CHEMISTRY THE CENTRAL SCIENCE
18 CHEMISTRY OF THE ENVIRONMENT
18.2 HUMAN ACTIVITIES AND EARTH'S ATMOSPHERE
Both natural and anthropogenic (human-caused) events can modify Earth's atmosphere. One impressive natural event was the eruption of Mount Pinatubo in June 1991 (![]() FIGURE 18.5). The volcano ejected approximately 10 km3 of material into the stratosphere, causing a 10% drop in the amount of sunlight reaching Earth's surface during the next 2 years. That drop in sunlight led to a temporary 0.5 °C drop in Earth's surface temperature. The volcanic particles that made it to the stratosphere remained there for approximately 3 years, raising the temperature of the stratosphere by several degrees due to light absorption. Measurements of the stratospheric ozone concentration showed significantly increased ozone decomposition in this 3-year period.
FIGURE 18.5). The volcano ejected approximately 10 km3 of material into the stratosphere, causing a 10% drop in the amount of sunlight reaching Earth's surface during the next 2 years. That drop in sunlight led to a temporary 0.5 °C drop in Earth's surface temperature. The volcanic particles that made it to the stratosphere remained there for approximately 3 years, raising the temperature of the stratosphere by several degrees due to light absorption. Measurements of the stratospheric ozone concentration showed significantly increased ozone decomposition in this 3-year period.

![]() FIGURE 18.5 Mount Pinatubo erupts, June 1991.
FIGURE 18.5 Mount Pinatubo erupts, June 1991.
Eruption of the Icelandic volcano Eyjafjallajökull in 2010, though not as large as the Pinatubo eruption, has similarly affected the atmosphere over large regions of the Northern Hemisphere.
The Ozone Layer and Its Depletion
The ozone layer protects Earth's surface from damaging ultraviolet (UV) radiation. Therefore, if the concentration of ozone in the stratosphere decreases substantially, more UV radiation will reach Earth's surface, causing unwanted photochemical reactions, including reactions correlated with skin cancer. Satellite monitoring of ozone, which began in 1978, has revealed a depletion of ozone in the stratosphere that is particularly severe over Antarctica, a phenomenon known as the ozone hole (![]() FIGURE 18.6). The first scientific paper on this phenomenon appeared in 1985, and the National Aeronautics and Space Administration (NASA) maintains an “Ozone Hole Watch” website with daily updates and data from 1999 to the present.
FIGURE 18.6). The first scientific paper on this phenomenon appeared in 1985, and the National Aeronautics and Space Administration (NASA) maintains an “Ozone Hole Watch” website with daily updates and data from 1999 to the present.
In 1995 the Nobel Prize in Chemistry was awarded to F. Sherwood Rowland, Mario Molina, and Paul Crutzen for their studies of ozone depletion. In 1970 Crutzen showed that naturally occurring nitrogen oxides catalytically destroy ozone. Rowland and Molina recognized in 1974 that chlorine from chlorofluorocarbons (CFCs) may deplete the ozone layer. These substances, principally CFCl3 and CF2Cl2, do not occur in nature and have been widely used as propellants in spray cans, as refrigerant and air-conditioner gases, and as foaming agents for plastics. They are virtually unreactive in the lower atmosphere. Furthermore, they are relatively insoluble in water and are therefore not removed from the atmosphere by rainfall or by dissolution in the oceans. Unfortunately, the lack of reactivity that makes them commercially useful also allows them to survive in the atmosphere and to diffuse into the stratosphere. It is estimated that several million tons of chlorofluorocarbons are now present in the atmosphere.
As CFCs diffuse into the stratosphere, they are exposed to high-energy radiation, which can cause photodissociation. Because C—Cl bonds are considerably weaker than C—F bonds, free chlorine atoms are formed readily in the presence of light with wavelengths in the range from 190 to 225 nm, as shown in this typical reaction:
![]()
Calculations suggest that chlorine atom formation occurs at the greatest rate at an altitude of about 30 km, the altitude at which ozone is at its highest concentration.
