CHEMISTRY THE CENTRAL SCIENCE
12 SOLIDS AND MODERN MATERIALS
EXERCISES
VISUALIZING CONCEPTS
12.1 For each of the two-dimensional structures shown here (a) draw the unit cell, (b) determine the type of two-dimensional lattice (from Figure 12.4), and (c) determine how many of each type of circle (white or black) there are per unit cell. [Section 12.2]

12.2 (a) What kind of packing arrangement is seen in the accompanying photo? (b) What is the coordination number of each cannonball in the interior of the stack? [Section 12.3]

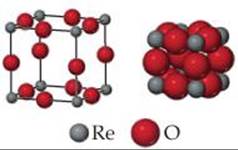
12.3 Rhenium oxide crystallizes with a structure that has a primitive cubic lattice, as shown here. In the image on the left, the sizes of the ions have been reduced to show the entire unit cell. (a) How many atoms of each type are there per unit cell? (b) Use the ionic radii of rhenium (0.70 Å) and oxygen (1.26 Å) to estimate the length of the edge of the unit cell. (c) Use your answers to parts (a) and (b) to estimate the density of this compound. [Section 12.5].
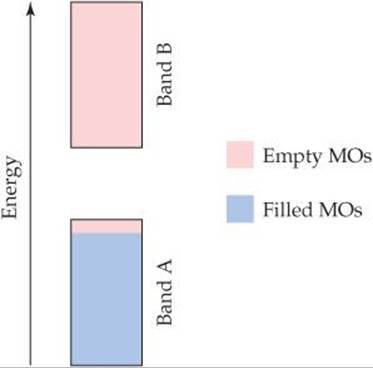
12.4 The electronic structure of a doped semiconductor is shown here. (a) Which band, A or B, is the valence band? (b) Which band is the conduction band? (c) Which band consists of bonding molecular orbitals? (d) Is this an example of an n-type or p-type doped semiconductor? (e) If the semiconductor is germanium, which of the following elements could be the dopant: Ga, Si, or P? [Section 12.7]
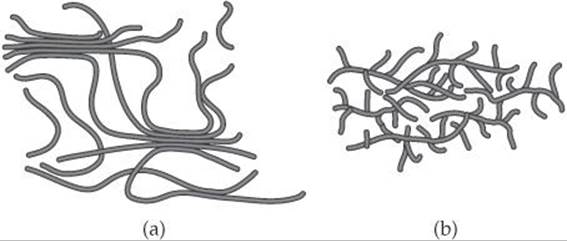
12.5 Shown here are cartoons of two different polymers. Based on these cartoons, which polymer would you expect to be denser? Which one would have the higher melting point? [Section 12.8]
12.6 The accompanying image shows photoluminescence from four different samples of CdTe nanocrystals, each embedded in a polymer matrix. The photoluminescence occurs because the samples are being irradiated by a UV light source. The nanocrystals in each vial have different average sizes. The sizes are 4.0, 3.5, 3.2, and 2.8 nm. (a) Which vial contains the 4.0-nm nanocrystals? (b) Which vial contains the 2.8-nm nanocrystals? (c) Crystals of CdTe that have sizes that are larger than approximately 100 nm have a band gap of 1.5 eV. What would be the wavelength and frequency of light emitted from these crystals? What type of light is this? [Sections 12.7 and 12.9]

CLASSIFICATIONS OF SOLIDS (section 12.1)
12.7 Covalent bonding occurs in both molecular and covalent-network solids. Why do these two kinds of solids differ so greatly in their hardness and melting points?
12.8 Silicon is the fundamental component of integrated circuits. Si has the same structure as diamond. Is Si a molecular, metallic, ionic, or covalent-network solid?
______
12.9 What kinds of attractive forces exist between particles in (a) molecular crystals, (b) covalent-network crystals, (c) ionic crystals, (d) metallic crystals?
12.10 Which type (or types) of crystalline solid is characterized by each of the following: (a) high mobility of electrons throughout the solid; (b) softness, relatively low melting point; (c) high melting point and poor electrical conductivity; (d) network of covalent bonds?
______
12.11 Indicate the type of crystal (molecular, metallic, ionic, or covalent-network) each of the following would form upon solidification: (a) CaCO3, (b) Pt, (c) ZrO2 (melting point, 2677 °C), (d) table sugar (C12H22O11), (e) benzene (C6H6), (f) I2.
12.12 Indicate the type of crystal (molecular, metallic, ionic, or cova-lent-network) each of the following would form upon solidification: (a) InAs, (b) MgO, (c) HgS, (d) In, (e) HBr.
