Cracking the AP Chemistry Exam
Part IV
Content Review for the AP Chemistry Exam
Chapter 5
Big Idea #3: Chemical Reactions, Energy Changes, and Redox Reactions
Changes in matter involve the rearrangement and/or reorganization of atoms and/or the transfer of electrons
TYPES OF REACTIONS
Reactions may be classified into several categories.
1. Synthesis Reactions − When simple compounds are combined to form a single, more complex compound.
2Mg(s) + O2(g) → 2 MgO(s)
2. Decomposition − The opposite of a synthesis. A reaction where a single compound is split into two or more simple compounds, usually in the presence of heat.
HgO(s) + Heat → Hg(s) + ½O2(g)
3. Acid-Base Reaction − A reaction when an acid (i.e. H+) reacts with a base (i.e. OH−) to form water and a salt.
HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l)
4. Oxidation-Reduction (Redox) Reaction − A reaction that results in the change of the oxidation states of some participating species.
Cu2+(aq) + 2e− → Cu(s)
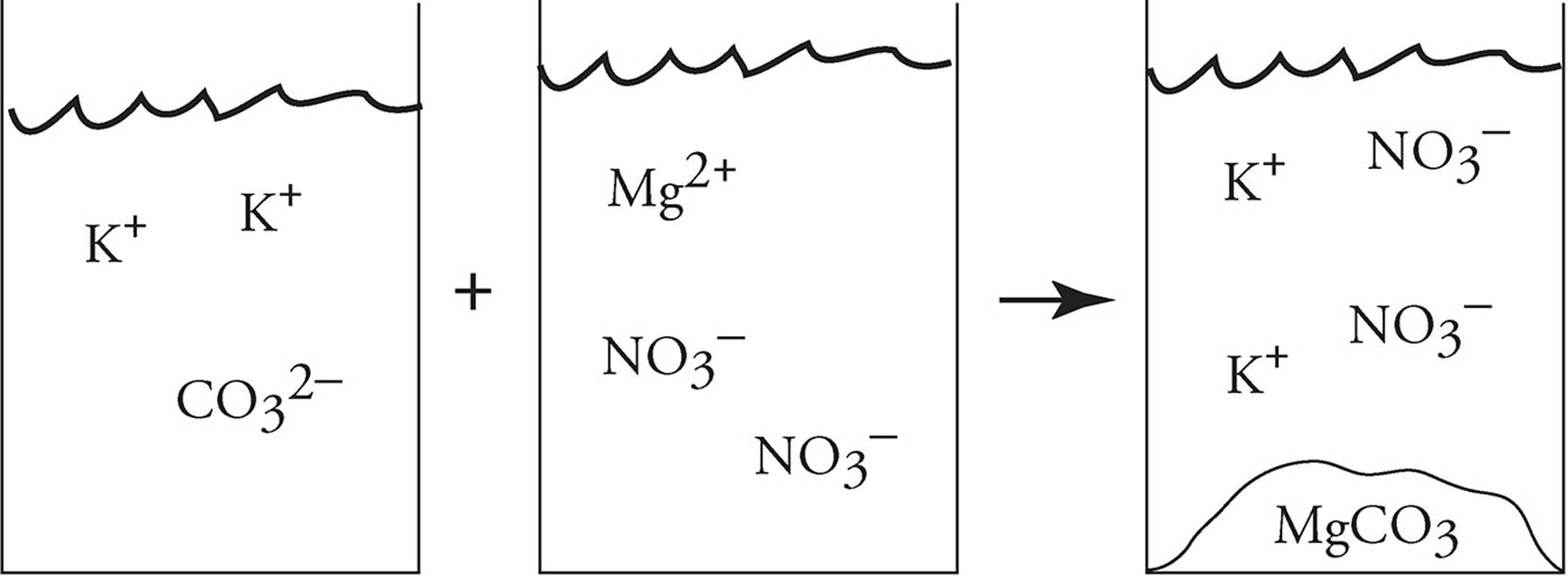
5. Precipitation − When two aqueous solutions mix, sometimes a new cation/anion pairing can create an insoluble salt. This type of reaction is called a precipitation reaction. When potassium carbonate and magnesium nitrate mix, a precipitate of magnesium carbonate will form as follows:
K2CO3(aq) + Mg(NO3)2(aq) → 2KNO3(aq) + MgCO3(s)
That can also be written as a net ionic equation. In solution, the potassium and nitrate ions do not actually take part in the reaction. They start out as free ions and end up as free ions. We call those ions spectator ions. The only thing that is changing is that carbonate and magnesium ions are bonding to form magnesium carbonate. The net ionic equation is
CO32− + Mg2+ → MgCO3(s)
Finally, this can be represented using particulate diagrams, as shown below:
You may have heard that you need to memorize solubility rules—that is, what ions will form insoluble precipitates when you combine. You DO NOT need to memorize most of these; for the most part the AP exam will provide you with them as needed. The only ones you need to know are:
1. Compounds with an alkali metal cation (Na+, Li+, K+, etc) or an ammonium cation (NH4+) are always soluble.
2. Compounds with a nitrate (NO3−) anion are always soluble.
That’s it! You should know how to interpret any solubility rules that you are given on the test, but you need not memorize anything beyond those two.