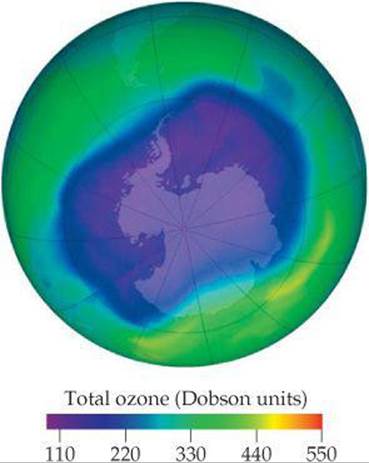
![]() FIGURE 18.6 Ozone present in the Southern Hemisphere, Sept. 24, 2006. This data was taken from an orbiting satellite. This day had the lowest stratospheric ozone concentration yet recorded. One “Dobson unit” corresponds to 2.69 × 1016 ozone molecules in a 1 cm2 column of atmosphere.
FIGURE 18.6 Ozone present in the Southern Hemisphere, Sept. 24, 2006. This data was taken from an orbiting satellite. This day had the lowest stratospheric ozone concentration yet recorded. One “Dobson unit” corresponds to 2.69 × 1016 ozone molecules in a 1 cm2 column of atmosphere.
Atomic chlorine reacts rapidly with ozone to form chlorine monoxide and molecular oxygen:
![]()
This reaction follows a second-order rate law with a very large rate constant:
![]()
Under certain conditions, the ClO generated in Equation 18.7 can react to regenerate free Cl atoms. One way that this can happen is by photodissociation of ClO:
![]()
The Cl atoms generated in Equations 18.6 and 18.9 can react with more O3, according to Equation 18.7. The result is a sequence of reactions that accomplishes the Cl-catalyzed decomposition of O3 to O2:

The equation can be simplified by eliminating like species from each side to give
![]()
Because the rate of Equation 18.7 increases linearly with [Cl], the rate at which ozone is destroyed increases as the quantity of Cl atoms increases. Thus, the greater the amount of CFCs that diffuse into the stratosphere, the faster the destruction of the ozone layer. Even though troposphere-to-stratosphere diffusion rates are slow, a thinning of the ozone layer over the South Pole has been observed, particularly during September and October (Figure 18.6).
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Since the rate of ozone destruction depends on [Cl], can Cl be considered a catalyst for the reaction of Equation 18.10?
Because of the environmental problems associated with CFCs, steps have been taken to limit their manufacture and use. A major step was the signing in 1987 of the Montreal Protocol on Substances That Deplete the Ozone Layer, in which participating nations agreed to reduce CFC production. More stringent limits were set in 1992, when representatives of approximately 100 nations agreed to ban the production and use of CFCs by 1996, with some exceptions for “essential uses.” Since then, the production of CFCs has indeed dropped precipitously, and the size of the ozone hole has leveled off. Nevertheless, because CFCs are unreactive and because they diffuse so slowly into the stratosphere, scientists estimate that ozone depletion will continue for many years to come. What substances have replaced CFCs? At this time, the main alternatives are hydrofluorocarbons, compounds in which C—H bonds replace the C—Cl bonds of CFCs. One such compound in current use is CH2FCF3, known as HFC-134a.
There are no naturally occurring CFCs, but some natural sources contribute chlorine and bromine to the atmosphere, and, just like halogens from CFC, these naturally occurring Cl and Br atoms can participate in ozone-depleting reactions. The principal natural sources are methyl bromide and methyl chloride, which are emitted from the oceans. It is estimated that these molecules contribute less than a third of the total Cl and Br in the atmosphere; the remaining two-thirds is a result of human activities.
Volcanoes are a source of HCl, but generally the HCl they release reacts with water in the troposphere and does not make it to the upper atmosphere.