______
12.13 A white substance melts with some decomposition at 730 °C. As a solid, it does not conduct electricity, but it dissolves in water to form a conducting solution. Which type of solid (molecular, metallic, covalent-network, or ionic) might the substance be?
12.14 You are given a white substance that sublimes at 3000 °C; the solid is a nonconductor of electricity and is insoluble in water. Which type of solid (molecular, metallic, covalent-network, or ionic) might this substance be?
STRUCTURES OF SOLIDS (section 12.2)
12.15 (a) Draw a picture that represents a crystalline solid at the atomic level. (b) Now draw a picture that represents an amorphous solid at the atomic level.
12.16 Amorphous silica, SiO2, has a density of about 2.2 g/cm3, whereas the density of crystalline quartz is 2.65 g/cm3. Account for this difference in densities.
______
12.17 Two patterns of packing different types of spheres are shown here. For each structure (a) draw the two-dimensional unit cell, (b) determine the angle between the lattice vectors, g, and whether the lattice vectors are the same length or of different lengths, (c) determine the type of two-dimensional lattice (from Figure 12.4).
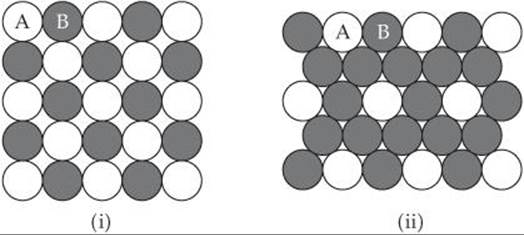
12.18 Two patterns of packing different types of spheres are shown here. For each structure (a) draw the two-dimensional unit cell, (b) determine the angle between the lattice vectors, γ, and whether the lattice vectors are the same length or of different lengths, (c) determine the type of two-dimensional lattice (from Figure 12.4).
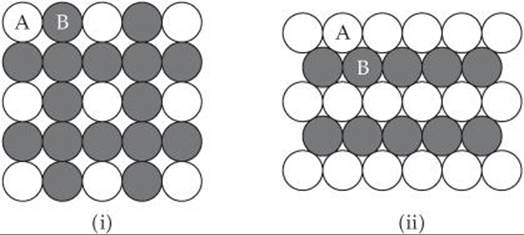
______
12.19 Of the seven three-dimensional primitive lattices, (a) which one has a unit cell where the a and b lattice vectors form a rectangular base, while the c lattice vector is perpendicular to the other two and of a different length? (b) What is the lattice if the a and b lattice vectors form a square base and the c lattice vector is perpendicular to the other two and of a different length?
12.20 Of the seven three-dimensional primitive lattices, (a) which one has a unit cell where the a and b lattice vectors form a base that is an arbitrary parallelogram (like the unit cell of a two-dimensional oblique lattice), while the c lattice vector is perpendicular to the other two? (b) What is the lattice if the a and b lattice vectors form a base that corresponds to the two-dimensional hexagonal unit cell and the c lattice vector is perpendicular to the other two?
______
12.21 Of the seven three-dimensional primitive lattices, which ones have a unit cell where no two lattice vectors are perpendicular to each other?
12.22 Of the seven three-dimensional primitive lattices, which ones have a unit cell where all three lattice vectors are of the same length?
______
12.23 What is the minimum number of atoms that could be contained in the unit cell of an element with a body-centered cubic lattice?
12.24 What is the minimum number of atoms that could be contained in the unit cell of an element with a face-centered cubic lattice?
______
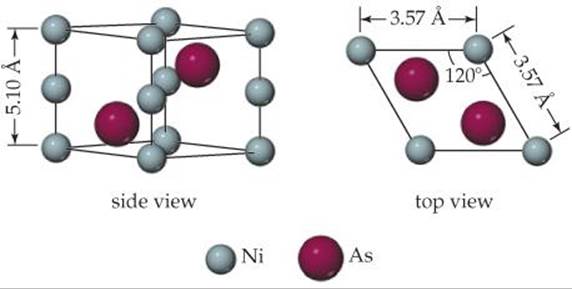
12.25 The unit cell of nickel arsenide is shown here. (a) What type of lattice does this crystal possess? (b) What is the empirical formula?
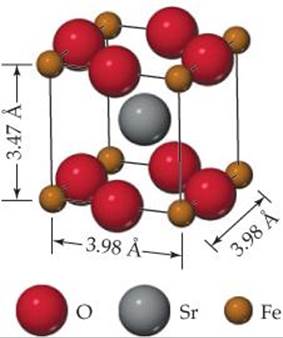
12.26 The unit cell of a compound containing strontium, iron, and oxygen is shown here. (a) What type of lattice does this crystal possess (all three lattice vectors are mutually perpendicular)? (b) What is the empirical formula?
