CHEMISTRY THE CENTRAL SCIENCE
11 LIQUIDS AND INTERMOLECULAR FORCES
11.6 PHASE DIAGRAMS
The equilibrium between a liquid and its vapor is not the only dynamic equilibrium that can exist between states of matter. Under appropriate conditions, a solid can be in equilibrium with its liquid or even with its vapor. A phase diagram is a graphic way to summarize the conditions under which equilibria exist between the different states of matter. Such a diagram also allows us to predict which phase of a substance is present at any given temperature and pressure.
The phase diagram for any substance that can exist in all three phases of matter is shown in ![]() FIGURE 11.27. The diagram contains three important curves, each of which represents the temperature and pressure at which the various phases can coexist at equilibrium. The only substance present in the system is the one whose phase diagram is under consideration. The pressure shown in the diagram is either the pressure applied to the system or the pressure generated by the substance. The curves may be described as follows:
FIGURE 11.27. The diagram contains three important curves, each of which represents the temperature and pressure at which the various phases can coexist at equilibrium. The only substance present in the system is the one whose phase diagram is under consideration. The pressure shown in the diagram is either the pressure applied to the system or the pressure generated by the substance. The curves may be described as follows:
![]() GO FIGURE
GO FIGURE
If the pressure exerted on a liquid is increased, while the temperature is held constant, what type of phase transition will eventually occur?
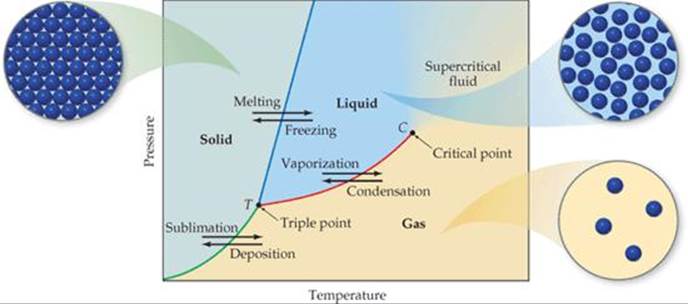
![]() FIGURE 11.27 Generic phase diagram for a pure substance. The green line is the sublimation curve, the blue line is the melting curve, and the red line is the vapor pressure curve.
FIGURE 11.27 Generic phase diagram for a pure substance. The green line is the sublimation curve, the blue line is the melting curve, and the red line is the vapor pressure curve.
1. The red curve is the vapor-pressure curve of the liquid, representing equilibrium between the liquid and gas phases. The point on this curve where the vapor pressure is 1 atm is the normal boiling point of the substance. The vapor-pressure curve ends at the critical point (C), which corresponds to the critical temperature and critical pressure of the substance. Beyond the critical point, the liquid and gas phases are indistinguishable from each other, and the substance is a supercritical fluid.
2. The green curve, the sublimation curve, separates the solid phase from the gas phase and represents the change in the vapor pressure of the solid as it sublimes at different temperatures.
3. The blue curve, the melting curve, separates the solid phase from the liquid phase and represents the change in melting point of the solid with increasing pressure. This curve usually slopes slightly to the right as pressure increases because for most substances the solid form is denser than the liquid form. An increase in pressure usually favors the more compact solid phase; thus, higher temperatures are required to melt the solid at higher pressures. The melting point at 1 atm is the normal melting point.
Point T, where the three curves intersect, is the triple point, and here all three phases are in equilibrium. Any other point on any of the three curves represents equilibrium between two phases. Any point on the diagram that does not fall on one of the curves corresponds to conditions under which only one phase is present. The gas phase, for example, is stable at low pressures and high temperatures, whereas the solid phase is stable at low temperatures and high pressures. Liquids are stable in the region between the other two.
The Phase Diagrams of H2O and CO2
![]() FIGURE 11.28 shows the phase diagram of H2O. Because of the large range of pressures covered in the diagram, a logarithmic scale is used to represent pressure. The melting curve (blue line) of H2O is atypical, slanting slightly to the left with increasing pressure, indicating that for water the melting point decreases with increasing pressure. This unusual behavior occurs because water is among the very few substances whose liquid form is more compact than its solid form, as we learned in Section 11.2.
