CHEMISTRY THE CENTRAL SCIENCE
14 CHEMICAL KINETICS
14.7 CATALYSIS
A catalyst is a substance that changes the speed of a chemical reaction without undergoing a permanent chemical change itself. Most reactions in the body, the atmosphere, and the oceans occur with the help of catalysts. Much industrial chemical research is devoted to the search for more effective catalysts for reactions of commercial importance. Extensive research efforts also are devoted to finding means of inhibiting or removing certain catalysts that promote undesirable reactions, such as those that corrode metals, age our bodies, and cause tooth decay.
Homogeneous Catalysis
A catalyst that is present in the same phase as the reactants in a reaction mixture is called a homogeneous catalyst. Examples abound both in solution and in the gas phase. Consider, for example, the decomposition of aqueous hydrogen peroxide, H2O2(aq), into water and oxygen:
![]()
In the absence of a catalyst, this reaction occurs extremely slowly. Many substances are capable of catalyzing the reaction, however, including bromide ion, which reacts with hydrogen peroxide in acidic solution, forming aqueous bromine and water (![]() FIGURE 14.22):
FIGURE 14.22):
![]()

![]() FIGURE 14.22 Homogeneous catalysis. Effect of catalyst on the speed of hydrogen peroxide decomposition to water and oxygen gas.
FIGURE 14.22 Homogeneous catalysis. Effect of catalyst on the speed of hydrogen peroxide decomposition to water and oxygen gas.
If this were the complete reaction, bromide ion would not be a catalyst because it undergoes chemical change during the reaction. However, hydrogen peroxide also reacts with the Br2(aq) generated in Equation 14.30:
![]()
The sum of Equations 14.30 and 14.31 is just Equation 14.29, a result which you can check for yourself.
When the H2O2 has been completely decomposed, we are left with a colorless solution of Br–(aq), which means that this ion is indeed a catalyst of the reaction because it speeds up the reaction without itself undergoing any net change. In contrast, Br2 is an intermediate because it is first formed (Equation 14.30) and then consumed (Equation 14.31). Neither the catalyst nor the intermediate appears in the equation for the overall reaction. Notice, however, that the catalyst is there at the start of the reaction, whereas the intermediate is formed during the course of the reaction.
How does a catalyst work? If we think about the general form of rate laws (Equation 14.7, rate = k[A]m[B]n), we must conclude that the catalyst must affect the numerical value of k, the rate constant. On the basis of the Arrhenius equation (Equation 14.19, k = Ae–Ea/RT), k is determined by the activation energy (Ea) and the frequency factor (A). A catalyst may affect the rate of reaction by altering the value of either Ea or A. We can envision this happening in two ways: The catalyst could provide a new mechanism for the reaction that has an Ea value lower than the Ea value for the uncatalyzed reaction, or the catalyst could assist in the orientation of reactants and so increase A. The most dramatic catalytic effects come from lowering Ea. As a general rule, a catalyst lowers the overall activation energy for a chemical reaction.
![]() GO FIGURE
GO FIGURE
Where are the intermediates and transition states in this diagram?
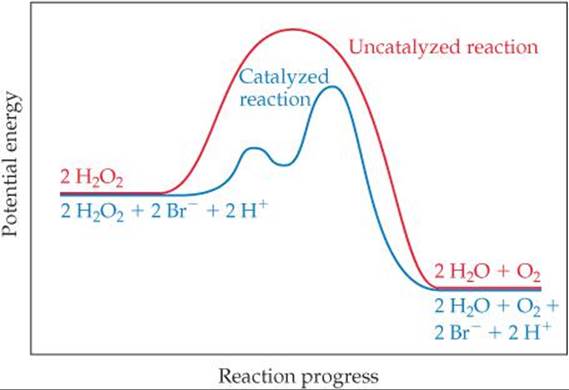
![]() FIGURE 14.23 Energy profiles for the uncatalyzed and bromide-catalyzed decomposition of H2O2.
FIGURE 14.23 Energy profiles for the uncatalyzed and bromide-catalyzed decomposition of H2O2.
