|
Further details |
Central nervous system (CNS), pp. 338–9; infection of skin, soft tissue, muscle and associated systems, p. 364; pathological consequences, p. 195; vaccination, p. 530. |
|
Clostridium Botulinum |
|
|
Characteristics |
Anaerobic Gram-positive rods. Not easily cultivated in competition with other organisms. Produces most potent toxins known to man. Seven immunologically distinct toxins (A to G) produced by different strains of C. botulinum. Types A, B, E and F are most commonly associated with human disease: serotypes A and B linked to a variety of foods (e.g. meat), serotype E especially associated with fish. |
|
Laboratory identification |
Requires strictly anaerobic conditions for isolation. Grows on blood agar, but very rarely isolated from human cases of disease. Detection of the toxin or organisms in the food or detection of the toxin or organisms in the serum or faeces of the patient, respectively, is the way of confirming the diagnosis. |
|
Diseases |
Major pathogen of birds and mammals, rare in humans. Botulism acquired by ingesting preformed toxin. Disease entirely due to effects of toxin. Infant botulism results from ingestion of organisms and production of toxin in infant”s gut. Associated with feeding honey contaminated with spores of C. botulinum. Wound botulism: toxin produced by organisms infecting a wound. Extremely rare. |
|
Transmission |
Soil is the normal habitat. Intoxication most often by ingestion of toxin in foods that have not been adequately sterilized (e.g. home-preserved foods) and improperly processed cans of food. Toxin is associated with germination of spores. There is no person-to-person spread. |
|
Pathogenesis |
Toxin released from organism as inactive protein and cleaved by proteases to uncover active site. It is acid stable and survives passage through stomach. Taken up through stomach and intestinal mucosa into bloodstream. Acts at neuromuscular junctions inhibiting acetylcholine release. Results in muscle paralysis and death from respiratory failure. |
|
Treatment and prevention |
Supportive therapy is paramount. Trivalent antitoxin is available. In the rare cases of infant and wound botulism (i.e. when the organism is growing in vivo), penicillin and metronidazole are effective. Prevention relates to good manufacturing practice. The toxin is not heat stable, therefore adequate cooking of food before consumption will destroy it. |
|
Further details |
Central nervous system (CNS), p. 349; gastrointestinal tract infections, p. 303; pathological consequences, p. 195. |
|
Clostridium Difficile |
|
|
Characteristics |
Slender Gram-positive anaerobic rod; spore-former; motile. |
|
Laboratory identification |
Difficult to isolate in ordinary culture because of overgrowth by other organisms; selective medium CCFA (cycloserine-cefoxitin-fructose agar) may be helpful; however, mere presence of the organism is not indicative of infection. Diagnosis by detection of toxin in faeces (usually immunoassay or, more rarely, the older tissue culture cytotoxicity assay). |
|
Diseases |
Pseudomembranous colitis (antibiotic-associated diarrhea). Can be rapidly fatal especially in the compromised host. |
|
Transmission |
Component of normal gut flora; flourishes under selective pressure of antibiotics. May also be spread from person to person by the faecal–oral route. |
|
Pathogenesis |
Toxin-mediated damage to gut wall. Produces both an enterotoxin (toxin A) and cytotoxin (toxin B). |
|
Treatment and prevention |
Oral vancomycin or metronidazole. Other antibiotics should be withheld if possible. Prevention of cross-infection in hospitals depends upon scrupulous attention to hygiene. |
|
Further details |
Diagnosis and control, pp. 489, 496, gastrointestinal tract infections, pp. 299–300; host–parasite relationship, p. 65. |
|
Genus Mycobacterium |
|
|
Mycobacteria are widespread both in the environment and in animals. The major human pathogens are M. tuberculosis and M. leprae, but awareness of the importance of other species (e.g. M. avium complex) is increasing with their recognition as pathogens in AIDS and other immunocompromised patients. |
|
|
Characteristics |
Aerobic rods with a Gram-positive cell wall structure, but stain with difficulty because of the long-chain fatty acids (mycolic acids) in the cell wall. Acid fastness can be demonstrated by resistance to decolorization by mineral acid and alcohol (Ziehl–Neelsen stain). Mycobacteria grow more slowly than many other bacteria of medical importance, but the genus can be divided into: rapid growers (form visible colonies within ca. 3–7 days); slow growers (form visible colonies only after ca. 2 weeks to 2 months” incubation). |
|
Laboratory identification |
Staining and microscopic examination of specimens for acid-fast rods is important because of the time required for culture results. All species except M. leprae can be grown in artificial culture, but they require complex media. Identification is based on rate of growth (rapid or slow), optimum temperature of growth and pigment production. Scotochromogenic species produce pigment in the absence of light whereas photochromogenic species require exposure to light before pigment becomes apparent. Further biochemical tests are required for full specification. Polymerase chain reaction methods, DNA probes and sequence-based approaches are available for identification purposes. |
|
Diseases |
M. tuberculosis causes tuberculosis in humans and animals. M. leprae is restricted to humans and causes leprosy. Mycobacteria other than tuberculosis (MOTT) are associated with a range of conditions, usually in immunocompromised hosts. M. avium-intracellulare (M. avium complex) has important associations with AIDS patients in the USA; in Africa M. tuberculosis is more common. |
|
Transmission |
Droplet spread aided by ability of organisms to survive in the environment (M. tuberculosis, M. leprae). Unpasteurized milk from cattle infected with M. bovis has been responsible for human infections in the past. Social and environmental factors and genetic predisposition all have a role. Leprosy requires close and prolonged contact for spread. |
|
Pathogenesis |
Both M. tuberculosis and M. leprae are intracellular parasites surviving within macrophages. They give rise to slowly developing, chronic conditions in which much of the pathology is attributable to host immune responsiveness rather than to direct bacterial toxicity. |
|
Treatment and prevention |
Prolonged treatment with combinations of antimycobacterial drugs is required. Bacille Calmette–Guérin (BCG) vaccination is valuable for prevention in endemic areas. Isoniazid prophylaxis used for contacts of cases of tuberculosis. Pasteurization of milk and improvement of living conditions have played a major role in prevention. |
|
Further details |
Central nervous system (CNS), p. 348; diagnosis and control, pp. 503–4; host–parasite relationship, pp. 158, 160, 165, 179, 184; immunocompromised host, p. 440; infection of skin, soft tissue, muscle and associated systems, pp. 365–8; lower respiratory tract infection, pp. 245–8; microbes as parasites, pp. 15, 18; pathologic consequences, p. 191; urinary tract infection (UTI), p. 258; vaccination, pp. 523, 530, 534. |
|
Genus Actinomyces |
|
The actinomycetes are true bacteria, although they have in the past been considered to resemble fungi because they form branching filaments. They are related to the corynebacteria and mycobacteria in the chemical structure of their cell walls. It is important to differentiate them from fungi because infections with actinomycetes should respond to antibacterial agents whereas similar clinical presentations caused by fungi are resistant to antibacterials (and extremely refractory to treatment by antifungal agents). This genus contains many species, some of which are important to humans as producers of antimicrobial agents. A few are pathogenic to humans and animals; A. israelii is a key cause of actinomycosis. |
|
Actinomyces Israelii |
|
|
Characteristics |
Gram-positive anaerobic filamentous branching rods. Non-sporing, non-acid fast. |
|
Laboratory identification |
Forms “sulphur granules” composed of a mass of bacterial filaments in pus. These can be identified by washing pus, squashing granules and observing in stained microscopic preparations. Gram-positive branching rods also visible in stained pus. Forms characteristic breadcrumb or “molar tooth” colonies on blood agar after 3–7 days anaerobic incubation at 35°C . |
|
Diseases |
Actinomycosis follows local trauma and invasion from normal flora. Hard non-tender swellings develop which drain pus through sinus tracts. Cervicofacial lesions are most common, but abdominal lesions after surgery and infection related to intrauterine contraceptive devices also occur. |
|
Transmission |
A. israelii is part of normal flora in mouth, gut and vagina. Infection is endogenous. There is no person-to-person spread. |
|
Pathogenesis |
Virulence factors not described. |
|
Treatment and prevention |
Penicillin is the drug of choice. Prolonged treatment is required, accompanied by surgical drainage. |
|
Further details |
Gastrointestinal tract infections, p. 321; immunocompromised host, pp. 438–9. |
|
Genus Nocardia |
|
|
Characteristics |
Aerobic Gram-positive rods that form thin branching filaments. Widespread in the environment. N. asteroides complex represents the important human pathogens. |
|
Laboratory identification |
Gram stains of pus may reveal Gram-positive filaments or rods. Sulphur granules not seen. Grow as “breadcrumb” colonies on blood agar within 2–10 days” incubation. Often weakly acid fast. Catalase positive. |
|
Diseases |
N. asterioides complex are opportunistic pathogens especially infecting immunocompromised patients; primarily a pulmonary infection, but secondary spread to form abscesses in brain or kidney is common. N. brasiliensis is the cause of actinomycetoma in Central and South America. |
|
Transmission |
Infection is acquired from the soil by the airborne route. Outbreaks of infection in renal transplant units have been associated with local building work. Actinomycetoma is acquired by implantation of organisms into wounds and progressive destruction of skin, fascia, bone and muscle. |
|
Pathogenesis |
Appears to be related to organism”s ability to survive the host”s inflammatory responses. Infection is controlled by cell-mediated immunity, but this may be defective in immunocompromised patients. |
|
Treatment and prevention |
Nocardiosis is often difficult to treat, but most regimens include sulphonamides as the drug of choice. |
|
Further details |
Immunocompromised host, pp. 439–9. |
|
GRAM-NEGATIVE RODS |
|
Enterobacteriaceae |
|
Most numerous facultative anaerobes in the human gut, comprising approximately 109/g of faeces. Outnumbered only by Gram-negative anaerobes (e.g. Bacteroides), which are present in numbers approximately 10 times those of the enterobacteria. Genera of the family Enterobacteriaceaeshare features that distinguish them from other families; can be distinguished from each other by biochemical tests. |
|
Genus Escherichia |
|
Genus contains only one species of medical importance: E. coli. |
|
Escherichia Coli |
|
|
Characteristics |
Gram-negative rod; motile; with or without capsule; non-fastidious, facultative anaerobe; bile tolerant; capable of growth at 44°C. |
|
Laboratory identification |
Grows readily on routine laboratory media and on bile-containing selective media. Lactose fermenter. Kits available for full identification. |
|
Diseases |
Urinary tract infection; diarrheal diseases; neonatal meningitis; septicaemia. |
|
Transmission |
Normal habitat is gut of humans and animals; may colonize lower end of urethra and vagina. Spread is by contact and ingestion (faecal–oral route); may be food-associated; may be endogenous. Possesses O (somatic), H (flagellar), K (capsular) and F (fimbrial) antigens, which can be used to characterize strains by serotyping (e.g. O157:H7 EHEC strains, see below). Pulsed-field gel electrophoresis most often used for epidemiologic analysis. |
|
Pathogenesis |
A variety of virulence factors have been identified, particularly in strains associated with diarrheal disease: • endotoxin: present in all strains • adhesins: P fimbriae (pili) associated with urinary tract infection; colonization factors (e.g. CFA I, II and III, K88, K99) associated with gastrointestinal tract infection in humans and animals • capsule present in some strains; may be associated with adhesion. K1 capsular type associated with neonatal meningitis • enterotoxins associated with diarrheal disease: ETEC (enterotoxigenic E. coli) produce heat-stable (ST) and cholera-like heat-labile (LT) toxins; EIEC (enteroinvasive E. coli) produce shiga-like cytotoxin; EHEC (enterohaemorrhagic E. coli) produce verotoxin-associated with haemolytic uremic syndrome. |
|
Treatment and prevention |
Wide range of antibacterial agents potentially available, but incidence of resistance variable and often plasmid-mediated; must be determined by susceptibility testing. Specific treatment of diarrheal disease usually not required. No currently available vaccine. |
|
Further details |
Diagnosis and control, pp. 551–2; gastrointestinal tract infections, pp. 289–92; microbes as parasites, pp. 17, 25, 31, urinary tract infection (UTI), p. 253; vaccination, p. 539. |
|
Genus Proteus |
|
|
This genus contains several species, of which two are of medical importance: P. mirabilis and P. vulgaris. |
|
|
Characteristics |
Gram-negative rod; non-fastidious; facultative anaerobe; bile tolerant; likes alkaline pH; characteristic unpleasant odour; highly motile and swarms on some media. |
|
Laboratory identification |
Lactose non-fermenter; produces urease; kits available for full identification. Species can be distinguished by indole test: P. mirabilis, indole-negative; P. vulgaris, indole-positive. O (somatic) and H (flagellar) antigens characterize. Proteus strains OX-19, OX-2 and OX-K share antigens with rickettsiae in the typhus and spotted fever groups and are agglutinated by antibodies produced by patients with these rickettsial infections (Weil–Felix test). Serologic response to Proteus infection not useful diagnostically. |
|
Diseases |
Urinary tract infection; hospital-acquired wound infection, septicaemia, pneumonia in the compromised host. |
|
Transmission |
Normal habitat is human gut, soil and water. Contact spread; infection often endogenous. |
|
Pathogenesis |
Characterized virulence factors include endotoxin, urease; possible role for bacteriocins. |
|
Treatment and prevention |
Range of agents available. Prevention is by good aseptic technique in hospitals. No vaccine available. |
|
Further details |
Antimicrobial agents, p. 503; obstetric and perinatal infections, p. 328; upper respiratory tract infection, p. 224; urinary tract infection (UTI), pp. 253, 256, 259. |
|
Genus Klebsiella and Related Enterobacteria Serratia and Enterobacter |
|
|
Unlike E. coli, species of the genera Klebsiella, Serratia and Enterobacter are rarely associated with infection except as opportunists in compromised patients. |
|
|
Characteristics |
Gram-negative rods, sometimes capsulate (usual for Klebsiella), non-fastidious growth requirements. Capable of aerobic and anaerobic respiration. |
|
Laboratory identification |
Lactose-fermenting, bile-tolerant organisms. Grow readily on routine laboratory media. Oxidase negative. Full identification based on biochemical reactions (commercial kits available). |
|
Diseases |
Opportunist infections in the compromised (usually hospitalized) host. Urinary and respiratory tracts most common sites of infection. Distinction between colonization and infection can sometimes be difficult. |
|
Transmission |
Normal habitat is gut of humans and animals and moist inanimate environments, especially soil and water. Infection may be endogenous or acquired by contact spread. Klebsiella have remarkable capacity for survival on hands. Pulsed-field gel electrophoresis most commonly used for epidemiologic investigation of healthcare-associated infection. |
|
Pathogenesis |
All possess endotoxin and fimbriae or other adhesins. Capsules, where present, are important in inhibiting phagocytosis. |
|
Treatment and prevention |
Multiple antibiotic resistance, usually plasmid-mediated, is common, and susceptibility must be determined by laboratory tests if treatment is indicated. Prevention depends upon scrupulous attention to aseptic techniques and to hand washing in hospitals. |
|
Further details |
Klebsiella: sexually transmitted disease, p. 272; Serratia: urinary tract infections (UTI), p. 253; Enterobacter: antimicrobial agents, pp. 482, 498–9; gastrointestinal tract infections, pp. 292, 294, 297, 313, 321; hospital infection, pp. 558, 560, 564; urinary tract infection (UTI), p. 253. |
|
Genus Shigella |
|
|
Contains four species of importance to humans as causes of bacillary dysentery: S. dysenteriae, S. boydii, S. flexneri and S. sonnei (in descending order of severity of symptoms). |
|
|
Characteristics |
Gram-negative rods. Non-motile (in contrast to salmonellae). Non-capsulate. Capable of aerobic and anaerobic respiration. |
|
Laboratory identification |
Non-fastidious, bile-tolerant. Lactose non-fermenters. Full identification requires use of biochemistry (commercial kits available) and serologic tests for O antigens. Serodiagnosis of disease not applicable. |
|
Diseases |
Bacillary dysentery. |
|
Transmission |
Human pathogens spread by faecal–oral route, especially in crowded conditions. Small infective dose. |
|
Pathogenesis |
Invasion of ileum and colon causes damage, which results in diarrhea. Intense inflammatory response involving neutrophils and macrophages characteristic. S. dysenteriae produces an exotoxin (Shiga toxin) causing damage to intestinal epithelial cells. In fewer instances, the toxin results in damage to glomerular endothelial cells, leading to haemolytic uremic syndrome (HUS). |
|
Treatment and prevention |
Antibiotic therapy (e.g. fluoroquinolones, trimethoprim-sulphamethoxazole) should only be given for severe diarrhea; usually not required. Many strains carry multiple antibiotic resistances, usually on plasmids; thus susceptibility testing is important. Prevention depends upon interrupting faecal–oral spread; hand hygiene important. No vaccine available. |
|
Further details |
Gastrointestinal tract infections, pp. 296–7; host–parasite relationship, pp. 132, 137; sexually transmitted disease, p. 285; vaccination, p. 539. |
|
Genus Pseudomonas and Related Organisms Burkholderia, Stenotrophomonas and Acinetobacter |
|
This group contains a large number of species, a few of which are human pathogens, some are animal pathogens and others are important pathogens of plants. Species also widely distributed and may contaminate the hospital environment and cause opportunist infections. Most important in humans are: • P. aeruginosa, important opportunist in a variety of compromised patients • Burkholderia pseudomallei, cause of melioidosis, a disease of restricted geographic distribution • Burkholderia cepacia, commonly associated with nosocomial infection and respiratory tract infections in cystic fibrosis patients • Stenotrophomonas maltophilia, an opportunistic pathogen also commonly associated with nosocomial infection • Acinetobacter baumannii (and other species), opportunistic pathogens causing a variety of infections (e.g. wound, respiratory tract, urinary tract); frequently antibiotic resistant. |
|
Pseudomonas Aeruginosa |
|
|
Characteristics |
Aerobic Gram-negative rod, motile by means of polar flagella. Able to utilize a very wide range of carbon and energy sources and to grow over a wide temperature range. Does not ferment carbohydrates. Does not grow anaerobically (except when nitrate is provided as a terminal electron acceptor). |
|
Laboratory identification |
Grows readily on routine media including bile-containing selective media. Produces irregular iridescent colonies and a characteristic smell. Most strains produce a blue-green pigment (pyocyanin; unique to P. aeruginosa) and a yellow-green pigment (pyoverdin). Pigment production is enhanced on special media (King”s A and B); oxidase positive. |
|
Diseases |
P. aeruginosa is an opportunist pathogen that can infect almost any body site given the right predisposing conditions. It causes infections of skin and burns, it is a major lung pathogen in cystic fibrosis, and can cause pneumonia in intubated patients. It can also cause urinary tract infections, septicaemia, osteomyelitis, and endocarditis. |
|
Transmission |
Carriage as part of the normal gut flora occurs in a small percentage of normal healthy people and in a higher proportion of hospital inpatients. Thus, endogenous infection may occur in compromised patients. P. aeruginosa is widespread in moist areas in the environment; patients usually become infected by contact spread, directly or indirectly, from these environmental sites. |
|
Pathogenesis |
A number of virulence factors have been identified, including endotoxin and exotoxin A, which acts as an inhibitor of elongation factor in eukaryotic protein synthesis. Extracellular polysaccharide capsule helps to prevent phagocytosis (e.g. massive amounts of alginate produced by strains specifically in cystic fibrosis patients). Pigments may have a role in pathogenicity, and pyoverdin acts as a siderophore. |
|
Treatment and prevention |
Resistant to many antibacterial agents; propensity to develop resistance during therapy. Combination antimicrobial chemotherapy based on susceptibility testing is required (e.g. aminoglycoside and beta-lactam antibiotic). Prevention depends upon good aseptic practice in hospitals, avoidance of unnecessary or prolonged broad spectrum antibiotic treatment and prophylaxis. |
|
Further details |
Diagnosis and control, pp. 491, 498, 501; immunocompromised host, p. 433. |
|
Curved Gram-Negative Rods |
|
There are several genera of curved Gram-negative rods that contain species that occur in humans as pathogens. Three of the most important are Vibrio, Campylobacter and Helicobacter. |
|
Genus Vibrio |
|
|
Most important species is V. cholerae. |
|
|
Characteristics |
Curved Gram-negative rods, highly motile by means of single polar flagellum. Capable of aerobic and anaerobic respiration (facultatively anaerobic). Many species are salt (NaCl) tolerant; some salt-requiring. |
|
Laboratory identification |
Grow in alkaline conditions (can be selected from other gut flora in alkaline peptone water). Oxidase positive. Grow on thiosulfate citrate bile salts sucrose (TCBS) medium to form yellow colonies (V. cholerae) or green colonies (other species). Biochemical tests and use of specific antisera required for complete identification. |
|
Diseases |
Cholera caused by V. cholerae. V. parahaemolyticus causes diarrheal disease. V. vulnificus causes wound infections and bacteraemia. |
|
Transmission |
V. cholerae is a human pathogen; no animal reservoir, but El Tor biotype survives better in the inanimate environment than classic V. cholerae. Infection is acquired from contaminated water (usually) or food (sometimes). V. parahaemolyticus and V. vulnificus acquired from consumption of contaminated fish and seafood. |
|
Pathogenesis |