FIGURE 11.28 shows the phase diagram of H2O. Because of the large range of pressures covered in the diagram, a logarithmic scale is used to represent pressure. The melting curve (blue line) of H2O is atypical, slanting slightly to the left with increasing pressure, indicating that for water the melting point decreases with increasing pressure. This unusual behavior occurs because water is among the very few substances whose liquid form is more compact than its solid form, as we learned in Section 11.2.
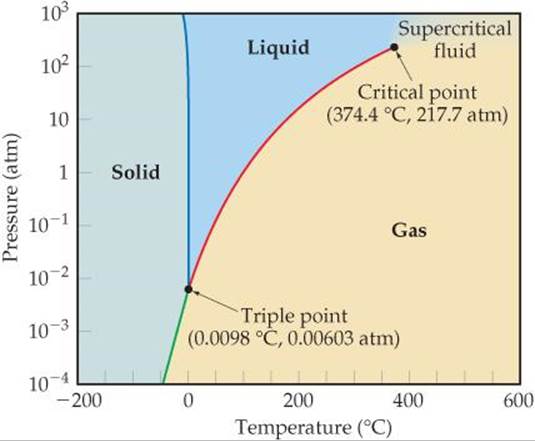
![]() FIGURE 11.28 Phase diagram of H2O. Note that a linear scale is used to represent temperature and a logarithmic scale to represent pressure.
FIGURE 11.28 Phase diagram of H2O. Note that a linear scale is used to represent temperature and a logarithmic scale to represent pressure.
If the pressure is held constant at 1 atm, it is possible to move from the solid to liquid to gaseous regions of the phase diagram by changing the temperature, as we expect from our everyday encounters with water. The triple point of H2O falls at a relatively low pressure, 0.00603 atm. Below this pressure, liquid water is not stable and ice sublimes to water vapor on heating. This property of water is used to “freeze-dry” foods and beverages. The food or beverage is frozen to a temperature below 0 °C. Next it is placed in a low-pressure chamber (below 0.00603 atm) and then warmed so that the water sublimes, leaving behind dehydrated food or beverage.
The phase diagram for CO2 is shown in ![]() FIGURE 11.29. The melting curve (blue line) behaves typically, slanting to the right with increasing pressure, telling us that the melting point of CO2 increases with increasing pressure. Because the pressure at the triple point is relatively high, 5.11 atm, CO2 does not exist as a liquid at 1 atm, which means that solid CO2 does not melt when heated, but instead sublimes. Thus, CO2 does not have a normal melting point; instead, it has a normal sublimation point, –78.5 °C. Because CO2 sublimes rather than melts as it absorbs energy at ordinary pressures, this makes solid CO2 (dry ice) a convenient coolant.
FIGURE 11.29. The melting curve (blue line) behaves typically, slanting to the right with increasing pressure, telling us that the melting point of CO2 increases with increasing pressure. Because the pressure at the triple point is relatively high, 5.11 atm, CO2 does not exist as a liquid at 1 atm, which means that solid CO2 does not melt when heated, but instead sublimes. Thus, CO2 does not have a normal melting point; instead, it has a normal sublimation point, –78.5 °C. Because CO2 sublimes rather than melts as it absorbs energy at ordinary pressures, this makes solid CO2 (dry ice) a convenient coolant.
SAMPLE EXERCISE 11.5 Interpreting a Phase Diagram
Use the phase diagram for methane, CH4, shown in ![]() FIGURE 11.30 to answer the following questions. (a) What are the approximate temperature and pressure of the critical point? (b) What are the approximate temperature and pressure of the triple point? (c) Is methane a solid, liquid, or gas at 1 atm and 0 °C? (d) If solid methane at 1 atm is heated while the pressure is held constant, will it melt or sublime? (e) If methane at 1 atm and 0 °C is compressed until a phase change occurs, in which state is the methane when the compression is complete?
FIGURE 11.30 to answer the following questions. (a) What are the approximate temperature and pressure of the critical point? (b) What are the approximate temperature and pressure of the triple point? (c) Is methane a solid, liquid, or gas at 1 atm and 0 °C? (d) If solid methane at 1 atm is heated while the pressure is held constant, will it melt or sublime? (e) If methane at 1 atm and 0 °C is compressed until a phase change occurs, in which state is the methane when the compression is complete?
