CHEMISTRY THE CENTRAL SCIENCE
16 ACID–BASE EQUILIBRIA
16.2 BRONSTED–LOWRY ACIDS AND BASES
The Arrhenius concept of acids and bases, while useful, has limitations. For one thing, it is restricted to aqueous solutions. In 1923 the Danish chemist Johannes Brønsted (1879–1947) and the English chemist Thomas Lowry (1874–1936) independently proposed a more general definition of acids and bases. Their concept is based on the fact that acid–base reactions involve the transfer of H+ions from one substance to another.
The H+ Ion in Water
We might at first imagine that ionization of HCl in water produces just H+ and Cl–. A hydrogen ion is no more than a bare proton, however, and would interact strongly with any source of electron density, such as the nonbonding electron pairs on the oxygen atoms of water molecules. For example, the interaction of a proton with water forms the hydronium ion, H3O+(aq):
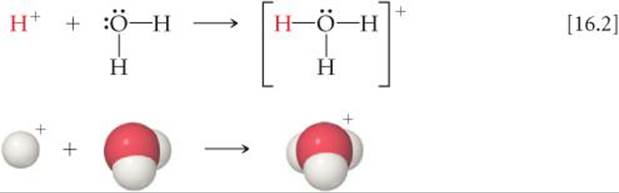
The behavior of H+ ions in liquid water is complex because hydronium ions are not the only polyatomic ions that form when H+ interacts with water molecules. The H3O+ ion can form hydrogen bonds to additional H2O molecules to generate such ions as H5O2+ and H9O4+ (![]() FIGURE 16.2)
FIGURE 16.2)
![]() GO FIGURE
GO FIGURE
Which type of intermolecular force do the dotted lines in this figure represent?
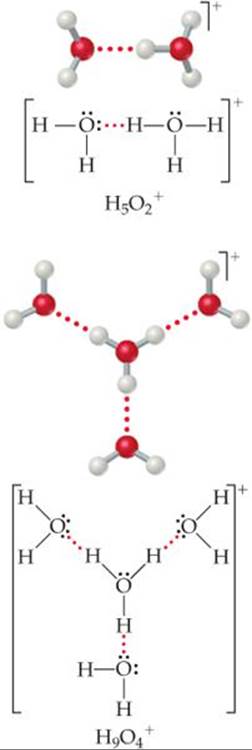
![]() FIGURE 16.2 Ball-and-stick models and Lewis structures for two hydrated hydronium ions.
FIGURE 16.2 Ball-and-stick models and Lewis structures for two hydrated hydronium ions.
Chemists use the notations H+(aq) and H3O+(aq) interchangeably to represent the hydrated proton responsible for the characteristic properties of aqueous solutions of acids. We often use the notation H+(aq) for simplicity and convenience, as we did in Chapter 4 and Equation 16.1. The notation H3O+(aq), however, more closely represents reality.
Proton-Transfer Reactions
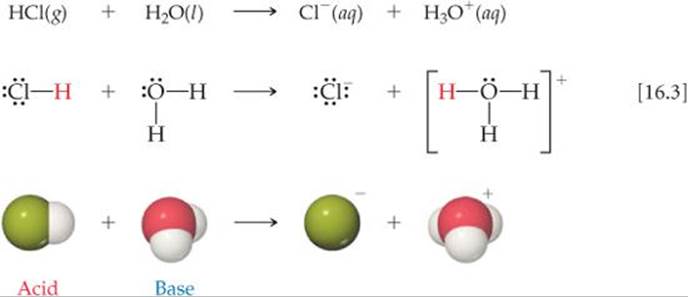
In the reaction that occurs when HCl dissolves in water, the HCl molecule transfers an H+ ion (a proton) to a water molecule. Thus, we can represent the reaction as occurring between an HCl molecule and a water molecule to form hydronium and chloride ions:
The polar H2O molecule promotes the ionization of acids in water solution by accepting a proton to form H3O+.
Brønsted and Lowry proposed definitions of acids and bases in terms of their ability to transfer protons:
• An acid is a substance (molecule or ion) that donates a proton to another substance.
• A base is a substance that accepts a proton.
Thus, when HCl dissolves in water (Equation 16.3), HCl acts as a Brønsted–Lowry acid (it donates a proton to H2O), and H2O acts as a Brønsted–Lowry base (it accepts a proton from HCl).