V. cholerae possesses several virulence factors (e.g. mucinase, adhesins and, most importantly, enterotoxin). Chromosomally encoded subunit toxin produced after cells bind to intestinal epithelium enters cells and binds to ganglioside receptors activating adenyl cyclase and causing fluid loss, resulting in massive watery diarrhea. V. parahaemolyticus produces a cytotoxin (which also haemolyses human red blood cells – the Kanagawa test). V. vulnificus produces cytolytic compounds and antiphagocytic polysaccharides. |
|
Treatment and prevention |
For cholera, fluid replacement (oral rehydration therapy: ORT) is of prime importance. Tetracycline shortens symptoms and duration of carriage. Some vaccine protection. Prevention of cholera depends upon provision of a clean (chlorinated) water supply and adequate sewage disposal. Specific treatment not indicated for V. parahaemolyticus diarrhea. Combination treatment (e.g. a tetracycline plus fluoroquinolone) used in treatment of V. vulnificus, V. parahaemolyticus and V. vulnificus infections can be prevented by adequate cooking of seafood. |
|
Further details |
Gastrointestinal tract infections, pp. 294–6, 297; host–parasite relationship, pp. 132, 133; microbes as parasites, p. 11, 24; pathological consequences, p. 195; vaccination, p. 524. |
|
Genus Campylobacter |
|
Curved Gram-negative rods, once classified as vibrios, campylobacters are primarily pathogens of animals, but several species also cause infections in humans. C. jejuni is a major cause of bacterial gastroenteritis in resource-rich countries. At a much lower frequency, C. coli also causes gastroenteritis. The infections caused by these organisms have an essentially identical clinical presentation, and laboratories generally do not distinguish between them. |
|
Campylobacter Jejuni |
|
|
Characteristics |
Slender, curved (seagull-shaped) Gram-negative rods. Motile by means of a polar flagellum at one or both ends. Microaerophiles. Do not utilize carbohydrate. |
|
Laboratory identification |
Require enriched media and moist microaerophilic environment (10% O2) for growth. Incubation at 42°C for 24–72 h. Colonies resemble water drops. Full identification by biochemical tests and characteristic antibiotic susceptibility pattern. |
|
Diseases |
Diarrhea. Can invade to give septicaemia. Guillain–Barré syndrome infrequently associated with Campylobacter disease. |
|
Transmission |
Animal reservoir. Organisms acquired from contaminated food and milk (but do not multiply in these vehicles). Person-to-person spread is rare. |
|
Pathogenesis |
Little known, but cytotoxin implicated. Also invasion and local destruction of gut mucosa. |
|
Treatment and prevention |
No specific treatment necessary for diarrhea. Erythromycin for invasive disease. Prevention depends upon good food hygiene. No vaccine. |
|
Further details |
Antimicrobial agents, p. 496; gastrointestinal tract infections, p. 294. |
|
Helicobacter Pylori |
|
|
Characteristics |
Associated with gastritis and duodenal ulcers; originally named C. pylori but now moved into the genus Helicobacter. Overall cellular morphology similar to Campylobacter. |
|
Laboratory identification |
Require enriched media and moist microaerophilic environment (10% O2) for growth. Incubation at 37°C for 24–72 h produces translucent colonies. Differentiated from Campylobacter by tests such as nitrate reduction (C. jejuni, positive; H. pylori, negative) and urease (C. jejuni, negative; H. pylori, positive). Full identification by biochemical tests and characteristic antibiotic susceptibility pattern. Organism in endoscopic biopsy specimens; positive urease test from endoscopic biopsy specimens or labelled urea breath-test also very useful. |
|
Diseases |
Gastritis and duodenal ulcers, associated with gastric carcinoma. |
|
Transmission |
Person-to-person transmission (faecal–oral) likely. Infections observed in multiple family members. |
|
Pathogenesis |
Both bacterial and host factors involved. Protease affects gastric mucosa; urease produces ammonia and buffers stomach acid. Some invasion of intestinal epithelium. |
|
Treatment and prevention |
Proton pump inhibitor plus antibiotics (e.g. clarithromycin, metronidazole, tetracycline). |
|
Further details |
Host–parasite relationship, pp. 133, 134. |
|
Gram-Negative Non-Spore-Forming Anaerobes |
|
Historically, all short Gram-negative anaerobic rods or coccobacilli have been classified in the genus Bacteroides and longer rods with tapering ends in the genus Fusobacterium. Recent applications of new techniques to the Bacteroides have resulted in the definition of two additional genera: Porphyromonas and Prevotella. The genus Bacteroides is now restricted to species found among the normal gut flora. Prevotella contains saccharolytic oral and genitourinary species, including P. melaninogenica (formerly B. melaninogenicus), which produces a characteristic black-brown pigment. The genus Porphyromonas contains asaccharolytic pigmented species, which form part of the normal mouth flora (P. gingivalis) and may be involved in endogenous infection within the oral cavity. The most important non-sporing anaerobe causing infection is B. fragilis although others are much more common (e.g. in gingivitis and other endogenous oral infections). |
|
Bacteroides Fragilis |
|
|
Characteristics |
Small pleomorphic Gram-negative rods or coccobacilli. Capable only of anaerobic respiration. Non-spore forming, non-motile. |
|
Laboratory identification |
Grows on blood agar incubated anaerobically and in other media designed for isolation of anaerobes. Plates may require up to 48 h incubation at 35°C for colonies to become visible. Cultures have a foul odour due to the fatty acid end-products of metabolism. These can be used as identifying characteristics by analysis of culture supernates by gas-liquid chromatography (GLC). The major products of Bacteroides are acetate and succinate. Full identification in the diagnostic laboratory is based on biochemical tests and antibiogram. Commercial kits are available. |
|
Diseases |
Intra-abdominal sepsis; liver abscesses; aspiration pneumonia; brain abscesses; wound infections. Infections often mixed with aerobic and microaerophilic bacteria. |
|
Transmission |
Endogenous infection arising from contamination by gut contents or faeces is most common route of acquisition. |
|
Pathogenesis |
Little is known about the virulence factors of B. fragilis. A polysaccharide capsule and production of extracellular enzymes (e.g. enterotoxin) are important features. An anaerobic environment is essential and in mixed infections growth of aerobic organisms probably helps the growth of Bacteroides by using up available oxygen. |
|
Treatment and prevention |
Metronidazole, imipenem, or beta-lactam-beta-lactamase inhibitor combinations used in therapy. Many strains produce beta-lactamases and thus susceptibility to penicillin and ampicillin is unreliable. Prevention of endogenous infection difficult; good surgical technique and appropriate use of prophylactic antibiotics important in abdominal surgery. |
|
Further details |
Gastrointestinal tract infections, p. 321. |
|
GRAM-NEGATIVE COCCI |
|
|
Genus Neisseria |
|
|
This genus contains several more or less fastidious species of which two, N. gonorrhoeae and N. meningitidis, are important human pathogens. |
|
|
Characteristics |
Non-motile Gram-negative diplococci with fastidious growth requirements: capnophilic; N. meningitidis is capsulate, N. gonorrhoeae is not. |
|
Laboratory identification |
Gram stains of pus or cerebrospinal fluid may reveal Gram-negative kidney-shaped diplococci, often intracellular (in polymorphs). Require supplemented media for growth (chocolate agar). N. gonorrhoeae easier to isolate on enriched media containing antibiotics to inhibit other organisms of normal flora from sample sites. The two species are differentiated by sugar utilization pattern. Kits available to detect N. gonorrhoeae nucleic acid in specimens. Latex agglutination test available for N. meningitidis. |
|
Diseases |
N. gonorrhoeae: gonorrhoea, and pelvic inflammatory disease and salpingitis in females; ophthalmia neonatorum in infants born to infected mothers. N. meningitidis: meningitis; occasionally septicaemia in absence of meningitis. |
|
Transmission |
Human pathogens; no animal reservoir. N. gonorrhoeae may be carried in genital tract, nasopharynx and anus. Spread by sexual or intimate contact. N. meningitidis carried in pharynx. Carriage rate in population increases during epidemics. Droplet spread. N. meningitidishas several immunologically distinct capsular types (e.g. A, B, C, Y, W135). |
|
Pathogenesis |
Several virulence factors have been identified. N. gonorrhoeae: pili or fimbriae act as adhesins; endotoxin; outer membrane proteins; protease production; resistance to lytic activity of serum; IgA proteases. N. meningitidis: the polysaccharide capsule is antiphagocytic; endotoxin and IgA protease also implicated. |
|
Treatment and prevention |
N. gonorrhoeae: resistance to first-line drugs now widespread; usual choice is beta-lactamase-stable cephalosporin (e.g. ceftriaxone). N. meningitidis: penicillin, ceftriaxone (or equivalent cephalosporin), or chloramphenicol. Prevention of gonorrhoea requires education, contact tracing. No vaccine available. Rifampicin is used for prophylaxis of close contacts of N. meningitidis meningitis. Tetravalent vaccine available (types A, C, Y, W135). |
|
Further details |
Central nervous system (CNS), pp. 335–7; host–parasite relationship, p. 182; sexually transmitted disease, pp. 266–8; vaccination, p. 537. |
|
Genus Moraxella |
|
Moraxella catarrhalis, previously classified as Branhamella catarrhalis, is a Gram-negative coccus morphologically similar to Neisseria, but with less fastidious growth requirements. Formerly regarded as a commensal in the respiratory tract, it has been associated with a variety of infections, including bronchitis, bronchopneumonia, sinusitis and otitis media. The majority of strains produce beta-lactamase and may be involved in the “protection” of more obvious pathogens, especially in the respiratory tract, by destroying penicillin or ampicillin administered as treatment. |
|
Genus Haemophilus |
|
The genus contains many species; H. influenzae and H. ducreyi are of medical importance. |
|
Haemophilus Influenzae |
|
|
Characteristics |
Small Gram-negative rods, frequently coccobacillary. Non-motile. Fastidious, capnophilic, facultative anaerobe. May be capsulate when isolated from site of infection. |
|
Laboratory identification |
Requires both haematin (X factor) and NAD (V factor) for growth (other species require one factor only). Grows on blood containing enriched media. Larger colonies around colonies of other organisms that secrete V factor (e.g. Staph. aureus) (satellitism). Dependence on X and V used as indicator of identity. H. influenzae can also be distinguished from other species by its inability to produce porphyrin. Six antigenically distinct capsular types recognized (a–f). Although type b has been most frequently found in disease this has changed with the introduction of vaccines against the type b strains. Capsulate organisms can be agglutinated by specific antisera and detected directly (e.g. by latex agglutination) in specimens. |
|
Diseases |
Capsular type b H. influenzae causes meningitis, osteomyelitis, epiglottitis, otitis. All are more common in children than older age groups. Non-capsulate strains associated with acute exacerbations of chronic bronchitis. Invasive disease due to type c and f isolates has increased. |
|
Transmission |
Normal habitat is upper respiratory tract in humans and associated animals. Transmitted from person to person by airborne route. Osteomyelitis probably follows septicaemia from respiratory focus. |
|
Pathogenesis |