SOLUTION
Analyze We are asked to identify key features of the phase diagram and to use it to deduce what phase changes occur when specific pressure and temperature changes take place.
Plan We must identify the triple and critical points on the diagram and also identify which phase exists at specific temperatures and pressures.
Solve
(a) The critical point is the point where the liquid, gaseous, and supercritical fluid phases coexist. It is marked point 3 in the phase diagram and located at approximately –80 °C and 50 atm.
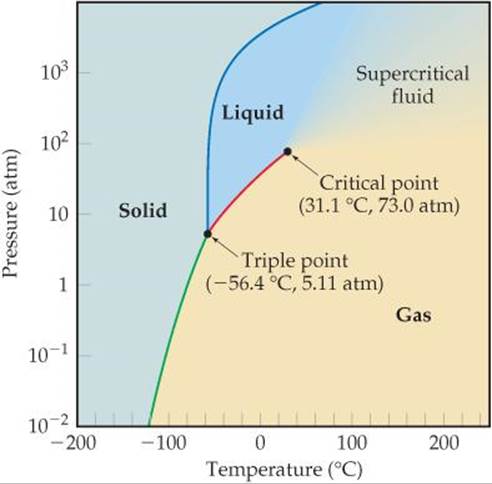
![]() FIGURE 11.29 Phase diagram of CO2. Note that a linear scale is used to represent temperature and a logarithmic scale to represent pressure.
FIGURE 11.29 Phase diagram of CO2. Note that a linear scale is used to represent temperature and a logarithmic scale to represent pressure.
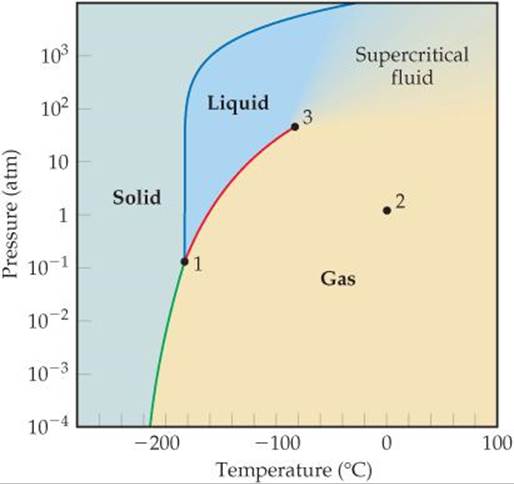
![]() FIGURE 11.30 Phase diagram of CH4. Note that a linear scale is used to represent temperature and a logarithmic scale to represent pressure.
FIGURE 11.30 Phase diagram of CH4. Note that a linear scale is used to represent temperature and a logarithmic scale to represent pressure.
(b) The triple point is the point where the solid, liquid, and gaseous phases coexist. It is marked point 1 in the phase diagram and located at approximately –180 °C and 0.1 atm.
(c) The intersection of 0 °C and 1 atm is marked point 2 in the phase diagram. It is well within the gaseous region of the phase diagram.
(d) If we start in the solid region at P = 1 atm and move horizontally (this means we hold the pressure constant), we cross first into the liquid region, at T ≈ –180 °C, and then into the gaseous region, at T ≈ –160 °C. Therefore, solid methane melts when the pressure is 1 atm. (In order for methane to sublime, the pressure must be below the triple point pressure.)
(e) Moving vertically up from point 2, which is 1 atm and 0 °C, the first phase change we come to is from gas to supercritical fluid. This phase change happens when we exceed the critical pressure (~50 atm).
Check The pressure and temperature at the critical point are higher than those at the triple point, which is expected. Methane is the principal component of natural gas. So it seems reasonable that it exists as a gas at 1 atm and 0 °C.
PRACTICE EXERCISE
Use the phase diagram of methane to answer the following questions. (a) What is the normal boiling point of methane? (b) Over what pressure range does solid methane sublime? (c) Liquid methane does not exist above what temperature?
Answers: (a) –162 °C; (b) It sublimes whenever the pressure is less than 0.1 atm; (c) The highest temperature at which a liquid can exist is defined by the critical temperature. So we do not expect to find liquid methane when the temperature is higher than –80 °C.