A catalyst can lower the activation energy for a reaction by providing a different mechanism for the reaction. In the decomposition of hydrogen peroxide, for example, two successive reactions of H2O2, first with bromide and then with bromine, take place. Because these two reactions together serve as a catalytic pathway for hydrogen peroxide decomposition, both of them must have significantly lower activation energies than the uncatalyzed decomposition (![]() FIGURE 14.23).
FIGURE 14.23).
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
How does a catalyst increase the rate of a reaction?
Heterogeneous Catalysis
A heterogeneous catalyst is one that exists in a phase different from the phase of the reactant molecules, usually as a solid in contact with either gaseous reactants or with reactants in a liquid solution. Many industrially important reactions are catalyzed by the surfaces of solids. For example, hydrocarbon molecules are rearranged to form gasoline with the aid of what are called “cracking” catalysts. Heterogeneous catalysts are often composed of metals or metal oxides. Because the catalyzed reaction occurs on the surface, special methods are often used to prepare catalysts so that they have very large surface areas.
The initial step in heterogeneous catalysis is usually adsorption of reactants. Adsorption refers to the binding of molecules to a surface, whereas absorption refers to the uptake of molecules into the interior of a substance. ![]() (Section 13.6) Adsorption occurs because the atoms or ions at the surface of a solid are extremely reactive. Unlike their counterparts in the interior of the substance, surface atoms and ions have unused bonding capacity that can be used to bond molecules from the gas or solution phase to the surface of the solid.
(Section 13.6) Adsorption occurs because the atoms or ions at the surface of a solid are extremely reactive. Unlike their counterparts in the interior of the substance, surface atoms and ions have unused bonding capacity that can be used to bond molecules from the gas or solution phase to the surface of the solid.
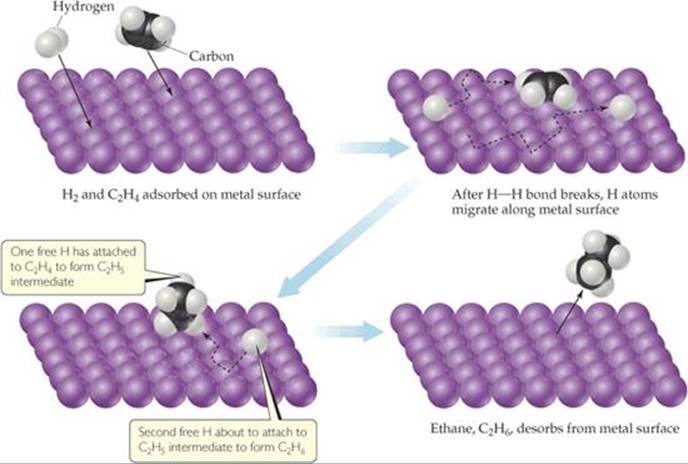
![]() FIGURE 14.24 Heterogeneous catalysis. Mechanism for reaction of ethylene with hydrogen on a catalytic surface.
FIGURE 14.24 Heterogeneous catalysis. Mechanism for reaction of ethylene with hydrogen on a catalytic surface.
The reaction of hydrogen gas with ethylene gas to form ethane gas provides an example of heterogeneous catalysis:
![]()
Even though this reaction is exothermic, it occurs very slowly in the absence of a catalyst. In the presence of a finely powdered metal, however, such as nickel, palladium, or platinum, the reaction occurs easily at room temperature via the mechanism diagrammed in ![]() FIGURE 14.24. Both ethylene and hydrogen are adsorbed on the metal surface. Upon adsorption, the H — H bond of H2 breaks, leaving two H atoms initially bonded to the metal surface but relatively free to move. When a hydrogen encounters an adsorbed ethylene molecule, it can form a σ bond to one of the carbon atoms, effectively destroying the C — C π bond and leaving an ethyl group (C2H5) bonded to the surface via a metal-to-carbon σ bond. This σ bond is relatively weak, so when the other carbon atom also encounters a hydrogen atom, a sixth C — H σ bond is readily formed, and an ethane molecule (C2H6) is released from the metal surface.