Sulfur Compounds and Acid Rain
Sulfur-containing compounds are present to some extent in the natural, unpolluted atmosphere. They originate in the bacterial decay of organic matter, in volcanic gases, and from other sources listed in Table 18.2. The amount of these compounds released into the atmosphere worldwide from natural sources is about 24 × 1012 g per year, less than the amount from human activities, about 79 × 1012 g per year (principally related to combustion of fuels).
TABLE 18.4 • Median Concentrations of Atmospheric Pollutants in a Typical Urban Atmosphere
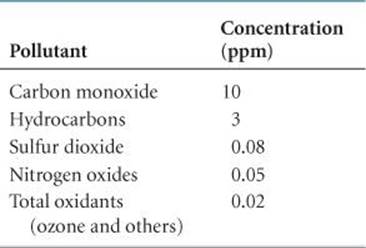
Sulfur compounds, chiefly sulfur dioxide, SO2, are among the most unpleasant and harmful of the common pollutant gases. ![]() TABLE 18.4 shows the concentrations of several pollutant gases in a typical urban environment (where by typical we mean one that is not particularly affected by smog). According to these data, the level of sulfur dioxide is 0.08 ppm or higher about half the time. This concentration is considerably lower than that of other pollutants, notably carbon monoxide. Nevertheless, SO2 is regarded as the most serious health hazard among the pollutants shown, especially for people with respiratory difficulties.
TABLE 18.4 shows the concentrations of several pollutant gases in a typical urban environment (where by typical we mean one that is not particularly affected by smog). According to these data, the level of sulfur dioxide is 0.08 ppm or higher about half the time. This concentration is considerably lower than that of other pollutants, notably carbon monoxide. Nevertheless, SO2 is regarded as the most serious health hazard among the pollutants shown, especially for people with respiratory difficulties.
Combustion of coal accounts for about 65% of the SO2 released annually in the United States, and combustion of oil accounts for another 20%. The majority of this amount is from coal-burning electrical power plants, which generate about 50% of our electricity. The extent to which SO2 emissions are a problem when coal is burned depends on the amount of sulfur in the coal. Because of concern about SO2 pollution, low-sulfur coal is in greater demand and is thus more expensive. Much of the coal from east of the Mississippi is relatively high in sulfur content, up to 6% by mass. Much of the coal from the western states has a lower sulfur content, but also a lower heat content per unit mass, so the difference in sulfur content per unit of heat produced is not as large as is often assumed.
China, which gets 70 percent of its energy from coal, is the world's largest generator of SO2, producing about 22 million tons annually. As a result, that nation has a major problem with SO2 pollution and has set targets to reduce emissions with some success. In 2010, the U.S. Environmental Protection Agency set new standards to reduce SO2 emissions, the first change in nearly 40 years. The old standard of 140 parts per billion, measured over 24 hours, will be replaced by a standard of 75 parts per billion, measured over 1 hour.
Sulfur dioxide is harmful to both human health and property; furthermore, atmospheric SO2 can be oxidized to SO3 by several pathways (such as reaction with O2 or O3). When SO3 dissolves in water, it produces sulfuric acid:
![]()
Many of the environmental effects ascribed to SO2 are actually due to H2SO4.
The presence of SO2 in the atmosphere and the sulfuric acid it produces result in the phenomenon of acid rain. (Nitrogen oxides, which form nitric acid, are also major contributors to acid rain.) Uncontaminated rainwater generally has a pH value of about 5.6. The primary source of this natural acidity is CO2, which reacts with water to form carbonic acid, H2CO3. Acid rain typically has a pH value of about 4. This acidity has affected many lakes in northern Europe, the northern United States, and Canada, reducing fish populations and affecting other parts of the ecological network in the lakes and surrounding forests.
