CHEMISTRY THE CENTRAL SCIENCE
17 ADDITIONAL ASPECTS OF AQUEOUS EQUILIBRIA
CHAPTER SUMMARY AND KEY TERMS
SECTION 17.1 In this chapter we have considered several types of important equilibria that occur in aqueous solution. Our primary emphasis has been on acid–base equilibria in solutions containing two or more solutes and on solubility equilibria. The dissociation of a weak acid or weak base is repressed by the presence of a strong electrolyte that provides an ion common to the equilibrium. This phenomenon is called the common-ion effect.
SECTION 17.2 A particularly important type of acid–base mixture is that of a weak conjugate acid–base pair. Such mixtures function as buffered solutions (buffers). Addition of small amounts of a strong acid or a strong base to a buffered solution causes only small changes in pH because the buffer reacts with the added acid or base. (Strong acid–strong base, strong acid–weak base, and weak acid–strong base reactions proceed essentially to completion.) Buffered solutions are usually prepared from a weak acid and a salt of that acid or from a weak base and a salt of that base. Two important characteristics of a buffered solution are its buffer capacity and its pH range. The optimal pH of a buffer is equal to pKa (or pKb) of the acid (or base) used to prepare the buffer. The relationship between pH, pKa, and the concentrations of an acid and its conjugate base can be expressed by the Henderson–Hasselbalch equation.
SECTION 17.3 The plot of the pH of an acid (or base) as a function of the volume of added base (or acid) is called a pH titration curve. The titration curve of a strong acid–strong base titration exhibits a large change in pH in the immediate vicinity of the equivalence point; at the equivalence point for such a titration pH = 7. For strong acid–weak base or weak acid–strong base titrations, the pH change in the vicinity of the equivalence point is not as large. Furthermore, the pH at the equivalence point is not 7 in either of these cases. Rather, it is the pH of the salt solution that results from the neutralization reaction. For this reason it is important to choose an indicator whose color change is near the pH at the equivalence point for titrations involving either weak acids or weak bases. It is possible to calculate the pH at any point of the titration curve by first considering the effects of the acid–base reaction on solution concentrations and then examining equilibria involving the remaining solute species.
SECTION 17.4 The equilibrium between a solid compound and its ions in solution provides an example of heterogeneous equilibrium. The solubility-product constant (or simply the solubility product), Ksp, is an equilibrium constant that expresses quantitatively the extent to which the compound dissolves. The Ksp can be used to calculate the solubility of an ionic compound, and the solubility can be used to calculate Ksp.
SECTION 17.5 Several experimental factors, including temperature, affect the solubilities of ionic compounds in water. The solubility of a slightly soluble ionic compound is decreased by the presence of a second solute that furnishes a common ion (the common-ion effect). The solubility of compounds containing basic anions increases as the solution is made more acidic (as pH decreases). Salts with anions of negligible basicity (the anions of strong acids) are unaffected by pH changes.
The solubility of metal salts is also affected by the presence of certain Lewis bases that react with metal ions to form stable complex ions. Complex-ion formation in aqueous solution involves the displacement by Lewis bases (such as NH3 and CN–) of water molecules attached to the metal ion. The extent to which such complex formation occurs is expressed quantitatively by the formation constant for the complex ion. Amphoteric oxides and hydroxides are those that are only slightly soluble in water but dissolve on addition of either acid or base.
SECTION 17.6 Comparison of the ion product, Q, with the value of Ksp can be used to judge whether a precipitate will form when solutions are mixed or whether a slightly soluble salt will dissolve under various conditions. Precipitates form when Q > Ksp. If two salts have sufficiently different solubilities, selective precipitation can be used to precipitate one ion while leaving the other in solution, effectively separating the two ions.
SECTION 17.7 Metallic elements vary a great deal in the solubilities of their salts, in their acid–base behavior, and in their tendencies to form complex ions. These differences can be used to separate and detect the presence of metal ions in mixtures. Qualitative analysis determines the presence or absence of species in a sample, whereas quantitative analysis determines how much of each species is present. The qualitative analysis of metal ions in solution can be carried out by separating the ions into groups on the basis of precipitation reactions and then analyzing each group for individual metal ions.
KEY SKILLS
• Describe the common-ion effect. (Section 17.1)
• Explain how a buffer functions. (Section 17.2)
• Calculate the pH of a buffered solution. (Section 17.2)
• Calculate the pH of a buffer after the addition of small amounts of a strong acid or a strong base. (Section 17.2)
• Calculate the pH at any point in a strong acid–strong base titration. (Section 17.3)
• Calculate the pH at any point in a weak acid–strong base or weak base-strong acid titration. (Section 17.3)
• Understand the differences between the titration curves for a strong acid–strong base titration and those when either the acid or base is weak. (Section 17.3)
• Given either Ksp, molar solubility or mass solubility for a substance calculate the other two quantities. (Section 17.4)
• Calculate molar solubility in the presence of a common ion. (Section 17.5)
• Predict the effect of pH on solubility. (Section 17.5)
• Predict whether a precipitate will form when solutions are mixed by comparing Q and Ksp. (Section 17.6)
• Calculate the ion concentrations required to begin precipitation. (Section 17.6)
• Explain the effect of complex-ion formation on solubility. (Section 17.6)
KEY EQUATIONS
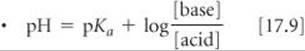
The Henderson–Hasselbalch equation, used to calculate the pH of a buffer from the concentrations of a conjugate acid–base pair