FIGURE 14.24. Both ethylene and hydrogen are adsorbed on the metal surface. Upon adsorption, the H — H bond of H2 breaks, leaving two H atoms initially bonded to the metal surface but relatively free to move. When a hydrogen encounters an adsorbed ethylene molecule, it can form a σ bond to one of the carbon atoms, effectively destroying the C — C π bond and leaving an ethyl group (C2H5) bonded to the surface via a metal-to-carbon σ bond. This σ bond is relatively weak, so when the other carbon atom also encounters a hydrogen atom, a sixth C — H σ bond is readily formed, and an ethane molecule (C2H6) is released from the metal surface.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
How does a homogeneous catalyst compare with a heterogeneous one regarding the ease of recovery of the catalyst from the reaction mixture?
Enzymes
The human body is characterized by an extremely complex system of interrelated chemical reactions, all of which must occur at carefully controlled rates to maintain life. A large number of marvelously efficient biological catalysts known as enzymes are necessary for many of these reactions to occur at suitable rates. Most enzymes are large protein molecules with molecular weights ranging from about 10,000 to about 1 million amu. They are very selective in the reactions they catalyze, and some are absolutely specific, operating for only one substance in only one reaction. The decomposition of hydrogen peroxide, for example, is an important biological process. Because hydrogen peroxide is strongly oxidizing, it can be physiologically harmful. For this reason, the blood and liver of mammals contain an enzyme, catalase, that catalyzes the decomposition of hydrogen peroxide into water and oxygen (Equation 14.29). ![]() FIGURE 14.26 shows the dramatic acceleration of this chemical reaction by the catalase in beef liver.
FIGURE 14.26 shows the dramatic acceleration of this chemical reaction by the catalase in beef liver.
 CHEMISTRY PUT TO WORK
CHEMISTRY PUT TO WORK
Catalytic Converters
Heterogeneous catalysis plays a major role in the fight against urban air pollution. Two components of automobile exhausts that help form photochemical smog are nitrogen oxides and unburned hydrocarbons. In addition, automobile exhaust may contain considerable quantities of carbon monoxide. Even with the most careful attention to engine design, it is impossible under normal driving conditions to reduce the quantity of these pollutants to an acceptable level in the exhaust gases. It is therefore necessary to remove them from the exhaust before they are vented to the air. This removal is accomplished in the catalytic converter.
The catalytic converter, which is part of an automobile's exhaust system, must perform two functions: (1) oxidation of CO and unburned hydrocarbons (CxHy) to carbon dioxide and water, and (2) reduction of nitrogen oxides to nitrogen gas:
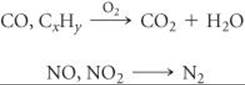
These two functions require different catalysts, so the development of a successful catalyst system is a difficult challenge. The catalysts must be effective over a wide range of operating temperatures. They must continue to be active despite the fact that various components of the exhaust can block the active sites of the catalyst. And the catalysts must be sufficiently rugged to withstand exhaust gas turbulence and the mechanical shocks of driving under various conditions for thousands of miles.
Catalysts that promote the combustion of CO and hydrocarbons are, in general, the transition-metal oxides and the noble metals. These materials are supported on a structure (![]() FIGURE 14.25) that allows the best possible contact between the flowing exhaust gas and the catalyst surface. A honeycomb structure made from alumina (Al2O3) and impregnated with the catalyst is employed. Such catalysts operate by first adsorbing oxygen gas present in the exhaust gas. This adsorption weakens the O — O bond in O2, so that oxygen atoms are available for reaction with adsorbed CO to form CO2. Hydrocarbon oxidation probably proceeds somewhat similarly, with the hydrocarbons first being adsorbed followed by rupture of a C — H bond.
FIGURE 14.25) that allows the best possible contact between the flowing exhaust gas and the catalyst surface. A honeycomb structure made from alumina (Al2O3) and impregnated with the catalyst is employed. Such catalysts operate by first adsorbing oxygen gas present in the exhaust gas. This adsorption weakens the O — O bond in O2, so that oxygen atoms are available for reaction with adsorbed CO to form CO2. Hydrocarbon oxidation probably proceeds somewhat similarly, with the hydrocarbons first being adsorbed followed by rupture of a C — H bond.