The pH of most natural waters containing living organisms is between 6.5 and 8.5, but as ![]() FIGURE 18.7 shows, freshwater pH values are far below 6.5 in many parts of the continental United States. At pH levels below 4.0, all vertebrates, most invertebrates, and many microorganisms are destroyed. The lakes most susceptible to damage are those with low concentrations of basic ions, such as HCO3–, that would act as a buffer to minimize changes in pH. Some of these lakes are recovering as sulfur emissions from fossil fuel combustion decrease, in part because of the Clean Air Act, which has resulted in a reduction of more than 40% in SO2 emissions from power plants since 1980.
FIGURE 18.7 shows, freshwater pH values are far below 6.5 in many parts of the continental United States. At pH levels below 4.0, all vertebrates, most invertebrates, and many microorganisms are destroyed. The lakes most susceptible to damage are those with low concentrations of basic ions, such as HCO3–, that would act as a buffer to minimize changes in pH. Some of these lakes are recovering as sulfur emissions from fossil fuel combustion decrease, in part because of the Clean Air Act, which has resulted in a reduction of more than 40% in SO2 emissions from power plants since 1980.
Because acids react with metals and with carbonates, acid rain is corrosive both to metals and to stone building materials. Marble and limestone, for example, whose major constituent is CaCO3, are readily attacked by acid rain (![]() FIGURE 18.8). Billions of dollars each year are lost because of corrosion due to SO2 pollution.
FIGURE 18.8). Billions of dollars each year are lost because of corrosion due to SO2 pollution.
One way to reduce the quantity of SO2 released into the environment is to remove sulfur from coal and oil before these fuels are burned. Although difficult and expensive, several methods have been developed. Powdered limestone (CaCO3), for example, can be injected into the furnace of a power plant, where it decomposes into lime (CaO) and carbon dioxide:
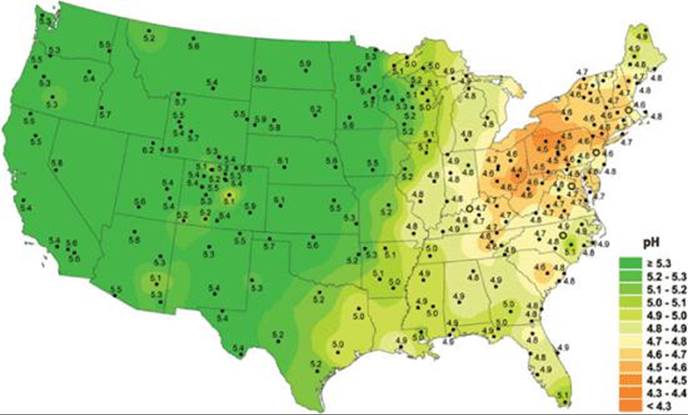
![]() FIGURE 18.7 Water pH values from freshwater sites across the United States, 2008. The numbered dots indicate the locations of monitoring stations.
FIGURE 18.7 Water pH values from freshwater sites across the United States, 2008. The numbered dots indicate the locations of monitoring stations.

![]() FIGURE 18.8 Damage from acid rain. The right photograph, recently taken, shows how the statue has lost detail in its carvings.
FIGURE 18.8 Damage from acid rain. The right photograph, recently taken, shows how the statue has lost detail in its carvings.
![]()
The CaO then reacts with SO2 to form calcium sulfite:
![]()
The solid particles of CaSO3, as well as much of the unreacted SO2, can be removed from the furnace gas by passing it through an aqueous suspension of CaO (![]() FIGURE 18.9). Not all the SO2 is removed, however, and given the enormous quantities of coal and oil burned worldwide, pollution by SO2 will probably remain a problem for some time.
FIGURE 18.9). Not all the SO2 is removed, however, and given the enormous quantities of coal and oil burned worldwide, pollution by SO2 will probably remain a problem for some time.
