CHEMISTRY THE CENTRAL SCIENCE
14 CHEMICAL KINETICS
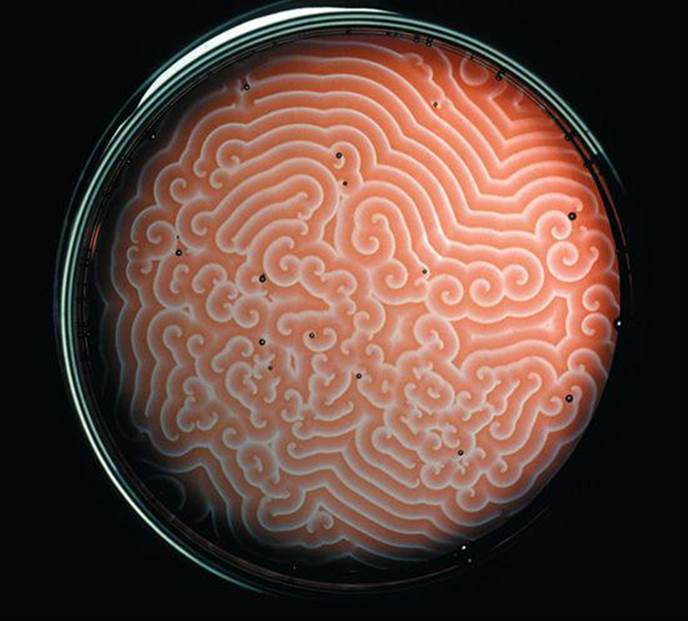
THE BELOUSOV-ZHABOTINSKY REACTION is one of the most famous reactions in the chemical world. The swirls of colors correspond to products forming at different places and times in the unstirred petri dish.
WHAT'S AHEAD
14.1 FACTORS THAT AFFECT REACTION RATES
We explore four variables that affect reaction rates: concentration, physical states of reactants, temperature, and presence of catalysts. These factors can be understood in terms of the collisions among reactant molecules that lead to reaction.
14.2 REACTION RATES
We consider how to express reaction rates and how reactant disappearance rates and product appearance rates are related to the reaction stoichiometry.
14.3 CONCENTRATION AND RATE LAWS
We then show that the effect of concentration on rate is expressed quantitatively by rate laws and show how rate laws and rate constants are determined experimentally.
14.4 THE CHANGE OF CONCENTRATION WITH TIME
We learn that rate equations can be written to express how concentrations change with time and look at several examples of rate equations: zero-order, first-order, and second-order reactions.
14.5 TEMPERATURE AND RATE
We next consider the effect of temperature on rate. In order to occur, most reactions require a minimum input of energy called the activation energy.
14.6 REACTION MECHANISMS
We then examine reaction mechanisms, the step-by-step molecular pathways leading from reactants to products.
14.7 CATALYSIS
We end the chapter with a discussion of how catalysts increase reaction rates, including a discussion of biological catalysts called enzymes.
CHEMICAL REACTIONS CONVERT SUBSTANCES that have well-defined properties into other substances that have different properties. The chapter-opening photograph, for instance, shows that we can watch complex reactions as they happen in time by observing the different colors of reactants and products. In this particular case, multiple redox reactions are occurring, with the products of one reaction becoming the reactants in other reactions.
So far, we have been concerned with the beginning and end of chemical reactions: We start with certain reactants and see what products they yield. This view is useful but does not tell us what happens in the middle, that is, which chemical bonds are broken, which are formed, and in what order these events occur. The speed at which a chemical reaction occurs is called the reaction rate. To investigate how reactions happen, we must examine the reaction rates and the factors that influence them. Experimental information on the rate of a given reaction provides important evidence that helps us formulate a reaction mechanism, which is a step-by-step, molecular-level view of the pathway from reactants to products.
Some chemical reactions are complete within a fraction of a second, such as explosions, whereas others can take thousands or even millions of years, such as the formation of minerals in Earth's crust (![]() FIGURE 14.1).
FIGURE 14.1).

![]() FIGURE 14.1 Reaction rates span an enormous range of time scales. The absorption of light by an atom or molecule is complete within one femtosecond; explosions occur within seconds; corrosion can occur over years; and the weathering of rocks can occur over millions of years.
FIGURE 14.1 Reaction rates span an enormous range of time scales. The absorption of light by an atom or molecule is complete within one femtosecond; explosions occur within seconds; corrosion can occur over years; and the weathering of rocks can occur over millions of years.
The area of chemistry concerned with the speeds, or rates, of reactions is chemical kinetics. Chemical kinetics relates to how quickly a medicine works in the body, to whether the processes that form and deplete ozone in the atmosphere are in balance, and to such industrial challenges as the development of new catalysts, materials that speed up reactions.
Our goal in this chapter is to understand how to determine reaction rates and to consider the factors that control these rates. What factors determine how rapidly food spoils, for instance, or how does one design a fast-setting material for dental fillings? What determines the rate at which steel rusts, or how can we remove hazardous pollutants in automobile exhaust before the exhaust leaves the tailpipe? Although we will not address these specific questions, we will see that the rates of all chemical reactions are subject to the same principles.
14.1 FACTORS THAT AFFECT REACTION RATES
Four factors allow us to change the rate at which any particular reaction occurs:
1. Physical state of the reactants. Reactants must come together to react. The more readily reactant molecules collide with one another, the more rapidly they react. Most of the reactions we consider are homogeneous, involving either all gases or all liquids. When reactants are in different phases, however, we have heterogeneous conditions, and the reaction is limited by the area of contact of the reactants. Thus, heterogeneous reactions that involve solids tend to proceed faster if the surface area of the solid is increased. For example, a medicine in the form of a fine powder dissolves in the stomach and enters the blood more quickly than the same medicine in the form of a tablet.
2. Reactant concentrations. Most chemical reactions proceed faster if the concentration of one or more reactants is increased. For example, steel wool burns only slowly in air, which contains 20% O2, but bursts into flame in pure oxygen(![]() FIGURE 14.2). As reactant concentration increases, the frequency with which the reactant molecules collide increases, leading to increased rates.
FIGURE 14.2). As reactant concentration increases, the frequency with which the reactant molecules collide increases, leading to increased rates.
3. Reaction temperature. Reaction rates generally increase as temperature is increased. The bacterial reactions that spoil milk, for instance, proceed more rapidly at room temperature than at the lower temperature of a refrigerator. Increasing temperature increases the kinetic energies of molecules. ![]() (Section 10.7) As molecules move more rapidly, they collide more frequently and with higher energy, leading to increased reaction rates.
(Section 10.7) As molecules move more rapidly, they collide more frequently and with higher energy, leading to increased reaction rates.

![]() FIGURE 14.2 Effect of concentration on reaction rate. The difference in behavior is due to the different concentrations of O2 in the two environments.
FIGURE 14.2 Effect of concentration on reaction rate. The difference in behavior is due to the different concentrations of O2 in the two environments.
4. The presence of a catalyst. Catalysts are agents that increase reaction rates without themselves being used up. They affect the kinds of collisions (and therefore alter the mechanism) that lead to reaction. Catalysts play many crucial roles in living organisms, including ourselves.
On a molecular level, reaction rates depend on the frequency of collisions between molecules. The greater the frequency of collisions, the higher the reaction rate. For a collision to lead to a reaction, however, it must occur with sufficient energy to break bonds and with suitable orientation for new bonds to form in the proper locations. We will consider these factors as we proceed through this chapter.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
In a reaction involving reactants in the gas state, how does increasing the partial pressures of the gases affect the reaction rate?