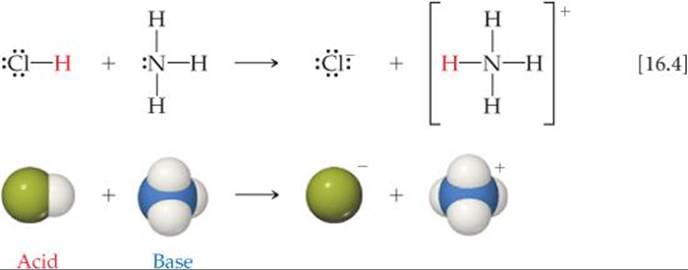
Because the emphasis in the Brønsted–Lowry concept is on proton transfer, the concept also applies to reactions that do not occur in aqueous solution. In the reaction between gas phase HCl and NH3, for example, a proton is transferred from the acid HCl to the base NH3:
The hazy film that forms on the windows of general chemistry laboratories and on glassware in the laboratory is largely solid NH4Cl formed by the gas-phase reaction between HCl and NH3.
Let's consider another example that compares the relationship between the Arrhe-nius and Brønsted–Lowry definitions of acids and bases—an aqueous solution of ammonia, in which we have the equilibrium:
![]()
Ammonia is a Brønsted–Lowry base because it accepts a proton from H2O. Ammonia is also an Arrhenius base because adding it to water leads to an increase in the concentration of OH–(aq).
An acid and a base always work together to transfer a proton. In other words, a substance can function as an acid only if another substance simultaneously behaves as a base. To be a Brønsted–Lowry acid, a molecule or ion must have a hydrogen atom it can lose as an H+ ion. To be a Brønsted–Lowry base, a molecule or ion must have a non-bonding pair of electrons it can use to bind the H+ ion.
Some substances can act as an acid in one reaction and as a base in another. For example, H2O is a Brønsted–Lowry base in Equation 16.3 and a Brønsted–Lowry acid in Equation 16.5. A substance capable of acting as either an acid or a base is called amphiprotic. An amphiprotic substance acts as a base when combined with something more strongly acidic than itself and as an acid when combined with something more strongly basic than itself.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
In the forward reaction of this equilibrium, which substance acts as the Brønsted–Lowry base?
![]()
Conjugate Acid–Base Pairs
In any acid–base equilibrium, both the forward reaction (to the right) and the reverse reaction (to the left) involve proton transfer. For example, consider the reaction of an acid HX with water:
![]()
In the forward reaction, HX donates a proton to H2O. Therefore, HX is the Brønsted–Lowry acid and H2O is the Brønsted–Lowry base. In the reverse reaction, the H3O+ ion donates a proton to the X– ion, so H3O+ is the acid and X– is the base. When the acid HX donates a proton, it leaves behind a substance, X–, that can act as a base. Likewise, when H2O acts as a base, it generates H3O+, which can act as an acid.
An acid and a base such as HX and X– that differ only in the presence or absence of a proton are called a conjugate acid–base pair.* Every acid has a conjugate base, formed by removing a proton from the acid. For example, OH– is the conjugate base of H2O, and X– is the conjugate base of HX. Every base has a conjugate acid, formed by adding a proton to the base. Thus, H3O+ is the conjugate acid of H2O, and HX is the conjugate acid of X–.
In any acid–base (proton-transfer) reaction, we can identify two sets of conjugate acid–base pairs. For example, consider the reaction between nitrous acid and water:
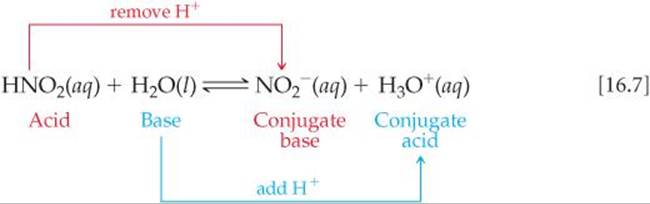
Likewise, for the reaction between NH3 and H2O (Equation 16.5), we have
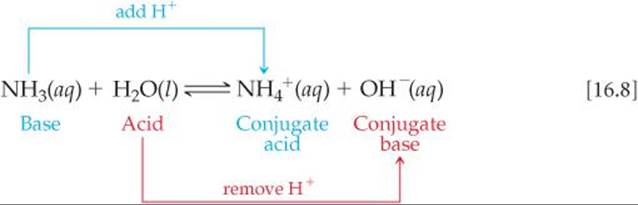
SAMPLE EXERCISE 16.1 Identifying Conjugate Acids and Bases
(a) What is the conjugate base of HClO4, H2S, PH4+, HCO3–? (b) What is the conjugate acid of CN–, SO42–, H2O, HCO3–?
