CHEMISTRY THE CENTRAL SCIENCE
11 LIQUIDS AND INTERMOLECULAR FORCES
11.7 LIQUID CRYSTALS
In 1888 Frederick Reinitzer, an Austrian botanist, discovered that the organic compound cholesteryl benzoate has an interesting and unusual property, shown in ![]() FIGURE 11.31. Solid cholesteryl benzoate melts at 145 °C, forming a viscous milky liquid; then at 179 °C the milky liquid becomes clear and remains that way at temperatures above 179 °C. When cooled, the clear liquid turns viscous and milky at 179 °C, and the milky liquid solidifies at 145 °C. Reinitzer's work represents the first systematic report of what we call a liquid crystal, the term we use today for the viscous, milky state.
FIGURE 11.31. Solid cholesteryl benzoate melts at 145 °C, forming a viscous milky liquid; then at 179 °C the milky liquid becomes clear and remains that way at temperatures above 179 °C. When cooled, the clear liquid turns viscous and milky at 179 °C, and the milky liquid solidifies at 145 °C. Reinitzer's work represents the first systematic report of what we call a liquid crystal, the term we use today for the viscous, milky state.
Instead of passing directly from the solid phase to the liquid phase when heated, some substances, such as cholesteryl benzoate, pass through an intermediate liquid crystalline phase that has some of the structure of solids and some of the freedom of motion of liquids. Because of the partial ordering, liquid crystals may be viscous and possess properties intermediate between those of solids and those of liquids. The region in which they exhibit these properties is marked by sharp transition temperatures, as in Reinitzer's sample.
Today liquid crystals are used as pressure and temperature sensors and as the display element in such devices as digital watches and laptop computers. They can be used for these applications because the weak intermolecular forces that hold the molecules together in the liquid crystalline phase are easily affected by changes in temperature, pressure, and electric fields.

![]() FIGURE 11.31 Cholesteryl benzoate in its liquid and liquid crystalline states.
FIGURE 11.31 Cholesteryl benzoate in its liquid and liquid crystalline states.
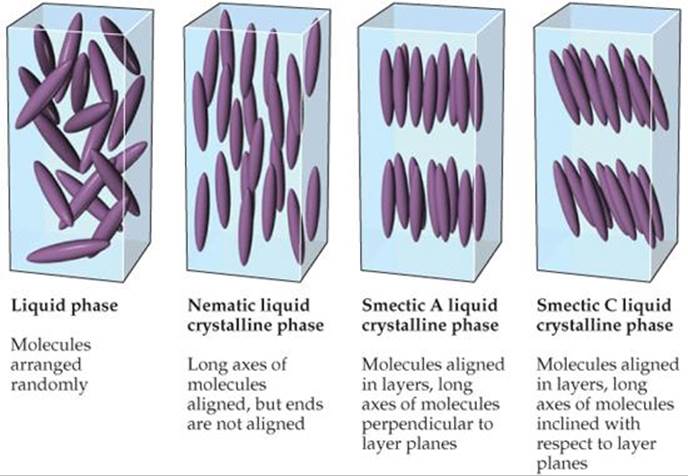
![]() FIGURE 11.32 Molecular order in nematic and smectic liquid crystals. In the liquid phase of any substance, the molecules are arranged randomly, whereas in the liquid crystalline phases the molecules are arranged in a partially ordered way.
FIGURE 11.32 Molecular order in nematic and smectic liquid crystals. In the liquid phase of any substance, the molecules are arranged randomly, whereas in the liquid crystalline phases the molecules are arranged in a partially ordered way.
Types of Liquid Crystals
Substances that form liquid crystals are often composed of rod-shaped molecules that are somewhat rigid in the middle. In the liquid phase, these molecules are oriented randomly. In the liquid crystalline phase, by contrast, the molecules are arranged in specific patterns as illustrated in ![]() FIGURE 11.32. Depending on the nature of the ordering, liquid crystals are classified as nematic, smectic A, smectic C, or cholesteric.
FIGURE 11.32. Depending on the nature of the ordering, liquid crystals are classified as nematic, smectic A, smectic C, or cholesteric.
In a nematic liquid crystal, the molecules are aligned so that their long axes tend to point in the same direction but the ends are not aligned with one another. In smectic A and smectic C liquid crystals, the molecules maintain the long-axis alignment seen in nematic crystals, but in addition they pack into layers.