METALLIC SOLIDS (section 12.3)
12.27 The densities of the elements K, Ca, Sc, and Ti are 0.86, 1.5, 3.2, and 4.5g/cm3, respectively. What factors are likely to be of major importance in determining this variation? Which factor do you think will be the most important?
12.28 Which of the following substances would you expect to possess metallic properties: (a) TiCl4, (b) NiCo alloy, (c) W, (d) Ge, (e) ScN?
______
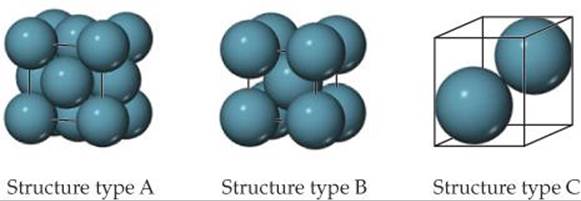
12.29 Consider the unit cells shown here for three different structures that are commonly observed for metallic elements. (a) Which structure(s) corresponds to the densest packing of atoms? (b) Which structure(s) corresponds to the least dense packing of atoms?
12.30 Sodium metal (atomic weight 22.99g/cm3) adopts a body-centered cubic structure with a density of 0.97g/cm3. (a) Use this information and Avogadro's number (NA = 6.022 × 1023) to estimate the atomic radius of sodium. (b) If it didn't react so vigorously, sodium could float on water. Use the answer from part (a) to estimate the density of Na if its structure were that of a cubic close-packed metal. Would it still float on water?
______
12.31 Iridium crystallizes in a face-centered cubic unit cell that has an edge length of 3.833 Å. (a) Calculate the atomic radius of an iridium atom. (b) Calculate the density of iridium metal.
12.32 Calcium crystallizes with a body-centered cubic structure. (a) How many Ca atoms are contained in each unit cell? (b) How many nearest neighbors does each Ca atom possess? (c) Estimate the length of the unit cell edge, a, from the atomic radius of calcium (1.97 Å). (d) Estimate the density of Ca metal.
______
12.33 Aluminum metal crystallizes in a cubic close-packed structure [face-centered cubic cell, Figure 12.14(a)]. (a) How many aluminum atoms are in a unit cell? (b) What is the coordination number of each aluminum atom? (c) Estimate the length of the unit cell edge, a, from the atomic radius of aluminum (1.43 Å). (d) Calculate the density of aluminum metal.
12.34 An element crystallizes in a body-centered cubic lattice. The edge of the unit cell is 2.86 Å, and the density of the crystal is 7.92 g/cm3. Calculate the atomic weight of the element.
______
12.35 Define the term alloy. Distinguish among solid solution alloys, heterogeneous alloys, and intermetallic compounds.
12.36 Distinguish between substitutional and interstitial alloys. What conditions favor formation of substitutional alloys?
______
12.37 For each of the following alloy compositions indicate whether you would expect it to be a substitutional alloy, an interstitial alloy, or an intermetallic compound: (a) Fe0.97Si0.03, (b) Fe0.60Ni0.40, (c) SmCo5.
12.38 For each of the following alloy compositions indicate whether you would expect it to be a substitutional alloy, an interstitial alloy, or an intermetallic compound:
(a) Cu0.66Zn0.34, (b) Ag3Sn, (c) Ti0.99O0.01.
______
12.39 Classify each of the following statements as true or false:
(a) Substitutional alloys tend to be more ductile than interstitial alloys.
(b) Interstitial alloys tend to form between elements with similar ionic radii.
(c) Nonmetallic elements are never found in alloys.
12.40 Classify each of the following statements as true or false:
(a) Intermetallic compounds have a fixed composition.
(b) Copper is the majority component in both brass and bronze.
(c) In stainless steel the chromium atoms occupy interstitial positions.
______
12.41 Which element or elements are alloyed with gold to make the following types of “colored gold” used in the jewelry industry? For each type indicate what type of alloy is formed: (a) white gold, (b) rose gold, (c) blue gold, (d) green gold.
12.42 What is the chemical composition of purple gold? Is the composition variable? Why don't jewelers use purple gold to make rings or necklaces?