Transition-metal oxides and noble metals are also the most effective catalysts for reduction of NO to N2 and O2. The catalysts that are most effective in one reaction, however, are usually much less effective in the other. It is therefore necessary to have two catalytic components.
Catalytic converters contain remarkably efficient heterogeneous catalysts. The automotive exhaust gases are in contact with the catalyst for only 100 to 400 ms, but in this very short time, 96% of the hydrocarbons and CO is converted to CO2 and H2O, and the emission of nitrogen oxides is reduced by 76%.
There are costs as well as benefits associated with the use of catalytic converters, one being that some of the metals are very expensive. Catalytic converters currently account for about 35% of the platinum, 65% of the palladium, and 95% of the rhodium used annually. All of these metals, which come mainly from Russia and South Africa, can be far more expensive than gold.
RELATED EXERCISES: 14.64, 14.87, 14.88

![]() FIGURE 14.25 Cross section of a catalytic converter.
FIGURE 14.25 Cross section of a catalytic converter.

![]() FIGURE 14.26 Enzymes speed up reactions.
FIGURE 14.26 Enzymes speed up reactions.
![]() GO FIGURE
GO FIGURE
Which molecules must bind more tightly to the active site, substrates or products?
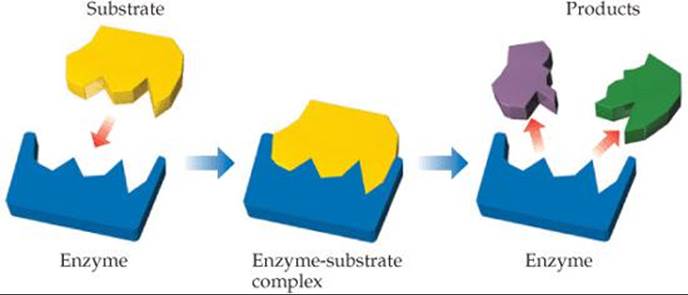
![]() FIGURE 14.27 Lock-and-key model for enzyme action.
FIGURE 14.27 Lock-and-key model for enzyme action.
The reaction any given enzyme catalyzes takes place at a specific location in the enzyme, the active site. The substances that react at this site are called substrates. The lock-and-key model provides a simple explanation for the specificity of an enzyme (![]() FIGURE 14.27). The substrate is pictured as fitting neatly into the active site, much like a key fits into a lock. Although this model oversimplifies the situation, since enzymes can “wiggle” to adjust the shape and size of the active site, the lock-and-key model is a good place to start understanding enzyme activity.
FIGURE 14.27). The substrate is pictured as fitting neatly into the active site, much like a key fits into a lock. Although this model oversimplifies the situation, since enzymes can “wiggle” to adjust the shape and size of the active site, the lock-and-key model is a good place to start understanding enzyme activity.
![]() FIGURE 14.28 shows a model of the enzyme lysozyme without and with a bound substrate molecule.
FIGURE 14.28 shows a model of the enzyme lysozyme without and with a bound substrate molecule.
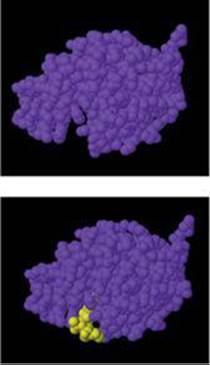
![]() FIGURE 14.28 Molecular model of lysozyme without and with a bound substrate molecule (yellow).
FIGURE 14.28 Molecular model of lysozyme without and with a bound substrate molecule (yellow).
The combination of enzyme and substrate is called the enzyme-substrate complex. Although Figure 14.27 shows both the active site and its substrate as having a fixed shape, the active site is often fairly flexible and so may change shape as it binds the substrate. The binding between substrate and active site involves dipole-dipole attractions, hydrogen bonds, and dispersion forces. ![]() (Section 11.2)
(Section 11.2)
As substrate molecules enter the active site, they are somehow activated so that they are capable of reacting rapidly. This activation process may occur, for example, by the withdrawal or donation of electron density from a particular bond or group of atoms in the enzyme's active site. In addition, the substrate may become distorted in the process of fitting into the active site and made more reactive. Once the reaction occurs, the products depart from the active site, allowing another substrate molecule to enter.
The activity of an enzyme is destroyed if some molecule other than the substrate specific to that enzyme binds to the active site and blocks entry of the substrate. Such substances are called enzyme inhibitors. Nerve poisons and certain toxic metal ions, such as lead and mercury, are believed to act in this way to inhibit enzyme activity. Some other poisons act by attaching elsewhere on the enzyme, thereby distorting the active site so that the substrate no longer fits.
Enzymes are enormously more efficient than nonbiochemical catalysts. The number of individual catalyzed reaction events occurring at a particular active site, called the turnover number, is generally in the range of 103 to 107 per second. Such large turnover numbers correspond to very low activation energies. Compared with a simple chemical catalyst, enzymes can increase the rate constant for a given reaction by a million-fold or more.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Is it reasonable to say that enzymes stabilize the transition state for a reaction? Explain your answer.
 CHEMISTRY AND LIFE
CHEMISTRY AND LIFE
NITROGEN FIXATION AND NITROGENASE
Nitrogen is one of the most essential elements in living organisms, found in many compounds vital to life, including proteins, nucleic acids, vitamins, and hormones. Nitrogen is continually cycling through the biosphere in various forms, as shown in ![]() FIGURE 14.29. For example, certain microorganisms convert the nitrogen in animal waste and dead plants and animals into N2(g), which then returns to the atmosphere. For the food chain to be sustained, there must be a means of converting atmospheric N2(g) in a form plants can use. For this reason, if a chemist were asked to name the most important chemical reaction in the world, she might easily say nitrogen fixation, the process by which atmospheric N2(g) is converted into compounds suitable for plant use. Some fixed nitrogen results from the action of lightning on the atmosphere, and some is produced industrially using a process we will discuss in Chapter 15. About 60% of fixed nitrogen, however, is a consequence of the action of the remarkable and complex enzyme nitrogenase. This enzyme is not present in humans or other animals; rather, it is found in bacteria that live in the root nodules of certain plants, such as the legumes clover and alfalfa.
FIGURE 14.29. For example, certain microorganisms convert the nitrogen in animal waste and dead plants and animals into N2(g), which then returns to the atmosphere. For the food chain to be sustained, there must be a means of converting atmospheric N2(g) in a form plants can use. For this reason, if a chemist were asked to name the most important chemical reaction in the world, she might easily say nitrogen fixation, the process by which atmospheric N2(g) is converted into compounds suitable for plant use. Some fixed nitrogen results from the action of lightning on the atmosphere, and some is produced industrially using a process we will discuss in Chapter 15. About 60% of fixed nitrogen, however, is a consequence of the action of the remarkable and complex enzyme nitrogenase. This enzyme is not present in humans or other animals; rather, it is found in bacteria that live in the root nodules of certain plants, such as the legumes clover and alfalfa.
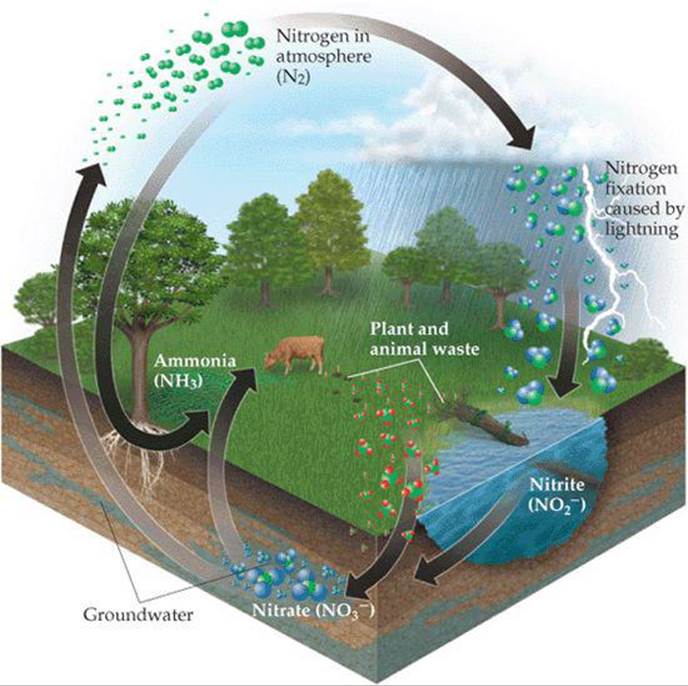
![]() FIGURE 14.29 Simplified picture of the nitrogen cycle.
FIGURE 14.29 Simplified picture of the nitrogen cycle.
Nitrogenase converts N2 into NH3, a process that, in the absence of a catalyst, has a very large activation energy. This process is a reduction reaction in which the oxidation state of N is reduced from 0 in N2 to –3 in NH3. The mechanism by which nitrogenase reduces N2 is not fully understood. Like many other enzymes, including catalase, the active site of nitrogenase contains transition-metal atoms; such enzymes are called metalloenzymes. Because transition metals can readily change oxidation state, metalloenzymes are especially useful for effecting transformations in which substrates are either oxidized or reduced.
It has been known for nearly 30 years that a portion of nitrogenase contains iron and molybdenum atoms. This portion, called the FeMo-cofactor, is thought to serve as the active site of the enzyme. The FeMo-cofactor of nitrogenase is a cluster of seven Fe atoms and one Mo atom, all linked by sulfur atoms (![]() FIGURE 14.30).
FIGURE 14.30).
It is one of the wonders of life that simple bacteria can contain beautifully complex and vitally important enzymes such as nitrogenase. Because of this enzyme, nitrogen is continually cycled between its comparatively inert role in the atmosphere and its critical role in living organisms. Without nitrogenase, life as we know it could not exist on Earth.
RELATED EXERCISES: 14.91, 14.126
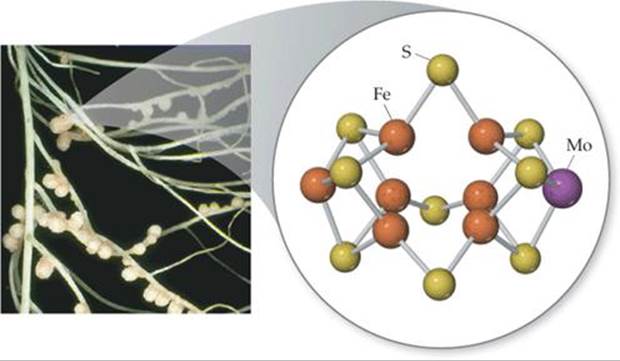
![]() FIGURE 14.30 The FeMo-cofactor of nitrogenase. Nitrogenase is found in nodules in the roots of certain plants, such as the white clover roots shown at the left. The cofactor, which is thought to be the active site of the enzyme, contains seven Fe atoms and one Mo atom, linked by sulfur atoms. The molecules on the outside of the cofactor connect it to the rest of the protein.
FIGURE 14.30 The FeMo-cofactor of nitrogenase. Nitrogenase is found in nodules in the roots of certain plants, such as the white clover roots shown at the left. The cofactor, which is thought to be the active site of the enzyme, contains seven Fe atoms and one Mo atom, linked by sulfur atoms. The molecules on the outside of the cofactor connect it to the rest of the protein.
SAMPLE INTEGRATIVE EXERCISE Putting Concepts Together
Formic acid (HCOOH) decomposes in the gas phase at elevated temperatures as follows:
HCOOH(g) → CO2(g) + H2(g)
The uncatalyzed decomposition reaction is determined to be first order. A graph of the partial pressure of HCOOH versus time for decomposition at 838 K is shown as the red curve in ![]() FIGURE 14.31. When a small amount of solid ZnO is added to the reaction chamber, the partial pressure of acid versus time varies as shown by the blue curve in Figure 14.31.
FIGURE 14.31. When a small amount of solid ZnO is added to the reaction chamber, the partial pressure of acid versus time varies as shown by the blue curve in Figure 14.31.
(a) Estimate the half-life and first-order rate constant for formic acid decomposition.
(b) What can you conclude from the effect of added ZnO on the decomposition of formic acid?
(c) The progress of the reaction was followed by measuring the partial pressure of formic acid vapor at selected times. Suppose that, instead, we had plotted the concentration of formic acid in units of mol/L. What effect would this have had on the calculated value of k?
(d) The pressure of formic acid vapor at the start of the reaction is 3.00 × 102 torr. Assuming constant temperature and ideal-gas behavior, what is the pressure in the system at the end of the reaction? If the volume of the reaction chamber is 436 cm3, how many moles of gas occupy the reaction chamber at the end of the reaction?
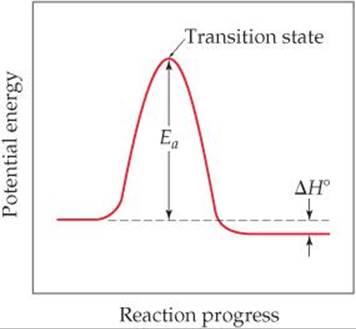
(e) The standard heat of formation of formic acid vapor is ΔH°f = –378.6 kJ/mol. Calculate ΔH° for the overall reaction. If the activation energy (Ea) for the reaction is 184 kJ/mol, sketch an approximate energy profile for the reaction, and label Ea, ΔH°, and the transition state.
SOLUTION
(a) The initial pressure of HCOOH is 3.00 × 102 torr. On the graph we move to the level at which the partial pressure of HCOOH is 1.50 × 102 torr, half the initial value. This corresponds to a time of about 6.60 × 102 s, which is therefore the half-life. The first-order rate constant is given by Equation 14.15: k = 0.693/t1/2 = 0.693/660 s = 1.05 × 10–3 s–1.
(b) The reaction proceeds much more rapidly in the presence of solid ZnO, so the surface of the oxide must be acting as a catalyst for the decomposition of the acid. This is an example of heterogeneous catalysis.
(c) If we had graphed the concentration of formic acid in units of moles per liter, we would still have determined that the half-life for decomposition is 660 s, and we would have computed the same value for k. Because the units for k are s–1, the value for k is independent of the units used for concentration.
(d) According to the stoichiometry of the reaction, two moles of product are formed for each mole of reactant. When reaction is completed, therefore, the pressure will be 600 torr, just twice the initial pressure, assuming ideal-gas behavior. (Because we are working at quite high temperature and fairly low gas pressure, assuming ideal-gas behavior is reasonable.) The number of moles of gas present can be calculated using the ideal-gas equation: ![]() (Section 10.4)
(Section 10.4)

(e) We first calculate the overall change in energy, ΔH° (![]() Section 5.7 and Appendix C), as in
Section 5.7 and Appendix C), as in
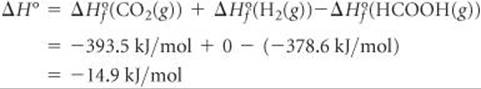
From this and the given value for Ea, we can draw an approximate energy profile for the reaction, in analogy to Figure 14.17.
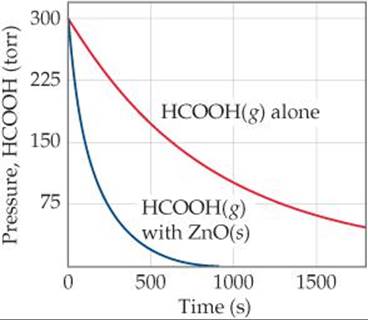
![]() FIGURE 14.31 Variation in pressure of HCOOH(g) as a function of time at 838 K.
FIGURE 14.31 Variation in pressure of HCOOH(g) as a function of time at 838 K.