CHEMISTRY THE CENTRAL SCIENCE
15 CHEMICAL EQUILIBRIUM
CHAPTER SUMMARY AND KEY TERMS
INTRODUCTION AND SECTION 15.1 A chemical reaction can achieve a state in which the forward and reverse processes are occurring at the same rate. This condition is called chemical equilibrium, and it results in the formation of an equilibrium mixture of the reac-tants and products of the reaction. The composition of an equilibrium mixture does not change with time if temperature is held constant.
SECTION 15.2 An equilibrium that is used throughout this chapter is the reaction N2(g) + 3 H2(g)![]() 2 NH3(g). This reaction is the basis of the Haber process for the production of ammonia. The relationship between the concentrations of the reactants and products of a system at equilibrium is given by the law of mass action. For an equilibrium equation of the form a A + b B
2 NH3(g). This reaction is the basis of the Haber process for the production of ammonia. The relationship between the concentrations of the reactants and products of a system at equilibrium is given by the law of mass action. For an equilibrium equation of the form a A + b B ![]() d D + e E, the equilibrium-constant expression is written as
d D + e E, the equilibrium-constant expression is written as
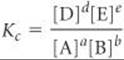
where Kc is a constant called the equilibrium constant. When the equilibrium system of interest consists of gases, it is often convenient to express the concentrations of reactants and products in terms of gas pressures:
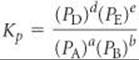
Kc and Kp are related by the expression Kp = Kc(RT)Δn.
SECTION 15.3 The value of the equilibrium constant changes with temperature. A large value of Kc indicates that the equilibrium mixture contains more products than reactants and therefore lies toward the product side of the equation. A small value for the equilibrium constant means that the equilibrium mixture contains less products than reactants and therefore lies toward the reactant side. The equilibrium-constant expression and the equilibrium constant of the reverse of a reaction are the reciprocals of those of the forward reaction. If a reaction is the sum of two or more reactions, its equilibrium constant will be the product of the equilibrium constants for the individual reactions.
SECTION 15.4 Equilibria for which all substances are in the same phase are called homogeneous equilibria; in heterogeneous equilibria two or more phases are present. The concentrations of pure solids and liquids are left out of the equilibrium-constant expression for a heterogeneous equilibrium.
SECTION 15.5 If the concentrations of all species in an equilibrium are known, the equilibrium-constant expression can be used to calculate the equilibrium constant. The changes in the concentrations of reac-tants and products on the way to achieving equilibrium are governed by the stoichiometry of the reaction.
SECTION 15.6 The reaction quotient, Q, is found by substituting reactant and product concentrations or partial pressures at any point during a reaction into the equilibrium-constant expression. If the system is at equilibrium, Q = K. If Q ≠ K, however, the system is not at equilibrium. When Q < K, the reaction will move toward equilibrium by forming more products (the reaction proceeds from left to right); when Q > K, the reaction will proceed from right to left. Knowing the value of K makes it possible to calculate the equilibrium amounts of reactants and products, often by the solution of an equation in which the unknown is the change in a partial pressure or concentration.
SECTION 15.7 Le Châtelier's principle states that if a system at equilibrium is disturbed, the equilibrium will shift to minimize the disturbing influence. By this principle, if a reactant or product is added to a system at equilibrium, the equilibrium will shift to consume the added substance. The effects of removing reactants or products and of changing the pressure or volume of a reaction can be similarly deduced. For example, if the volume of the system is reduced, the equilibrium will shift in the direction that decreases the number of gas molecules. The enthalpy change for a reaction indicates how an increase in temperature affects the equilibrium: For an endothermic reaction, an increase in temperature shifts the equilibrium to the right; for an exothermic reaction, a temperature increase shifts the equilibrium to the left. Catalysts affect the speed at which equilibrium is reached but do not affect the magnitude of K.
KEY SKILLS
• Understand what is meant by chemical equilibrium and how it relates to reaction rates (Section 15.1).
• Write the equilibrium-constant expression for any reaction (Section 15.2).
• Relate Kc and Kp (Section 15.2).
• Relate the magnitude of an equilibrium constant to the relative amounts of reactants and products present in an equilibrium mixture (Section 15.3).
• Manipulate the equilibrium constant to reflect changes in the chemical equation (Section 15.3).
• Write the equilibrium-constant expression for a heterogeneous reaction (Section 15.4).
• Calculate an equilibrium constant from concentration measurements (Section 15.5).
• Predict the direction of a reaction given the equilibrium constant and the concentrations of reactants and products (Section 15.6).
• Calculate equilibrium concentrations given the equilibrium constant and all but one equilibrium concentration (Section 15.6).
• Calculate equilibrium concentrations, given the equilibrium constant and the starting concentrations (Section 15.6).
• Understand how changing the concentrations, volume, or temperature of a system at equilibrium affects the equilibrium position (Section 15.7).
KEY EQUATIONS
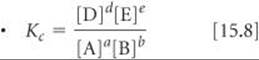
The equilibrium-constant expression for a general reaction of the type a A + b B ![]() d D + e E; the concentrations are equilibrium concentrations only
d D + e E; the concentrations are equilibrium concentrations only
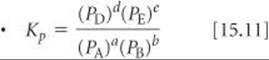
The equilibrium-constant expression in terms of equilibrium partial pressures
![]()
Relating the equilibrium constant based on pressures to the equilibrium constant based on concentration
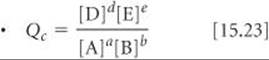
The reaction quotient. The concentrations are for any time during a reaction. If the concentrations are equilibrium concentrations, then Qc = Kc.