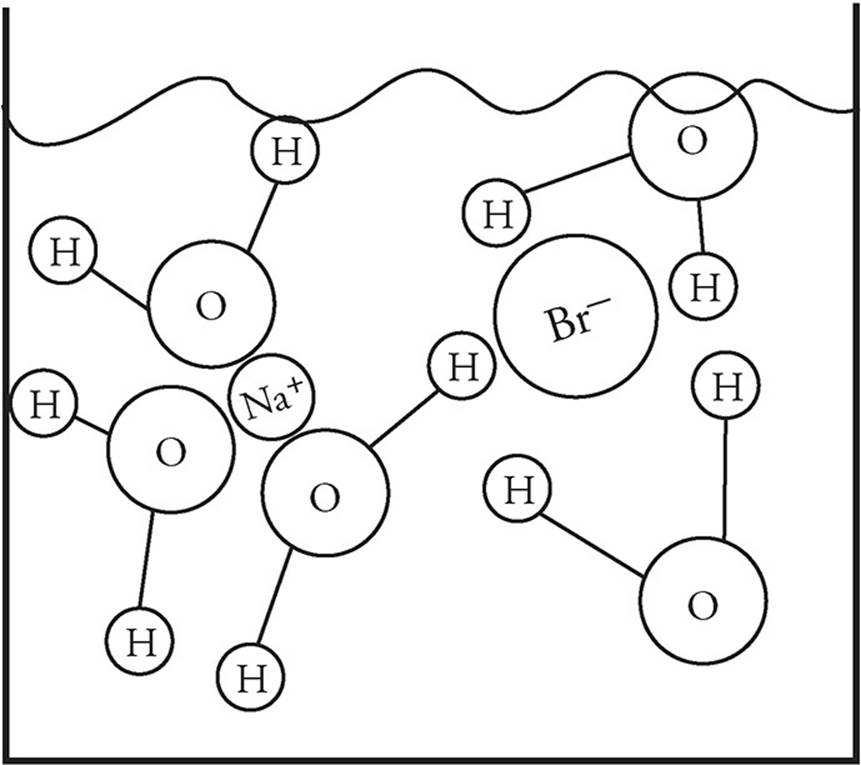
Ionic substances that dissolve in water do so because the attraction of the ions to the dipoles of the water molecules. Let’s take NaBr for example:
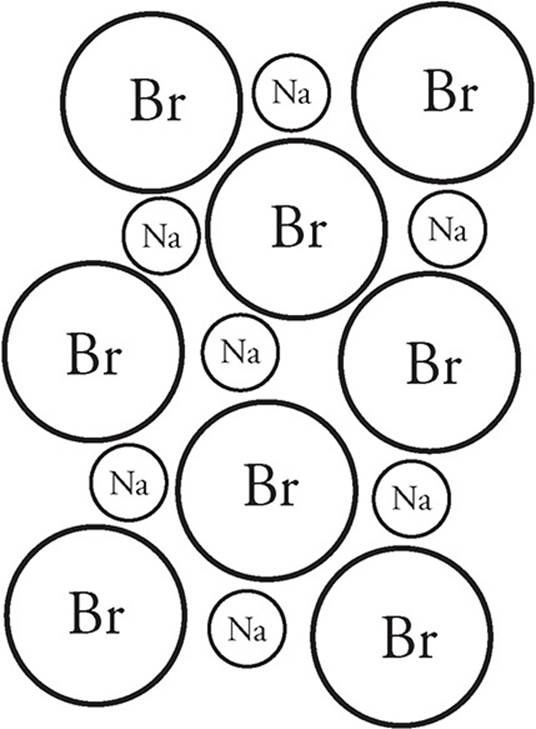
When it dissolves in water, the positive Na+ cations are attracted to the negative (oxygen) ends of the water molecules. The negative Br− anions are attracted to the positive (hydrogen) ends of the water molecules. Thus, when the substance dissolves it looks like this on the particulate level: