CHEMISTRY THE CENTRAL SCIENCE
17 ADDITIONAL ASPECTS OF AQUEOUS EQUILIBRIA

THE GREAT BARRIER REEF stretches for more than 2600 km off of the eastern coast of Australia. Coral reefs such as this are among the most diverse ecosystems on the planet.
WHAT'S AHEAD
17.1 THE COMMON-ION EFFECT
We begin by considering a specific example of Le Châtelier's principle known as the common-ion effect.
17.2 BUFFERED SOLUTIONS
We consider the composition of buffered solutions and learn how they resist pH change when small amounts of a strong acid or strong base are added to them.
17.3 ACID–BASE TITRATIONS
We examine acid–base titrations and explore how to determine pH at any point in an acid–base titration.
17.4 SOLUBILITY EQUILIBRIA
We learn how to use solubility-product constants to determine to what extent a sparingly soluble salt dissolves in water.
17.5 FACTORS THAT AFFECT SOLUBILITY
We investigate some of the factors that affect solubility, including the common-ion effect and the effect of acids.
17.6 PRECIPITATION AND SEPARATION OF IONS
We learn how differences in solubility can be used to separate ions through selective precipitation.
17.7 QUALITATIVE ANALYSIS FOR METALLIC ELEMENTS
We explain how the principles of solubility and complexation equilibria can be used to identify ions in solution.
WATER, THE MOST COMMON AND most important solvent on Earth, occupies its position of importance because of its abundance and its exceptional ability to dissolve a wide variety of substances. Coral reefs are a striking example of aqueous chemistry at work in nature. Coral reefs are built by tiny animals called stony corals, which secrete a hard calcium carbonate exoskeleton. Over time, the stony corals build up large networks of calcium carbonate upon which a reef is built. The size of such structures can be immense, as illustrated by the Great Barrier Reef.
Stony corals make their exoskeletons from dissolved Ca2+ and CO32– ions. This process is aided by the fact that the CO32– concentration is supersaturated in most parts of the ocean. However, well-documented increases in the amount of CO2 in the atmosphere threaten to upset the aqueous chemistry that stony corals depend on. As atmospheric CO2 levels increase, the amount of CO2 dissolved in the ocean also increases. This lowers the pH of the ocean and leads to a decrease in the CO32– concentration. As a result it becomes more difficult for stony corals and other important ocean creatures to maintain their exoskeletons. We will take a closer look at the consequences of ocean acidification later in the chapter.
To understand the chemistry that underlies coral reef formation, we must understand the concepts of aqueous equilibria. In this chapter we take a step toward understanding such complex solutions by looking first at further applications of acid–base equilibria. The idea is to consider not only solutions in which there is a single solute but also those containing a mixture of solutes. We then broaden our discussion to include two additional types of aqueous equilibria: those involving slightly soluble salts and those involving the formation of metal complexes in solution. For the most part, the discussions and calculations in this chapter are an extension of those in Chapters 15 and 16.
17.1 THE COMMON-ION EFFECT
In Chapter 16 we examined the equilibrium concentrations of ions in solutions containing a weak acid or a weak base. We now consider solutions that contain a weak acid, such as acetic acid (CH3COOH), and a soluble salt of that acid, such as sodium acetate (CH3COONa). Notice that these solutions contain two substances that share a common ion, CH3COO–. It is instructive to view these solutions from the perspective of Le Châtelier's principle. ![]() (Section 15.7)
(Section 15.7)
Sodium acetate is a soluble ionic compound and therefore a strong electrolyte. ![]() (Section 4.1) Consequently, it dissociates completely in aqueous solution to form Na+ and CH3COO– ions:
(Section 4.1) Consequently, it dissociates completely in aqueous solution to form Na+ and CH3COO– ions:
![]()
In contrast, CH3COOH is a weak electrolyte that ionizes only partially, represented by the equilibrium
![]()
When we have sodium acetate and acetic acid in the same solution, the CH3COO– from CH3COONa causes the equilibrium of Equation 17.1 to shift to the left, thereby decreasing the equilibrium concentration of H+(aq):

In other words, the presence of the added acetate ion causes the acetic acid to ionize less than it normally would.
Whenever a weak electrolyte and a strong electrolyte containing a common ion are together in solution, the weak electrolyte ionizes less than it would if it were alone in solution. We call this observation the common-ion effect.
SAMPLE EXERCISE 17.1 Calculating the pH When a Common Ion Is Involved
What is the pH of a solution made by adding 0.30 mol of acetic acid and 0.30 mol of sodium acetate to enough water to make 1.0 L of solution?
SOLUTION
Analyze We are asked to determine the pH of a solution of a weak electrolyte (CH3COOH) and a strong electrolyte (CH3COONa) that share a common ion, CH3COO–.
Plan In any problem in which we must determine the pH of a solution containing a mixture of solutes, it is helpful to proceed by a series of logical steps:
1. Consider which solutes are strong electrolytes and which are weak electrolytes, and identify the major species in solution.
2. Identify the important equilibrium that is the source of H+ and therefore determines pH.
3. Tabulate the concentrations of ions involved in the equilibrium.
4. Use the equilibrium-constant expression to calculate [H+] and then pH.
Solve First, because CH3COOH is a weak electrolyte and CH3COONa is a strong electrolyte, the major species in the solution are CH3COOH (a weak acid), Na+ (which is neither acidic nor basic and is therefore a spectator in the acid–base chemistry), and CH3COO– (which is the conjugate base of CH3COOH).
Second, [H+] and, therefore, the pH are controlled by the dissociation equilibrium of CH3COOH:
![]()
[We have written the equilibrium using H+(aq) rather than H3O+(aq), but both representations of the hydrated hydrogen ion are equally valid.]
Third, we tabulate the initial and equilibrium concentrations as we did in solving other equilibrium problems in Chapters 15 and 16:

The equilibrium concentration of CH3COO– (the common ion) is the initial concentration that is due to CH3COONa (0.30 M) plus the change in concentration (x) that is due to the ionization of CH3COOH.
Now we can use the equilibrium-constant expression:
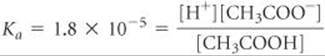
The dissociation constant for CH3COOH at 25 °C is from Appendix D; addition of CH3COONa does not change the value of this constant. Substituting the equilibrium-constant concentrations from our table into the equilibrium expression gives:
![]()
Because Ka is small, we assume that x is small compared to the original concentrations of CH3COOH and CH3COO– (0.30 M each). Thus, we can ignore the very small x relative to 0.30 M, giving:
![]()
The resulting value of x is indeed small relative to 0.30, justifying the approximation made in simplifying the problem.
![]()
Finally, we calculate the pH from the equilibrium concentration of H+(aq):
![]()
Comment In Section 16.6 we calculated that a 0.30 M solution of CH3COOH has a pH of 2.64, corresponding to [H+] = 2.3 × 10–3M. Thus, the addition of CH3COONa has substantially decreased [H+], as we expect from Le Châtelier's principle.
PRACTICE EXERCISE
Calculate the pH of a solution containing 0.085 M nitrous acid (HNO2, Ka = 4.5 × 10–4) and 0.10 M potassium nitrite (KNO2).
Answer: 3.42
SAMPLE EXERCISE 17.2 Calculating Ion Concentrations When a Common Ion Is Involved
Calculate the fluoride ion concentration and pH of a solution that is 0.20 M in HF and 0.10 M in HCl.
SOLUTION
Analyze We are asked to determine the concentration of F– and the pH in a solution containing the weak acid HF and the strong acid HCl. In this case the common ion is H+.
Plan We can again use the four steps outlined in Sample Exercise 17.1.
Solve Because HF is a weak acid and HCl is a strong acid, the major species in solution are HF, H+, and Cl–. The Cl–, which is the conjugate base of a strong acid, is merely a spectator ion in any acid–base chemistry. The problem asks for [F–], which is formed by ionization of HF. Thus, the important equilibrium is
![]()
The common ion in this problem is the hydrogen (or hydronium) ion. Now we can tabulate the initial and equilibrium concentrations of each species involved in this equilibrium:

The equilibrium constant for the ionization of HF, from Appendix D, is 6.8 × 10–4. Substituting the equilibrium-constant concentrations into the equilibrium expression gives
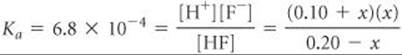
If we assume that x is small relative to 0.10 or 0.20 M, this expression simplifies to
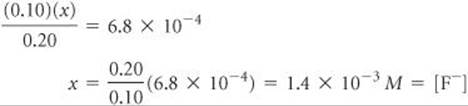
This F– concentration is substantially smaller than it would be in a 0.20 M solution of HF with no added HCl. The common ion, H+, suppresses the ionization of HF. The concentration of H+(aq) is
![]()
Thus,
![]()
Comment Notice that for all practical purposes, the hydrogen ion concentration is due entirely to the HCl; the HF makes a negligible contribution by comparison.
PRACTICE EXERCISE
Calculate the formate ion concentration and pH of a solution that is 0.050 M in formic acid (HCOOH, Ka = 1.8 × 10–4) and 0.10 M in HNO3.
Answer: [HCOO–] = 9.0 × 10–5, pH = 1.00
Sample Exercises 17.1 and 17.2 both involve weak acids. The ionization of a weak base is also decreased by the addition of a common ion. For example, the addition of NH4+ (as from the strong electrolyte NH4Cl) causes the base-dissociation equilibrium of NH3 to shift to the left, decreasing the equilibrium concentration of OH– and lowering the pH:

![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
If solutions of NH4Cl(aq) and NH3(aq) are mixed, which ions in the resulting solution are spectator ions in any acid–base chemistry occurring in the solution? What equilibrium reaction determines [OH–] and, therefore, the pH of the solution?