Polysaccharide capsule is important virulence factor. Outer membrane proteins and endotoxin may play a part, but no known exotoxin. |
|
Treatment and prevention |
Frequent beta-lactamase-producing strains. Ampicillin (or amoxicillin) may be used if isolates are susceptible. Third-generation cephalosporin (e.g. cefotaxime or ceftriaxone) are usual alternatives. All children should be immunized with Hib vaccine. Rifampicin prophylaxis recommended for close contacts of Haemophilus meningitis. |
|
Further details |
Central nervous system (CNS), p. 337; upper respiratory tract infection, p. 224; vaccination, pp. 524, 538. |
|
Haemophilus Ducreyi |
|
Cause of the genital tract infection “soft chancre”. Slender Gram-negative rods appearing in pairs or chains. Direct microscopic examination of smear from chancre can be diagnostic. Organism very susceptible to dehydration; inoculate plates in clinic. Requires enriched medium (as for H. influenzae, but with addition of antibiotics to inhibit growth of other genital tract organisms). |
|
Genus Bordetella |
|
|
There are three species, of which one, B. pertussis, is of greatest medical importance. |
|
|
Characteristics |
Small Gram-negative coccobacilli. Slow growing and fastidious in its growth requirements. |
|
Laboratory identification |
Requires enriched medium (e.g. Bordet–Gengou or blood charcoal agar). Intolerant of fatty acids in medium. Fails to grow on routine blood agar (i.e. 5–7% blood). Requires 3–7 days incubation in moist atmosphere. Iridescent bisected pearl colony type characteristic on Bordet–Gengou. Further identification by reaction with specific antisera. Nucleic acid amplification tests in development. |
|
Diseases |
Whooping cough (pertussis). |
|
Transmission |
Human pathogen spread by airborne route from cases of disease (healthy carriage not documented). |
|
Pathogenesis |
Several virulence factors, including tracheal cytotoxin, fimbrial antigen and endotoxin. Stimulates a lymphocytic response. |
|
Treatment and prevention |
Macrolides erythromycin or clarithromycin for cases and close contacts of whooping cough. Antibacterial therapy has little effect on clinical course, but may reduce infectivity and incidence of superinfection. Vaccine administered to young children in five doses together with diphtheria and tetanus toxoids. |
|
Further details |
Host–parasite relationship, pp. 131, 160, 181; lower respiratory tract infection, pp. 228–9; microbes as parasites, p. 23; vaccination, pp. 524, 530. |
|
Genus Brucella |
|
|
There are several species of the genus Brucella, each characteristically associated with an animal species. Four species: B. abortus from cattle, B. suis from pigs, B. canis from dogs, and B. melitensis from goats, are most often found causing human zoonotic infections. |
|
|
Characteristics |
Small Gram-negative coccobacilli. Intracellular pathogens. Growth enhanced by erythritol in placenta of animals (not in humans). |
|
Laboratory identification |
Some strains slow-growing and fastidious, requiring complex growth media. Isolation from blood cultures improved by use of biphasic systems (e.g. Castaneda bottles containing both broth and agar). Usually require 3–5 days” incubation in CO2-enriched environment, but some strains of B. abortus may take up to 4 weeks – important in investigation of fever of unknown origin (FUO). Identification is by biochemical reactions, patterns of resistance to certain dyes, and serologic tests. The disease may be diagnosed by examination of patient”s serum for antibodies. |
|
Diseases |
Undulant fever (brucellosis). Patients frequently present with FUO. Infection may become chronic if not adequately treated. |
|
Transmission |
Zoonotic infections transmitted to humans through consumption of contaminated milk or other unpasteurized dairy products (increasingly seen in individuals who prefer untreated products) and by direct contact (occupational hazard for veterinarians, abattoir workers and farmers). |
|
Pathogenesis |
Virulence associated with ability to survive intracellularly, especially in bone marrow, liver and spleen, and thus “hide” from host defences. Erythritol is a growth stimulant for the organism in animals and accounts for the tropism of the organisms to the placenta and fetus. This is not true in humans. |
|
Treatment and prevention |
Doxycycline alone or in combination (e.g. with rifampin). Tetracyclines may not be tolerated during long treatment courses required; trimethoprim-sulphamethoxazole also effective. Recrudescence of infection is common. Prevention depends upon eliminating the disease from domestic animals by vaccination and pasteurization of milk. |
|
Further details |
Multisystem zoonoses, pp. 417–8. |
|
Francisella Tularensis |
|
|
Characteristics |
Small Gram-negative coccobacilli. Strict aerobe. Intracellular pathogen. The organism is found worldwide and occurs in a variety of wild and domestic animals. |
|
Laboratory identification |
Requires specialized medium (e.g. chocolate agar plates supplemented with cysteine) and lengthy incubation. Identification is by reaction with specific (i.e. anti-Francisella) antiserum. The diagnosis of disease may be aided by examination of patient”s serum for antibodies. However, the long-term persistence of antibody may cloud discrimination of current from past disease. Antibody against Brucella may cross react with Francisella. |
|
Diseases |
Francisella tularensis causes tularaemia (also known as glandular fever, deerfly fever or tick fever). Human disease is most commonly acquired from bite of an infected tick or contact with an infected animal (e.g. infected squirrels and rabbits). Tularaemia quickly develops after a short period of incubation (e.g. 3–4 days), potentially leading to high fever, chills, myalgia, malaise depending on the specific form of the disease (i.e. ulceroglandular, glandular, oculoglandular, oropharyngeal, pneumonic, gastrointestinal, and typhoidal). |
|
Transmission |
Zoonotic infections transmitted to humans through contact with infected animals, the bite of infected fleas or ticks, or ingestion of contaminated meat. |
|
Pathogenesis |
Virulence associated with an antiphagocytic capsule and the ability to survive intracellularly in macrophages. Francisella tularensis is highly infectious with as few as 10 organisms causing disease. For this reason, the public health agencies such as the World Health Organization and the US Centers for Disease Control are concerned about its potential use as an agent of bioterrorism. |
|
Treatment and prevention |
Gentamicin, fluoroquinolones, or doxycycline. Prevention depends upon avoiding the vectors and reservoirs of infection and use of protective clothing and gloves. In the USA, a live attenuated vaccine is available for at-risk individuals (e.g. laboratory workers, hunters, trappers, etc.). |
|
Further details |
Multisystem zoonoses, pp. 414–5. |
|
Pasteurella Multocida |
|
|
Characteristics |
Facultatively anaerobic, small Gram-negative coccobacilli. Occurs as a commensal in the upper respiratory tract of many animals including livestock, poultry, and domestic pets. |
|
Laboratory identification |
Gram stain of pus or other fluid specimen. Organisms grow well on ordinary bacteriologic media at 37°C. Oxidase-positive and catalase-positive. Bipolar staining enhanced by Wright, Giemsa, or Wayson stains. |
|
Diseases |
Infected animal (e.g. cat or dog) bite. Acute onset of redness, pain, and swelling. |
|
Transmission |
Zoonotic (animal bite) infection. |
|
Pathogenesis |
Capsule. |
|
Treatment and prevention |
Treat animal bite as polymicrobial infection (e.g. a beta-lactam antibiotic such as amoxicillin combined with a beta-lactam inhibitor). |
|
Further details |
Multisystem zoonoses, p. 416. |
|
Genus Yersinia |
|
A member of the family Enterobacteriaceae. This genus contains a variety of species, only a few of which are considered important human pathogens. |
|
Yersinia Pestis |
|
|
Characteristics |
Gram-negative rods, facultatively anaerobic, zoonotic. |
|
Laboratory identification |
Exhibits bipolar staining with special (e.g. Wright–Giemsa, Wayson”s) stains. Grows best on media containing blood or tissue fluids. Tentative identification by biochemical reactions. Definitive identification by immunofluorescence. |
|
Diseases |
Bubonic plague results from multiplication within monocytes with production of antiphagocytic proteins. Reaching the lymph nodes an intense haemorrhagic inflammation develops. Dissemination via the bloodstream leads to haemorrhagic and necrotic lesions in multiple organs. Pneumonic plague results from inhalation leading to haemorrhagic consolidation and sepsis. |
|
Transmission |
Zoonotic infection transmitted to humans through the bite of fleas carried by rodents. |
|
Pathogenesis |
Multiple virulence factors including lipopolysaccharides with endotoxic activity, antiphagocytic envelope protein, and plasmid-encoded virulence factors. Concern has been expressed regarding the possible use of this organism as an agent of bioterrorism. |
|
Treatment and prevention |
Broad-spectrum cephalosporins, doxycycline, trimethoprim-sulphamethoxazole. Control and eradication of infected animals is important. |
|
Further details |
Multisystem zoonoses, pp. 413–4. |
|
Yersinia Enterocolitica |
|
|
Characteristics |
Gram-negative rods, zoonotic. A multitude of serotypes exist; however, depending on geographic origin, most causing human disease are serotype 03, 08, or 09. |
|
Laboratory identification |
Non-lactose-fermenting Gram-negative rods; urease-positive and oxidase-negative. Bipolar staining. Facultative anaerobe that grows best and is motile at 25°C but non-motile at 37°C. Diagnosis involves isolation of the organism from the patient”s faeces or other body fluid (blood, vomit, etc.). Confirmation by biochemical and serological tests. |
|
Diseases |
Y. enterocolitica most commonly causes enterocolitis although extraintestinal infections may also (rarely) occur. |
|
Transmission |
Infection results from ingestion of contaminated food and drink (e.g. unpasteurized milk, raw pork, etc.). The organism adheres to and penetrates the terminal ileum leading to non-specific ileocolitis with potential lymph node infection and bacteraemia. Symptoms include fever, abdominal pain and diarrhea which may be watery or bloody. Y. enterocolitica can grow at refrigeration temperatures and transmission by blood transfusion has been observed. |
|
Pathogenesis |
Multiple virulence factors including plasmid-encoded proteins related to adherence and invasion. |
|
Treatment and prevention |
Most enteric infections are self-limited. When necessary, treatment is usually with doxycycline, aminoglycosides, trimethoprim-sulphamethoxazole, or third-generation cephalosporins. Prevention includes avoiding contaminated food and drink. |
|
Further details |
Gastrointestinal tract infections, pp. 293, 297–8. |
|
Genus Legionella |
|
In the overall history of microbiology, this is one of the more recent discoveries, originally demonstrated by techniques used for virus isolation (e.g. growth in embryonated hens” eggs). In free-living state, can grow in water, but difficult to cultivate on routine laboratory media. L. pneumophila is the pathogen of greatest medical importance. |
|
Legionella Pneumophila |
|
|
Characteristics |
In tissue appear as Gram-negative coccobacilli; pleomorphic on laboratory media; stain poorly with Gram”s stain (and therefore easily missed). Fastidious growth requirements in laboratory. |
|
Laboratory identification |