Two molecules that exhibit liquid crystalline phases are shown in ![]() FIGURE 11.33. The lengths of these molecules are much greater than their widths. The double bonds, including those in the benzene rings, add rigidity to the molecules, and the rings, because they are flat, help the molecules stack with one another. The polar CH3O and COOH groups give rise to dipole–dipole interactions and promote alignment of the molecules. Thus, the molecules order themselves quite naturally along their long axes. They can, however, rotate around their axes and slide parallel to one another. In smectic liquid crystals, the intermolecular forces (dispersion forces, dipole-dipole attractions, and hydrogen bonding) limit the ability of the molecules to slide past one another.
FIGURE 11.33. The lengths of these molecules are much greater than their widths. The double bonds, including those in the benzene rings, add rigidity to the molecules, and the rings, because they are flat, help the molecules stack with one another. The polar CH3O and COOH groups give rise to dipole–dipole interactions and promote alignment of the molecules. Thus, the molecules order themselves quite naturally along their long axes. They can, however, rotate around their axes and slide parallel to one another. In smectic liquid crystals, the intermolecular forces (dispersion forces, dipole-dipole attractions, and hydrogen bonding) limit the ability of the molecules to slide past one another.
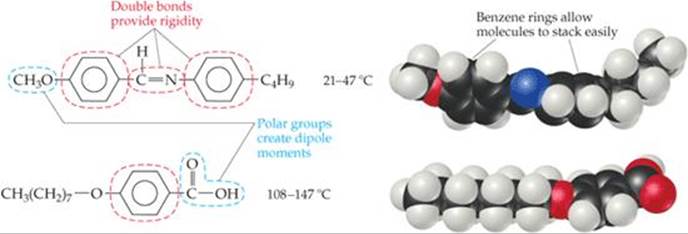
![]() FIGURE 11.33 Molecular structure and liquid crystal temperature range for two typical liquid crystalline materials.
FIGURE 11.33 Molecular structure and liquid crystal temperature range for two typical liquid crystalline materials.
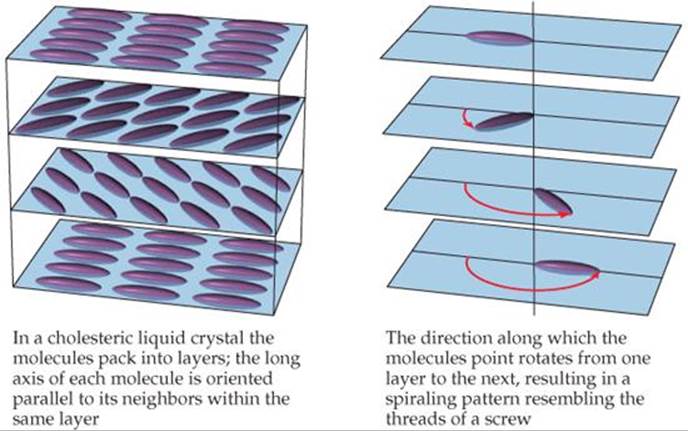
![]() FIGURE 11.34 Molecular order in a cholesteric liquid crystal.
FIGURE 11.34 Molecular order in a cholesteric liquid crystal.
In a cholesteric liquid crystal, the molecules are arranged in layers, with their long axes parallel to the other molecules within the same layer.* Upon moving from one layer to the next, the orientation of the molecules rotates, resulting in the spiral pattern shown in ![]() FIGURE 11.34. These liquid crystals are so named because many derivatives of cholesterol adopt this structure.
FIGURE 11.34. These liquid crystals are so named because many derivatives of cholesterol adopt this structure.
The molecular arrangement in cholesteric liquid crystals produces unusual coloring patterns with visible light. Changes in temperature and pressure change the order and, hence, the color. Cholesteric liquid crystals are used to monitor temperature changes in situations where conventional methods are not feasible. For example, they can detect hot spots in microelectronic circuits, which may signal the presence of flaws. They can also be fashioned into thermometers for measuring the skin temperature of infants. Because cholesteric liquid crystal displays can be built that draw very little power, they are also being investigated for use in electronic paper (![]() FIGURE 11.35).
FIGURE 11.35).

![]() FIGURE 11.35 Electronic paper (e-paper) based on cholesteric liquid crystal technology.
FIGURE 11.35 Electronic paper (e-paper) based on cholesteric liquid crystal technology.
