CHEMISTRY THE CENTRAL SCIENCE
13 PROPERTIES OF SOLUTIONS
13.4 EXPRESSING SOLUTION CONCENTRATION
The concentration of a solution can be expressed either qualitatively or quantitatively. The terms dilute and concentrated are used to describe a solution qualitatively. A solution with a relatively small concentration of solute is said to be dilute; one with a large concentration is said to be concentrated. Chemists use various ways to express concentration quantitatively, and we examine several of these next.
Mass Percentage, ppm, and ppb
One of the simplest quantitative expressions of concentration is the mass percentage of a component in a solution, given by
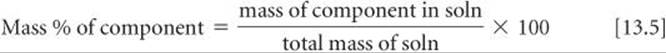
Because percent means “per hundred,” a solution of hydrochloric acid that is 36% HCl by mass contains 36 g of HCl for each 100 g of solution.
We often express the concentration of very dilute solutions in parts per million (ppm) or parts per billion (ppb). These quantities are similar to mass percentage but use 106 (a million) or 109 (a billion), respectively, in place of 100 as a multiplier for the ratio of the mass of solute to the mass of solution. Thus, parts per million is defined as
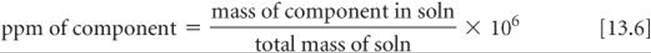
A solution whose solute concentration is 1 ppm contains 1 g of solute for each million (106) grams of solution or, equivalently, 1 mg of solute per kilogram of solution. Because the density of water is 1 g/mL, 1 kg of a dilute aqueous solution has a volume very close to 1 L. Thus, 1 ppm also corresponds to 1 mg of solute per liter of aqueous solution.
The acceptable maximum concentrations of toxic or carcinogenic substances in the environment are often expressed in ppm or ppb. For example, the maximum allowable concentration of arsenic in drinking water in the United States is 0.010 ppm; that is, 0.010 mg of arsenic per liter of water. This concentration corresponds to 10 ppb.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
A solution of SO2 in water contains 0.00023 g of SO2 per liter of solution. What is the concentration of SO2 in ppm? In ppb?
SAMPLE EXERCISE 13.3 Calculation of Mass-Related Concentrations
(a) A solution is made by dissolving 13.5 g of glucose (C6H12O6) in 0.100 kg of water. What is the mass percentage of solute in this solution? (b) A 2.5-g sample of groundwater was found to contain 5.4 μg of Zn2+. What is the concentration of Zn2+ in parts per million?
SOLUTION
(a) Analyze We are given the number of grams of solute (13.5 g) and the number of grams of solvent (0.100 kg = 100 g). From this we must calculate the mass percentage of solute.
Plan We can calculate the mass percentage by using Equation 13.5. The mass of the solution is the sum of the mass of solute (glucose) and the mass of solvent (water).
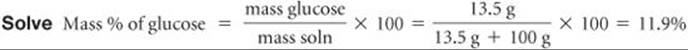
Comment The mass percentage of water in this solution is (100–11.9)% = 88.1%.
(b) Analyze In this case we are given the number of micrograms of solute. Because 1 μg is 1 × 10–6 g, 5.4 μg = 5.4 × 10–6 g.
Plan We calculate the parts per million using Equation 13.6.
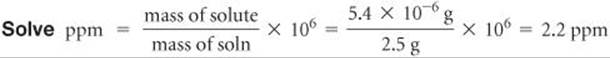
PRACTICE EXERCISE
(a) Calculate the mass percentage of NaCl in a solution containing 1.50 g of NaCl in 50.0 g of water. (b) A commercial bleaching solution contains 3.62 mass % sodium hypochlorite, NaOCl. What is the mass of NaOCl in a bottle containing 2.50 kg of bleaching solution?
Answers: (a) 2.91%, (b) 90.5 g of NaOCl
Mole Fraction, Molarity, and Molality
Concentration expressions are often based on the number of moles of one or more components of the solution. Recall from Section 10.6 that the mole fraction of a component of a solution is given by