METALLIC BONDING (section 12.4)
12.43 Explain how the electron-sea model accounts for the high electrical and thermal conductivity of metals.
12.44 (a) Compare the electronic structures of atomic chromium and atomic selenium. In what respects are they similar, and in what respects do they differ? (b) Chromium is a metal, and selenium is a nonmetal. What factors are important in determining this difference in properties?
______
12.45 The molecular-orbital diagrams for two- and four-atom linear chains of lithium atoms are shown in Figure 12.23. Construct a molecular-orbital diagram for a chain containing six lithium atoms and use it to answer the following questions. (a) How many molecular orbitals are there in the diagram? (b) How many nodes are in the lowest-energy molecular orbital? (c) How many nodes are in the highest-energy molecular orbital? (d) How many nodes are in the highest-energy occupied molecular orbital (HOMO)? (e) How many nodes are in the lowest-energy unoccupied molecular orbital (LUMO)?
12.46 Repeat Exercise 12.45 for a linear chain of eight lithium atoms.
______
12.47 Which would you expect to be the more ductile element, (a) Ag or Mo, (b) Zn or Si? In each case explain your reasoning.
12.48 How do you account for the observation that the alkali metals, like sodium and potassium, are soft enough to be cut with a knife?
______
12.49 Explain this trend in melting points: Y 1522 °C, Zr 1852 °C, Nb 2468 °C, Mo 2617 °C.
12.50 For each of the following groups which metal would you expect to have the highest melting point; (a) gold (Au), rhenium (Re), or cesium (Cs); (b) rubidium (Rb), molybdenum (Mo), or indium (In); (c) ruthenium (Ru), strontium (Sr), or cadmium (Cd)?
IONIC AND MOLECULAR SOLIDS (section 12.5 and 12.6)
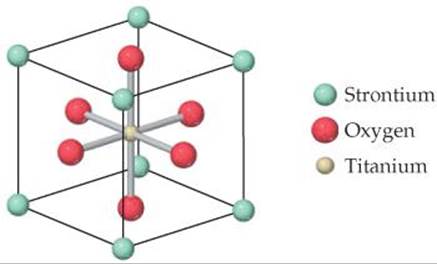
12.51 Tausonite, a mineral composed of Sr, O, and Ti, has the cubic unit cell shown in the drawing. (a) What is the empirical formula of this mineral? (b) It is easy to see that Ti is coordinated by six oxygen atoms because the Ti atom is located at the center of the unit cell. To see the full coordination environment of the other ions, we have to consider neighboring unit cells. How many oxygens are coordinated to strontium?
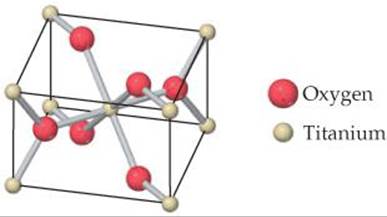
12.52 Rutile is a mineral composed of Ti and O. Its unit cell, shown in the drawing, contains Ti atoms at each corner and a Ti atom at the center of the cell. Four O atoms are on the opposite faces of the cell, and two are entirely within the cell. (a) What is the chemical formula of this mineral? (b) What is the coordination number of each atom?
______
12.53 NaF has the same structure as NaCl. (a) Use ionic radii from Chapter 7 to estimate the length of the unit cell edge for NaF. (b) Use the unit cell size calculated in part (a) to estimate the density of NaF.
12.54 Clausthalite is a mineral composed of lead selenide (PbSe). The mineral adopts a NaCl-type structure. The density of PbSe at 25 °C is 8.27 g/cm3. Calculate the length of an edge of the PbSe unit cell.
______
12.55 A particular form of cinnabar (HgS) adopts the zinc blende structure, Figure 12.26. The length of the unit cell edge is 5.852 Å. (a) Calculate the density of HgS in this form. (b) The mineral tiemmanite (HgSe) also forms a solid phase with the zinc blende structure. The length of the unit cell edge in this mineral is 6.085 Å. What accounts for the larger unit cell length in tiemmanite? (c) Which of the two substances has the higher density? How do you account for the difference in densities?
12.56 At room temperature and pressure RbI crystallizes with the NaCl-type structure. (a) Use ionic radii to predict the length of the cubic unit cell edge. (b) Use this value to estimate the density. (c) At high pressure the structure transforms to one with a CsCl-type structure. (c) Use ionic radii to predict the length of the cubic unit cell edge for the high-pressure form of RbI. (d) Use this value to estimate the density. How does this density compare with the density you calculated in part (b)?