Direct fluorescent antibody tests performed on sputum samples have the advantage of specificity, distinguishing L. pneumophila from environmental contaminants. However, relatively few organisms may be present in expectorated sputum. Silver staining techniques are better than standard Gram staining method. Require enriched media containing iron and cysteine and absorbents to remove fatty acids. Most require incubation for 3–5 days for growth. Produces small tenacious colonies. Further identification based on requirement for cysteine and serologic characteristics. Diagnosis is often based on antibody detection rather than culture. |
|
Diseases |
Legionnaires” disease; one of the causes of atypical pneumonia. Pontiac fever which may be caused by other species is a less severe flu-like illness. |
|
Transmission |
Environmental saprophyte acquired by inhalation of contaminated water from showers, air conditioning systems, cooling towers. |
|
Pathogenesis |
Virulence factors unclear, but intracellular survival in alveolar macrophages important. Host predisposition (e.g. immunocompromise, chronic lung disease) important. |
|
Treatment and prevention |
Fluoroquinolone or newer macrolide (azithromycin). No vaccine available; prevention depends upon maintenance of hot water and air-conditioning systems, particularly in large buildings, such as offices, hospitals and hotels. |
|
Further details |
Antimicrobial agents, p. 496; lower respiratory tract infection, pp. 233, 235. |
|
Gardnerella Vaginalis |
|
|
Characteristics |
Formerly known as Haemophilus vaginalis and Corynebacterium vaginalis. Gram-variable facultatively anaerobic rods. |
|
Laboratory identification |
Special culture requirements (e.g. increased levels of carbon dioxide). Vaginal epithelial cells covered with “clue cells” (Gram-variable coccobacilli) and the amine or “whiff” test (i.e. presence of fishy odour after addition of potassium hydroxide to a sample of vaginal discharge) helpful in diagnosis. |
|
Diseases |
Cause a variety of genitourinary infections but one of a number of organisms commonly associated with bacterial vaginosis. To a lesser extent may also be associated with genitourinary infections (i.e. lower urinary tract) in men. |
|
Transmission |
Transmitted by sexual contact. |
|
Pathogenesis |
Poorly understood. |
|
Treatment and prevention |
Metronidazole, clindamycin. Condom use may aid in prevention. |
|
Further details |
Sexually transmitted disease, p. 273. |
|
Spiral Bacteria |
|
There are three genera of medical importance: Treponema, Leptospira and Borrelia. |
|
Genus Treponema |
|
|
Regularly coiled spirochetes with a longer wavelength than Leptospira. Several species and subspecies are important human pathogens; others are members of the normal flora, especially in the mouth. T. pallidum and its subspecies pertenue, and T. carateum are the most important species. |
|
|
Characteristics |
Individual cells too small to visualize by direct light microscopy; can be seen with dark ground (darkfield) illumination or after silver impregnation or immunofluorescent staining. Cells are actively motile by means of flagella contained within the periplasmic sheath. |
|
Laboratory identification |
T. pallidum and closely related species cannot be grown in artificial media; diagnosis of infection depends upon microscopic examination of fluid from primary lesions and on serology. |
|
Diseases |
T. pallidum: syphilis. T. pallidum-pertenue and T. carateum: the non-sexually transmitted treponematoses, yaws and pinta. |
|
Transmission |
Very susceptible to heat and drying, so successful transmission depends upon very close contact. T. pallidum is spread by close sexual contact and may also be vertically transmitted in utero. Yaws and pinta spread by direct contact from infected skin lesions. No animal reservoir. |
|
Pathogenesis |
Study of virulence factors hampered by the inability to grow T. pallidum in artificial culture media. Disease presents characteristically in three phases: after local primary infection, organisms widely disseminated in the body and may become quiescent for months or years. Immunopathology plays a major role in causing damage to the host, particularly in the tertiary stage of disease. |
|
Treatment and prevention |
Penicillin is the treatment of choice for syphilis. Doxycycline or tetracycline may be given to penicillin-allergic patients. Prevention depends upon detection and treatment of cases, contact tracing and serologic testing of pregnant women. Possible cross-reactions between T. pallidum and the species causing yaws and pinta must be noted. |
|
Further details |
Congenital infections, p. 327; sexually transmitted disease, pp. 261–6. |
|
Genus Leptospira |
|
Two species: L. interrogans and L. biflexa; the former is parasitic, the latter contains free-living species. Within the species interrogans there are several different serogroups and serovars responsible for disease in humans and animals. |
|
Leptospira Interrogans |
|
|
Characteristics |
Finely coiled spirochetes with hooked ends. Cells 0.1–0.2 μm in diameter, up to 20 μm in length. Not visible by direct light microscopy unless stained by silver impregnation or immunofluorescent methods. Dark ground (darkfield) microscopy reveals rotational and directional motility by means of periplasmic flagella. |
|
Laboratory identification |
Direct microscopy of blood and urine possible, but difficult to interpret. Leptospira can be grown, with difficulty, in special serum-containing media. Serologic diagnosis is usual. Commercial kits available. |
|
Diseases |
Leptospirosis or Weil”s disease in humans and animals. |
|
Transmission |
Leptospirosis in humans is a zoonosis, usual hosts being rodents, bats, cattle, sheep, goats and other domestic animals. Leptospires excreted in urine contaminate food and water. Infection occurs by contact either through occupation (e.g. sewer workers, farmers, abattoir workers) or recreation (e.g. canoeing, windsurfing on inland waters). Organisms may penetrate unabraded skin and conjunctiva. |
|
Pathogenesis |
After initial invasion, there is haematogenous spread before the organisms localize in various organs including the liver and kidney. Subclinical infection is common in endemic areas. |
|
Treatment and prevention |
Penicillin; doxycycline in penicillin-allergic patients. Disease may be prevented after exposure by doxycycline. |
|
Further details |
Multisystem zoonoses, pp. 416–7. |
|
Genus Borrelia |
|
|
Two species of Borrelia are of importance in humans: B. burgdorferi causes Lyme disease; B. recurrentis causes relapsing fever. |
|
|
Characteristics |
Less finely coiled than the leptospires. Cells 0.2–0.5 μm in diameter; stain readily, so are visible by light microscopy. |
|
Laboratory identification |
Microaerophilic, complex nutritional requirements, long growth time (weeks) thus culture is not routinely used for identification. B. recurrentis demonstrated in blood smears by staining with Giemsa or acridine orange. B. burgdorferi much more difficult to visualize. Culture from biopsy material possible, but difficult; diagnosis usually by serology. |
|
Diseases |
In relapsing fever, the relapsing element may be due to antigen switching. Lyme disease slowly progressive rather than relapsing. Characteristic “bull”s eye” skin lesion (erythema chronicum migrans) commonly occurs. Joint pains and fatigue common and later, in untreated cases, neurologic and cardiac manifestations. |
|
Transmission |
B. recurrentis spread from person to person by lice. Lyme disease is a zoonosis transmitted to humans by hard ticks (lxodes spp.) associated with deer. Ticks are found on bracken and undergrowth and attach to exposed skin. Tick bite is often unnoticed, but less than a minute is required for the organisms to enter the host. |
|
Pathogenesis |
Little is known about the pathogenesis of either disease. Antigen switching in B. recurrentis presumably allows evasion of host”s antibody response. |
|
Treatment and prevention |
Doxycycline; erythromycin and amoxicillin have also been used successfully. Prevention depends upon avoiding contact with vectors (e.g. protective clothing for walkers and forestry workers). |
|
Further details |
Host–parasite relationship, p. 174; vector-borne, pp. 397–9. |
|
OTHER BACTERIA |
|
|
Mycoplasmas |
|
|
Characteristics |
Distinguished from other prokaryotes and placed in the class Mollicutes because they lack a true cell wall and consequent rigidity. This is a stable characteristic exhibited by genera such as Mycoplasma and Ureaplasma and is distinct from cell wall-deficient and L-forms of other species. The outer membrane, the outermost layer, functions as the major antigenic interface. It is a flexible triple-layered structure of proteins and lipids. Many species also contain cholesterol in the membrane, which is absent from other bacterial cells. The important species is M. pneumoniae, but M. hominis and U. urealyticum may cause genital tract infections. |
|
Laboratory identification |
Many species are fastidious, and complex media and soft agar may be required for satisfactory culture. Cultures incubated for at least 7 days, although some species (e.g. M. hominis) grow readily on moist blood agar plates within 48 h. Cells variable in size (up to 100 μm but many smaller than 0.5 μm) and morphology; cannot be stained by Gram”s stain (no cell wall), but impressions of colonies can be stained with Dienes” or Romanowsky”s stains. Diagnosis of infection based on serology because of difficulties of culture. |
|
Diseases |
M. pneumoniae is an important cause of “atypical pneumonia”. Mycoplasmas are also associated with genital infections (e.g. non-gonococcal urethritis) and with joint and other inflammatory infections. Other mycoplasmas are important pathogens of animals and birds. |
|
Transmission |
Transmission of M. pneumoniae is from person to person by airborne route. Other mycoplasmas and ureaplasmas can be transmitted by sexual contact. |
|
Pathogenesis |
Surface protein adhesin binds M. pneumoniae to sialoglycolipids on respiratory epithelium of host. Other virulence factors are not yet clearly understood. |
|
Treatment and prevention |
Doxycycline or erythromycin (note that the lack of cell wall target means lack of susceptibility to beta-lactams). No vaccine currently available. Prevention by interruption of spread is difficult. |
|
Further details |
Sexually transmitted diseases, p. 272. |
|
Rickettsiae |
|
|
Characteristics |
These organisms have requirement for coenzyme A, NAD and ATP, which they cannot supply themselves, and are therefore obligate intracellular parasites; with rare exceptions they need to be grown in cell cultures or experimental animals. |
|
Laboratory identification |
Small (0.7–2 μm diameter), Gram-negative bacteria. Isolation in laboratory is difficult for the reasons outlined above (and may carry a high risk of laboratory-acquired infection); therefore rarely attempted outside specialized facilities. Diagnosis of infection based on serology. |
|
Diseases |
Typhus; Rocky Mountain, Mediterranean and other spotted fevers; Q fever. |
|
Transmission |
Maintained in animal reservoirs and transmitted by bites of ticks, fleas, mites and lice. In contrast, Coxiella burnetii (a related organism now moved to a separate genus) survives drying and is transmitted in aerosols from animals or materials contaminated by infected animals and inhaled. |
|
Pathogenesis |
Mechanisms unclear, but organisms have a predilection for endothelial cells, giving rise to characteristic primary skin lesion (in spotted fevers) and vasculitis. The intracellular habitat is important to the organism”s survival in the face of host defences. |
|
Treatment and prevention |
Tetracyclines generally used. Beta-lactams ineffective. Infection prevented by avoiding contact with vectors. Vaccines available for at-risk groups (e.g. veterinarians, farm workers). |
|