SAMPLE EXERCISE 11.6 Properties of Liquid Crystals
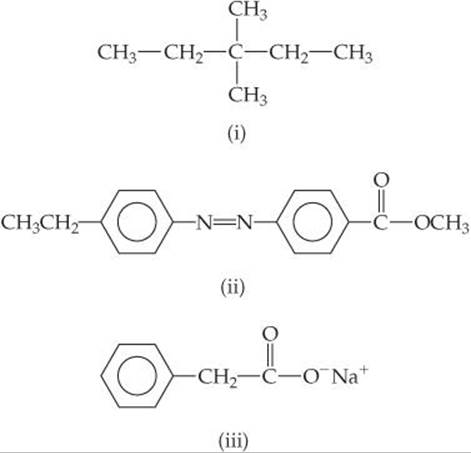
Which of these substances is most likely to exhibit liquid crystalline behavior?
SOLUTION
Analyze We have three molecules with different structures, and we are asked to determine which one is most likely to be a liquid crystalline substance.
Plan We need to identify all structural features that might induce liquid crystalline behavior.
Solve Molecule (i) is not likely to be liquid crystalline because the absence of double and/or triple bonds make this molecule flexible rather than rigid. Molecule (iii) is ionic and the generally high melting points of ionic materials make it unlikely that this substance is liquid crystalline. Molecule (ii) possesses the characteristic long axis and the kinds of structural features often seen in liquid crystals: The molecule has a rodlike shape, the double bonds and benzene rings provide rigidity, and the polar COOCH3 group creates a dipole moment.
PRACTICE EXERCISE
Suggest a reason why decane
CH3CH2CH2CH2CH2CH2CH2CH2CH2CH3
does not exhibit liquid crystalline behavior.
Answer: Because rotation can occur about carbon–carbon single bonds, molecules whose backbone consists predominantly of C—C single bonds are too flexible; the molecules tend to coil in random ways and, thus, are not rodlike.
 CHEMISTRY PUT TO WORK
CHEMISTRY PUT TO WORK
Liquid Crystal Displays
Liquid crystals displays (LCDs) are widely used in electronic devices such as watches, calculators, and computer screens. These applications are possible because an applied electrical field changes the orientation of liquid crystal molecules and thus affects the optical properties of the device.
LCDs come in a variety of designs, but the structure illustrated in ![]() FIGURE 11.36 is typical. A thin layer (5–20 μm) of liquid crystalline material is placed between electrically conducting, transparent glass electrodes. Ordinary light passes through a vertical polarizer that permits light in only the vertical plane to pass. Using a special process during fabrication, the liquid crystal molecules are oriented so that the molecules at the front electrode are oriented vertically and those at the back electrode horizontally. The orientation of the molecules in between the two electrodes varies systematically from vertical to horizontal, as shown in Figure 11.36(a). The plane of polarization of the light is turned by 90° as it passes through the liquid crystal layer and is thus in the correct orientation to pass through the horizontal polarizer. In a watch display, a mirror reflects the light back, and the light retraces its path, allowing the device to look bright. When a voltage is applied to the plates, the liquid crystalline molecules align with the voltage, as shown in Figure 11.36(b). The light rays thus are not properly oriented to pass through the horizontal polarizer, and the device appears dark. Displays of this kind are called “twisted nematic.” As the name implies, materials that order as nematic liquid crystals are used for this application.
FIGURE 11.36 is typical. A thin layer (5–20 μm) of liquid crystalline material is placed between electrically conducting, transparent glass electrodes. Ordinary light passes through a vertical polarizer that permits light in only the vertical plane to pass. Using a special process during fabrication, the liquid crystal molecules are oriented so that the molecules at the front electrode are oriented vertically and those at the back electrode horizontally. The orientation of the molecules in between the two electrodes varies systematically from vertical to horizontal, as shown in Figure 11.36(a). The plane of polarization of the light is turned by 90° as it passes through the liquid crystal layer and is thus in the correct orientation to pass through the horizontal polarizer. In a watch display, a mirror reflects the light back, and the light retraces its path, allowing the device to look bright. When a voltage is applied to the plates, the liquid crystalline molecules align with the voltage, as shown in Figure 11.36(b). The light rays thus are not properly oriented to pass through the horizontal polarizer, and the device appears dark. Displays of this kind are called “twisted nematic.” As the name implies, materials that order as nematic liquid crystals are used for this application.