______
12.57 CuI, CsI, and NaI each adopt a different type of structure. The three different structures are those shown in Figure 12.26. (a) Use ionic radii, Cs+(r = 1.81 Å), Na+(r = 1.16 Å), Cu+(r = 0.74 Å), and I-(r = 2.06 Å), to predict which compound will crystallize with which structure. (b) What is the coordination number of iodide in each of these structures?
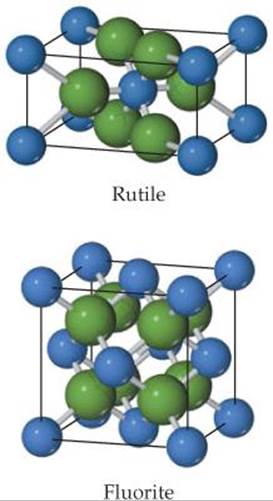
12.58 The rutile and fluorite structures, shown here (anions are colored green), are two of the most common structure types for ionic compounds where the cation to anion ratio is 1:2. (a) For CaF2 and ZnF2 use ionic radii, Ca2+(r = 1.14 Å), Zn2+(r = 0.88 Å), F–(r = 1.19 Å), to predict which compound is more likely to crystallize with the fluorite structure and which with the rutile structure. (b) What are the coordination numbers of the cations and anions in each of these structures?
______
12.59 The coordination number for Mg2+ ion is usually six. Assuming this assumption holds, determine the anion coordination number in the following compounds: (a) MgS, (b) MgF2, (c) MgO.
12.60 The coordination number for the Al3+ ion is typically between four and six. Use the anion coordination number to determine the Al3+ coordination number in the following compounds: (a) AlF3 where the fluoride ions are two coordinate, (b) Al2O3 where the oxygen ions are six coordinate, (c) AlN where the nitride ions are four coordinate.
______
12.61 Classify each of the following statements as true or false:
(a) Although both molecular solids and covalent-network solids have covalent bonds, the melting points of molecular solids are much lower because their covalent bonds are much weaker.
(b) Other factors being equal, highly symmetric molecules tend to form solids with higher melting points than asymmetrically shaped molecules.
12.62 Classify each of the following statements as true or false:
(a) For molecular solids the melting point generally increases as the strengths of the covalent bonds increase.
(b) For molecular solids the melting point generally increases as the strengths of the intermolecular forces increase.
COVALENT-NETWORK SOLIDS (section 12.7)
12.63 Both covalent-network solids and ionic solids can have melting points well in excess of room temperature, and both can be poor conductors of electricity in their pure form. However, in other ways their properties are quite different.
(a) Which type of solid is more likely to dissolve in water?
(b) Which type of solid can become an electrical conductor via chemical substitution?
12.64 Which of the following properties are typical characteristics of a covalent-network solid, a metallic solid, or both: (a) ductility, (b) hardness, (c) high melting point?
______
12.65 For each of the following pairs of semiconductors, which one will have the larger band gap: (a) CdS or CdTe, (b) GaN or InP, (c) GaAs or InAs?
12.66 For each of the following pairs of semiconductors, which one will have the larger band gap: (a) InP or InAs, (b) Ge or AlP, (c) AgI or CdTe?
______
12.67 If you want to dope GaAs to make an n-type semiconductor with an element to replace Ga, which element(s) would you pick?
12.68 If you want to dope GaAs to make a p-type semiconductor with an element to replace As, which element(s) would you pick?
______
12.69 Silicon has a band gap of 1.1 eV at room temperature. (a) What wavelength of light would a photon of this energy correspond to? (b) Draw a vertical line at this wavelength in the figure shown, which shows the light output of the sun as a function of wavelength. Does silicon absorb all, none, or a portion of the visible light that comes from the sun?
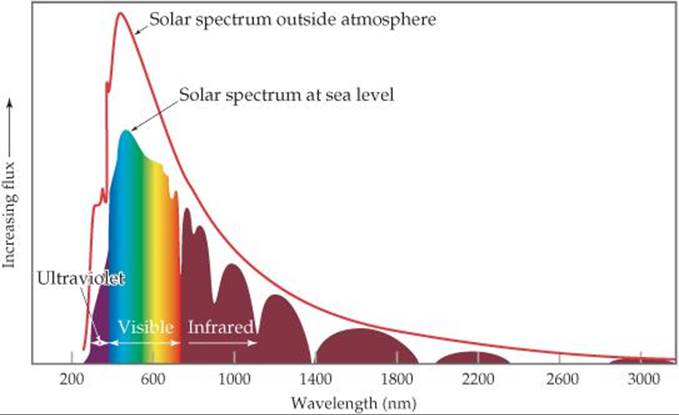
12.70 Cadmium telluride is an important material for solar cells. (a) What is the band gap of CdTe? (b) What wavelength of light would a photon of this energy correspond to? (c) Draw a vertical line at this wavelength in the figure shown with Exercise 12.69, which shows the light output of the sun as a function of wavelength. (d) With respect to silicon, does CdTe absorb a larger or smaller portion of the solar spectrum?