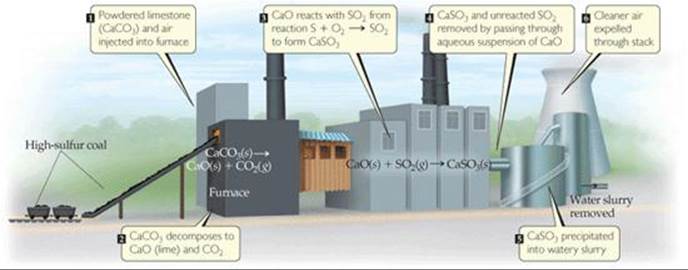
![]() FIGURE 18.9 One method for removing SO2 from combusted fuel.
FIGURE 18.9 One method for removing SO2 from combusted fuel.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What chemical behavior associated with sulfur oxides gives rise to acid rain?
Nitrogen Oxides and Photochemical Smog
Nitrogen oxides are primary components of smog, a phenomenon with which city dwellers are all too familiar. The term smog refers to the pollution condition that occurs in certain urban environments when weather conditions produce a relatively stagnant air mass. The smog made famous by Los Angeles, but now common in many other urban areas as well, is more accurately described as photochemical smog because photochemical processes play a major role in its formation (![]() FIGURE 18.10).
FIGURE 18.10).

![]() FIGURE 18.10 Photochemical smog is produced largely by the action of sunlight on vehicle exhaust gases.
FIGURE 18.10 Photochemical smog is produced largely by the action of sunlight on vehicle exhaust gases.
The majority of nitrogen oxide emissions (about 50%) comes from cars, buses, and other forms of transportation. Nitric oxide, NO, forms in small quantities in the cylinders of internal combustion engines in the reaction
![]()
As noted in the “Chemistry Put to Work” box in Section 15.7, the equilibrium constant for this reaction increases from about 10–15 at 300 K to about 0.05 at 2400 K (approximate temperature in the cylinder of an engine during combustion). Thus, the reaction is more favorable at higher temperatures. In fact, some NO is formed in any high-temperature combustion. As a result, electrical power plants are also major contributors to nitrogen oxide pollution.
Before the installation of pollution-control devices on automobiles, typical emission levels of NOx were 4 grams per mile. (The x is either 1 or 2 because both NO and NO2 are formed, although NO predominates.) Starting in 2004, the auto emission standards for NOx called for a phased-in reduction to 0.07 g/mi by 2009, which was achieved.
In air, nitric oxide is rapidly oxidized to nitrogen dioxide:
![]()
The equilibrium constant for this reaction decreases from about 1012 at 300 K to about 10–5 at 2400 K.
The photodissociation of NO2 initiates the reactions associated with photochemical smog. Dissociation of NO2 requires 304 kJ/mol, which corresponds to a photon wavelength of 393 nm. In sunlight, therefore, NO2 dissociates to NO and O:
![]()
The atomic oxygen formed undergoes several reactions, one of which gives ozone, as described earlier:
![]()
Although it is an essential UV screen in the upper atmosphere, ozone is an undesirable pollutant in the troposphere. It is extremely reactive and toxic, and breathing air that contains appreciable amounts of ozone can be especially dangerous for asthma sufferers, exercisers, and the elderly. We therefore have two ozone problems: excessive amounts in many urban environments, where it is harmful, and depletion in the stratosphere, where its presence is vital.
In addition to nitrogen oxides and carbon monoxide, an automobile engine also emits unburned hydrocarbons as pollutants. These organic compounds are the principal components of gasoline and of many compounds we use as fuel (propane, C3H8, and butane, C4H10; for example), but are major ingredients of smog. A typical engine without effective emission controls emits about 10 to 15 g of hydrocarbons per mile. Current standards require that hydrocarbon emissions be less than 0.075 g per mile. Hydrocarbons are also emitted naturally from living organisms (see “A Closer Look” box later in this section).
Reduction or elimination of smog requires that the ingredients essential to its formation be removed from automobile exhaust. Catalytic converters reduce the levels of NOx and hydrocarbons, two of the major ingredients of smog. (See the “Chemistry Put to Work: Catalytic Converters” in Section 14.7.)
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
What photochemical reaction involving nitrogen oxides initiates the formation of photochemical smog?