Further details |
Diagnosis and control, p. 468; gastrointestinal tract infections, p. 311; multisystem zoonoses, p. 412; vector-borne, pp. 393–7. |
|
Chlamydiae |
|
|
Characteristics |
Obligate intracellular parasites (unable to synthesize ATP) with distinct life cycle involving elementary bodies and reticulate bodies. Small cells with genome approximately 25% of that of E. coli. Important species are Chlamydia trachomatis, Chlamydophila psittaci andChlamydophila pneumoniae. |
|
Laboratory identification |
Must be grown in cell culture, so cultural techniques are limited to specialized laboratories. In cell cultures, C. trachomatis forms characteristic, glycogen-containing inclusion bodies, which can be stained with iodine. Both C. psittaci and C. trachomatis contain specific surface antigens that allow detection by immunofluorescent antibody techniques. Nucleic acid based tests for C. trachomatis are also available. C. pneumoniae is currently detectable only by serology. |
|
Diseases |
C. trachomatis causes trachoma (eye infection), urethritis and other infections of the genital tract, and pneumonitis in newborns, acquired during birth from infected mothers. C. pneumoniae, described more recently, now recognized as important cause of atypical pneumonia. C. psittaci causes the atypical pneumonia, psittacosis. |
|
Transmission |
C. pneumoniae and C. psittaci are acquired by inhalation, the latter from infected birds or contaminated bird litter. C. trachomatis is spread by direct contact and is sexually transmitted. |
|
Pathogenesis |
Virulence factors remain unclear, but the intracellular habitat and different life cycle forms help organisms to evade host defences. Uptake into cells may be by parasite-encoded mechanisms. |
|
Treatment and prevention |
Tetracyclines, erythromycin (tetracycline should not be used in children). Vaccines not available and may not be useful because of the immunopathologic element of the infections. |
|
Further details |
Diagnosis and control, pp. 472, 468–9; eye infections, pp. 351–2; host–parasite relationship, p. 135; sexually transmitted disease, pp. 268–70. |
Fungi
|
SUPERFICIAL MYCOSES |
|
|
Dermatophytes |
|
|
General term for species invading superficial layers of skin. Of the many species involved, those belonging to Epidermophyton, Microsporum and Trichophyton are of greatest importance. |
|
|
Characteristics |
Filamentous fungi invading surface keratinized structures: skin, hair, nails. Hyphae penetrate between cells. |
|
Laboratory identification |
Examination of KOH-treated skin scrapings for hyphae; fluorescence under Wood”s lamp. Culture on media useful in identifying species. Both Sabouraud dextrose agar (SDA) and dermatophyte test medium (DTM) can be used. |
|
Diseases |
Tinea, ringworm, athlete”s foot. |
|
Transmission |
By fungal material on skin scales. |
|
Pathogenesis |
Skin inflammation, pruritus – sometimes localized hypersensitivity reactions. |
|
Treatment and prevention |
Topical (imidazoles) and oral antifungal agents (griseofulvin, itraconazole, terbinafine). Improved skin care and hygiene. |
|
Further details |
Host–parasite relationship, pp. 141–2; infection of skin, soft tissue, muscle and associated systems, pp. 369–71. |
|
Sporothrix Schenckii |
|
|
Characteristics |
Dimorphic fungus (capable of growing as both single-celled yeast and multicelled hyphae). Occurs in external environment. Invades subcutaneous tissues. |
|
Laboratory identification |
Budding cells in inflammatory exudate from lesions. Culture on SDA. |
|
Diseases |
Sporotrichosis. |
|
Transmission |
Direct fungal contamination of wounds in skin (e.g. those made by thorns). |
|
Pathogenesis |
Ulceration or abscess formation in draining lymphatics. |
|
Treatment and prevention |
Oral potassium iodide, itraconazole. |
|
Further details |
Infection of skin, soft tissue, muscle and associated systems, pp. 372–3. |
|
DEEP MYCOSES |
|
|
ASPERGILLUS |
|
|
A. fumigatus is the most important of three common species, the others being A. flavus and A. niger. |
|
|
Characteristics |
Filamentous fungi causing opportunistic infections in immunocompromised patients. Occur widely in external environment. Invade lungs and blood vessels. |
|
Laboratory identification |
Presence of hyphae in tissues. Culture on SDA. PCR. Serology. |
|
Diseases |
Aspergillosis. |
|
Transmission |
Inhalation of airborne stages (conidia). |
|
Pathogenesis |
Causes thrombosis and infarction when blood vessels invaded. Partial blockage of airways from fungal mass. Allergic bronchopulmonary reactions. |
|
Treatment and prevention |
Amphotericin B or its lipid complexes. High doses needed for neutropenic patients. Voriconazole or caspofungin are alternatives. |
|
Further details |
Diagnosis and control, p. 559; general, pp. 47–8; immunocompromised host, p. 437; lower respiratory tract infection, pp. 249–50. |
|
Blastomyces Dermatitidis |
|
|
Characteristics |
Dimorphic fungus. Invades through lungs, can become widely disseminated in body. |
|
Laboratory identification |
Yeast cells in sputum or skin lesions. Culture on SDA. |
|
Diseases |
Blastomycosis. |
|
Transmission |
Inhalation of air-borne spores. |
|
Pathogenesis |
Fungal infection in lungs. Presentation may be confused with tuberculosis. Skin involved in 40–80% of cases. Can produce abscesses. |
|
Treatment and prevention |
If needed (e.g. progressive disease) Amphotericin B, itraconazole. |
|
Further details |
Infection of skin, soft tissue, muscle and associated systems, p. 373. |
|
Candida Albicans |
|
|
Characteristics |
Dimorphic fungus, occurring as yeast on mucosal surfaces as component of normal flora, but forms hyphae when invasive. Produces opportunistic infections in stressed, suppressed and antibiotic-treated individuals. Paracoccidioides brasiliensis in central and South America has many similarities. |
|
Laboratory identification |
Fungal stages in tissues. Culture on SDA. Isolates may be typed by molecular techniques. Serological methods can be used for disseminated disease, but less helpful in neutropenic patients. |
|
Diseases |
Candidiasis, thrush. |
|
Transmission |
Part of normal flora of skin, mouth and intestine. |
|
Pathogenesis |
Localized mucocutaneous lesions; invasion of all major organs in the disseminated condition. |
|
Treatment and prevention |
Topical and oral antifungals (e.g. nystatin, miconazole). Fluconazole, itraconazole, amphotericin B for disseminated disease. |
|
Further details |
General, p. 47; host–parasite relationship, pp. 63–5; immunocompromised host, pp. 436–7; infection of skin, soft tissue, muscle and associated systems, p. 372; sexually transmitted disease, p. 272; upper respiratory tract infection, p. 225. |
|
Coccidioides Immitis |
|
|
Characteristics |
Dimorphic fungus, growing as hyphae in soils, but as yeast-like endospores within capsules (spherules) in tissues. Invasion through lungs; can become widely disseminated in body. |
|
Laboratory identification |
In sputum or tissues. Culture on SDA. PCR. Serology. |
|
Diseases |
Coccidioidomycosis. Indigenous to the Americas. |
|
Transmission |
Inhalation of airborne stages (arthroconidia). |
|
Pathogenesis |
Lung infections give mild, influenza-like condition, but serious illness may follow dissemination. |
|
Treatment and prevention |
Amphotericin B, itraconazole, fluconazole for at-risk patients. |
|
Further details |
Central nervous system (CNS), p. 340; infection of skin, soft tissue, muscle and associated systems, p. 373. |
|
Cryptococcus Neoformans |
|
|
Characteristics |
Encapsulated yeast-like fungus common in soils where there are bird droppings. Invades through lungs; can spread to CNS. |
|
Laboratory identification |
Encapsulated yeast cells in sputum or cerebrospinal fluid. Culture on SDA. Molecular methods. Serology. |
|
Diseases |
Cryptococcosis. |
|
Transmission |
Inhalation of airborne cells. |
|
Pathogenesis |
Lung infection may result in influenza-like condition or pneumonia. In immunocompromised patients, CNS involvement leads to meningitis. |
|
Treatment and prevention |
Amphotericin B plus flucytosine, followed by azole therapy. |
|
Further details |
Central nervous system (CNS), pp. 339–40; general, pp. 47–8; immunocompromised host, p. 437; infection of skin, soft tissue, muscle and associated systems, p. 373. |
|
Histoplasma Capsulatum |
|
|
Characteristics |
Dimorphic fungus, growing as hyphae in soil where there are bird droppings. Invades through lungs and grows as yeast cells, which can survive intracellularly after phagocytosis. Can become widely disseminated in body. |
|
Laboratory identification |
Yeast cells in sputum or tissues. Culture on SDA. Molecular typing of isolates. Serology. |
|
Diseases |
Histoplasmosis. |
|
Transmission |
Inhalation of airborne spores. |
|
Pathogenesis |
Many infections are asymptomatic. Can produce acute and chronic pulmonary disease. Serious illness results from dissemination into other organs. |
|
Treatment and prevention |
Amphotericin B, itraconazole. |
|
Further details |
General, p. 47; immunocompromised host, p. 437. |
|
Pneumocystis JirovecIi (Carinii) |
|
|
Characteristics |
Respiratory organism previously classed as a sporozoan protozoan, now classified as a fungus. Lives extracellularly within alveoli. |
|
Laboratory identification |
Histologic identification of organisms in sputum, bronchial lavage or tissues. PCR. |
|
Diseases |
Pneumonia-like condition, severe in immunocompromised patients. Worldwide distribution. |
|
Transmission |
Assumed to be by droplets. |
|
Pathogenesis |
Inflammation in lung. |
|
Treatment and prevention |
Trimethoprim-sulphamethoxazole or pentamidine. |
|
Further details |
General, p. 48; immunocompromised host, pp. 437–8; lower respiratory tract infection, p. 250. |
Protozoa
|
Cryptosporidium Hominis and C. Parvum |
|
|
Characteristics |
Intestinal coccidian, invades and reproduces in epithelial cells of small intestine. Forms oocysts, which are passed in faeces. |
|
Laboratory identification |
Small (5 μm) oocysts in faeces, detected by acid-fast staining and/or immunofluorescent staining. PCR. |
|
Diseases |
Cryptosporidiosis. Worldwide distribution. |
|
Transmission |
Faecal–oral. Swallowing infective oocysts, usually in contaminated water. Animal reservoirs of infection for C. parvum. |
|
Pathogenesis |
Invasion of epithelial cells causes diarrhea; can be profuse in immunocompromised patients. |
|
Treatment and prevention |
Nitazoxanide. Paromomycin is of limited value. Self-limiting in those with normal immunity. Treatment often required in immunocompromised patients, but antiparasitic drugs less effective in that group. Improved sanitation. |
|
Further details |
Gastrointestinal tract infections, pp. 307–8; general, p. 51. |
|
Cyclospora Cayetanensis |
|
|
Characteristics |
Intestinal coccidian. Forms oocysts, which are passed in faeces. |
|
Laboratory identification |
8–10 μm oocysts with two sporocysts found in faeces, detected by microscopy of faecal concentrate and/or acid-fast staining. PCR in some centres. |
|
Diseases |
Cyclosporiasis. |
|
Transmission |
Faecal–oral. Swallowing infective oocysts in contaminated food. It is unclear whether or not animals act as reservoir hosts. |
|
Pathogenesis |
Diarrhea. Infection can be serious in immunocompromised patients. |
|
Treatment and prevention |
Infections may be self-limiting. Trimethoprim-sulphamethoxazole if treatment required. Washing of fruit and vegetables. |
|
Further details |
Gastrointestinal tract infections, p. 308. |
|
Entamoeba Histolytica/Dispar |
|
|
Characteristics |
Intestinal amoeba, lives in intestine as trophozoite; produces resistant cysts, which are passed in faeces. Entamoeba histolytica and E. dispar have morphologically identical faecal cyst stages, but only E. histolytica is pathogenic. |