The symbol X is commonly used for mole fraction, with a subscript to indicate the component of interest. For example, the mole fraction of HCl in a hydrochloric acid solution is represented as XHCl. Thus, if a solution contains 1.00 mol of HCl (36.5 g) and 8.00 mol of water (144 g), the mole fraction of HCl is XHCl = (1.00 mol)/(1.00 mol + 8.00 mol) = 0.111. Mole fractions have no units because the units in the numerator and the denominator cancel. The sum of the mole fractions of all components of a solution must equal 1. Thus, in the aqueous HCl solution, XH2O = 1.000 – 0.111 = 0.889. Mole fractions are very useful when dealing with gases, as we saw in Section 10.6, but have limited use when dealing with liquid solutions.
Recall from Section 4.5 that the molarity (M) of a solute in a solution is defined as
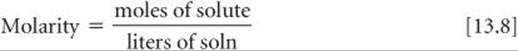
For example, if you dissolve 0.500 mol of Na2CO3 in enough water to form 0.250 L of solution, the molarity of Na2CO3 in the solution is (0.500 mol)/(0.250 L) = 2.00 M. Molarity is especially useful for relating the volume of a solution to the quantity of solute contained in that volume, as we saw in our discussions of titrations. (![]() Section 4.6)
Section 4.6)
The molality of a solution, denoted m, is a concentration unit that is also based on moles of solute. Molality equals the number of moles of solute per kilogram of solvent:

Thus, if you form a solution by mixing 0.200 mol of NaOH (8.00 g) and 0.500 kg of water (500 g), the concentration of the solution is (0.200 mol)/(0.500 kg) = 0.400 m (that is, 0.400 molal) in NaOH.
The definitions of molarity and molality are similar enough that they can be easily confused. Molarity depends on the volume of solution, whereas molality depends on the mass of solvent. When water is the solvent, the molality and molarity of dilute solutions are numerically about the same because 1 kg of solvent is nearly the same as 1 kg of solution, and 1 kg of the solution has a volume of about 1 L.
The molality of a given solution does not vary with temperature because masses do not vary with temperature. The molarity of the solution does change with temperature, however, because the volume of the solution expands or contracts with temperature. Thus, molality is often the concentration unit of choice when a solution is to be used over a range of temperatures.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
If an aqueous solution is very dilute, will its molality be greater than its molarity, nearly the same as its molarity, or smaller than its molarity?
SAMPLE EXERCISE 13.4 Calculation of Molality
A solution is made by dissolving 4.35 g glucose (C6H12O6) in 25.0 mL of water at 25 °C. Calculate the molality of glucose in the solution. Water has a density of 1.00 g/mL.
SOLUTION
Analyze We are asked to calculate a molality. To do this, we must determine the number of moles of solute (glucose) and the number of kilograms of solvent (water).
Plan We use the molar mass of C6H12O6 to convert grams to moles. We use the density of water to convert milliliters to kilograms. The molality equals the number of moles of solute divided by the number of kilograms of solvent (Equation 13.9).
Solve Use the molar mass of glucose, 180.2 g/mol, to convert grams to moles:
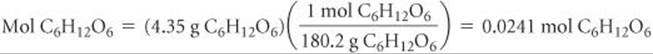
Because water has a density of 1.00 g/mL, the mass of the solvent is
(25.0 mL)(1.00 g/mL) = 25.0 g = 0.0250 kg
Finally, use Equation 13.9 to obtain the molality:

PRACTICE EXERCISE
What is the molality of a solution made by dissolving 36.5 g of naphthalene (C10H8) in 425 g of toluene (C7H8)?
Answer: 0.670 m
Converting Concentration Units
Sometimes the concentration of a given solution needs to be known in several concentration units. It is possible to interconvert concentration units as shown in Sample Exercise 13.5.
SAMPLE EXERCISE 13.5 Calculation of Mole Fraction and Molality
An aqueous solution of hydrochloric acid contains 36% HCl by mass. (a) Calculate the mole fraction of HCl in the solution. (b) Calculate the molality of HCl in the solution.
SOLUTION
Analyze We are asked to calculate the concentration of the solute, HCl, in two related concentration units, given only the percentage by mass of the solute in the solution.
Plan In converting concentration units based on the mass or moles of solute and solvent (mass percentage, mole fraction, and molality), it is useful to assume a certain total mass of solution. Let's assume that there is exactly 100 g of solution. Because the solution is 36% HCl, it contains 36 g of HCl and (100 – 36) g = 64 g of H2O. We must convert grams of solute (HCl) to moles to calculate either mole fraction or molality. We must convert grams of solvent (H2O) to moles to calculate mole fractions and to kilograms to calculate molality.
Solve (a) To calculate the mole fraction of HCl, we convert the masses of HCl and H2O to moles and then use Equation 13.7:

(b) To calculate the molality of HCl in the solution, we use Equation 13.9. We calculated the number of moles of HCl in part (a), and the mass of solvent is 64 g = 0.064 kg:
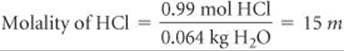
PRACTICE EXERCISE
A commercial bleach solution contains 3.62 mass % NaOCl in water. Calculate (a) the mole fraction and (b) the molality of NaOCl in the solution.
Answers: (a) 9.00 × 10–3, (b) 0.505 m
To interconvert molality and molarity, we need to know the density of the solution. ![]() FIGURE 13.20 outlines the calculation of the molarity and molality of a solution from the mass of solute and the mass of solvent. The mass of the solution is the sum of masses of the solvent and solute. The volume of the solution can be calculated from its mass and density.
FIGURE 13.20 outlines the calculation of the molarity and molality of a solution from the mass of solute and the mass of solvent. The mass of the solution is the sum of masses of the solvent and solute. The volume of the solution can be calculated from its mass and density.
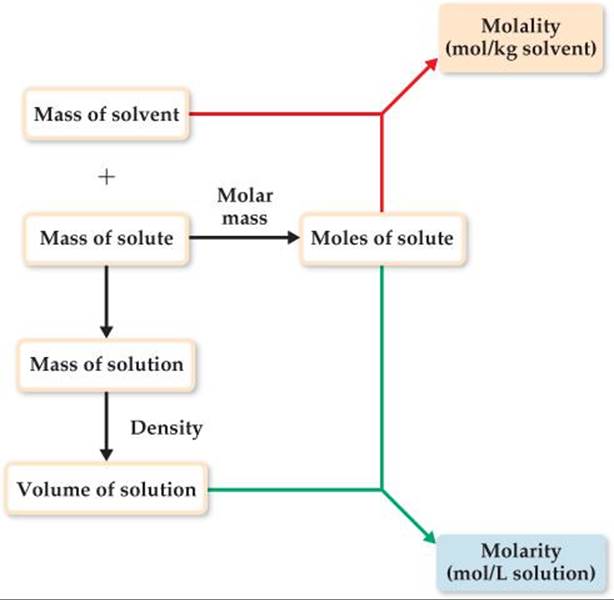
![]() FIGURE 13.20 Calculating molality and molarity from solute mass, solvent mass, and solution density.
FIGURE 13.20 Calculating molality and molarity from solute mass, solvent mass, and solution density.
SAMPLE EXERCISE 13.6 Calculation of Molarity Using the Density of the Solution
A solution with a density of 0.876 g/mL contains 5.0 g of toluene (C7H8) and 225 g of benzene. Calculate the molarity of the solution.
SOLUTION
Analyze Our goal is to calculate the molarity of a solution, given the masses of solute (5.0 g) and solvent (225 g) and the density of the solution (0.876 g/mL).
Plan The molarity of a solution is the number of moles of solute divided by the number of liters of solution (Equation 13.8). The number of moles of solute (C7H8) is calculated from the number of grams of solute and its molar mass. The volume of the solution is obtained from the mass of the solution (mass of solute + mass of solvent = 5.0 g + 225 g = 230 g) and its density.
Solve The number of moles of solute is
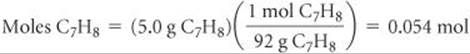
The density of the solution is used to convert the mass of the solution to its volume:
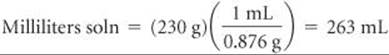
Molarity is moles of solute per liter of solution:

Check The magnitude of our answer is reasonable. Rounding moles to 0.05 and liters to 0.25 gives a molarity of
(0.05 mol)/(0.25 L) = 0.2 M
The units for our answer (mol/L) are correct, and the answer, 0.21, has two significant figures, corresponding to the number of significant figures in the mass of solute (2).
Comment Because the mass of the solvent (0.225 kg) and the volume of the solution (0.263) are similar in magnitude, the molarity and molality are also similar in magnitude:
(0.054 mol C7H8)/(0.225 kg solvent) = 0.24 m
PRACTICE EXERCISE
A solution containing equal masses of glycerol (C3H8O3) and water has a density of 1.10 g/mL. Calculate (a) the molality of glycerol, (b) the mole fraction of glycerol, (c) the molarity of glycerol in the solution.
Answers: (a) 10.9 m, (b) XC3H8O3 = 0.163, (c) 5.97 M