Greenhouse Gases: Water Vapor, Carbon Dioxide, and Climate
In addition to screening out harmful short-wavelength radiation, the atmosphere is essential in maintaining a reasonably uniform and moderate temperature on Earth's surface. Earth is in overall thermal balance with its surroundings. This means that the planet radiates energy into space at a rate equal to the rate at which it absorbs energy from the Sun. ![]() FIGURE 18.11 shows the distribution of radiation to and from Earth's surface, and
FIGURE 18.11 shows the distribution of radiation to and from Earth's surface, and ![]() FIGURE 18.12 shows which portion of the infrared radiation leaving the surface is absorbed by atmospheric water vapor and carbon dioxide. In absorbing this radiation, these two atmospheric gases help maintain a livable uniform temperature at the surface by holding in, as it were, the infrared radiation, which we feel as heat.
FIGURE 18.12 shows which portion of the infrared radiation leaving the surface is absorbed by atmospheric water vapor and carbon dioxide. In absorbing this radiation, these two atmospheric gases help maintain a livable uniform temperature at the surface by holding in, as it were, the infrared radiation, which we feel as heat.
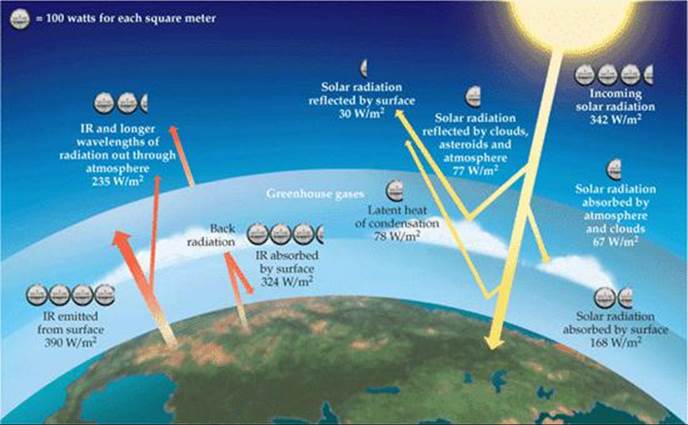
![]() FIGURE 18.11 Earth's thermal balance. The amount of radiation reaching the surface of the planet is approximately equal to the amount radiated back into space.
FIGURE 18.11 Earth's thermal balance. The amount of radiation reaching the surface of the planet is approximately equal to the amount radiated back into space.
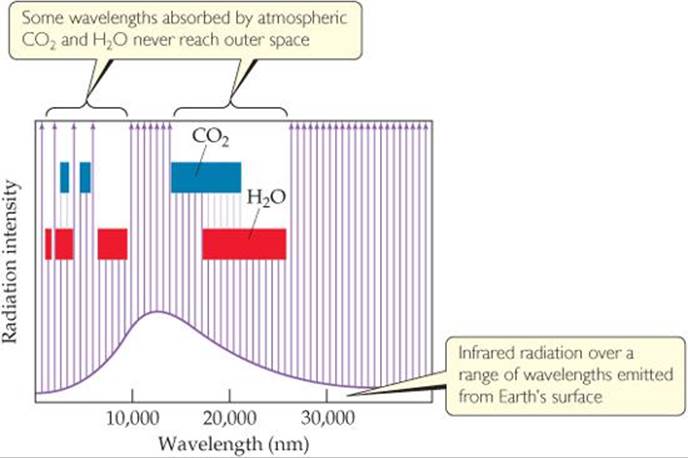
![]() FIGURE 18.12 Portions of the infrared radiation emitted by Earth's surface that are absorbed by atmospheric CO2 and H2O.
FIGURE 18.12 Portions of the infrared radiation emitted by Earth's surface that are absorbed by atmospheric CO2 and H2O.
The influence of H2O, CO2, and certain other atmospheric gases on Earth's temperature is called the greenhouse effect because in trapping infrared radiation these gases act much like the glass of a greenhouse. The gases themselves are called greenhouse gases.