|
Laboratory identification |
Microscopic identification of cysts in faeces. ELISA or PCR to distinguish E. histolytica from E. dispar. Only E. histolytica produces haematophagous trophozoites in the faeces. Serology. |
|
Diseases |
Amoebic dysentery, liver abscess. Worldwide distribution, commonest in tropical and subtropical countries. |
|
Transmission |
Faecal–oral. Swallowing cysts in contaminated water or food. |
|
Pathogenesis |
Invasion of large bowel mucosa causes ulceration and diarrhea, often bloody. Spread to liver causes formation of bacteriologically sterile abscess. |
|
Treatment and prevention |
Metronidazole or tinidazole to kill invasive amoebae, followed by diloxanide furoate to kill amoebae in gut lumen. Hygiene and sanitation. |
|
Further details |
Gastrointestinal tract infections, pp. 305–6. |
|
Giardia Intestinalis (Formerly G. Lamblia) |
|
|
Characteristics |
Intestinal flagellate; lives on mucosa of small bowel. Produces cysts, which are passed in faeces. |
|
Laboratory identification |
Trophozoites in faeces, detected in fixed stained smears. Cysts in faeces seen in faecal concentrates. Direct recovery of trophozoites from duodenal aspirate. PCR. |
|
Diseases |
Giardiasis. Worldwide distribution. |
|
Transmission |
Faecal–oral. Swallowing cysts, usually in contaminated water. Animal reservoirs of infection. |
|
Pathogenesis |
Large numbers of trophozoites can cause severe diarrhea and impaired absorption. Most severe in immunocompromised patients. |
|
Treatment and prevention |
Metronidazole, tinidazole. Improved sanitation, water treatment. |
|
Further details |
General, p. 285; gastrointestinal tract infections, pp. 306–7; host–parasite relationship, p. 133. |
|
Genus Leishmania |
|
|
Genus contains several species, of which L. Viannia braziliensis and L. donovani complex cause serious disease. |
|
|
Characteristics |
Tissue flagellates living intracellularly in macrophages as amastigote stage. Transmitted by phlebotomine sandflies. |
|
Laboratory identification |
Presence of amastigotes in stained biopsy material, in-vitro culture of tissue specimens to obtain promastigotes. PCR of tissue specimens. |
|
Diseases |
Visceral (donovani complex), cutaneous (tropica, major) and mucosal (Viannia) leishmaniasis. Disease also known by many local names (e.g. Kala-azar, Oriental sore, Espundia). Commonest in tropical and subtropical countries. |
|
Transmission |
By bite of infected sandfly. |
|
Pathogenesis |
Visceral: hepatosplenomegaly from invasion of macrophages in liver and spleen; sometimes dermal nodules after treatment. Cutaneous: localized ulcers, which resolve. Mucosal: progressive invasion of mucosal tissues in nose and mouth. |
|
Treatment and prevention |
Liposomal amphotericin for visceral disease, Oral miltefosine in some cases. Local or systemic antimonials for cutaneous form. Avoidance of vectors. |
|
Further details |
Diagnosis and control, p. 468; host–parasite relationship, p. 159; infection of skin, soft tissue, muscle and associated systems, p. 373; vaccination, p. 541; vector-borne, pp. 404–5. |
|
Genus Plasmodium |
|
|
Genus contains five species causing disease: P. falciparum, P. malariae, P. ovale, P. vivax and P. knowlesi. P. falciparum and P. vivax are commonest. P. falciparum is by far the most dangerous, but P. knowlesi can cause illness of equal severity. |
|
|
Characteristics |
Sporozoa living intracellularly in liver and primarily in red blood cells. |
|
Laboratory identification |
Parasites in red blood cells in stained blood smear. Rapid antigen detection tests. PCR. |
|
Diseases |
Malaria. Commonest in tropical and subtropical countries. |
|
Transmission |
By bite of infected anopheline mosquito. |
|
Pathogenesis |
Bursting of infected red cells causes periodic fevers. In falciparum malaria, sequestration of infected cells in brain capillaries can cause fatal cerebral malaria; this infection is sometimes associated with intravascular haemolysis. Infection with P. malariae can lead to nephritis due to immune complex deposition. |
|
Treatment and prevention |
Many antimalarial drugs, but P. falciparum parasites show considerable drug resistance. Avoidance of vectors. Mosquito control. Field trials of vaccines underway. |
|
Further details |
Central nervous system (CNS), p. 347; diagnosis and control, p. 515; host–parasite relationship, p. 170; pathological consequences, pp. 198, 202; vector-borne (malaria), pp. 399–402. |
|
Toxoplasma Gondii |
|
|
Characteristics |
Coccidian living intracellularly, forming large tissue cysts. Natural host is cat, where parasite has enteric cycle, producing oocysts in faeces. In humans, organisms can invade many tissues. |
|
Laboratory identification |
Serology; need repeated tests to establish current infection or estimate when infection may have taken place. PCR in special cases. |
|
Diseases |
Toxoplasmosis. Worldwide distribution. |
|
Transmission |
Swallowing oocysts passed by cats; ingestion of tissue cysts in raw or undercooked meat; transplacental. |
|
Pathogenesis |
In adults, many cases are asymptomatic. Also causes mild influenza-like illness; lymph nodes may be enlarged. Symptoms more severe in immunocompromised patients. Congenital infections can damage eye or brain and prove fatal. |
|
Treatment and prevention |
In adults with normal immunity, specific treatment is not usually required. Pyrimethamine plus sulfadiazine plus folinic acid when indicated. Hygiene, cooking of meat. |
|
Further details |
General, pp. 327–8; central nervous system (CNS), p. 347; eye infections, pp. 353–4. |
|
Trichomonas Vaginalis |
|
|
Characteristics |
Flagellate living in genitourinary system of females and males. Trophozoite form only, no cyst. |
|
Laboratory identification |
Identification of trophozoites in stained material from vaginal smears. Culture. PCR. |
|
Diseases |
Trichomoniasis. Worldwide distribution. |
|
Transmission |
Venereal. |
|
Pathogenesis |
Mild in males; causes vaginitis with discharge in females. |
|
Treatment and prevention |
Metronidazole or tinidazole. Use of condoms. |
|
Further details |
Diagnosis and control, pp. 468, 502; sexually transmitted disease, p. 273. |
|
Genus Trypanosoma |
|
|
Genus contains three species that cause disease: T. brucei gambiense, T. brucei rhodesiense (African trypanosomiasis) and T. cruzi (American trypanosomiasis). |
|
|
Characteristics |
Flagellates living in blood and tissues. T. cruzi has intracellular stages. |
|
Laboratory identification |
Organisms in blood or cerebrospinal fluid (African) or blood or tissue biopsy (American). Serology (especially for T. cruzi). PCR. |
|
Diseases |
African trypanosomiasis (sleeping sickness): sub-Saharan Africa. American trypanosomiasis (Chagas disease): South America. |
|
Transmission |
By introduction of trypanosomes in the bite of an infected tsetse fly (African). American transmitted by trypanosomes in faeces of infected reduviid bug entering the site of its bite. |
|
Pathogenesis |
African: infection of CNS causing meningoencephalitis. American: destruction of infected cells, especially neurones, megacolon, megaoesophagus, sudden death from arrhythmia or ruptured cardiac aneurysm, cardiac failure. |
|
Treatment and prevention |
Various antitrypanosomal drugs, all toxic (e.g. arsenicals). Avoidance of vectors. Vector control. |
|
Further details |
Diagnosis and control, p. 468; infection of skin, soft tissue, muscle and associated systems, pp. 386–7; vector-borne, pp. 402–4. |
|
Microsporidia (Contains a number of species) |
|
|
Characteristics |
Intracellular pathogens, in intestine and other organs, characteristic spores. |
|
Laboratory identification |
Detection of organisms in faeces, urine, biopsies. Gram, modified trichrome and other stains can be used. PCR. |
|
Diseases |
Microsporidiosis. |
|
Pathogenesis |
Diarrhea, other symptoms dependent on organ infected. Infection can be serious in immunocompromised patients. |
|
Treatment and prevention |
Albendazole (response is species-variable and may be poor). Hygiene and sanitation. |
|
Further details |
Gastrointestinal tract infections, pp. 305, 308. |
|
Isospora Belli |
|
|
Characteristics |
A coccidian parasite infecting epithelial cells of the small intestine. |
|
Laboratory identification |
Diagnosis of infection based on observation of oocysts in faeces. Internal morphology of the oocyst helpful in identification. |
|
Diseases |
Chronic intestinal disease especially in immunocompromised patients. Symptoms of infection include diarrhea, malaise, and abdominal pain. |
|
Transmission |
Infection occurs by ingestion of sporulated oocysts from the faeces of another host. |
|
Pathogenesis |
Invasion of epithelial cells in the small intestine. Cycles of reproduction (asexual to sexual reproduction) with additional epithelial cell invasion and further production of oocysts, which are excreted in the stool. |
|
Treatment and prevention |
Trimethoprim-sulphamethoxazole. Hygiene and improved sanitation. |
|
Further details |
Gastrointestinal tract infections, p. 308; immunocompromised host, p. 440. |
Helminths
|
TAPEWORMS |
|
|
Diphyllobothrium Latum |
|
|
Characteristics |
Large adult tapeworm in intestine. Scolex with sucking grooves not suckers. Eggs released and passed in faeces. |
|
Laboratory identification |
Faecal concentrates. Eggs in faeces have characteristic operculum (lid). |
|
Diseases |
Diphyllobothriasis (fish tapeworm). Worldwide distribution. Commonest where fresh water fish eaten raw. |
|
Transmission |
Larval stages in fish. Adult worm acquired when infected fish eaten raw or undercooked. |
|
Pathogenesis |
Usually harmless; may be associated with vitamin B12 deficiency. |
|
Treatment and prevention |
Niclosamide, praziquantel. Cooking of fish. Sanitation. |
|
Further details |
Gastrointestinal tract infections, p. 311. |
|
Echinococcus Granulosus |
|
|
Characteristics |
Large fluid-filled (hydatid) cysts, in liver (approximately 66%), lungs (approximately 10%). Can occur in any organ or system, e.g. bone, CNS. |
|
Laboratory identification |
Scans, serology. |
|
Diseases |
Hydatidosis, hydatid disease. Worldwide distribution, commonest in sheep-rearing countries. |
|
Transmission |
Swallowing eggs released from adult tapeworms in dogs. Natural cycle is adult (dog), larval cysts (sheep). |
|
Pathogenesis |
Cysts exert pressure on internal organs. Release of cyst fluid can cause anaphylaxis. |
|
Treatment and prevention |
Albendazole. Surgical removal or therapeutic injection of cysts. Prevention of dogs eating infected viscera from sheep. Hygiene after handling dogs. |
|
Further details |
Central nervous system (CNS), p. 347; host–parasite relationship, p. 178; multisystem zoonoses, p. 419. |
|
Hymenolepis Nana and Hymenolepis Diminuta |
|
|
Characteristics |
Small (2–4 cm) adult tapeworms in intestine. Scolex with suckers and hooks. Eggs passed in faeces. Life cycle is direct for H. nana and via rat flea intermediate host for H. diminuta. |
|
Laboratory identification |
Microscopy of faecal concentrates. Thin-shelled eggs in faeces. |
|
Diseases |
Hymenolepiasis (dwarf tapeworm). Worldwide distribution. |
|
Transmission |
Swallowing eggs (H. nana). Accidental ingestion of larvae in rat fleas (H. diminuta). |
|
Pathogenesis |
Usually harmless. Numbers of H. nana can build up by autoinfection in children and enteritis may result. |
|
Treatment and prevention |
Niclosamide, praziquantel. Hygiene and sanitation. |
|
Further details |
Gastrointestinal tract infections, p. 311. |
|
Genus Taenia |
|
|
Two species of this genus infect humans: T. saginata and T. solium. |
|
|
Characteristics |