12.71 The semiconductor GaP has a band gap of 2.2 eV. Green LEDs are made from pure GaP. What wavelength of light would be emitted from an LED made from GaP?
12.72 The first LEDs were made from GaAs, which has a band gap of 1.43 eV. What wavelength of light would be emitted from an LED made from GaAs? What region of the electromagnetic spectrum does this light correspond to: UV, visible, or IR?
______
12.73 GaAs and GaP (Eg = 2.26 eV) make solid solutions that have the same crystal structure as the parent materials, with As and P randomly distributed throughout the crystal. GaPxAs1–x exists for any value of x. If we assume that the band gap varies linearly with composition betweenx = 0 and x = 1, estimate the band gap for GaP0.5As0.5. What wavelength of light does this correspond to?
12.74 Red light-emitting diodes are made from GaAs and GaP solid solutions, GaPxAs1-x (see Exercise 12.73). The original red LEDs emitted light with a wavelength of 660 nm. If we assume that the band gap varies linearly with composition between x = 0 and x = 1, estimate the composition (the value of x) that is used in these LEDs.
POLYMERIC SOLIDS (section 12.8)
12.75 What is a monomer? Give three examples of monomers taken from the examples given in this chapter.
12.76 The molecular formula of n-decane is CH3(CH2)8CH3. Decane is not considered a polymer, whereas polyethylene is. What is the distinction?
______
12.77 An ester is a compound formed by a condensation reaction between a carboxylic acid and an alcohol. Read the discussion of esters in Section 24.4 and then give an example of a reaction forming an ester. How might this kind of reaction be extended to form a polymer (a polyester)?
12.78 Write a chemical equation for formation of a polymer via a condensation reaction from the monomers succinic acid (HOOCCH2CH2COOH) and ethylenediamine (H2NCH2CH2NH2).
______
12.79 Draw the structure of the monomer(s) employed to form each of the following polymers shown in Table 12.5: (a) polyvinyl chloride, (b) nylon 6,6, (c) polyethylene terephthalate.
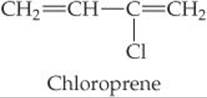
12.80 Write the chemical equation that represents the formation of (a) polychloroprene from chloroprene (polychloroprene is used in highway-pavement seals, expansion joints, conveyor belts, and wire and cable jackets);
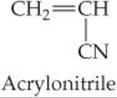
(b) polyacrylonitrile from acrylonitrile (polyacrylonitrile is used in home furnishings, craft yarns, clothing, and many other items).
12.81 The nylon Nomex®, a condensation polymer, has the following structure:
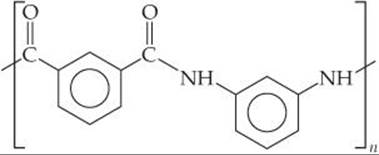
Draw the structures of the two monomers that yield Nomex®.
12.82 Proteins are polymers formed by condensation reactions of amino acids, which have the general structure
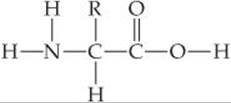
In this structure, R represents—H,—CH3, or another group of atoms. Draw the general structure for a polyamino acid polymer formed by condensation polymerization of the molecule shown here.
______
12.83 What molecular features make a polymer flexible? Explain how cross-linking affects the chemical and physical properties of the polymer.
12.84 What molecular structural features cause high-density polyethylene to be denser than low-density polyethylene?
______
12.85 Are high molecular weights and a high degree of crystallinity always desirable properties of a polymer? Explain.
12.86 Briefly describe each of the following: (a) elastomer, (b) thermoplastic, (c) thermosetting plastic.
NANOMATERIALS (section 12.9)
12.87 Explain why “bands” may not be the most accurate description of bonding in a solid when the solid has nanoscale dimensions.
12.88 CdS has a band gap of 2.4 eV. If large crystals of CdS are illuminated with ultraviolet light, they emit light equal to the band gap energy. (a) What color is the emitted light? (b) Would appropriately sized CdS quantum dots be able to emit blue light? (c) What about red light?
______
12.89 True or false:
(a) The band gap of a semiconductor decreases as the particle size decreases in the 1–10-nm range.