Liquid crystal displays for computer and televisions employ a light source in place of the reflector, but the principle is the same. The screen is divided into a large number of tiny cells, with the voltages at points on the screen surface controlled by transistors made from thin films of amorphous silicon. Red-green-blue color filters are employed to provide full color. The entire display is refreshed at a frequency of about 60 Hz, so the display can change rapidly with respect to the response time of the human eye. Displays of this kind are remarkable technical achievements based on a combination of basic scientific discovery and creative engineering.
RELATED EXERCISES: 11.68, 11.86
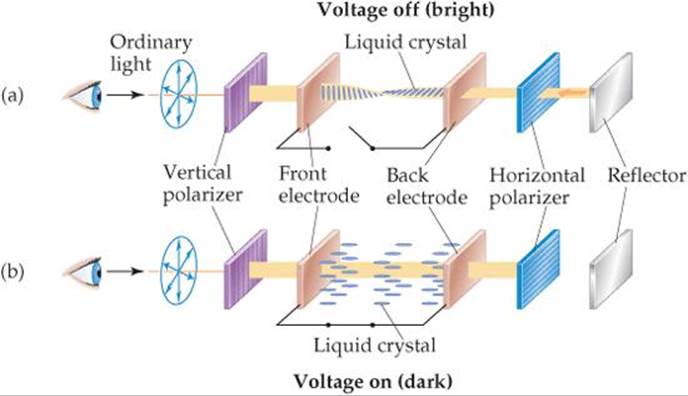
![]() FIGURE 11.36 Schematic illustration of the operation of a twisted nematic liquid crystal display (LCD). (a) When the voltage is off, the molecules in the liquid crystal are aligned so that they rotate the polarization of the light by 90°. This alignment allows light to pass through both the vertical and horizontal polarizers before being reflected and retracing its path to give a bright display. (b) When a voltage is applied to the electrodes, the liquid crystal molecules align parallel to the light path. In this state the light retains the vertical polarization and cannot pass through the horizontal polarizer. The area covered by the front electrode therefore appears dark.
FIGURE 11.36 Schematic illustration of the operation of a twisted nematic liquid crystal display (LCD). (a) When the voltage is off, the molecules in the liquid crystal are aligned so that they rotate the polarization of the light by 90°. This alignment allows light to pass through both the vertical and horizontal polarizers before being reflected and retracing its path to give a bright display. (b) When a voltage is applied to the electrodes, the liquid crystal molecules align parallel to the light path. In this state the light retains the vertical polarization and cannot pass through the horizontal polarizer. The area covered by the front electrode therefore appears dark.
SAMPLE INTEGRATIVE EXERCISE Putting Concepts Together
The substance CS2 has a melting point of –110.8 °C and a boiling point of 46.3 °C. Its density at 20 °C is 1.26 g/cm3. It is highly flammable. (a) What is the name of this compound? (b) List the intermolecular forces that CS2 molecules exert on one other. (c) Write a balanced equation for the combustion of this compound in air. (You will have to decide on the most likely oxidation products.) (d) The critical temperature and pressure for CS2 are 552 K and 78 atm, respectively. Compare these values with those for CO2 (Table 11.5) and discuss the possible origins of the differences. (e) Would you expect the density of CS2 at 40 °C to be greater or less than at 20 °C? What accounts for the difference?
SOLUTION
(a) The compound is named carbon disulfide, in analogy with the naming of other binary molecular compounds such as carbon dioxide. ![]() (Section 2.8)
(Section 2.8)
(b) Only dispersion forces affect CS2; it does not have a dipole moment, based upon its molecular shape, and obviously cannot undergo hydrogen bonding.
(c) The most likely products of the combustion will be CO2 and SO2. ![]() (Section 3.2) Under some conditions SO3 might be formed, but this would be the less likely outcome. Thus, we have the following equation for combustion:
(Section 3.2) Under some conditions SO3 might be formed, but this would be the less likely outcome. Thus, we have the following equation for combustion:
CS2(l) + 3 O2(g) → CO2(g) + 2 SO2(g)
(d) The critical temperature and pressure of CS2 (552 K and 78 atm) are both higher than those given for CO2 in Table 11.5 (304 K and 73 atm). The difference in critical temperatures is especially notable. The higher values for CS2 arise from the greater dispersion attractions between the CS2 molecules compared with CO2. These greater attractions are due to the larger size of the sulfur compared to oxygen and, therefore, its greater polarizability.
(e) The density would be lower at the higher temperature. Density decreases with increasing temperature because the molecules possess higher kinetic energies. Their more energetic movements result in larger average distances between molecules, which translate into lower densities.