Water vapor makes the largest contribution to the greenhouse effect. The partial pressure of water vapor in the atmosphere varies greatly from place to place and time to time but is generally highest near Earth's surface and drops off with increasing elevation. Because water vapor absorbs infrared radiation so strongly, it plays the major role in maintaining the atmospheric temperature at night, when the surface is emitting radiation into space and not receiving energy from the Sun. In very dry desert climates, where the water-vapor concentration is low, it may be extremely hot during the day but very cold at night. In the absence of a layer of water vapor to absorb and then radiate part of the infrared radiation back to Earth, the surface loses this radiation into space and cools off very rapidly.
Carbon dioxide plays a secondary but very important role in maintaining the surface temperature. The worldwide combustion of fossil fuels, principally coal and oil, on a prodigious scale in the modern era has sharply increased carbon dioxide levels in the atmosphere. To get a sense of the amount of CO2 produced—for example, by the combustion of hydrocarbons and other carbon-containing substances, which are the components of fossil fuels—consider the combustion of butane, C4H10. Combustion of 1.00 g of C4H10 produces 3.03 g of CO2. ![]() (Section 3.6) Similarly, a gallon (3.78 L) of gasoline (density 0.7 g/mL, approximate composition C8H18) produces about 8 kg of CO2. Combustion of fossil fuels releases about 2.2 × 1016 g (22 billion tons) of CO2 into the atmosphere annually, with the largest quantity coming from transportation vehicles.
(Section 3.6) Similarly, a gallon (3.78 L) of gasoline (density 0.7 g/mL, approximate composition C8H18) produces about 8 kg of CO2. Combustion of fossil fuels releases about 2.2 × 1016 g (22 billion tons) of CO2 into the atmosphere annually, with the largest quantity coming from transportation vehicles.
Much CO2 is absorbed into oceans or used by plants. Nevertheless, we are now generating CO2 much faster than it is being absorbed or used. Analysis of air trapped in ice cores taken from Antarctica and Greenland makes it possible to determine the atmospheric levels of CO2 during the past 160,000 years. These measurements reveal that the level of CO2 remained fairly constant from the last Ice Age, some 10,000 years ago, until roughly the beginning of the Industrial Revolution, about 300 years ago. Since that time, the concentration of CO2 has increased by about 30% to a current high of about 386 ppm (![]() FIGURE 18.13).
FIGURE 18.13).
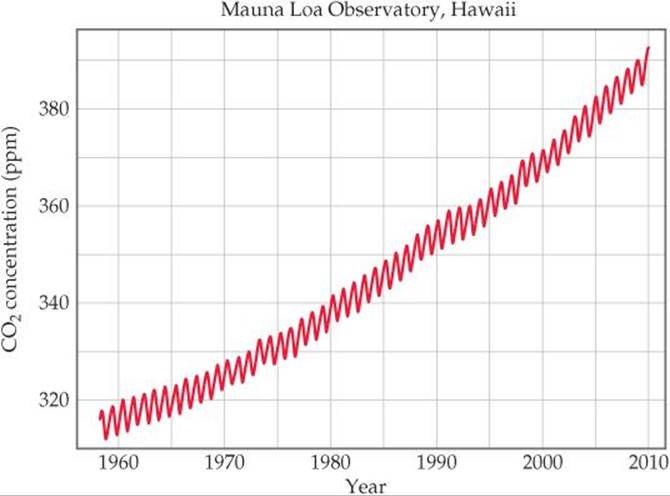
![]() FIGURE 18.13 Rising CO2 levels. The sawtooth shape of the graph is due to regular seasonal variations in CO2 concentration for each year.
FIGURE 18.13 Rising CO2 levels. The sawtooth shape of the graph is due to regular seasonal variations in CO2 concentration for each year.