(b) The light that is emitted from a semiconductor, upon external stimulation, becomes longer in wavelength as the particle size of the semiconductor decreases.
12.90 True or false:
If you want a semiconductor that emits blue light, you could either use a material that has a band gap corresponding to the energy of a blue photon or you could use a material that has a smaller band gap but make an appropriately sized nanoparticle of the same material.
______
12.91 Gold adopts a face-centered cubic structure with a unit cell edge of 4.08 Å (Figure 12.11). How many gold atoms are there in a sphere that is 20 nm in diameter? Recall that the volume of a sphere is ![]() .
.
12.92 Cadmium telluride, CdTe, takes the zinc blende structure (Figure 12.26) with a unit cell edge length of 6.49 Å. There are four cadmium atoms and four tellurium atoms per unit cell. How many of each type of atom are there in a cubic crystal with an edge length of 5.00 nm?
ADDITIONAL EXERCISES
[12.93] A face-centered tetragonal lattice is not one of the 14 three-dimensional lattices. Show that a face-centered tetragonal unit cell can be redefined as a body-centered tetragonal lattice with a smaller unit cell.
12.94 Pure iron crystallizes with a body-centered cubic structure, but small amounts of impurities can stabilize a face-centered cubic structure. Which form of iron has a higher density?
[12.95] Introduction of carbon into a metallic lattice generally results in a harder, less ductile substance with lower electrical and thermal conductivities. Explain why this might be so.
12.96 As mentioned in Section 12.3, Ni3Al is used in the turbines of aircraft engines because of its strength and low density. Nickel metal has a cubic close-packed structure with a face-centered cubic unit cell, while Ni3Al has the ordered cubic structure shown in Figure 12.17. The length of the cubic unit cell edge is 3.53 Å for nickel and 3.56 Å for Ni3Al. Use these data to calculate and compare the densities of these two materials.
12.97 For each of the intermetallic compounds shown in Figure 12.17 determine the number of each type of atom in the unit cell. Do your answers correspond to ratios expected from the empirical formulas: Ni3Al, Nb3Sn, and SmCo5?
12.98 What type of lattice—primitive cubic, body-centered cubic, or face-centered cubic—does each of the following structure types possess: (a) CsCl, (b) Au, (c) NaCl, (d) Po, (e) ZnS?
12.99 Tin exists in two allotropic forms: Gray tin has a diamond structure, and white tin has a close-packed structure. One of these allotropic forms is a semiconductor with a small band gap while the other is a metal. Which one is which? Which form would you expect to have the longer Sn — Sn bond distance?
[12.100] The electrical conductivity of titanium is approximately 2500 times greater than that of silicon. Titanium has a hexagonal close-packed structure, and silicon has the diamond structure. Explain how the structures relate to the relative electrical conductivities of the elements.
12.101 Silicon carbide, SiC, has the three-dimensional structure shown in the figure.
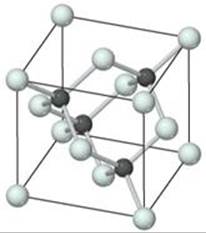
(a) Name another compound that has the same structure.
(b) Would you expect the bonding in SiC to be predominantly ionic, metallic, or covalent? (c) How do the bonding and structure of SiC lead to its high thermal stability (to 2700 °C) and exceptional hardness?
[12.102] Unlike metals, semiconductors increase their conductivity as you heat them (up to a point). Suggest an explanation.
12.103 Teflon® is a polymer formed by the polymerization of F2C═CF2. Draw the structure of a section of this polymer. What type of polymerization reaction is required to form it?
12.104 Hydrogen bonding between polyamide chains plays an important role in determining the properties of a nylon such as nylon 6,6 (Table 12.5). Draw the structural formulas for two adjacent chains of nylon 6,6 and show where hydrogen-bonding interactions could occur between them.
12.105 Explain why X-rays can be used to measure atomic distances in crystals but visible light cannot be used for this purpose.
12.106 In their study of X-ray diffraction, William and Lawrence Bragg determined that the relationship among the wavelength of the radiation (λ), the angle at which the radiation is diffracted (θ), and the distance between planes of atoms in the crystal that cause the diffraction (d) is given by nλ = 2d sin θ. X-rays from a copper X-ray tube that have a wavelength of 1.54 Å are diffracted at an angle of 14.22 degrees by crystalline silicon. Using the Bragg equation, calculate the distance between the planes of atoms responsible for diffraction in this crystal, assuming n = 1 (first-order diffraction).