SOLUTION
Analyze We are asked to give the conjugate base for several acids and the conjugate acid for several bases.
Plan The conjugate base of a substance is simply the parent substance minus one proton, and the conjugate acid of a substance is the parent substance plus one proton.
Solve
(a) HClO4 less one proton H+ is ClO4–. The other conjugate bases are HS–, PH3, and CO32–.
(b) CN– plus one proton H+ is HCN. The other conjugate acids are HSO4–, H3O+, and H2CO3. Notice that the hydrogen carbonate ion (HCO3–) is amphiprotic. It can act as either an acid or a base.
PRACTICE EXERCISE
Write the formula for the conjugate acid of each of the following: HSO3–, F–, PO43–, CO.
Answers: H2SO3, HF, HPO42–, HCO+
Once you become proficient at identifying conjugate acid–base pairs it is not difficult to write equations for reactions involving Brønsted–Lowry acids and bases (proton-transfer reactions).
SAMPLE EXERCISE 16.2 Writing Equations for Proton-Transfer Reactions
The hydrogen sulfite ion (HSO3–) is amphiprotic. Write an equation for the reaction of HSO3– with water (a) in which the ion acts as an acid and (b) in which the ion acts as a base. In both cases identify the conjugate acid–base pairs.
SOLUTION
Analyze and Plan We are asked to write two equations representing reactions between HSO3– and water, one in which HSO3– should donate a proton to water, thereby acting as a Brønsted–Lowry acid, and one in which HSO3– should accept a proton from water, thereby acting as a base. We are also asked to identify the conjugate pairs in each equation.
Solve
![]()
The conjugate pairs in this equation are HSO3– (acid) and SO32– (conjugate base), and H2O (base) and H3O+ (conjugate acid).
![]()
The conjugate pairs in this equation are H2O (acid) and OH– (conjugate base), and HSO3– (base) and H2SO3 (conjugate acid).
PRACTICE EXERCISE
When lithium oxide (Li2O) is dissolved in water, the solution turns basic from the reaction of the oxide ion (O2–) with water. Write the equation for this reaction and identify the conjugate acid–base pairs.
Answer: ![]() . The OH– is both the conjugate acid of O2– and the conjugate base of H2O.
. The OH– is both the conjugate acid of O2– and the conjugate base of H2O.
Relative Strengths of Acids and Bases
Some acids are better proton donors than others, and some bases are better proton acceptors than others. If we arrange acids in order of their ability to donate a proton, we find that the more easily a substance gives up a proton, the less easily its conjugate base accepts a proton. Similarly, the more easily a base accepts a proton, the less easily its conjugate acid gives up a proton. In other words, the stronger an acid, the weaker its conjugate base, and the stronger a base, the weaker its conjugate acid. Thus, if we know something about the strength of an acid (its ability to donate protons), we also know something about the strength of its conjugate base (its ability to accept protons).
The inverse relationship between the strengths of acids and their conjugate bases is illustrated in ![]() FIGURE 16.3. Here we have grouped acids and bases into three broad categories based on their behavior in water:
FIGURE 16.3. Here we have grouped acids and bases into three broad categories based on their behavior in water:
1. A strong acid completely transfers its protons to water, leaving no undissociated molecules in solution. ![]() (Section 4.3) Its conjugate base has a negligible tendency to be protonated (to accept protons) in aqueous solution. (The conjugate base of a strong acid shows negligible basicity.)
(Section 4.3) Its conjugate base has a negligible tendency to be protonated (to accept protons) in aqueous solution. (The conjugate base of a strong acid shows negligible basicity.)
2. A weak acid only partially dissociates in aqueous solution and therefore exists in the solution as a mixture of the acid and its conjugate base. The conjugate base of a weak acid shows a slight ability to remove protons from water. (The conjugate base of a weak acid is a weak base.)
3. A substance with negligible acidity contains hydrogen but does not demonstrate any acidic behavior in water. Its conjugate base is a strong base, reacting completely with water, abstracting protons to form OH– ions. (The conjugate base of a substance with negligible acidity is a strong base.)