A consensus is emerging among scientists that this increase in atmospheric CO2 is perturbing Earth's climate and may be responsible for the observed increase in the average global air temperature of 0.3 °C to 0.6 °C over the past century. Scientists often use the term climate changeinstead of global warming to refer to this effect because as the Earth's temperature increases, it affects winds and ocean currents in ways that can cool some areas and warm others.
On the basis of present and expected future rates of fossil-fuel use, the atmospheric CO2 level is expected to double from its present level sometime between 2050 and 2100. Computer models predict that this increase will result in an average global temperature increase of 1 °C to 3 °C. Because so many factors go into determining climate, we cannot predict with certainty what changes will occur because of this warming. Clearly, however, humanity has acquired the potential, by changing the concentrations of CO2 and other heat-trapping gases in the atmosphere, to substantially alter the climate of the planet.
The climate change threat posed by atmospheric CO2 has sparked considerable research into ways of capturing the gas at its largest combustion sources and storing it under ground or under the seafloor. There is also much interest in developing new ways to use CO2 as a chemical feedstock. However, the approximately 115 million tons of CO2 used annually by the global chemical industry is but a small fraction of the approximately 24 billion tons of annual CO2 emissions. The use of CO2 as a raw material will probably never be great enough to reduce its atmospheric concentration.
![]() GIVE IT SOME THOUGHT
GIVE IT SOME THOUGHT
Explain why nighttime temperatures remain higher in locations where there is higher humidity.
 A CLOSER LOOK
A CLOSER LOOK
OTHER GREENHOUSE GASES
Although CO2 receives most of the attention, other gases contribute to the greenhouse effect, including methane, CH4, hydrofluorocarbons (HFCs), and chlorofluorocarbons (CFCs).
HFCs have replaced CFCs in a host of applications, including refrigerants and air-conditioner gases. Although they do not contribute to the depletion of the ozone layer, HFCs are nevertheless strong greenhouse gases. Their total concentration in the atmosphere is still small (40 parts per trillion), but this amount is increasing about 10% per year. Thus, these substances are becoming increasingly important contributors to the greenhouse effect.
Methane already makes a significant contribution to the greenhouse effect. Each methane molecule has about 25 times the greenhouse effect of a CO2 molecule. Studies of atmospheric gas trapped long ago in the Greenland and Antarctic ice sheets show that the atmospheric methane concentration has increased from pre-industrial values of 0.3 to 0.7 ppm to the present value of about 1.8 ppm. The major sources of methane are associated with agriculture and fossil-fuel use.
Methane is formed in biological processes that occur in low-oxygen environments. Anaerobic bacteria, which flourish in swamps and landfills, near the roots of rice plants, and in the digestive systems of cows and other ruminant animals, produce methane (![]() FIGURE 18.14). It also leaks into the atmosphere during naturalgas extraction and transport. It is estimated that about two-thirds of present-day methane emissions, which are increasing by about 1% per year, are related to human activities.
FIGURE 18.14). It also leaks into the atmosphere during naturalgas extraction and transport. It is estimated that about two-thirds of present-day methane emissions, which are increasing by about 1% per year, are related to human activities.
Methane has a half-life in the atmosphere of about 10 years, whereas CO2 is much longer-lived. This might seem a good thing, but there are indirect effects to consider. Methane is oxidized in the stratosphere, producing water vapor, a powerful greenhouse gas that is otherwise virtually absent from the stratosphere. In the troposphere, methane is attacked by reactive species such as OH radicals or nitrogen oxides, eventually producing other greenhouse gases, such as O3. It has been estimated that the climate-changing effects of CH4 are more than half those of CO2. Given this large contribution, important reductions of the greenhouse effect could be achieved by reducing methane emissions or capturing the emissions for use as a fuel.

![]() FIGURE 18.14 Methane production. Ruminant animals, such as cows and sheep, produce methane in their digestive systems.
FIGURE 18.14 Methane production. Ruminant animals, such as cows and sheep, produce methane in their digestive systems.