12.107 Germanium has the same structure as silicon, but the unit cell size is different because Ge and Si atoms are not the same size. If you were to repeat the experiment described in the previous problem but replace the Si crystal with a Ge crystal, would you expect the X-rays to be diffracted at a larger or smaller angle, θ?
[12.108] (a) The density of diamond [Figure 12.30(a)] is 3.5 g/cm3 and that of graphite [Figure 12.30(b)] is 2.3 g/cm3. Based on the structure of buckminsterfullerene (Figure 12.47), what would you expect its density to be relative to these other forms of carbon? (b) X-ray diffraction studies of buckmin-sterfullerene show that it has a face-centered cubic lattice of C60 molecules. The length of an edge of the unit cell is 14.2 Å. Calculate the density of buckminsterfullerene.
12.109 The valence band of a semiconductor is made up of bonding molecular orbitals and the conduction band of antibonding molecular orbitals. (a) Given this fact would you expect n-type doping to strengthen or weaken the bonds in a semiconductor? (b) Would you expect p-type doping to strengthen or weaken the bonds in a semiconductor?
INTEGRATIVE EXERCISES
12.110 The karat scale used to describe gold alloys is based on mass percentages. (a) If an alloy is formed that is 50 mol% silver and 50 mol% gold, what is the karat number of the alloy? Use Figure 12.18 to estimate the color of this alloy. (b) If an alloy is formed that is 50 mol% copper and 50 mol% gold, what is the karat number of the alloy? What is the color of this alloy?
12.111 Spinel is a mineral that contains 37.9% Al, 17.1% Mg, and 45.0% O, by mass, and has a density of 3.57 g/cm3. The unit cell is cubic with an edge length of 8.09 Å. How many atoms of each type are in the unit cell?
12.112 (a) What are the C—C—C bond angles in diamond? (b) What are they in graphite?
[12.113] Employing the bond enthalpy values listed in Table 8.4, estimate the molar enthalpy change occurring upon (a) polymerization of ethylene, (b) formation of nylon 6,6, (c) formation of polyethylene terephthalate (PET).
[12.114] Although polyethylene can twist and turn in random ways, the most stable form is a linear one with the carbon backbone oriented as shown in the following figure:
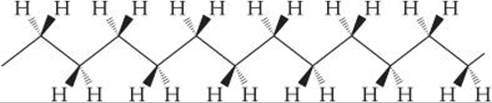
The solid wedges in the figure indicate bonds from carbon that come out of the plane of the page; the dashed wedges indicate bonds that lie behind the plane of the page.
(a) What is the hybridization of orbitals at each carbon atom? What angles do you expect between the bonds?
(b) Now imagine that the polymer is polypropylene rather than polyethylene. Draw structures for polypropylene in which (i) the CH3 groups all lie on the same side of the plane of the paper (this form is called isotactic polypropylene); (ii) the CH3 groups lie on alternating sides of the plane (syndiotac-tic polypropylene); or (iii) the CH3 groups are randomly distributed on either side (atactic polypropylene). Which of these forms would you expect to have the highest crystallinity and melting point, and which the lowest? Explain in terms of intermolecular interactions and molecular shapes.
(c) Polypropylene fibers have been employed in athletic wear. The product is said to be superior to cotton or polyester clothing in wicking moisture away from the body through the fabric to the outside. Explain the difference between polypropylene and polyester or cotton (which has many — OH groups along the molecular chain) in terms of intermolecular interactions with water.
12.115 (a) In polyvinyl chloride shown in Table 12.5, which bonds have the lowest average bond enthalpy? (b) When subjected to high pressure and heated polyvinyl chloride converts to diamond. During this transformation which bonds are most likely to break first? (c) Employing the values of average bond enthalpy in Table 8.4, estimate the overall enthalpy change for converting PVC to diamond.
[12.116] Silicon has the diamond structure (Figure 12.30(a)) with unit cell edge length of 5.43 Å and eight atoms per unit cell. (a) How many silicon atoms are there in 1 cm3 of material? (b) Suppose you dope that 1-cm3 sample of silicon with 1 ppm of phosphorus that will increase the conductivity by a factor of a million. How many milligrams of phosphorus are required?
12.117 KCl has the same structure as NaCl. The length of the unit cell is 6.28 Å. The density of KCl is1.984 g/cm3, and its formula mass is 74.55 amu. Using this information, calculate Avogadro's number.
12.118 Look up the diameter of a silicon atom, in Å. The channel length in a Pentium 4 processor chip is 65 nm long. How many silicon atoms does this correspond to?