The ions H+(aq) and OH–(aq) are, respectively, the strongest possible acid and strongest possible base that can exist at equilibrium in aqueous solution. Stronger acids react with water to produce H+(aq) ions, and stronger bases react with water to produce OH–(aq) ions. This effect is known as the leveling effect.
![]() GO FIGURE
GO FIGURE
If O2– ions are added to water, what reaction, if any, occurs?
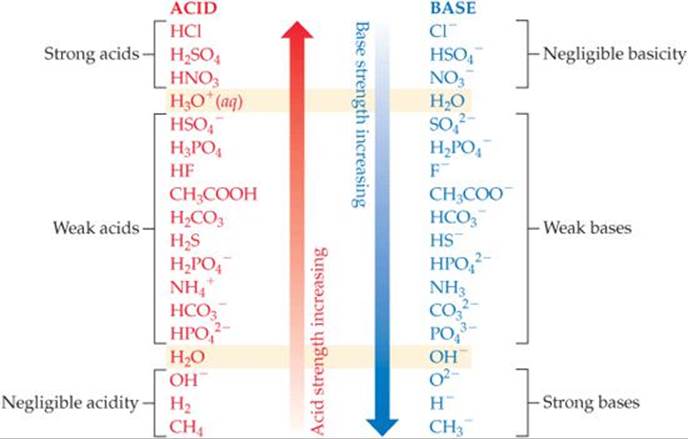
![]() FIGURE 16.3 Relative strengths of select conjugate acid–base pairs. The two members of each pair are listed opposite each other in the two columns.
FIGURE 16.3 Relative strengths of select conjugate acid–base pairs. The two members of each pair are listed opposite each other in the two columns.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Given that HClO4 is a strong acid, how would you classify the basicity of ClO4–?
We can think of proton-transfer reactions as being governed by the relative abilities of two bases to abstract protons. For example, consider the proton transfer that occurs when an acid HX dissolves in water:
![]()
If H2O (the base in the forward reaction) is a stronger base than X– (the conjugate base of HX), H2O abstracts the proton from HX to produce H3O+ and X–. As a result, the equilibrium lies to the right. This describes the behavior of a strong acid in water. For example, when HCl dissolves in water, the solution consists almost entirely of H3O+ and Cl– ions with a negligible concentration of HCl molecules:
![]()
H2O is a stronger base than Cl– (Figure 16.3), so H2O acquires the proton to become the hydronium ion.
When X– is a stronger base than H2O, the equilibrium lies to the left. This situation occurs when HX is a weak acid. For example, an aqueous solution of acetic acid consists mainly of CH3COOH molecules with only a relatively few H3O+ and CH3COO– ions:
![]()
The CH3COO– ion is a stronger base than H2O (Figure 16.3) and therefore abstracts the proton from H3O+.
From these examples, we conclude that in every acid–base reaction, equilibrium favors transfer of the proton from the stronger acid to the stronger base to form the weaker acid and the weaker base.
SAMPLE EXERCISE 16.3 Predicting the Position of a Proton-Transfer Equilibrium
For the following proton-transfer reaction use Figure 16.3 to predict whether the equilibrium lies to the left (Kc < 1) or to the right (Kc > 1):
![]()
SOLUTION
Analyze We are asked to predict whether an equilibrium lies to the right, favoring products, or to the left, favoring reactants.
Plan This is a proton-transfer reaction, and the position of the equilibrium will favor the proton going to the stronger of two bases. The two bases in the equation are CO32–, the base in the forward reaction, and SO42–, the conjugate base of HSO4–. We can find the relative positions of these two bases in Figure 16.3 to determine which is the stronger base.
Solve The CO32– ion appears lower in the right-hand column in Figure 16.3 and is therefore a stronger base than SO42–. CO32–, therefore, will get the proton preferentially to become HCO3–, while SO42– will remain mostly unprotonated. The resulting equilibrium lies to the right, favoring products (that is, Kc > 1):
![]()
Comment Of the two acids HSO4– and HCO3–, the stronger one (HSO4–) gives up a proton more readily, and the weaker one (HCO3–) tends to retain its proton. Thus, the equilibrium favors the direction in which the proton moves from the stronger acid and becomes bonded to the stronger base.
PRACTICE EXERCISE
For each reaction, use Figure 16.3 to predict whether the equilibrium lies to the left or to the right:
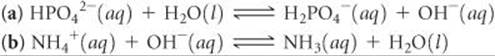
Answers: (a) left, (b) right