Large (metres) adult tapeworms in intestine. Scolices with suckers (saginata) or suckers and hooks (solium). Proglottids (segments) passed in faeces. Small cysts (larval stages of solium) in muscles, CNS, and eyes. |
|
Laboratory identification |
Proglottids in faeces. Species identifiable on basis of number of branches to uterus (T. saginata 15–30; T. solium 7–12). Eggs in faeces (morphologically identical for both species). Scans and serology for cysticercosis. |
|
Diseases |
Taeniasis (beef and pork tapeworms). Cysticercosis (larval stages in tissue) occurs with T. solium only. Worldwide distribution. |
|
Transmission |
Adult worms acquired by eating raw or undercooked meat (beef, saginata; pork, solium) from animals infected with larval stages. Cysticercosis via ingestion of T. solium eggs in food contaminated with faecal matter from a person infected with adult T. solium. |
|
Pathogenesis |
Adult worms essentially asymptomatic. In cysticercosis, cysts in brain can result in convulsions or other neurologic symptoms. |
|
Treatment and prevention |
Niclosamide, praziquantel for intestinal worms. Albendazole or praziquantel under steroid cover for cerebral cysticercosis. Adequate cooking of meat. Prevention of human faeces contaminating grazing and feeding areas of cattle and pigs. Hygiene. |
|
Further details |
Central nervous system (CNS), pp. 347–8; gastrointestinal tract infections, p. 311; infection of skin, soft tissue, muscle and associated systems, p. 387; multisystem zoonoses, p. 418. |
|
FLUKES |
|
|
Clonorchis Sinensis |
|
|
Characteristics |
Liver fluke. Narrow elongated worms in bile ducts. |
|
Laboratory identification |
Eggs in faecal concentrates. |
|
Diseases |
Clonorchiasis (Asia). |
|
Transmission |
Larval stages in fish; adult flukes acquired when infected fish eaten raw or undercooked. |
|
Pathogenesis |
Inflammation and distortion of bile ducts. Long-standing heavy infections can result in cholangiocarcinoma. |
|
Treatment and prevention |
Praziquantel. Cooking of fish. Sanitation. |
|
Further details |
Gastrointestinal tract infections, p. 320. |
|
Paragonimus Westermani |
|
|
Characteristics |
Lung fluke. Thick fleshy worms living as pairs in cysts. |
|
Laboratory identification |
Eggs in sputum or faeces. |
|
Diseases |
Paragonimiasis (Asia). |
|
Transmission |
Larval stages in crabs; adult flukes acquired when infected crab meat eaten raw or undercooked. |
|
Pathogenesis |
Inflammation of lungs, secondary bacterial infections. Cavities may be confused with tuberculosis. |
|
Treatment and prevention |
Praziquantel. Cooking of crab meat. Sanitation. |
|
Further details |
Lower respiratory tract infection, p. 251. |
|
Genus Schistosoma |
|
|
Genus contains several species able to infect humans. Three are of major importance: S. haematobium, S. japonicum and S. mansoni. |
|
|
Characteristics |
Blood flukes; adult worms in blood vessels around intestine (S. japonicum, S. mansoni) or bladder (S. haematobium). Eggs in tissues. |
|
Laboratory identification |
Microscopy of faecal concentrates and urine for eggs. Spined eggs in faeces (S. japonicum, small lateral spine; S. mansoni, large lateral spine). Eggs in urine (S. haematobium, terminal spine). Serology. |
|
Diseases |
Schistosomiasis. Widely distributed in tropical/subtropical countries (S. mansoni, Africa, S. America; S. haematobium, Africa, Middle East; S. japonicum, Asia). |
|
Transmission |
Larvae released from eggs infect aquatic snails. These release infective cercariae, which actively penetrate human skin. |
|
Pathogenesis |
Hypersensitivity responses to eggs cause inflammation, granuloma formation, fibrosis and obstructive disease in intestine, bladder and liver. |
|
Treatment and prevention |
Praziquantel. Avoidance of infected waters. Snail control. Sanitation. |
|
Further details |
General, p. 55; host–parasite relationship, p. 180; infection of skin, soft tissue, muscle and associated systems, p. 373; pathological consequences, p. 203; urinary tract infections (UTI), p. 258; vector-borne, pp. 405–6. |
|
NEMATODES |
|
|
Ascaris Lumbricoides |
|
|
Characteristics |
Large (up to 30 cm) intestinal roundworm; migratory stages pass through the lungs. |
|
Laboratory identification |
Microscopy of faecal concentrates. Thick-shelled eggs in faeces; worms also passed occasionally. |
|
Diseases |
Ascariasis. Worldwide distribution. Commonest in tropical and subtropical countries. |
|
Transmission |
Swallowing infective eggs in contaminated soil, food or water. |
|
Pathogenesis |
Migrating larvae cause pneumonitis. Adults can obstruct intestine, interfere with digestion and absorption of food, migrate into bile or pancreatic ducts. Allergic symptoms may occur in the acute phase. |
|
Treatment and prevention |
Mebendazole, piperazine. Hygiene and sanitation. |
|
Further details |
Gastrointestinal tract infections, pp. 308–9; host–parasite relationship, p. 133; pathological consequences, pp. 200–1. |
|
Enterobius Vermicularis |
|
|
Characteristics |
Small (1 cm) roundworm in large bowel. Worms emerge from anus at night to lay eggs. |
|
Laboratory identification |
Eggs recovered from perianal skin by saline swab or adhesive slide test; adult worms in faeces. |
|
Diseases |
Enterobiasis, threadworm, pinworm. Worldwide distribution. Commonest in children. |
|
Transmission |
Swallowing eggs, which can be carried on fingers and in dust. Eggs infective when laid, so direct reinfection is common. |
|
Pathogenesis |
Perianal pruritus. |
|
Treatment and prevention |
Mebendazole, pyrantel, piperazine. Hygiene. |
|
Further details |
Gastrointestinal tract infections, p. 308. |
|
Filarial Nematodes |
|
|
Large group. Most important species living in lymphatic tissues (Wuchereria bancrofti, Brugia malayi) or in skin (Onchocerca volvulus). |
|
|
Characteristics |
Adults very long, thin worms, living in lymphatics with microfilariae (larvae) in blood (Wuchereria, Brugia) or in subcutaneous nodules with microfilariae in skin (Onchocerca). |
|
Laboratory identification |
Detection of microfilariae in stained blood smear, fresh filtered blood or fresh skin snip. Serology less helpful. Antigen detection for Wuchereria. |
|
Diseases |
Lymphatic filariasis (Wuchereria, Brugia). Onchocerciasis or river blindness (Onchocerca). |
|
Transmission |
Microfilariae taken up by blood-feeding insects (mosquitoes, Wuchereria, Brugia; Simulium blackflies, Onchocerca), develop to infective stage and reintroduced into humans at the next blood meal. Widely distributed in tropical and subtropical countries. |
|
Pathogenesis |
In lymphatic filariasis, adult worms cause inflammation of lymph nodes and blockage of lymphatics, sometimes causing elephantiasis (big leg). In onchocerciasis, hypersensitivity to microfilariae leads to skin and eye lesions. |
|
Treatment and prevention |
Diethyl carbamazine (lymphatic) and ivermectin (onchocerciasis). Avoidance of vectors. Vector control. |
|
Further details |
Eye infections, p. 355; infection of skin, soft tissue, muscle and associated systems, pp. 373–4; vector-borne, pp. 406–7. |
|
Hookworms |
|
|
General term for intestinal bloodsucking worms. Two major species: Ancylostoma duodenale and Necator americanus. |
|
|
Characteristics |
Small (1 cm) intestinal roundworms; migratory stages pass through skin and lungs. Adult worms have expanded mouths for attachment to intestinal mucosa. |
|
Laboratory identification |
Microscopy of faecal concentrates. Thin-shelled eggs in faeces. |
|
Diseases |
Hookworm disease. Widespread in tropical and subtropical countries. |
|
Transmission |
Infective larvae penetrate skin (both species) or mucous membranes after ingestion (Ancylostoma). |
|
Pathogenesis |
Bloodsucking of worms can lead to anaemia and protein loss. Larval penetration associated with local dermatitis (“ground itch”). |
|
Treatment and prevention |
Mebendazole, pyrantel. Hygiene and sanitation. |
|
Further details |
Gastrointestinal tract infections, p. 309; infection of skin, soft tissue, muscle and associated systems, p. 373. |
|
Strongyloides Stercoralis |
|
|
Characteristics |
Minute (2 mm) intestinal roundworm, living in humans only as larvae and parthenogenetic females. Migratory stages pass through skin and lungs, often seen in lungs in hyperinfestation. Eggs hatch in intestine; rhabditiform larvae in faeces may become infective filariform larvae directly or initiate a free-living generation in soil, from which infective filariform larvae develop. |
|
Laboratory identification |
Larvae in fresh faecal specimens. Charcoal culture. Serology. |
|
Diseases |
Strongyloidiasis. Widespread in tropical and subtropical countries. |
|
Transmission |
Infective larvae penetrate skin. |
|
Pathogenesis |
In immunocompromised patients, repeated autoinfection (development of larvae released from females in the intestine) can lead to hyperinfection (disseminated strongyloidiasis), with larvae invading all body tissues. Hyperinfection can be fatal. Diarrhea and malabsorption accompany heavy intestinal infections. |
|
Treatment and prevention |
Ivermectin. Hygiene and sanitation. |
|
Further details |
Gastrointestinal tract infections, p. 309; general, p. 53; host–parasite relationship, p. 70; multisystem zoonoses, pp. 419–20. |
|
Toxocara Canis |
|
|
Characteristics |
Invasion of larvae of roundworm Toxocara canis normally maturing in intestine of dogs. |
|
Laboratory identification |
Serology. |
|
Diseases |
Toxocariasis, visceral larva migrans, ocular larva migrans. Worldwide distribution. |
|
Transmission |
Swallowing infective eggs passed by dogs in contaminated soil, food or water. |
|
Pathogenesis |
Invasion of body tissues causing granulomatous inflammatory responses. Larvae in liver may lead to hepatomegaly, in lungs to pneumonitis, and in CNS to convulsions. Ocular granulomas may cause unilateral visual loss. |
|
Treatment and prevention |
Albendazole. Hygiene. Routine deworming of puppies and pregnant bitches. |
|
Further details |
Central nervous system (CNS), p. 347; eye infections, pp. 354–5; pathological consequences, p. 203. |
|
Trichinella Spiralis |
|
|
Characteristics |
Minute (2–3 mm) roundworms, living as adults in the intestine. Coiled larvae in muscles. Low host specificity; infects and matures in wide variety of mammals. |
|
Laboratory identification |
Clinical signs, serology, muscle biopsy. |
|
Diseases |
Trichinellosis (trichinosis). Worldwide distribution. |
|
Transmission |
Acquired by eating raw or undercooked meat (usually pork) containing infective larvae. |
|
Pathogenesis |
Diarrhea during intestinal phase. Allergic symptoms, skeletal muscle pain, myocarditis; can be fatal. |
|
Treatment and prevention |
Early treatment with albendazole or mebendazole. Adjunctive corticosteroids if symptoms severe. Cooking of meat. |
|
Further details |
Host–parasite relationship, p. 133; infection of skin, soft tissue, muscle and associated systems, p. 387; multisystem zoonoses, p. 419. |
|
Trichuris Trichiura |
|
|
Characteristics |
Medium size (3–5 cm) roundworms in large bowel. Body of characteristic “whipworm” form, with long thin anterior and short, thicker posterior. |
|
Laboratory identification |
Microscopy of faecal concentrate. Eggs in faeces have characteristic shape, oval with plug at each pole. |
|
Diseases |
Trichuriasis. Worldwide distribution. Commonest in tropical and subtropical countries. |
|
Transmission |
Swallowing infective eggs in contaminated soil, food or water. |
|
Pathogenesis |
Light infections asymptomatic. Heavy infections may produce trichuris dysentery syndrome with rectal prolapse, rectal bleeding, anaemia, growth stunting and growth retardation in children. |
|
Treatment and prevention |
Mebendazole. Hygiene and sanitation. |
|
Further details |
Gastrointestinal tract infections, pp. 308–10. |